Abstract
Background:
Anti-signal recognition particle immune-mediated necrotizing myopathy (anti-SRP IMNM) is a rare autoimmune disorder characterized by muscle weakness and necrosis. Identifying clinical subgroups within this patient population could facilitate the management of the disease.
Objectives:
To identify distinct clinical subgroups of anti-SRP IMNM patients.
Design:
A retrospective study was conducted on anti-SRP IMNM patients treated at West China Hospital of Sichuan University between January 2010 and October 2023.
Methods:
Clinical data were collected. Unsupervised cluster analysis was conducted to classify patients into distinct subgroups based on their clinical features. Statistical analyses were performed to compare the clinical characteristics and outcomes among the identified clusters.
Results:
A total of 116 patients were included in the study, and 3 distinct clinical subgroups were identified: Cluster 3 (acute), Cluster 2 (subacute), and Cluster 1 (poor prognosis). Patients in Cluster 3 exhibited a short disease course (median 3 months), severe muscle weakness (78.38% with Medical Research Council (MRC) score ⩽3), high muscle enzyme levels, and a good response to treatment. Cluster 2 patients were younger (mean age 45.83 years), had a longer disease course (median 6.5 months), milder muscle damage, and lower autoantibody titers. Cluster 1 patients were older (mean age 58.10 years), predominantly male (70.97%), and had higher incidences of interstitial lung disease (70.97%) and cardiac injury (45.16%). In Cluster 1, 16.13% of cases were refractory, and the relapse rate was 38.71%, which was significantly higher compared to the other two clusters.
Conclusion:
This study highlights the clinical heterogeneity among anti-SRP IMNM patients and identifies three distinct clinical subgroups with unique characteristics. These findings provide insights for personalized management.
Plain Language Summary
Anti-SRP positive immune-mediated necrotizing myopathy (anti-SRP IMNM) is a rare and serious muscle disease that causes significant disability. This study aimed to identify different patient groups within this disease using a statistical method called cluster analysis. By examining the medical records of patients from West China Hospital of Sichuan University between January 2010 and October 2023, researchers identified three distinct patient groups, each with different characteristics and treatment responses. The first group, referred to as the “acute group,” had a short disease duration of about 3 months, severe muscle weakness, and higher levels of muscle enzymes, but they responded well to treatment. The second group, the “subacute group,” consisted of younger patients with a slightly longer disease course and milder muscle damage. The third group, labeled the “poor prognosis group,” included older patients who were mostly male, with a higher likelihood of lung and heart complications. This group had the highest number of difficult-to-treat cases and a significant relapse rate. The findings of this study show that anti-SRP IMNM patients are not all the same and that understanding these differences can help doctors provide more personalized and effective treatment options.
Keywords
Introduction
Idiopathic inflammatory myopathies (IIMs) are a rare group of autoimmune diseases that can cause inflammation of skeletal muscle and muscle weakness, and may also affect multiple organs including the skin, joints, lungs, and heart. 1 The muscle involvement in IIMs can lead to disability, even life-threatening complications. 2
Initially, IIMs were classified into dermatomyositis (DM) and polymyositis (PM), primarily distinguished by the presence of a characteristic skin rash in DM. 3 With the identification of an anti-signal recognition particle (anti-SRP) as a myositis-specific antibody (MSA), a new subgroup known as immune-mediated necrotizing myopathy (IMNM) was classified. 4 The muscle pathology of anti-SRP IMNM patients is characterized by predominant muscle fiber necrosis accompanied by few or mild lymphocytic infiltrates. 5 Subsequently, another MSA, anti-3-hydroxy-3-methylglutaryl-coA reductase (anti-HMGCR), was subsequently identified as characteristic of IMNM. 6 So far, IMNM can be classified into three subtypes: anti-HMGCR IMNM, anti-SRP IMNM, and seronegative IMNM (autoantibodies negative).7–9
The clinical manifestations of anti-SRP IMNM include severe proximal muscle weakness, elevated serum creatine kinase (CK) levels, and a poor response to conventional immunosuppressive therapy. 10 Patients may also present with extramuscular symptoms such as interstitial lung disease (ILD) and cardiac involvement.11,12 Given the heterogeneity in disease presentation and progression, it is crucial to understand the different clinical phenotypes of anti-SRP IMNM to effectively tailor treatment strategies.
The objective of this study is to utilize unsupervised cluster analysis to identify distinct clinical subgroups of anti-SRP IMNM patients and offer valuable insights into the therapeutic strategies of anti-SRP IMNM.
Methods
Study design and population
All IMNM patients who visited West China Hospital of Sichuan University from January 2010 to October 2023 were screened for this study. The inclusion criteria included the following: The diagnosis of IMNM must meet the criteria established by the 119th or 224th European Neuromuscular Centre workshop; anti-SRP autoantibodies must be positive; and the patient must be over 18 years of age.13,14 The exclusion criteria include the following: patients with muscle damage due to other known causes and patients with incomplete data.
“Refractory” patients are defined as individuals who, following treatment with an adequate dose of corticosteroids and at least one adequate immunosuppressant (IS), experience less than a 20% improvement in the Medical Research Council sum score (MRC, including limb and neck muscle groups), or a decrease in CK levels of less than 50%, or no improvement in severe respiratory or swallowing muscle dysfunction. An “adequate” glucocorticoid regimen comprises 1 mg/kg/day of prednisone administered daily for at least 1 month. An adequate IS regimen involves a 3-month course of the agent (e.g., azathioprine, methotrexate, mycophenolate mofetil, cyclosporine, tacrolimus, cyclophosphamide) at a known effective dose. The relapse can be defined as the occurrence of any one of the following three conditions after reaching the stable phase: (1) Re-admission for treatment due to aggravation of the original symptoms of the disease; (2) CK level increases more than five times compared to the stable period; and (3) the emergence of new clinical symptoms associated with IMNM.
All subjects completed the electrocardiogram and echocardiography examinations. The definition of cardiac involvement is based on the following: (1) diagnostic evidence of structural or functional changes in the heart or (2) manifestations related to cardiac involvement, such as arrhythmia, orthopnea, and dyspnea after activity, with other associated factors excluded.
A subject is defined as having ILD if they meet the following criteria: the presence of respiratory symptoms such as shortness of breath or a persistent dry cough, along with radiological evidence of interstitial lung abnormalities on high-resolution computed tomography scans.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 15
Data collection
All laboratory test results are from enrolled patients at the first visit. Clinical data, including symptoms, manual muscle testing, other physical signs, laboratory examination results, and treatment regimens, were retrieved from electronic medical records. The neutrophil-to-lymphocyte ratio was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count. The De Ritis Ratio (DRR) is defined as the ratio of aspartate aminotransferase (AST) to alanine aminotransferase (ALT).
Detection of autoantibodies
Myositis-specific autoantibodies and myositis-associated antibodies, including anti-SRP, anti-Jo-1, anti-PL-7, anti-PL-12, anti-EJ, anti-Mi2, anti-NXP2, anti-MDA5, anti-TIF1-γ, anti-SAE, anti-Ku, anti-PM-Scl 75, and anti-Ro-52, were detected with immunoblot testing (anti-HMGCR autoantibodies were measured using an ELISA test), while antinuclear antibodies (ANA) were identified with the Nova Lite Hep-2 ANA kit (EUROIMMUN AG, Lübeck, Germany). Positive findings in patients were validated in duplicate.
Cluster analysis
Variable selection was conducted using theoretical, correlation, and enumeration methods. Correlation analysis was conducted to identify strongly correlated variables, among which representative variables were selected based on clinical significance (see Supplemental Figure 1 for details). Variables with missing data exceeding 5% were excluded from the analysis. For other variables with missing values, imputation was carried out using the median for continuous variables or the mode for categorical variables.
Hopkins Statistic is calculated to evaluate the clustering trend of the data. Gower distance was calculated. Partitioning Around Medoids (PAM) clustering was employed to segment the data. PAM, facilitated by the Gower distance metric, partitions the dataset into “n” clusters, offering insights into inherent patterns and structures within the data. A delineation of how data points are grouped based on their similarities. The quality of the clusters was assessed by the Dunn index. In addition, silhouette plots were used to demonstrate clustering efficacy and data point cohesion. A heatmap displays the differences between the clusters.
The decision tree model was constructed using the “rpart” package in R, with the dataset partitioned into 75% for training and 25% for testing. The model was trained on the training data using the “class” method and the “information” splitting criterion, with a complexity parameter set to 0.0001. The model’s performance was evaluated using confusion matrices and multiclass ROC analysis on both the training and testing subsets.
In addition, Kaplan–Meier (KM) analysis and the log-rank test were used to assess the cumulative relapse-free survival probability across different clusters.
Statistical analysis
The ANOVA analysis was performed to discern variances among the groups. Continuous variables exhibiting a normal distribution pattern were analyzed using the T-test, whereas the U-test was applied to data that deviated from normality. Comparisons of categorical variables were conducted using the Chi-square test. In cases where a group’s numbers fell below 5 or the total count was under 40, Fisher’s exact test was employed. A statistical significance of p < 0.05 was considered. The aforementioned analyses were implemented using R version 4.2.3.
Results
Subgroups by cluster analysis
A total of 256 patients diagnosed with IMNM underwent thorough screening for this study. Of these patients, 27 patients were excluded due to comorbidities that could cause muscle damage. In addition, 29 patients were excluded due to lacking complete data required for the study. Finally, a total of 116 patients with anti-SRP IMNM were included in the study. Twelve variables were selected for cluster analysis (Supplemental Table 1). The Hopkins Statistic was calculated to be 0.679 (Supplemental Figure 2).
The optimal clustering occurred at k = 3, with an average silhouette width value of 0.194 (Figure 1, Supplemental Figure 3). The Dunn index was found to be 0.131. The characteristics of the three clusters were further analyzed.

K-means clustering analysis results. (a) The x-axis represents the number of clusters, while the y-axis represents the silhouette width. Peaks (k = 3) in the plot suggest the optimal number of clusters, with an average silhouette width value of 0.194. (b) This scatter plot visualizes the clustering of data points based on the k-means algorithm. Each point represents an enrolled patient, plotted with respect to variables X and Y. The different colors (red, green, blue) correspond to different clusters, labeled as 1, 2, and 3, respectively.
Demographic and clinical characteristics
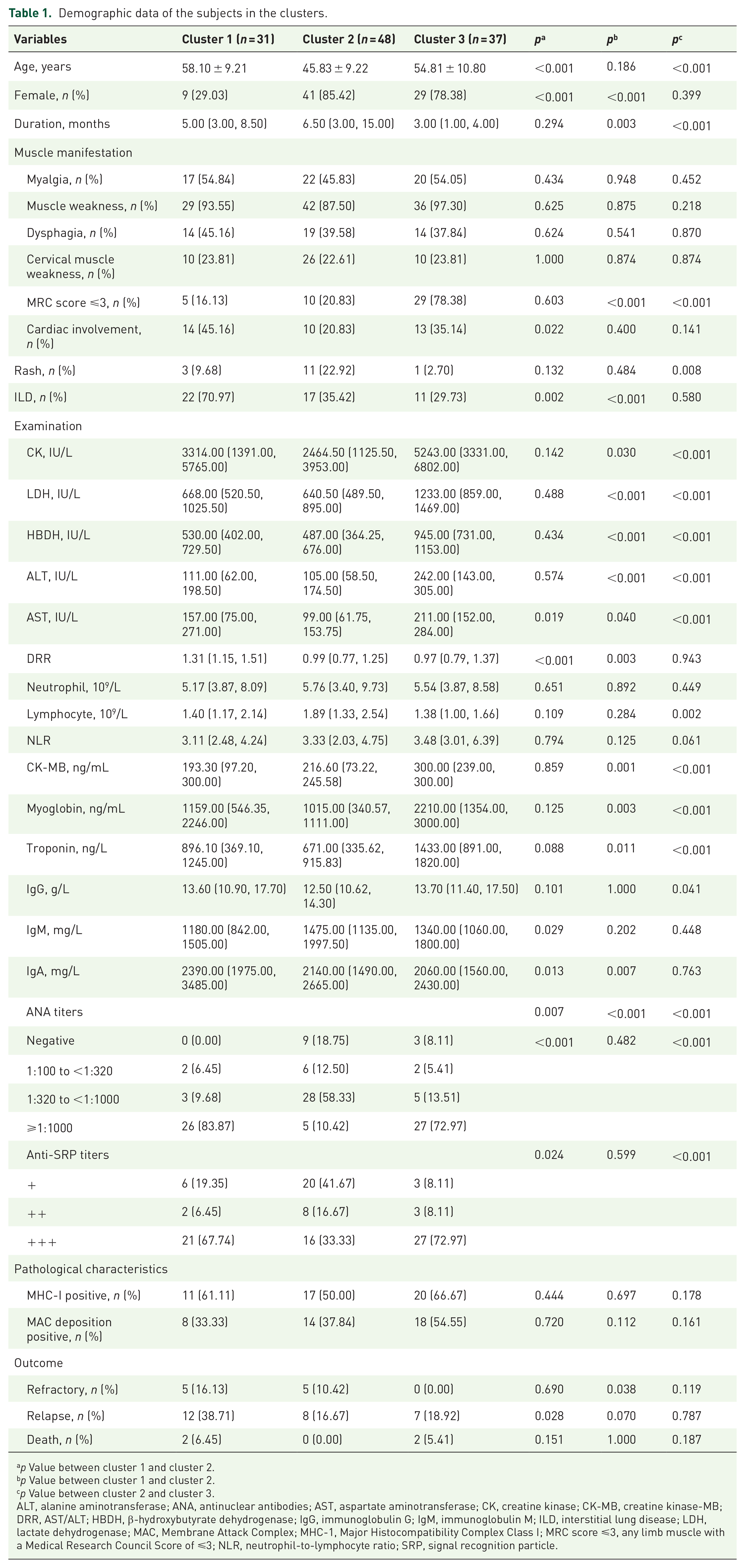
Significant differences were observed in the mean age among the clusters, with Cluster 1 having the highest mean age (58.10 ± 9.21 years) compared to Cluster 2 (45.83 ± 9.22 years) and Cluster 3 (54.81 ± 10.80 years; p < 0.001). In addition, there was a significantly higher percentage of males in Cluster 1 (70.97%) compared to Cluster 2 (14.58%) and Cluster 3 (21.62%; p < 0.001)(Table 1).
Demographic data of the subjects in the clusters.
p Value between cluster 1 and cluster 2.
p Value between cluster 1 and cluster 2.
p Value between cluster 2 and cluster 3.
ALT, alanine aminotransferase; ANA, antinuclear antibodies; AST, aspartate aminotransferase; CK, creatine kinase; CK-MB, creatine kinase-MB; DRR, AST/ALT; HBDH, β-hydroxybutyrate dehydrogenase; IgG, immunoglobulin G; IgM, immunoglobulin M; ILD, interstitial lung disease; LDH, lactate dehydrogenase; MAC, Membrane Attack Complex; MHC-1, Major Histocompatibility Complex Class I; MRC score ⩽3, any limb muscle with a Medical Research Council Score of ⩽3; NLR, neutrophil-to-lymphocyte ratio; SRP, signal recognition particle.
The median duration of disease varied among the clusters, with Cluster 2 having the longest duration (6.50 months), followed by Cluster 1 (5.00 months) and Cluster 3 (3.00 months) (p = 0.003 between Cluster 1 and Cluster 3; p < 0.001 between Cluster 2 and Cluster 3). Muscle manifestations, including myalgia, muscle weakness, and dysphagia, did not show significant differences across the clusters. However, the proportion of subjects with an MRC score of ⩽3 was significantly higher in Cluster 3 (78.38%) compared to Cluster 1 (16.13%) and Cluster 2 (20.83%; p < 0.001) (Table 1).
Cardiac involvement was more common in Cluster 1 (45.16%) compared to Cluster 2 (20.83%; p = 0.022). The prevalence of rash was higher in Cluster 2 (22.92%) than in Cluster 1 (9.68%) and Cluster 3 (2.70%), with a significant difference between Cluster 2 and Cluster 3 (p = 0.008). The incidence of ILD was significantly higher in Cluster 1 (70.97%) compared to Cluster 2 (35.42%) and Cluster 3 (29.73%; p = 0.002 between Cluster 1 and Cluster 2; p < 0.001 between Cluster 1 and Cluster 3)(Table 1).
Laboratory findings
Significant differences in laboratory findings were observed among the clusters. Cluster 3 had the highest median levels of CK, lactate dehydrogenase, β-hydroxybutyrate dehydrogenase, ALT, and AST (p < 0.001). Cluster 1 had the highest median DRR (1.31), significantly different from Cluster 2 (0.99) and Cluster 3 (0.97; p < 0.001).
The ANA titers also showed significant differences across the clusters, with Cluster 1 having the highest proportion of high ANA titers (ANA ⩾1:1000, 83.87%), which was significantly different from both Cluster 2 and Cluster 3 (p < 0.001). In addition, the proportion of high anti-SRP autoantibodies titers was significantly lower in Cluster 2 compared to Cluster 1 and Cluster 3 (p = 0.024 between Cluster 1 and Cluster 2; p < 0.001 between Cluster 2 and Cluster 3).
No significant differences were found in the positive rates of MHC-I and MAC deposition in muscle biopsies among the clusters.
Outcomes
A total of 100 patients were included in the follow-up study, with a period of 39.43 (IQR 22.27, 61.37) months. Among them, 27 patients experienced relapse. The relapse rates were significantly higher in Cluster 1 (38.71%) compared to Cluster 2 (16.67%; p = 0.028). The KM analysis of relapse-free survival probability showed no statistical differences between the three clusters. However, a statistical significance was found between Cluster 1 patients and non-Cluster 1 patients (p# = 0.035, Figure 2).

Kaplan–Meier analysis of relapse-free survival probability.
In all, the demographic, clinical, and laboratory characteristics, as well as outcomes, varied significantly among the three clusters. Patients in Cluster 1 are characterized by being elderly, male, having cardiac injury and ILD, poor response to treatment, and a high risk of relapse, classified as the “poor prognosis” type. Cluster 2 is characterized by younger age, longer disease duration, and mild symptoms, classified as the “subacute” type. Cluster 3 is characterized by a short disease duration, high titers of anti-SRP autoantibodies, and severe muscle damage, classified as the “acute” type. The differences in clinical characteristics between clusters are represented using a heatmap (Figure 3).
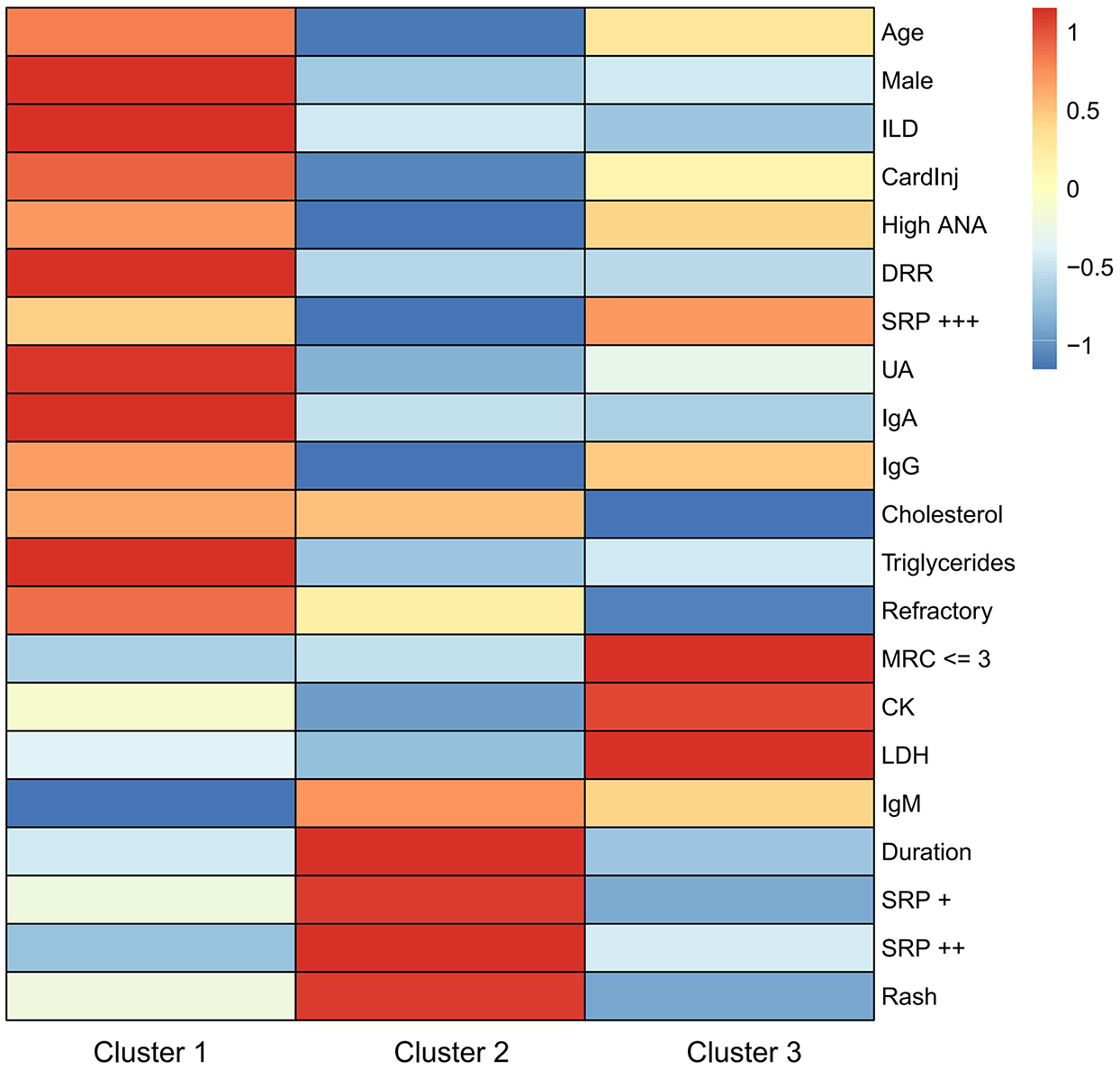
Heatmap of cluster analysis.
A decision tree model was trained to distinguish key features from clusters (accuracy: 0.784, 95% confidence interval: 0.684, 0.865, Figure 4). CP values and variable weights are shown in Supplemental Figure 4.
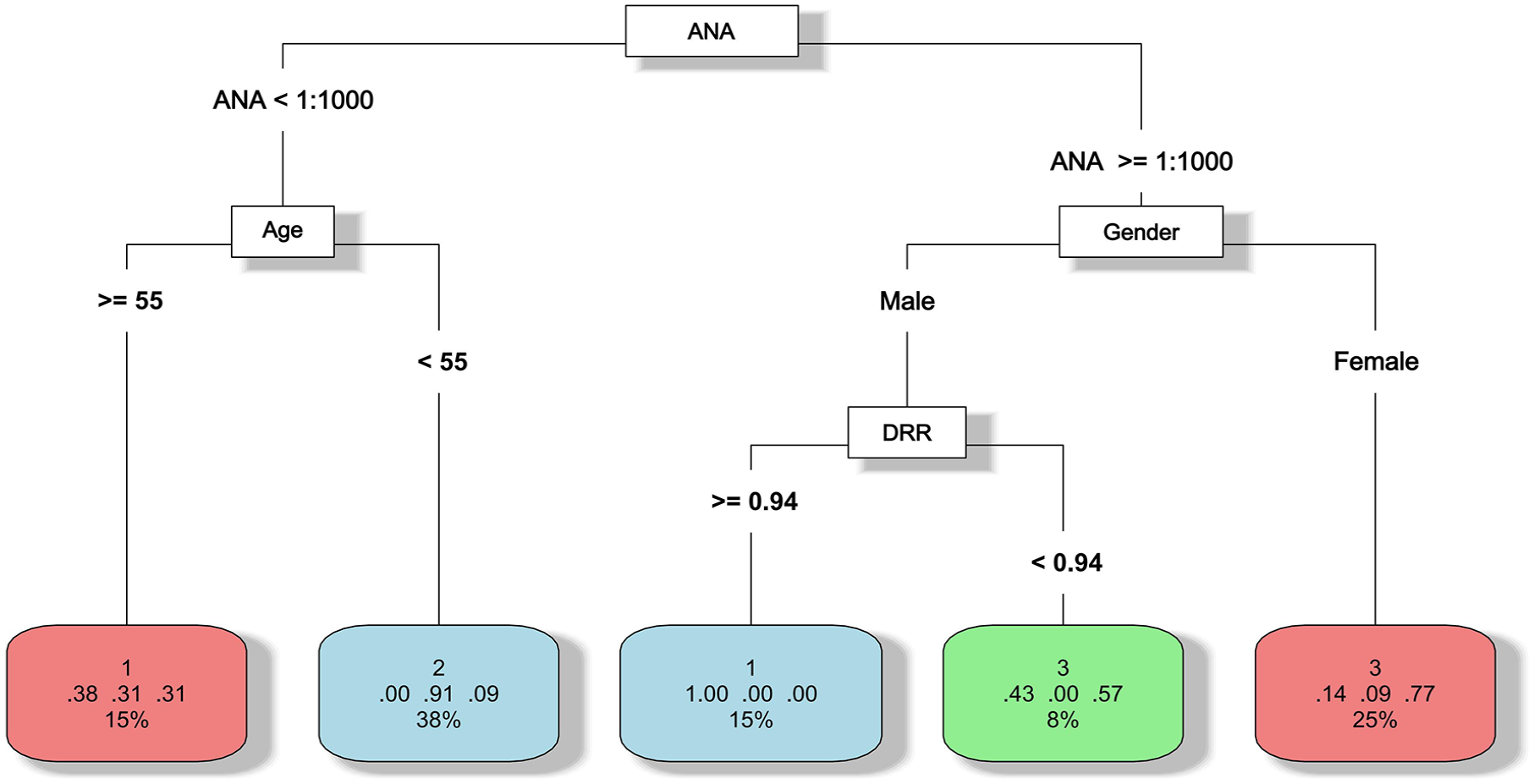
Decision tree model for key feature distinction.
Discussion
To our knowledge, this is the largest retrospective study on anti-SRP IMNM. Through unsupervised cluster analysis, the study identified that anti-SRP IMNM patients can be distinctly categorized into three groups based on their clinical characteristics, labeled as “acute,” “subacute,” and “poor prognosis.”
The progression of anti-SRP IMNM is variable, manifesting as either an acute or subacute onset.9,16 In our study, patients in cluster 3 demonstrated the acute type. Compared to the other two groups, these patients had a shorter disease course, more pronounced muscle weakness, and higher levels of muscle enzymes. In addition, this group showed high titers of ANA and anti-SRP autoantibodies. Interestingly, all patients in this group responded well to initial treatment. The association between serum CK levels and anti-SRP autoantibodies in anti-SRP IMNM remains unclear. One study found no significant correlation between CK levels and the titers of anti-SRP autoantibodies. 16 Conversely, another study demonstrated a strong association between autoantibody levels and CK levels in anti-SRP IMNM patients undergoing treatment. 17
IMNM is generally considered to be a disease with a poor prognosis, especially for anti-SRP IMNM patients.18–20 According to previous studies, the incidence rate of refractory cases in IMNM is approximately 8.8%–59.37%, and the relapse rate is approximately 38.6%–53.8%.20,21 This study found that the prognosis of anti-SRP IMNM patients varies across different clusters. Patients in cluster 3 responded adequately to treatment. Compared to the other two clusters, patients in cluster 1 showed a poorer prognosis. This group presented notable characteristics: male, older age, ILD, cardiac injury, and a high proportion of refractory and relapse cases.
The association between gender and prognosis in anti-SRP IMNM has been reported. According to previous studies, the female-to-male ratio in anti-SRP IMNM ranges from 0.89 to 4.24.18,19 Female gender was associated with a greater improvement in serum CK levels. 19 In another study, male gender is associated with refractory cases, with an odds ratio (OR) value of 19.57. 20 Conversely, one study showed that female patients had higher modified Rankin Scale scores during follow-up. 16
This study found a relatively high occurrence rate of ILD in anti-SRP IMNM patients, especially in cluster 1, which is consistent with the previous study. 20 ILD is also considered to be associated with a poor prognosis in IMNM.21,22 In a retrospective study on IMNM, ILD was found to be strongly associated with refractory patients, with an OR value of 39.70. 19 In addition, the occurrence of ILD may be associated with older age. 23 Therefore, through cluster analysis, we identified high-risk factors associated with poor prognosis in anti-SRP IMNM patients, including male gender, older age, and ILD. This subset of patients is concentrated in cluster 1.
The induction therapy for anti-SRP IMNM includes glucocorticoids and immunosuppressants, such as methotrexate. 14 Anti-SRP IMNM may exhibit a poor response to treatment and have a high recurrence rate. For patients who respond poorly to glucocorticoids and immunosuppressants, combination therapy with rituximab should be considered. 24 This study provides a clue for stratified treatment of anti-SRP IMNM. According to this study, patients who are male, older, or have concurrent ILD are indicative of a poor treatment response and a higher recurrence rate. Therefore, these patients should receive more intensive induction therapy and be closely monitored during follow-up.
This study demonstrates the diversity of clinical characteristics among anti-SRP IMNM patients but it also has some limitations. Although the sample size in this study is larger than that of previous research, it is still insufficient for machine learning applications. Consequently, further validation is needed to generalize the research findings.
Conclusion
In conclusion, this study represents the largest retrospective analysis of anti-SRP IMNM to date. We use unsupervised cluster analysis to reveal the clinical characteristics of subgroups among anti-SRP IMNM patients. Key findings include that the acute group had a shorter disease course, pronounced muscle weakness, high muscle enzyme levels, and responded well to treatment; the subacute group had a longer disease course and milder muscle damage; the poor prognosis group featured high-risk factors such as male gender, older age, and ILD, leading to worse outcomes. This study highlights the clinical diversity of anti-SRP IMNM patients and provides valuable insights for personalized management and treatment strategies.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251314697 – Supplemental material for Identification of clinical subgroups in anti-SRP positive immune-mediated necrotizing myopathy patients using cluster analysis
Supplemental material, sj-docx-1-tab-10.1177_1759720X251314697 for Identification of clinical subgroups in anti-SRP positive immune-mediated necrotizing myopathy patients using cluster analysis by Beibei Cui, Hongjiang Liu, Ruiting Liu, Geng Yin and Qibing Xie in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
