Abstract
Rheumatologists are increasingly utilizing ultrasound for suspected giant cell arteritis (GCA) or Takayasu arteritis (TAK). This enables direct confirmation of a suspected diagnosis within the examination room without further referrals. Rheumatologists can ask additional questions and explain findings to their patients while performing ultrasound, preferably in fast-track clinics to prevent vision loss. Vascular ultrasound for suspected vasculitis was recently integrated into rheumatology training in Germany. New European Alliance of Associations for Rheumatology recommendations prioritize ultrasound as the first imaging tool for suspected GCA and recommend it as an imaging option for suspected TAK alongside magnetic resonance imaging, positron emission tomography and computed tomography. Ultrasound is integral to the new classification criteria for GCA and TAK. Diagnosis is based on consistent clinical and ultrasound findings. Inconclusive cases require histology or additional imaging tests. Robust evidence establishes high sensitivities and specificities for ultrasound. Reliability is good among experts. Ultrasound reveals a characteristic non-compressible ‘halo sign’ indicating intima-media thickening (IMT) and, in acute disease, artery wall oedema. Ultrasound can further identify stenoses, occlusions and aneurysms, and IMT can be measured. In suspected GCA, ultrasound should include at least the temporal and axillary arteries bilaterally. Nearly all other arteries are accessible except the descending thoracic aorta. TAK mostly involves the common carotid and subclavian arteries. Ultrasound detects subclinical GCA in over 20% of polymyalgia rheumatica (PMR) patients without GCA symptoms. Patients with silent GCA should be treated as GCA because they experience more relapses and require higher glucocorticoid doses than PMR patients without GCA. Scores based on intima-thickness (IMT) of temporal and axillary arteries aid follow-up of GCA, particularly in trials. The IMT decreases more rapidly in temporal than in axillary arteries. Ascending aorta ultrasound helps monitor patients with extracranial GCA for the development of aneurysms. Experienced sonologists can easily identify pitfalls, which will be addressed in this article.
Plain language summary
Rheumatologists use ultrasound to diagnose two types of blood vessel inflammation: giant cell arteritis (GCA) or Takayasu arteritis (TAK). They can do this right in their office during the examination, without sending patients elsewhere. During the ultrasound, rheumatologists can talk with patients about what they see. This is especially helpful in fast-track clinics to prevent vision loss. In Germany, doctors training to become rheumatologists learn how to use ultrasound to check for problems like these. An organization called ‘European Alliance of Associations for Rheumatology (EULAR)’ recommends using ultrasound as the main way to look for GCA and, if needed, for TAK. Ultrasound is also an important part of the new classification criteria for GCA and TAK. However, doctors do not rely on ultrasound alone. They also look what patients are feeling and do other medical tests. If ultrasound is not clear enough, doctors might need to do more tests like taking a small piece of tissue (biopsy) or using other kinds of imaging like MRI or CT scans. Ultrasound can show some characteristic signs of blood vessel inflammation, like a ‘halo sign,’ which tells doctors that the blood vessel walls are thicker than normal. It can also spot other problems like blockages or bulges in the blood vessels. When doctors suspect GCA, they should at least examine the arteries at the forehead and at the armpit. Most of the time, these areas are easy to see with ultrasound, but some areas might be harder to reach. Sometimes, people can have blood vessel inflammation without feeling any typical symptoms. Ultrasound can still find this silent inflammation in more than 20% of people with a condition called polymyalgia rheumatica (PMR). Even though these patients do not have typical symptoms of GCA, it is important to treat them the same way as those with GCA. Otherwise, they may have more flare-ups and need higher doses of glucocorticoids. Doctors may measure the thickness of the artery walls over time in research studies. This helps them to see if treatments are working well. The wall thickness decreases faster in arteries of the head than in larger arteries outside the head. Ultrasound of the aorta close to heart helps to find out if a widening of the aorta develops. This can be dangerous because of rupture.
Keywords
Introduction
Rheumatologists routinely diagnose and manage vasculitides. The Chapel Hill nomenclature categorizes primary vasculitides based on vessel size. It distinguishes vasculitides affecting large, medium, small and variable vessels. 1 This review focuses on large-vessel vasculitis (LVV), specifically giant cell arteritis (GCA) and Takayasu arteritis (TAK), where ultrasound effectively depicts distinct pathology. Other large-vessel-affecting vasculitides exist. Active vasculitis poses a risk factor for venous thrombosis. 2 Thrombosis is caused directly by vasculitis in Behçet’s syndrome, where increased vein wall diameters are noted even in veins without thrombosis. 3
Musculoskeletal ultrasound has become an important diagnostic tool for rheumatologists, playing a significant role in rheumatology training across many countries. 4 With widespread ultrasound equipment and expertise in rheumatology practice, it is increasingly employed for examining various organs in conditions such as Sjögren’s syndrome affecting salivary glands, 5 interstitial lung disease, 6 systemic sclerosis affecting the skin, 7 and connective tissue diseases through echocardiography. 8
Temporal artery colour Doppler ultrasound in GCA was initially described in 1995,9–11 and many subsequent studies have solidified its role. Ultrasound is now integral to the 2022 American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) classification criteria for GCA 12 and TAK. 13
The 2018 EULAR recommendations on imaging in LVV were the first to recommend imaging as equivalent to histology in diagnosis of GCA. 14 The 2023 update strengthened the role of ultrasound, recommending it as the primary imaging modality to investigate for suspected GCA. 15 Various other European guidelines including those from EULAR on management of LVV, 16 the European Headache Federation, 17 the British Society for Rheumatology, 18 the Swedish Society of Rheumatology, 19 the Norwegian Society of Rheumatology 20 and German societies 21 recommend ultrasound for confirming GCA.
Probably the first country to include ultrasound for suspected vasculitis in the curriculum for rheumatology training was Germany in 2023. 22 This review aims to provide an overview of current literature and practical aspects of ultrasound scanning in clinical practice.
Giant cell arteritis and Takayasu arteritis
Both diseases share similarities, with CD4+ T cells and macrophages forming granulomatous lesions in the walls of large arteries, particularly in the media. However, the composition of the wall-infiltrating immune cell compartment differs between TAK and GCA. The ratio of CD4/CD8 T cells is high in GCA and low in TAK. TAK aortitis exhibits a higher proportion of natural killer cells, and the adventitia is typically more expanded in TAK than in GCA, suggesting distinct inflammatory effector pathways. 23
Some clinical features also differ between GCA and TAK:
Age: GCA predominantly affects individuals around 70 years, with almost all patients over 50. TAK has a mean onset age of approximately 25 years, affecting mostly individuals under 40.
Artery involvement: While extracranial artery involvement may be similar, temporal, facial and occipital arteries are never involved in TAK. Axillary vasculitis is more common in GCA, whereas the abdominal aorta, mesenteric and renal arteries are more frequently affected in TAK. The subclavian and carotid arteries and the aorta are the most affected arteries in TAK.24,25
Prognosis: The disease course is longer, and stenoses of extracranial arteries are more prevalent in TAK.
Treatment: Glucocorticoids are affective in both diseases, but responses to other treatments vary. For instance, tumor necrose factor (TNF) inhibitors are effective in TAK, but not in GCA.26,27
In the author’s GCA fast-track clinic in Berlin-Buch, out of 367 GCA patients with newly diagnosed GCA in a 4-year period, 48% had cranial GCA, 21% of them had extracranial GCA and 32% of them had mixed GCA involving both cranial and extracranial arteries. 28 Extracranial vasculitis, mostly of the aorta, subclavian and axillary arteries, correlates with younger age and female sex, with less common symptoms like headache, jaw claudication and vision loss.29,30 Patients with extracranial GCA tend to require glucocorticoids for a longer duration with a higher cumulative dose. 31
ACR/EULAR classification criteria for giant cell arteritis. 12
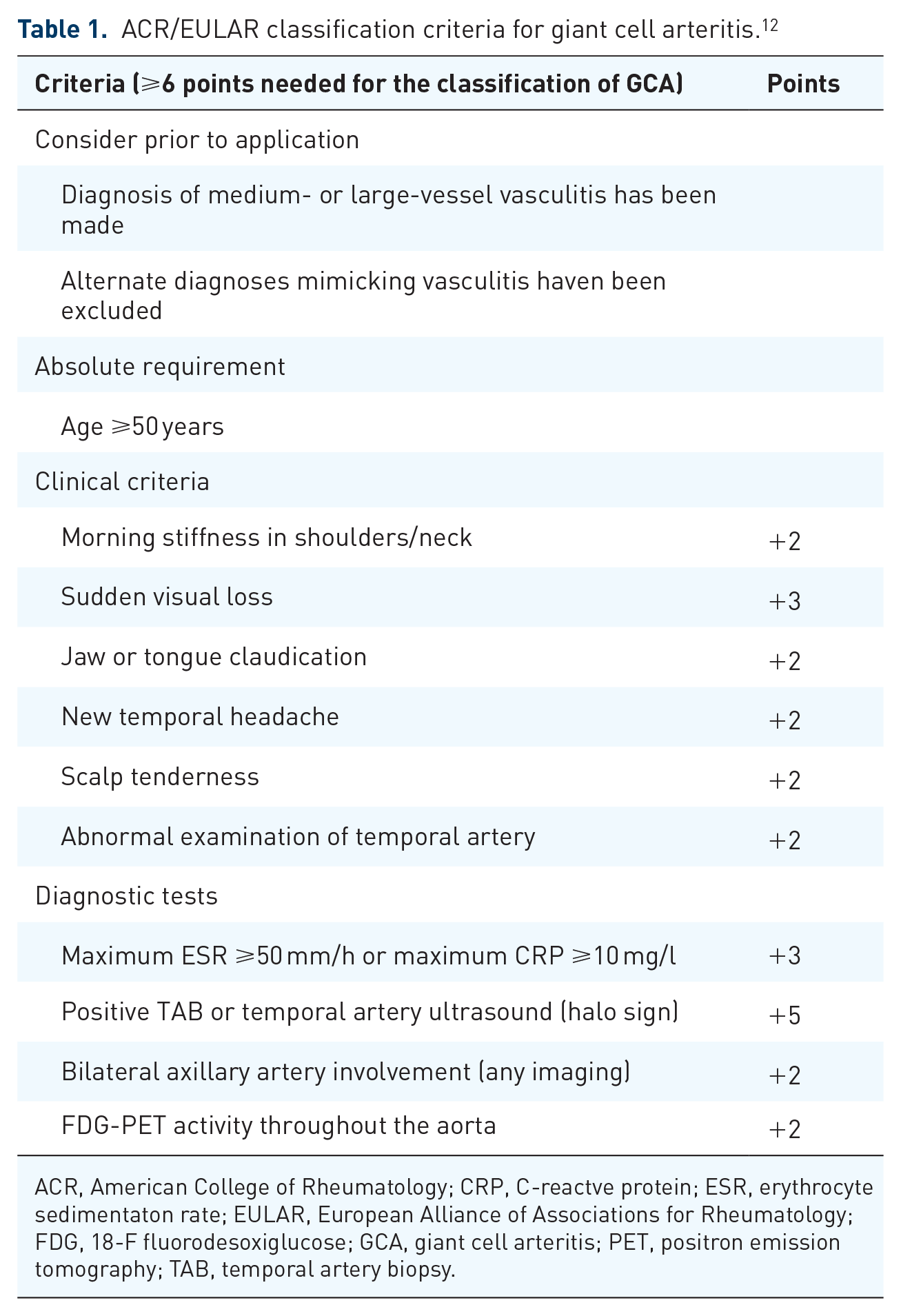
ACR, American College of Rheumatology; CRP, C-reactve protein; ESR, erythrocyte sedimentaton rate; EULAR, European Alliance of Associations for Rheumatology; FDG, 18-F fluorodesoxiglucose; GCA, giant cell arteritis; PET, positron emission tomography; TAB, temporal artery biopsy.
For
ACR/EULAR classification criteria for Takayasu arteritis. 13

ACR, American College of Rheumatology; CT, computed tomography; EULAR, European Alliance of Associations for Rheumatology; MRI, magnetic resonance imaging; PET, positron emission tomography; TAK, Takayasu arteritis.
Diagnosing GCA and TAK relies on
Technical requirements and settings for temporal artery ultrasound
Table 3 lists the technical requirements and settings for temporal artery ultrasound. Facial arteries require similar settings. For the occipital arteries, image depth needs to be slightly increased, Doppler frequency should be low and pulse repetition frequency (PRF) should be decreased to 1–2 kHz for achieving sufficient Doppler sensitivity. 15
Technical requirements and settings for temporal artery ultrasound. 15

PRF, pulse repetition frequency.
Small foot print or hockey stick probes with frequencies exceeding 20 MHz are increasingly prevalent in rheumatology practice, offering excellent resolution, particularly for visualizing temporal arteries. These probes allow measuring intima-media thickening (IMT) even in normal temporal arteries.
Probes with frequencies ranging from 50 to 70 MHz are also available, providing an axial resolution of about 0.04 mm. This high resolution allows for the distinction of thickened intima, media and adventitia in GCA, exhibiting excellent correlation with histological measurements. 40 Occasionally, such pathology can also be displayed using 20–30 MHz footprint probes in temporal and axillary arteries.28,41 However, very high frequency probes are expensive and cannot be used for musculoskeletal ultrasound because of low penetration.
Ultrasound findings
A normal artery exhibits a distinctive anechoic (black) lumen representing fluid (blood). Ultrasound renders anatomical interfaces as hyperechoic (bright) lines. The interface between the arterial lumen and the intima appears as a thin hyperechoic line. Another hyperechoic line represents the interface between media and adventitia. The region between these lines is hypoechoic (dark) representing the intima media complex (IMC; Figure 1). Ultrasound cannot discern the thin intima in normal arteries. The interface between adventitia and the surrounding tissue is usually invisible. The normal IMC is defined as a ‘homogenous, hypoechoic, or anechoic echostructure delineated by two parallel hyperechoic margins (“double line pattern”), which is surrounded by mid-echoic to hyperechoic tissue’. 42
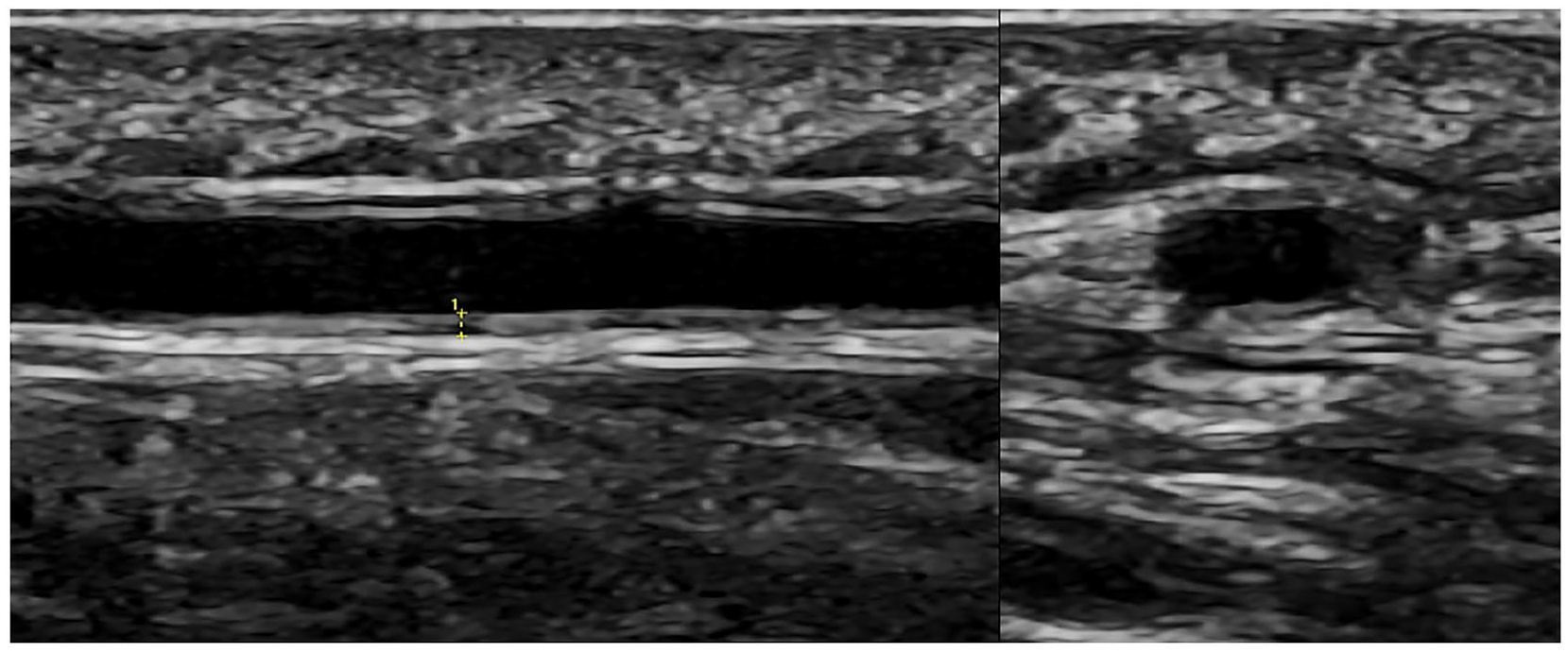
Normal temporal artery frontal branch in longitudinal (left) and transverse planes examined with a 6–24 MHz hockey stick probe. The callipers in the longitudinal view indicate a normal IMT of 0.28 mm.
In LVV, the artery wall is thickened due to infiltration with lymphocytes and macrophages leading to myointimal hyperplasia. In acute disease, artery wall oedema contributes to the hypoechoic appearance on ultrasound. Ultrasound shows hypoechoic, non-compressible material causing a thickening of the IMC. It is uncompressible while the artery lumen can be compressed. Colour Doppler facilitates the detection of smaller arteries like the temporal arteries, with colour coding correlating with blood flow. Transverse scans show a dark ring around the artery lumen, termed the ‘
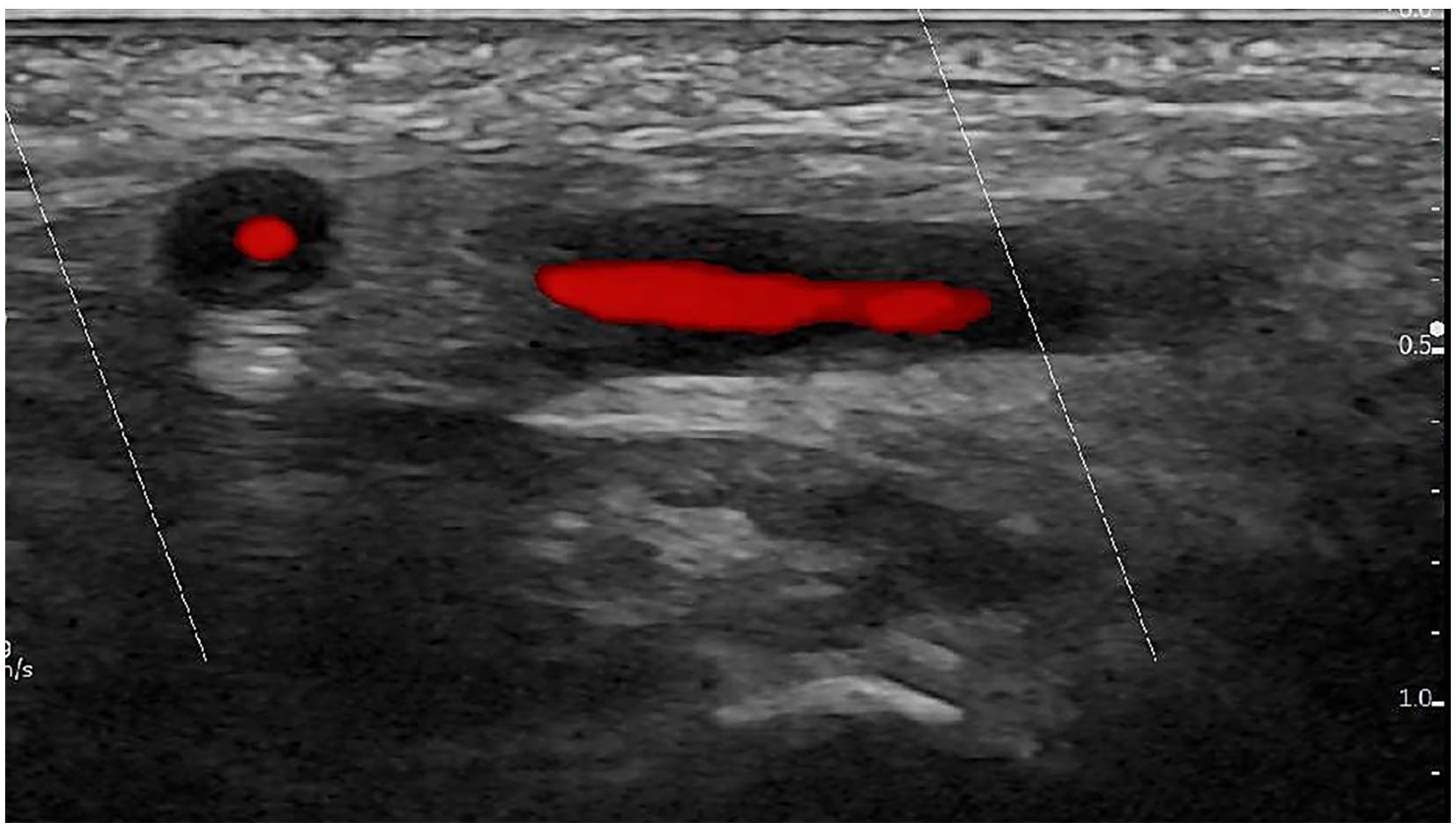
Halo sign in the parietal and frontal branch of a temporal artery near the bifurcation in GCA, using the same probe as in Figure 1.
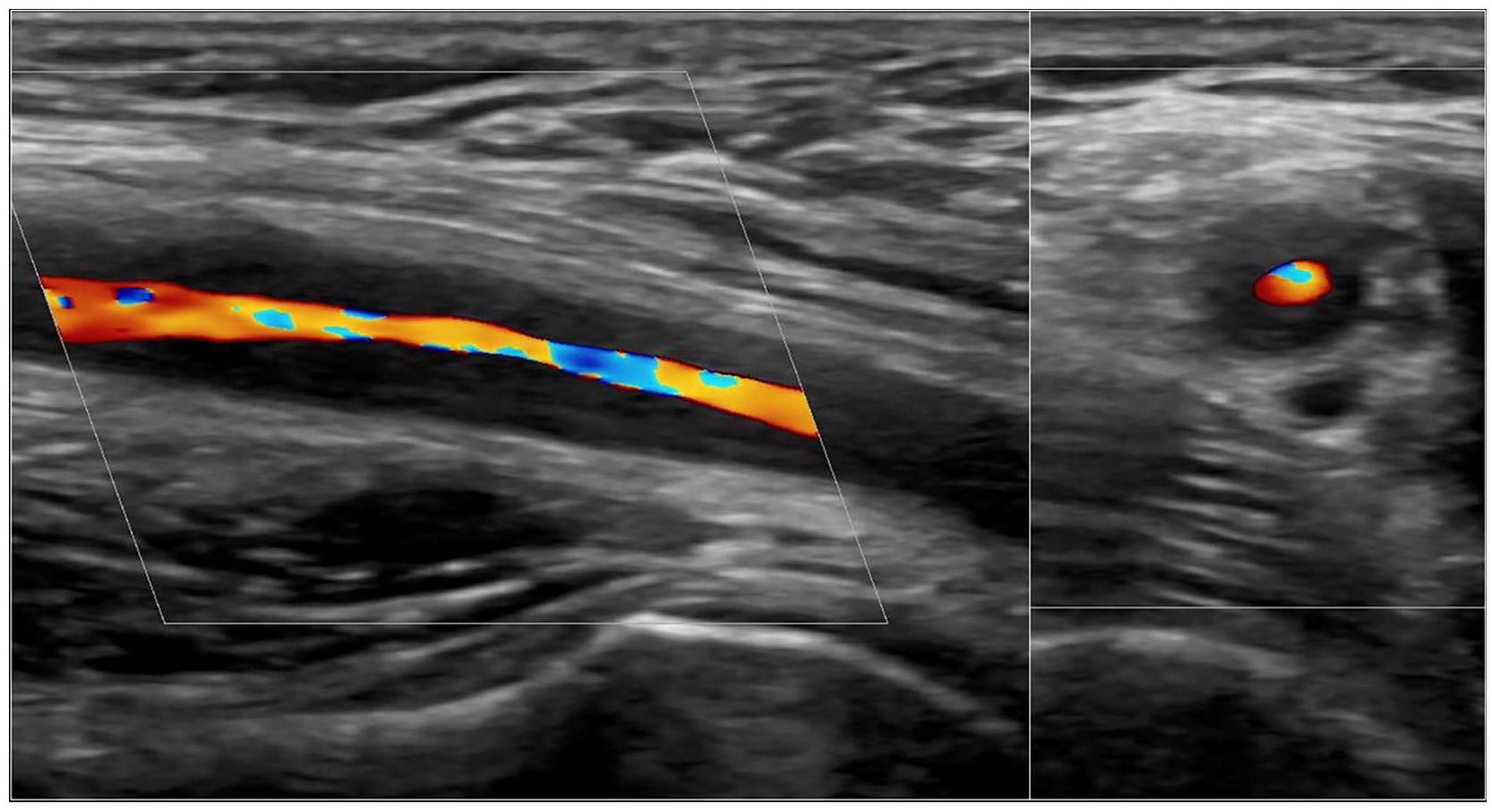
Halo sign in the axillary artery in extracranial GCA in longitudinal (left) and transverse planes (right) with a 2–9 MHz linear probe. Mixed colour signals suggest turbulent flow (aliasing) with increased velocities due to stenosis. The hyperechoic line at the bottom represents the humeral head and neck, with the right side being proximal in the longitudinal image.
Vasculitic wall thickening may lead to

Pulsed-wave Doppler curves in the same artery as in Figure 4, typical for stenosis. Peak systolic flow velocity is elevated (217 cm/s), the spectrum is broader, and the systolic flow velocity increases more slowly while remaining high for a longer duration. The colour signal exhibits no aliasing because the PRF is adjusted for high velocity (9.1 kHz).
How extensively should ultrasound be performed?
In suspected GCA, at least the common superficial
We examine the axillary arteries from the axillary side if the patient can abduct the arm. This scan accesses the middle and the distal segments. The axillary artery is also visible from anterior at the cranio-lateral aspects of the subscapularis fossa. As the axillary artery lies deeper when examined anteriorly, a lower ultrasound frequency is necessary leading to lower resolution. However, the anterior scan provides access also to the proximal segments of the axillary artery, but it does not increase sensitivities for GCA or extracranial in GCA in established GCA. 49
Protocols may additionally include the extracranial
Common carotid arteries are easily accessible with ultrasound. In TAK, exclusive vasculitis of the carotid arteries is considered specific. However, in suspected GCA patients, arteriosclerosis in carotid arteries is common, potentially leading to confusion with vasculitis, particularly when soft plaques are present. Exclusive GCA involvement of carotid arteries is uncommon, and vasculitis should be detected in additional arteries. Plaques are particularly common at the carotid bifurcation. The proximal internal and external carotid arteries are rarely involved in GCA and TAK, but in the age group of GCA patients, arteriosclerotic stenoses are common. 28 Stenoses in common carotid arteries occur rarely in GCA but more frequently in TAK.
Other arteries will be listed in the ‘standard scans’ section.
Does limiting the ultrasound examination to temporal and axillary arteries overlook patients with GCA or GCA patients with extracranial involvement? A study with 72 consecutive, newly diagnosed GCA patients found that while PET-CT detected aortitis in 33%, ultrasound of temporal and axillary arteries only missed two patients, one with isolated aortitis, actually not classified as GCA, 1 and one with aortitis and vasculitis of the iliac arteries. 51
Standard scans
Prior to the ultrasound examination,
For examining the
The

Panoramic ultrasound image displaying normal axillary artery anatomy seen from the axillary recess. The undulating walls reflect pulsations while generating the panoramic image.
Examination of
Examination of other arteries is optional. The patient should be supine except for the popliteal arteries which are examined in the prone position.
The
The
The
The first 6–8 cm of the
The
The pedal pulses should be palpated in newly diagnosed LVV. If absent, the
Indications, clinical correlation and probes for arteries involved in LVV.

GCA, giant cell arteritis; LVV, large-vessel vasculitis; TAK, Takayasu arteritis.
Other arteries are accessible with ultrasound. Abdominal ultrasound can access
Advantages and disadvantages of ultrasound and other imaging techniques
Many studies report a high accuracy of ultrasound. A meta-analysis, which informed the 2023 EULAR recommendations on imaging in LVV, demonstrated also high specificity for MRI and PET, with ultrasound exhibiting greater sensitivity (Table 5). 43 The sensitivity of PET may further increase with newer technology concerning access to cranial arteries.
Pooled sensitivities of low-risk of bias studies on ultrasound, MRI and PET-CT for the diagnosis of GCA. 43

GCA, giant cell arteritis; MRI, magnetic resonance imaging; PET-CT, positron emission tomography-computed tomography.
Some studies have directly compared ultrasound with other imaging techniques in GCA. One study observed a very good agreement between ultrasound and PET-CT. 45 Another study described comparable diagnostic accuracy for GCA. However, PET/CT and ultrasound were often discrepant within single vascular regions. 57 High agreement of ultrasound and MRI was observed in another study. 59
In TAK, fewer studies have been conducted. A meta-analysis of mostly small retrospective studies, comparing imaging with clinical criteria or conventional angiography, yielded pooled sensitivities of 81%, 81% and 92% for ultrasound, PET and MRI, respectively. Pooled specificities were >90% for ultrasound and MRI and 74% for PET. 60
Despite ultrasound being recommended as the preferred diagnostic test for GCA, alternative diagnostic techniques can be applied in the absence of expertise or adequate equipment. Table 6 summarizes the advantages and disadvantages of diagnostic techniques. 61
Advantages and disadvantages of imaging techniques for LVV.

CT, computed tomography; GCA, giant cell arteritis; LVV, large-vessel vasculitis; MRI, magnetic resonance imaging; PET, positron emission tomography; PMR, polymyalgia rheumatic.
Ultrasound is often perceived as highly investigator dependent. However, all diagnostic modalities require training, standard operating procedures and adequate equipment. 15 Exercises with ultrasound have demonstrated good reliability in suspected GCA with trained examiners.42,62 Currently, ultrasound machines can be connected to Picture Archiving and Communication Systems. Findings should be archived in two planes. For temporal arteritis at least one video showing an incompressible halo sign together with a longitudinal still image should be stored.
Transcranial ultrasound can visualize intracerebral arteries, but its resolution is too low for detecting a halo sign. High-resolution MRI could identify at least one vasculitic intracranial artery in 15% of GCA patients, a prevalence higher than previously suspected. 63
How rapidly should the diagnosis be confirmed?
How rapidly does pathology normalize with glucocorticoid treatment? This depends on the diagnostic method, equipment quality and artery size (Table 7). PET normalizes most rapidly. In a study with repeated PET scans 100% remained positive after 3 days, but only 36% remained positive after 10 days. 64 PET should preferably be performed within the first 3 days of glucocorticoid treatment.
Estimation of time intervals for pathology normalization for PET-CT, ultrasound, MRI and histology.

MRI, magnetic resonance imaging; PET-CT, positron emission tomography-computed tomography.
Temporal arteries normalize more rapidly than extracranial arteries. In the first study on ultrasound in GCA, the halo sign persisted for a mean of 16 days. 10 With newer equipment, pathology may be remain visible for a longer duration. Other studies comparing the length of glucocorticoid treatment in cohorts with suspected GCA noted a decrease in sensitivity already after 2 days.68,69 In this regard, MRI and ultrasound of temporal arteries exhibit similar trends. 68 Consequently, ultrasound or MRI of temporal arteries should be performed within the initial 7 days of glucocorticoid treatment.
Histological pathology of TAB persists for a longer time. In a study with sequential TAB, 75% remained positive after 6 months, and 44% were positive after 9 and 12 months, respectively. Notably, none of the positive specimens after 12 months exhibited giant cells any more.
Axillary artery wall thickening persists even longer, with pathology still visible in 70% of patients after a mean of 50 months. 67 In another cohort, IMT of axillary arteries exceeded the cut-off for chronic vasculitic axillary artery changes of 0.9 mm in half of the patients after 4 years.70,71
Nevertheless, diagnosis becomes more challenging with increasing length of glucocorticoid treatment, leading to a decrease of diagnostic certainty. Slight wall changes of vessel wall morphology observed through ultrasound, MRI or CT, a slight increase in glucose metabolism in PET, or the absence of giant cells in histology bear a risk of misinterpretation. Therefore, confirming the suspected diagnosis of LVV, particularly of GCA, is crucial as soon as possible. However, diagnostic procedures should not delay the initiation of treatment. 15
Pitfalls
Several pitfalls may be encountered during ultrasound examinations:
Poor quality of colour Doppler signals when sound waves are perpendicular to the artery. Therefore, the colour box should be angled in longitudinal scans, and the probe should be slightly tilted in transverse scans [Figure 6(a)].
Pseudo-halo: Colour Doppler signals appearing only in the middle of the artery lumen may leave an anechoic area around this signal, creating a false impression of vasculitis [Figure 6(b)]. This issue can be resolved by compressing the temporal artery, increasing the Doppler gain, and focusing on the B-mode image showing hypoechoic, but not anechoic wall swelling.
Colour signals extend over the artery walls because the colour gain is too high, leading to overlooking the halo-sign [Figure 6(c)].
No colour signal and poor visibility of the arteries below hair. This challenge can be overcome by using more ultrasound gel or focusing on compression sonography in B-mode [Figure 6(d)].
Arteriosclerosis: Arteriosclerotic vessel wall changes may also cause wall thickening of the temporal and other arteries. Unlike vasculitis, arteriosclerotic plaques are hyperechoic, irregular and asymmetric. Arteriosclerosis is rare in temporal arteries [Figure 6(e)]. However, it can be misinterpreted, particularly in the carotid arteries and at the root of the right subclavian artery [Figure 6(e)].
Skip lesions: Vasculitis may be limited to a few segments, leading to potential oversight if arteries are not thoroughly scanned [Figure 6(f)].
Normal findings due to previous glucocorticoid treatment. This is particularly relevant to the temporal, facial and occipital arteries. Ultrasound examination can be extended to extracranial arteries where pathology may persist for a longer duration.
True halo sign due to a diagnosis other than GCA or TAK. This aspect is further explained in the next paragraph.
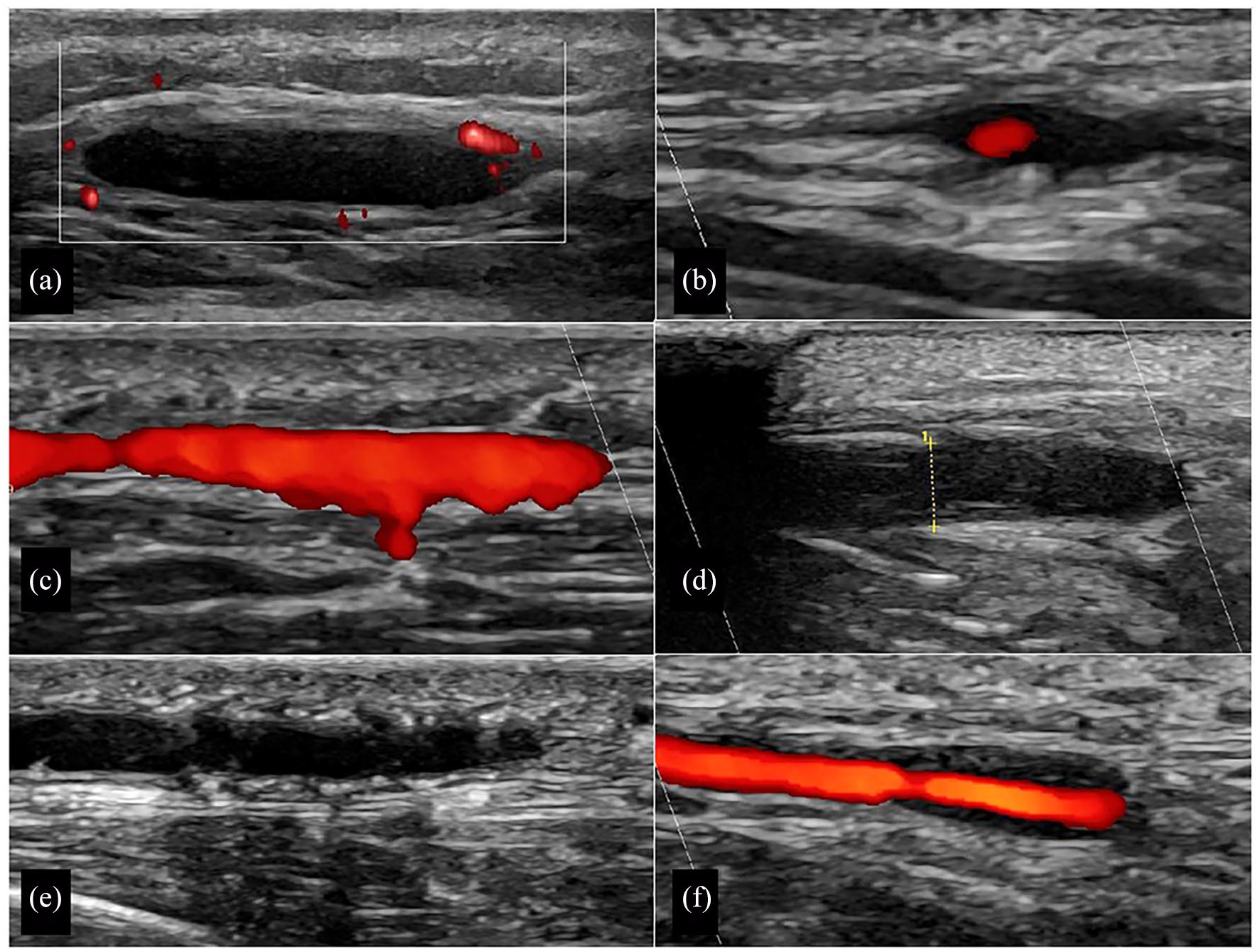
Pitfalls: (a) The colour box is not angled resulting in poor colour Doppler signals. (b) Pseudo-halo because colour gain is too low. (c) Colour gain is too high, while colour signals extend over the artery wall. (d) Hair impairs sound waves on the left side of the image, but vasculitic artery is not compressible on its right side. The callipers indicate a thickness of 2.2 mm for the compressed artery, meaning that each IMT is 1.1 mm. (e) Arteriosclerosis of a temporal artery, displayed as hyperechoic (white) irregular spots in the IMT of both sides. (f) Focal vasculitis on right side with normal temporal artery segment on left side.
Differential diagnosis
An experienced sonologist using modern technology and adhering to standard operating procedures can hardly overlook or misdiagnose vasculitis. However, other conditions causing arterial wall thickening may
Aortitis and periaortitis appear differently. Aortitis typically correlates with the characteristic halo sign. In case of arteriosclerosis, hypoechoic material extends around the hyperechoic rim of plaques. 28 Chronic periaortitis due to Ormond’s disease or IgG4 syndrome is less distinct, appearing hyperechoic with often poorly defined boundaries. 77 Especially in younger TAK patients, infectious disease such as lues or mycobacterial infection should be considered. Furthermore, TAK may co-occur with axial spondyloarthritis, Cogan syndrome, relapsing polychondritis and other autoimmune diseases. 78
Ultrasound fast-track clinics
Rheumatologists increasingly offer fast-track clinics for suspected GCA, providing a service within 24 h on working days for patients in whom a physician suspects GCA. The fastest route is to contact the nurse of the fast-track clinic by telephone. Initially, an experienced rheumatologist conducts a brief, structured history and a focused clinical examination, followed by an ultrasound examination of at least the temporal and axillary arteries. Ideally, two to three experts should be trained within a centre.
The implementation of fast-track clinics correlated with a substantial
In Europe, mostly the
A GCA
For patients with a high clinical suspicion of GCA and positive ultrasound findings, an immediate diagnosis of GCA can be established. Likewise, low clinical probability combined with negative ultrasound excludes GCA. TAB or further imaging is warranted if findings are inconclusive, a situation encountered in less than 5% in our fast-track clinics.
Vascular ultrasound in PMR
PMR and GCA are closely related conditions characterized by systemic inflammation, a predominant interleukin-6 signature, an excellent response to glucocorticoids, a tendency to a chronic and relapsing course and older age of the affected population. 86 Despite these commonalities, differences exist in pathogenesis. For instance, inflamed arteries in GCA exhibit extensive infiltrates of T helper-1 and -17 cells, whereas T-cell infiltrates in PMR synovium are limited, predominantly consisting of T helper-1 cells. 87 However, a significant clinical connection between PMR and GCA exists. This relationship has implications for disease stratification, 88 leading to introduction of the term ‘GCA-PMR Spectrum Disease’. 86 New treat-to-target Recommendations in GCA and PMR emphasize that clinical management of GCA and PMR should be driven by the awareness that they are closely interrelated conditions in a common spectrum of inflammatory diseases and can occur separately, simultaneously, or in temporal sequence to each other. 89
In a cohort of over 1000 newly diagnosed GCA patients in Berlin-Buch and in other cohorts, approximately 50% exhibited symptoms of PMR characterized by shoulder or pelvic girdle pain and morning stiffness.86,87 ‘Pure PMR’ is characterized by PMR symptoms without clinical features of GCA, and PMR may precede, coincide with or follow the clinical manifestations of GCA. 87
How many PMR patients have subclinical GCA? A meta-analysis of 13 cohorts, utilizing various diagnostic tests such as histology, ultrasound and PET in PMR, found that 22% of PMR patients with and without GCA symptoms were ultimately diagnosed with GCA. 90 In a monocentre study involving ultrasound examinations of temporal, facial, axillary and carotid arteries, 22% of 60 patients with newly diagnosed ‘pure’ PMR had subclinical GCA. 91 In a multicentre study, examining temporal, axillary, common carotid and subclavian arteries with ultrasound in 346 patients with newly diagnosed PMR, and subclinical GCA was found in 23%. PMR patients with subclinical GCA more frequently exhibited an extracranial large vessel pattern compared to classical GCA. 92 Follow-up assessments of 100 patients with non-vasculitic PMR and 50 patients with subclinical GCA revealed that the latter experienced more relapses (62% versus 16%) and required higher doses of prednisone equivalent after 12 months (mean dose, 7.5 versus 2.4 mg/day). 93 In conclusion, PMR patients with subclinical GCA necessitate treatment as GCA, highlighting the importance of searching for GCA in PMR for effective disease management.
Should all patients with suspected new PMR be referred to a rheumatologist? An international survey involving 394 general practitioners (GPs) and 937 rheumatologists concluded that GPs only referred 25% of their patients to rheumatologists. 94 A systematic literature review found only sparse evidence on early management and referral strategies. Four studies on shared care and fast-track clinics revealed promising results including reduced hospitalization rates, lower starting glucocorticoid doses and faster PMR diagnosis. 95 Based on these outcomes, an international GCA/PMR study group recommends early referral of individuals with suspected PMR to specialist care. Patients should also be informed about the potential overlap with GCA. 96
Ultrasound for disease monitoring
Ultrasound can be applied for monitoring disease activity or damage in both GCA and in TAK. Disease activity may correlate with IMT. Damage refers to stenoses, occlusions and particularly to aortic aneurysms.
For monitoring IMT, normal and cut-off IMT values have been established for GCA. However, such values have not yet been determined for TAK. Younger individuals reveal lower IMT. Several studies reveal consistent results for suspected GCA (Table 8).97–101
Normal and cut-off values in the age of patients with suspected GCA. 28
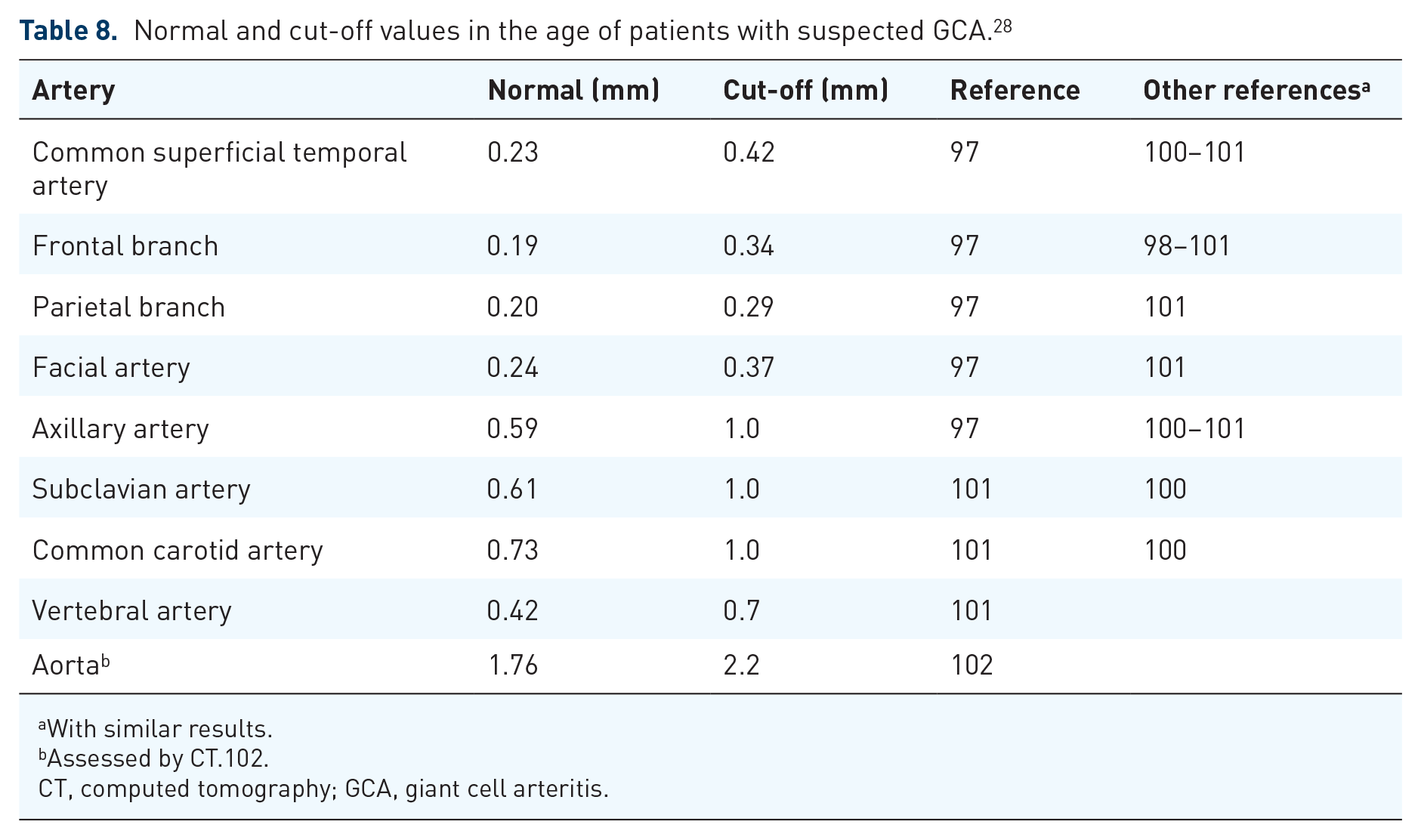
With similar results.
Assessed by CT.102.
CT, computed tomography; GCA, giant cell arteritis.
For diagnosing GCA or TAK, the presence or absence of the halo sign, which is uncompressible in cranial arteries, is crucial. IMT scores were not developed for diagnosis, but for disease monitoring, although cut-off values may be sometimes helpful.
A comparable score based on IMT of the same segments, known as the Outcome Measures in Rheumatology (OMERACT) Giant Cell Arteritis Ultrasonography Score (
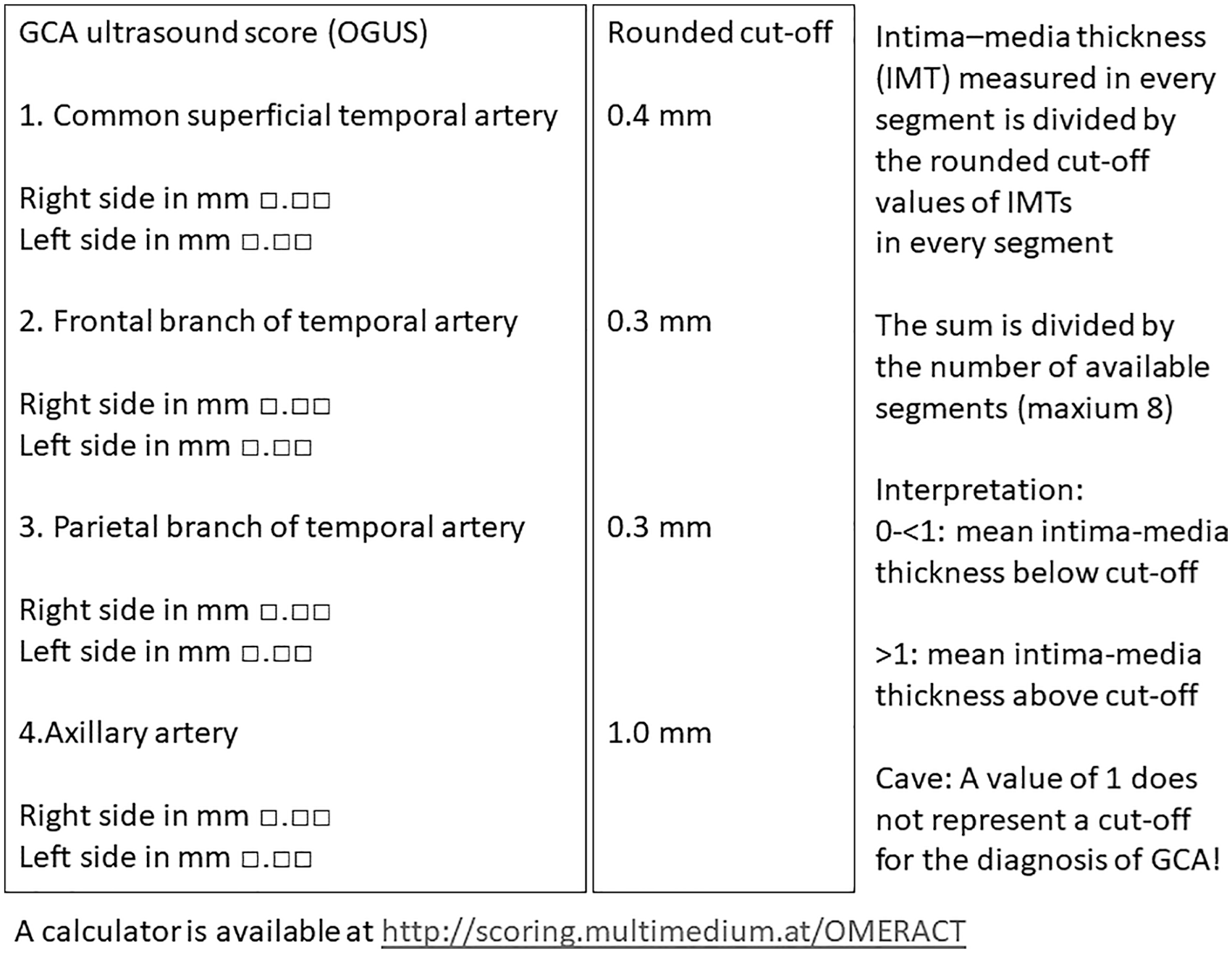
Calculation of the OGUS.
Sensitivity to change and convergent construct validity of the OGUS were tested in a prospective cohort of new GCA patients undergoing ultrasound-based IMT measurements at baseline, weeks 1, 3, 6, 12 and 24. This study found a continuous and significant reduction of the halo size of temporal arteries, whereas the reduction in the axillary arteries was less pronounced and insignificant. The reduction in the temporal artery halo size correlated with ESR, CRP, Birmingham Vasculitis Score and cumulative glucocorticoid dose. 65
Alternative to the OGUS, the number of vasculitic segments can be counted (
In an ultrasound substudy of the GUSTO trial from Switzerland, a score based on normal IMT was applied, easily convertible to the OGUS. IMT in both temporal and axillary arteries closely correlated with disease activity in a cohort of GCA patients treated with glucocorticoid pulse therapy for only 3 days followed by tocilizumab. 108
Another independent cohort from Denmark, comprising 47 newly diagnosed GCA patients tested several scores. Similar to the other cohorts, the OGUS exhibited a large magnitude of change. It was considered the score least prone to potential bias. All scores demonstrated the potential to discriminate between remission and relapse.109,110
Another study revealed a decrease of abnormal temporal and extracranial artery IMT in 85% and 45%, respectively, within 24 months. 111
The IMT decreased more rapidly in the temporal arteries, particularly in the first 24 weeks. A study investigating GCA patients for up to 12 years identified also a substantial decrease in IMT in the axillary arteries. 70 Relapses correlated with a significant increase of axillary artery IMT. 31
Confirming new drug’s positive on structural vascular pathology, not just on inflammation, is crucial. The most significant score reductions occur in the first few weeks of treatment. Therefore, trials should retrospectively include IMT measurements of the initial diagnostic ultrasound scan if available. 110
Although primarily developed for trials, the OGUS may be useful in clinical practice. The benefit of regular IMT monitoring for all LVV patients is yet unclear. However, monitoring appears relevant in uncertain clinical situations, such as tocilizumab treatment, unclear symptoms and normal CRP. The interpretation of ultrasound findings both at diagnosis and follow-up should always consider clinical and laboratory features.
Only few prospective studies exist on
Ultrasound can detect the development of structural damage. New arterial stenoses develop particularly in TAK and, to some extent, in GCA, due to active disease.
A study using PET-CT at baseline and yearly CT scans found a tenfold increase in thoracic aorta aneurysms in GCA patients with aortitis compared to those without. 119 Nevertheless, it remains unclear how many patients need to be regularly screened with which imaging technique in which intervals for preventing severe complications or death caused by aortic dissection or rupture. Therefore, the EULAR recommendations on imaging in LVV do not yet advocate routinely screening all patients for aneurysm development, but decide on an individual basis. 15
In Berlin-Buch, we annually determine the diameter of the ascending aorta with ultrasound in patients with extracranial GCA.28,41 This examination takes about 10 s when performed by an experienced sonologist with a sector probe available for echocardiography. The first 6–8 cm of the ascending aorta are well visible with ultrasound. The cut-off value for dilatation depends on body size and patient age. As a rule of thumb, MRI or CT should be initiated if the diameter of the ascending aorta increases above 38 mm to provide information on the whole thoracic aorta. Further studies are needed to evaluate this very simple concept.
Ultrasound for prediction of outcome
Predicting the outcome of GCA and TAK remains challenging. Ultrasound findings poorly correlate with disease outcome,65,111,120 and treatment recommendations for cranial and extracranial GCA have not yet differentiated, despite slightly longer and higher-dose glucocorticoid treatment in extracranial GCA patients. 16
Conclusion
Ultrasound is recommended as the first diagnostic tool in GCA and proves valuable in TAK. Ultrasound in suspected GCA should at least include the temporal and axillary arteries. Nearly all other arteries are accessible with ultrasound except the descending thoracic aorta. Fast-track clinics led to a reduction of vision loss in GCA. If clinical and ultrasound findings align, no further confirmatory tests are needed. Ultrasound detects subclinical GCA in over 20% of PMR patients. These ‘silent’ GCA patients need to be treated as GCA. Ultrasound is also useful for follow-up with IMT measurements. Scores have been developed primarily for research studies.
