Abstract
Background:
Real-world evidence supporting a safe and effective transition from rituximab originator (RTX-O) to its biosimilars (RTX-B) in autoimmune rheumatic diseases (ARDs) is still limited.
Objectives:
The primary aims of this study were to evaluate the long-term persistence of RTX-B after the non-medical switch (NMS) from RTX-O in ARD patients, and to explore the RTX-B safety profile. The secondary aims were to evaluate the impact of different factors on RTX-B drug retention rate (DRR) and to identify any factors associated with RTX-B discontinuation.
Design:
Retrospective observational study.
Methods:
We included consecutive ARD patients undergoing NMS from RTX-O to GP2013 or CT-P10 from January 2018 to December 2020. RTX-B DRR was estimated by Kaplan–Meier plot analysis and compared according to different factors by the Log-rank test; the Cox proportional hazard model was used to detect factors associated with RTX-B discontinuation in the first 36 months.
Results:
We enrolled 181 patients switching to RTX-B: GP2013 in 143 (79.0%) cases and CT-P10 in 38 (21.0%). The estimated DRR for RTX-B was 81.5% at 12 months, 80.6% at 24 months, and 77.4% at 36 months. The incidence of adverse events with RTX-B was 12.6/100 patients/year. In the Log-rank test, no statistically significant differences were observed in the RTX-B DRR according to sex (p = 0.171), ARD diagnosis (p = 0.281), and concomitant immunosuppressive therapy (p = 0.054); on the contrary, patients on GP2013 showed a higher DRR than those on CT-P10 (p < 0.001). In the Cox proportional hazard analysis, the switch to CT-P10 was associated with a higher probability of stopping treatment (hazard ratio, 1.83 (confidence interval, 1.10–3.04), p = 0.02).
Conclusion:
NMS to RTX-B is associated with a high chance of retaining the drug for up to 36 months, irrespective of the diagnosis. GP2013 showed a higher retention rate than CT-P10.
Introduction
The market availability of biosimilar drugs offers the potential for broader access to biologic therapy for various autoimmune rheumatic diseases (ARDs) globally, due to the increased sustainability of pharmaceutical costs. 1 On one hand, the commercialization of biosimilars reduces the cost of expensive originator drugs through market competition, making these treatments more affordable. On the other hand, the savings generated from biosimilars can be used to fund access to new medications, including advanced biotechnological drugs, thereby enhancing the availability of therapeutic innovation.2,3
Rituximab (RTX), a chimeric anti-CD20 antibody, is licensed for the treatment of rheumatoid arthritis (RA) and antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis (AAV) and is also used off-label for connective tissue diseases (CTDs).4 –8 In Europe, two RTX biosimilars (RTX-B) are available: GP2013 and CT-P10. These biosimilars exhibit comparable physicochemical and pharmacodynamic properties to the RTX originator (RTX-O), demonstrating equivalent efficacy in treating both ARDs and hematologic disorders. Moreover, these biosimilars have not introduced any new safety concerns.9,10
As of the current date, no established recommendations exist regarding the transition from originator biologics to biosimilars, or among biosimilars themselves, despite this being a widespread practice in numerous countries.11,12 The Italian Medicines Agency posits that biosimilar drugs cannot be equated with generic drugs. Therefore, the selection of treatment remains a clinical decision entrusted to the prescribing physician, who is also responsible for ensuring the sustainable use of healthcare resources within the specific regional regulations, and for providing patients with accurate information (https://www.aifa.gov.it/position-paper accessed on July 2, 2024).
This study provides real-world data on long-term persistence with RTX-B treatment, as well as their safety profile, in subjects with ARDs (including RA, AAV, and CTDs) who switched from RTX-O to GP2013 or CT-P10 for non-medical reasons.
Methods
Study objectives
This is a multicentric retrospective observational study. The primary objectives of the study were as follows: (1) to evaluate the long-term persistence with RTX-B treatment after the non-medical switch (NMS) in patients with ARDs and (2) to explore the RTX-B safety profile.
The secondary objectives were as follows: (1) to evaluate the impact of sex, diagnosis, RTX-B molecule employed, and concomitant immunosuppressive treatment on the long-term persistence with RTX-B treatment; (2) to identify any factors associated with RTX-B discontinuation; and (3) to explore the safety profile of GP2013 and CT-P10 separately.
Inclusion criteria, operative procedures, and data collection
Consecutive adult patients affected by ARDs treated with RTX-O at two Italian rheumatological centers (Siena and Florence University Hospitals) were screened for possible inclusion in the study. The diagnosis of specific ARDs was established clinically and corroborated by meeting the classification criteria set forth by the American College of Rheumatology or the European League Against Rheumatism for each disease. Patients qualified for inclusion if they had been receiving treatment with RTX-O for a minimum duration of 6 months, equivalent to one treatment cycle, prior to switching to one of the two available RTX-B (GP2013 or CT-P10) between January 2018 and December 2020.
All enrolled patients signed informed consent before undergoing RTX treatment. All treatment switches were conducted based on non-medical considerations, in accordance with the Italian healthcare system’s regional regulations. The choice between GP2013 and CT-P10 depended on local availability at the treatment site at the time of the switch. Information about NMS was provided to every patient during clinical consultations according to best clinical practices. The NMS was performed with the treating physician’s approval, following patient consent and a careful evaluation of the clinical state. RTX-O treatment schedules (two 1 g infusions separated by 2 weeks/a single 1 g infusion/two 500 mg infusions separated by 2 weeks/a single 500 mg infusion/others) were assigned to each patient according to the treating physician’s experience and disease-specific recommendations, to achieve disease remission or minimal disease activity, as appropriate for each specific patient. RTX was re-applied with >6-month intervals based on disease activity, as determined by the clinical evaluation of the treating physician. At the time of the switch to RTX-B, the previous therapeutic scheme was maintained in all patients. Patients’ consent and regulatory approval were obtained in accordance with the clinical practices of the treating hospitals for off-label RTX treatment schedules. This includes off-label indications, monotherapy (necessary in cases of methotrexate intolerance or contraindication), and the use of RTX as a first-line biologic therapy when required to tailor treatment to the specific clinical needs of the patient.
The following data were retrospectively collected at the time of NMS: age, sex, diagnosis, disease duration, concomitant treatment with conventional synthetic disease-modifying anti-rheumatic drugs (csDMARDs), RTX-O treatment schedule, line of treatment, RTX-B molecule, number of RTX-O and RTX-B infusions, and number and type of adverse events (AEs) reported during the entire RTX therapy.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 13
Statistical analysis
In alignment with the study’s primary objectives, the following primary endpoints were defined: (1) the RTX-B drug retention rate (DRR) over a 36-month treatment period and (2) the incidence and type of AEs during RTX-B therapy. In alignment with the secondary aims of the study, the following secondary endpoints were defined: (1) a statistically significant difference in the DRR of RTX-B according to the mentioned modifying factors; (2) the risk of experiencing RTX-B discontinuation, measured as hazard ratios (HR) with 95% confidence intervals (CI), considering the RTX-B molecules, sex, and diagnosis as factors; and (3) the incidence and type of AEs during GP2013 and CT-P10 therapy.
Data were computed using IBM SPSS Statistics for Windows, version 28. Descriptive statistics were applied to calculate absolute and relative frequencies, mean and standard deviation, or median and interquartile range, as appropriate. The Shapiro–Wilk test was used to analyze the normality distribution of the data. The RTX-B DRR was estimated by a Kaplan–Meier plot, with the event being RTX-B discontinuation. When examining treatment retention, the time of RTX-B withdrawal was defined as the date of the patient’s last received infusion. The Log-rank test was employed to assess any potential differences in terms of DRR among factors, including sex, type of ARD diagnosis (RA vs other diagnoses), and concomitant therapy with csDMARD at the time of NMS. Event-free survival was studied with a Cox proportional hazard model, using 95% CI for HR to detect factors associated with RTX-B discontinuation, including the RTX-B molecules, sex, and ARD diagnosis. The assumption of proportionality over time for different strata was assessed. The incidence of AEs was determined based on the actual duration of treatment for subjects on RTX-O, RTX-B, GP2013, and CT-P10. Subjects with missing data/lost to follow-up were excluded from the analysis. The threshold for statistical significance was set at p < 0.05, and all p-values were two-sided.
Results
Description of the population on RTX-O
The study included 181 participants (144 female and 37 male patients). Their baseline information, including demographics, diagnoses, and details of RTX-O treatment, is provided in Table 1.
Demographic, clinical, and therapeutic data at the start of RTX originator.
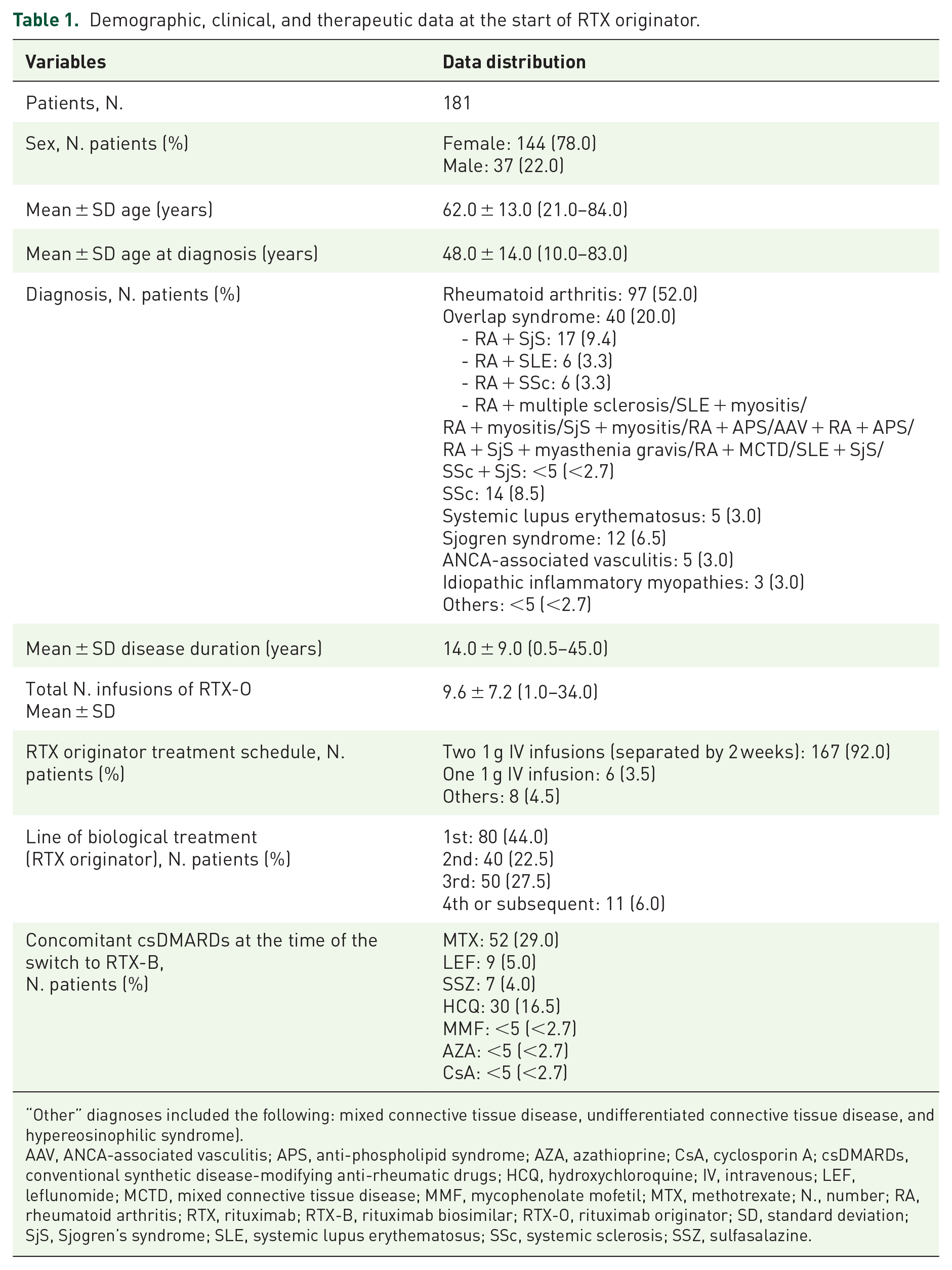
“Other” diagnoses included the following: mixed connective tissue disease, undifferentiated connective tissue disease, and hypereosinophilic syndrome).
AAV, ANCA-associated vasculitis; APS, anti-phospholipid syndrome; AZA, azathioprine; CsA, cyclosporin A; csDMARDs, conventional synthetic disease-modifying anti-rheumatic drugs; HCQ, hydroxychloroquine; IV, intravenous; LEF, leflunomide; MCTD, mixed connective tissue disease; MMF, mycophenolate mofetil; MTX, methotrexate; N., number; RA, rheumatoid arthritis; RTX, rituximab; RTX-B, rituximab biosimilar; RTX-O, rituximab originator; SD, standard deviation; SjS, Sjogren’s syndrome; SLE, systemic lupus erythematosus; SSc, systemic sclerosis; SSZ, sulfasalazine.
DRR of RTX-B
A NMS was made from RTX-O to GP2013 in 143 cases (79.0%), while in 38 cases (21.0%), patients received CT-P10. At the time of the switch to RTX-B, 89 (49.0%) patients were concomitantly treated with csDMARDs.
The estimated cumulative DRR for RTX-B was 81.5% at 12 months, 80.6% at 24 months, and 77.4% at 36 months since the time of NMS, with a mean follow-up of 17 ± 24 months (range 1–247) (Figure 1(a)).

(a) Rituximab biosimilar cumulative drug retention rate: 81.5% at 12 months, 80.6% at 24 months, and 77.4% at 36 months. (b) Comparison of GP2013 and CT-P10 survival curves: GP2013 showed a higher drug survival (Log-rank test: p < 0.001). (c) Survival curves of rituximab biosimilars by stratifying the population according to disease diagnosis (rheumatoid arthritis vs other diseases) (Log-rank test: p = 0.281). (d) Survival curves of rituximab biosimilars stratified by sex (Log-rank test: p = 0.171).
According to the Log-rank test, subjects treated with GP2013 showed a higher DRR than those on CT-P10 (p < 0.001) (Figure 1(b)). Patients treated with concomitant csDMARDs at the time of NMS did not show a statistically significant difference in the DRR compared to those on monotherapy (p = 0.054). No statistically significant differences were observed in DRR when stratifying the population according to sex (p = 0.171) or ARD diagnosis (RA vs other diagnoses, p = 0.281) (Figure 1(c) and (d)).
The Cox proportional hazard analysis, including the RTX-B molecules, sex, and ARD diagnosis showed that the switch to CT-P10 was associated with a higher probability of stopping treatment (HR, 1.83 (CI, 1.10–3.04), p = 0.02) while the ARD diagnosis (RA vs others) or sex did not impact treatment retention (p = 0.393 and p = 0.239, respectively).
Analysis of AEs
Patients undergoing GP2013 or CT-P10 reported AEs in 21 (14.7%) cases (10.6 events/100 patients/year) and 12 (31.6%) cases (26.0 events/100 patients/year), respectively, with an overall incidence of 12.6 events/100 patients/year with RTX-B.
Specifically, 22 patients experienced infusion reactions (12.2%), while <5 patients (<2.7%) had infections, neoplasia (thyroid, breast), disease relapses, lymphopenia, hypogammaglobulinemia, or abdominal pain. For infections, cases of pleurisy, pharyngotonsillitis, and urinary infection were reported in less than five cases each, all of which were treated with antibiotic therapies. Infusion reactions included throat/ears tickling in eight cases, skin rash in five cases, and anaphylaxis, localized or diffuse itching, dyspnea, asthenia, nausea, vomiting, diarrhea, vertigo, tachycardia, cough, or hypotension in less than five cases each. In patients experiencing infusion reactions, the drug infusion rate was reduced or temporarily stopped in 11 cases, RTX dosage was reduced in <5 cases, systemic glucocorticoids were administered in <5 cases, and no intervention was needed in <5 cases. All patients who developed neoplastic or hematologic AEs discontinued RTX-B treatment.
Table 2 provides a detailed breakdown of AEs leading to treatment discontinuation in subjects treated with GP2013 and CT-P10, along with details of the GP2013 and CT-P10 subgroups.
Details of the GP2013 and CT-P10 subgroups and respective treatment courses.

AEs, adverse events; N., number; RTX-B, rituximab biosimilar; RTX-O, rituximab originator; SD, standard deviation.
Discussion
Our findings indicate that RTX-B exhibits a favorable DRR following NMS from RTX-O, with an anticipated continuation of treatment in over 77% of patients at a follow-up period of 36 months. Specifically, patients undergoing treatment with GP2013 demonstrated a superior retention rate compared to those treated with CT-P10, which might be negatively affected by a higher incidence of AEs. Furthermore, the survival of RTX-B did not appear to be influenced by the specific diagnosis (RA vs other ARDs) or by concomitant treatment with csDMARDs at the time of the switch from RTX-O. However, this latter factor warrants further exploration in a larger cohort to enable a more detailed stratification of patients, especially considering the borderline statistical significance observed in this cohort.
In a large retrospective study, Melville et al. found that RA patients switching from RXT-O to CT-P10 showed a similar change in DAS28-CRP compared to those remaining on RTX-O, with 16.5% of subjects withdrawing RTX-B for loss of efficacy and 2% for AEs. They also observed a higher risk of CT-P10 discontinuation in subjects with comorbidities and more than two previous biologics taken. 14 Similarly, during the extension period of a randomized controlled trial (RCT) in RA patients, the switch from RTX-O to CT-P10 and vice versa was well tolerated, without significant differences in efficacy, pharmacodynamics, immunogenicity, and safety outcomes in all the study arms. 15 As for GP2013, Antonelou et al. 16 estimated a drug survival rate aligned with our findings (80.0% at 1 year, 57.7% at 2 years), with 38.6% of RA patients discontinuing the drug for clinical remission, 27.1% for a not better-defined physician’s decision, and 14.3% for AEs (14.3%). Regarding more heterogeneous cohorts in terms of ARD diagnoses, a large Swedish observational study involving 834 first-ever starters of RTX found indicated no major differences in 1-year treatment retention for RTX-O and RTX-B. 17 In addition, 360 subjects from the same cohort were switched for non-medical reasons from RTX-O to GP2013 or CT-P10, showing a comparable 1-year retention rate, reaching 88.0% for GP2013 and 83.0% for CT-P10. 17 In comparison to our experience, these studies exhibit a limitation due to their shorter duration of observation; nonetheless, their major strength lies in the inclusion of one or more control groups, which further corroborates the results of our study. Collectively, the existing evidence endorses the effectiveness of switching to RTX-B within a clinical context.
The safety of NMS from RTX-O to RTX-B deserves special consideration. Our findings suggest a higher incidence of AEs in subjects switching to CT-P10 than those receiving GP2013, excluding instances of disease relapse. This phenomenon may be reflected in a lower retention rate for CT-P10. It is important to recognize the uneven patient allocation between the CT-P10 and GP2013 groups as a potential source of bias, along with the lack of information about patients’ comorbidities, disease activity status at the time of the switch, and the exact therapeutic interval for each patient. Nonetheless, it is plausible to consider that minor variations in the excipients of these biosimilar drugs, despite their analogous pharmacological actions, may potentially influence clinical outcomes, their safety profile, and consequently their drug survival. This phenomenon has been previously postulated in the context of adalimumab biosimilars within a Danish cohort study. 18
Although the literature offers limited data from direct comparisons between the two RTX-B, studies on the safety of the switch from RTX-O to RTX-B provide reassuring results. In their study of CT-P10, Bahap-Kara et al. reported findings indicating a safety profile for CT-P10 that is comparable, if not superior, to that of RTX-O across 128 subjects with ARDs. This comparison took into account infusion reactions (occurring in 39.1% of patients treated with CT-P10 vs 46.8% with RTX-O), infections (33.9% for CT-P10 vs 35.0% for RTX-O), and hypogammaglobulinemia. 19 In a large multicentric study, Campochiaro et al. 20 evaluated the safety and efficacy of CT-P10 in subjects with systemic sclerosis over a 6-month follow-up, reporting transient neutropenia in one patient as the only AE in the cohort, alongside a remarkable improvement of skin sclerosis and articular involvement. Furthermore, in the context of AAV, CT-P10 demonstrated equivalence to RTX-O with respect to the incidence of hypogammaglobulinemia, B-cell depletion, and the frequency of infusion reactions. 16 On the other hand, in a subset of 38 patients diagnosed with IgG4-related disease, predominantly treatment-naïve to RTX, approximately 37.0% experienced AEs on CT-P10, in line with previous experiences with RTX-O. 21 As for GP2013, patients with RA switched from RTX-O faced a comparable frequency of AEs compared to patients continuing RTX-O, with regard to hypersensitivity (9.4% and 11.1%, respectively) and infusion-related reactions (11.3% and 18.5%, respectively), in a multinational, randomized, double-blind, parallel-group safety study. 22 Furthermore, according to the latest Periodic Safety Update Report, safety concerns for GP2013 match those of the reference biologic, with a favorable overall benefit–risk profile. 23
This study has several limitations, primarily due to its retrospective nature, which may have introduced recall bias and affected the accuracy of the drug safety profile assessment. A potential source of bias in estimating the DRRs for CT-P10 and GP2013 treatments may arise from the uneven allocation of patients between these groups, along with the lack of information regarding comorbidities, disease activity status at the time of the switch, and the exact therapeutic interval for RTX-B in each patient. Furthermore, the study design assumed all participants switched from RTX-O to RTX-B, precluding a direct comparison of AE incidence between RTX-O and RTX-B. This limitation arises because subjects who experienced serious AEs on RTX-O likely discontinued the drug and were thus not included in the study. In addition, the potential for reporting bias due to more rigorous monitoring of patients transitioning to biosimilars immediately following their market introduction cannot be disregarded.
Conclusion
In conclusion, our data suggest that patients switching from RTX-O to RTX-B for non-medical reasons may be likely to persist on therapy for 36 months irrespective of their specific diagnosis of ARDs, with GP2013 potentially showing a higher DRR and a lower incidence of AEs than CT-P10.
Supplemental Material
sj-pdf-1-tab-10.1177_1759720X241292312 – Supplemental material for Switching from rituximab originator to GP2013 or CT-P10 biosimilars in autoimmune rheumatic diseases: drug retention rate and safety data from a multicentric retrospective cohort
Supplemental material, sj-pdf-1-tab-10.1177_1759720X241292312 for Switching from rituximab originator to GP2013 or CT-P10 biosimilars in autoimmune rheumatic diseases: drug retention rate and safety data from a multicentric retrospective cohort by Stefano Gentileschi, Cosimo Bruni, Carla Gaggiano, Roberto D’Alessandro, Giovanni Pacini, Jurgen Sota, Serena Guiducci, Marco Matucci Cerinic and Bruno Frediani in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
