Abstract
High Energy Musculoskeletal Traumas (HEMTs) represent a relevant problem for healthcare systems, considering the high social costs, and both the high morbidity and mortality. The poor outcomes associated with HEMT are related to the high incidence of complications, including bone infection, fracture malunion and non-union. The treatment of each of these complications could be extremely difficult. Limb reconstruction often needs multiple procedures, rising some questions on the opportunity in perseverate to try to save the affected limb. In fact, theoretically, amputation may guarantee better function and lower complications. However, amputation is not free of complication, and a high long-term social cost has been reported. A comprehensive literature review was performed to suggest possible ways to optimize the limb preservation surgeries of HEMT’s complications in order to ameliorate their management.
Keywords
Introduction
High Energy Musculoskeletal Traumas (HEMTs) are a prominent problem for healthcare systems, considering both the social cost and the high morbidity and mortality. 1 Several factors contribute to generate the considerable healthcare cost associated with HEMT, that was estimated to be between 1% and 3% of the gross domestic product in the United States of America.1,2 Indeed, only 58% of patients with a HEMT are able to return to their work activities in the first year after the injury. Moreover, disability, patient death and some other direct medical costs also act in increasing the reported economic burden. 2
In many cases, HEMTs severely affect also the vascular, nervous, and other soft tissues. Therefore, a multidisciplinary evaluation of HEMTs is often necessary to plan the limb reconstruction, generally involving the orthopedic, the plastic and the vascular surgeons.2–4 The occurrence of a severe vascular injury may be a dreadful event, often representing an emergency. In fact, in these cases the revascularization must be performed in the first 6 h considering the relevant risk of necrosis after this time point. An appropriate evaluation of the vascular injury should be constantly performed by looking for high suspicious signs (such as pallor, decreased temperature, reduced pulses, delayed capillary refill), also after gross realignment of the skeletal segment. A strict collaboration between the trauma leader and vascular surgeon is essential in case of HEMTs to organize the most convenient sequence of surgical procedures to minimize the risk of long ischemia.2,3 Another actor in this multidisciplinary framework is the plastic surgeon, preferably with experience in microsurgery. In fact, the use of complex reconstructive techniques, such as muscle flaps, free flaps, vascularized bone graft, may considerably improve the outcomes, thanks to the notable effects of filling bone and/or skin defects using vascularized tissues. Furthermore, some techniques are able to provide for the association of muscle-cutaneous flaps containing nervous structures to be anastomosed with peripheral injured nerves.5–7
Anyway, the high morbidity and poor outcomes associated with HEMT are mostly related to the high incidence of complications, including wound infection (28.3%) and fracture non-union (23.7%). 8 Treatment decision-making of HEMT-related complications may be extremely difficult. Limb preservation options often need multiple surgical procedures, rising some questions on the opportunity in perseverate to try to save the affected limb. In fact, amputation has proven to be a valid alterative that guarantees good function and reduced complications. 9 Moreover, some studies have shown that quality of life is not directly dependent on limb salvage. 9 Several parameters have been described to help in the decision to save or amputate a severely injured limb.10–12 The main criteria to be considered may be age, general condition, time of ischemia, extent of soft tissue damage and bilateral amputations. These criteria are part of some scores that may further aid in the treatment decision-making, reducing the rate of complications following a limb salvage attempt. Particularly, Battiston et al. 10 proposed a modified mangled extremity severity score (MESS) in which a score <5 clearly indicate a reimplantation, while a score >8 an amputation (see Table 1). 10 Moreover, the authors described a high rate of secondary complications requiring a subsequent amputation, in a case series of 12 patients with a reimplanted limb. 10 Chen and Chen described a grade system, according to which it is possible to foresee the functional outcomes of a reimplanted limb. 11
MESS score modified by Battiston et al. 10
If the score is >8, contraindication for replantation; 6–7, possible replantation, poor functional result; ⩽5, indication for replantation.
MESS, mangled extremity severity score.
However, amputee long-term management is not cost and risk free.13,14 Recently, in the United Kingdom, a total 40-year £288 million cost (equal to USD 444 million) has been estimated among the UK Afghanistan lower limb amputee cohort. 13 Moreover, a loss of function related to a gait imbalance, was demonstrated even among patients with transtibial amputation (one of the most efficient level of amputation). 14
Therefore, to ameliorate patients’ outcomes, the limb salvage procedures of HEMT complications should be carried out carefully and patients appropriately selected.
Considering the multiple unmet needs related to the surgical procedures available to treat HEMT-related complications, we decided to perform a comprehensive evaluation of the available literature to guide the orthopedic surgeon throughout the treatment decision-making.
Fracture Non-Union
The failure of the bone healing process occurs in up to 10% of patients with a fracture, 15 but HEMTs consistently raises up the probability of observing this complication. The incidence of non-unions depends also on the bone involved. In fact, carpal scaphoid fractures are associated with a risk of non-union of 15.5%, whereas tibial and femoral ones of 14.4% and 13.9% respectively. 15
Multiple definitions of fracture non-union were reported in the current literature. Indeed, the Food and Drug Administration defined a non-union as a fracture that was not healed at 9 months after the injury. 16 Some other authors, instead, described it as a fracture that was not healed in the expected time and/or with no healing progression on plain radiographs, or a fracture that needs additional procedures to achieve union. 17
Both patient- and surgical-related factors could negatively affect the bone healing and they must all be considered before starting to treat a fracture non-union. 18 The evaluation of the previous treatment, the morphology of the original fracture, the appropriateness of the previous fracture fixation and the quality of reduction are all surgical-related factors that have to be taken into account. Moreover, a global health status evaluation of the patient-related factors, including comorbidities, life habits, as well as bone metabolism and bone quality should be performed. Obviously, bone and soft tissue infections must be excluded, considering that they lead to a different diagnosis and dramatically change the management.
Classification of Non-Union
The classification of non-union is not univocal. The most popular was described by Weber and Cech, 19 that distinguished three types of non-unions, basically biologically active (hypertrophic) and inactive (oligotrophic and atrophic) (see Table 2). However, available evidence did not completely support this classification. In fact, some histological studies did not report relevant differences between hypertrophic and atrophic non-unions. 20 Moreover, this kind of classification did not consider patients’ comorbidities.
Most widely used classifications of fracture non-unions. In both of them, reading from the left to the right, a greater need for biological supplementation is required.

NUSS, Non Union Scoring System.
In bold there are reported the categories of each classification.
For this reasons, other authors proposed more complex classification systems.19,22 One of the most used is the Non Union Scoring System. 21 This classification included patient-related factors, soft tissue status, Weber and Cech classification, previous treatment, bone quality and fracture-related factors (see Table 2 for further details).
Non-Surgical Treatments
If a biologically active non-union is suspected, some non-surgical treatments may be attempted.
Recently, a relevant role has been suggested for bone anabolic drugs for the treatment of delayed consolidations and non-unions. Specifically, teriparatide, a parathyroid hormone (PTH) derivate, has been suggested to be able to improve bone healing, 23 although the scientific evidence is still poor and mainly based on case reports and small clinical series.24–29 Particularly, Gariffo et al. 27 in a recent case series of 20 delayed/non-unions reported a union rate of 85%, most of which occurred after 3 months of teriparatide use.
Abaloparatide, a synthetic analog of a PTH-related peptide with a weaker binding to the R° conformation of the PTH receptor compared to teriparatide, has been recently proposed as a more effective anabolic drug in osteoporotic patients. 30 Recent preclinical studies supported its role also on fracture healing. 31
Physical agent modalities too may act as non-surgical means to improve bone healing. In fact, a recent systematic review on Pulsed Electromagnetic Fields demonstrated a low to moderate quality of evidence supporting its role in increasing bone healing rate and accelerating time to healing. 32
Low-intensity pulsed ultrasound (LIPUS) is another physical-agent modality with promising results in the treatment of non-unions. 33 The authors of a recent systematic review reported a success rate of 82% and proposed LIPUS as an alternative to surgery in fracture non-union in elderly, or in patients with severe comorbidities. 34 Moreover, some evidence supported their use also in case of hypertrophic non-unions. 34
Surgical Management of Non-Unions
The surgical management of non-unions may be extremely difficult, and often requires a comprehensive approach focused on all those factors that could affect bone healing. This kind of approach has been recently summarized in the ‘diamond concept’ conceived by Andrzejowski and Giannoudis 35 According to this concept, a successful fracture healing depends on both the biological and the mechanical environment observable at the fracture site as well as the vascularization. In fact, according to the authors, a viable biological environment is essential to provide mediators and cells, as well as osteoconductive matrix necessary to the osteoprogenitors to act, while the need of a consistent mechanical environment, underline the relevance of a proper fracture stability to aid cells in appropriately complete the callus formation. 36 Andrzejowski and Giannoudis 35 affirmed that a rigorous application of all the aspects of the diamond concept could ensure a success rate of 89–100% in the treatment of long-bone non-union.
To achieve the needed focal bone revitalization for the bone union, some biologically active materials and techniques are available. 37 These materials and techniques may be divided into two main categories: vascularized and non-vascularized. The first ones are represented by autologous vascularized graft and distraction osteogenesis, while the second typically include allogenic graft, autologous non-vascularized graft, and bone substitutes (scaffolds). 37
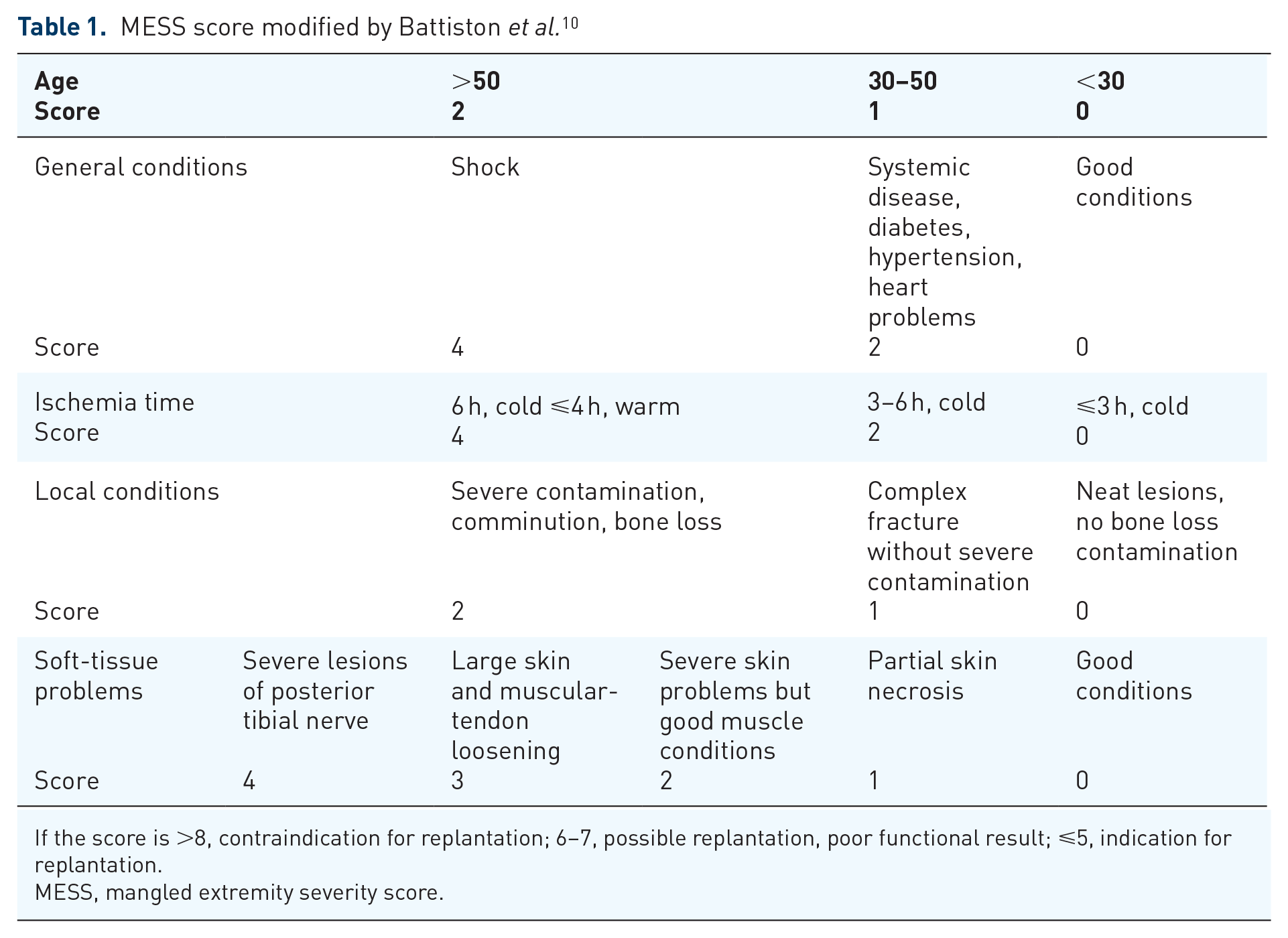
In the context of vascularized grafts, the use of the vascularized fibular graft can provide both an adequate mechanical stability and biology in case of non-unions with large segmental defects. 38 Moreover, its use is possible also for the management of infected non-unions. However, the harvesting of a vascularized bone graft may be extremely difficult and requires microvascular environment. When biological support is strongly recommended but microsurgical supplies are not available, the historical Huntington’s ipsilateral vascularized fibular transposition is still a viable option in selected patients.39–42 Figure 1 shows a clinical case of a recalcitrant middle tibia non-union treated using the Huntington’s procedure.
A clinical case of a 42 years old male with a middle shaft tibia septic non-union following an open fracture treated with intramedullary nailing (a). The patient was treated through septic bone resection and subsequent bone transport (b). In (c) non-union of the docking point, treated through a new bone resection and bone transport for definitive infection control (d). In (e) X-ray at 7 months suggestive of docking point union. In (f) recurrence of an aseptic atrophic non-union treated using the Huntington’s procedure (g). In (h) X-ray at 12 months after the procedure, note the full integration and partial hypertrophy of the fibula.
Moreover, recent studies supported the use of Bone Marrow (BM) Concentrate to treat long-bone non-unions, 43 especially when non-vascularized biological materials are used. In fact, as suggested by pre-clinical studies, the application of BM is able to enhance bone vascularization. 44 Moreover, the simultaneous use of internal fixation, cortical allograft and BM-stem cells has been recently demonstrated to be a viable option to treat humeral shaft non-union also in the elderly patient. 45
The management of non-unions with severe bone loss following HEMTs might be addressed also using an external fixation,17,20 considering its reported ability to provide stability, correct alignment and biological stimulation at the non-union site. Moreover, the massive bone loss might be replaced using the bone transport technique. Theoretically, it leaves intact the soft tissues around the fracture site, being a percutaneous device. This leads to a further preservation of the biological and vascularization environment, that might aid also for the reconstruction of any soft tissue injury. However, pin tract infections, docking-site non-union are among the complications reported for the external fixator use in fracture non-unions.46–48 To reduce their incidence an accurate surgical technique and post-operative follow-up is needed, especially for pin tract-related complications. 49 On the other hand, acute shortening or reintervention with the application of bone stimulating substances are among the proposed protocols to treat docking-site non-union.46,50
Recently, the adoption of a contemporary use of internal and external fixations in the same patient is gaining popularity, thanks also to the reported reduction in time of use of the external fixator.51–53 Particularly, Kadhim et al., 54 in their Systematic review, reported that the combined use of circular fixation and intramedullary nail provided the highest success rate for the treatment of tibial non-union with segmental bone defect.
Fracture Malunion
Malunion is an extremely common complication of HEMTs, with a reported incidence of about 22%.8,55
The treatment of malunion is often required because it may severely affect limb function or increase the risk of early osteoarthritis. Particularly, a 10° varus/valgus malunion has been observed to lead to a critical increase in medial and lateral knee cartilage stress, respectively. 56
Several techniques had been described to manage fracture malunion, substantially based on corrective osteotomy and application of both internal and external fixation.57,58 Infection, delayed healing and non-union are among the reported complications of corrective osteotomies of the lower limbs.58,59 In order to reduce bone healing complications and improving patients’ comfort the clamshell and chipping corrective osteotomies had been proposed.60–63 Clamshell osteotomy requires the identification of the malunited segment and to perform two subsequent osteotomies, one proximally and one other distally the center of the deformity. After that, the malunited segment is again osteotomized along its axis and wedged open. The three resulting fragments are then aligned with an intramedullary rod.
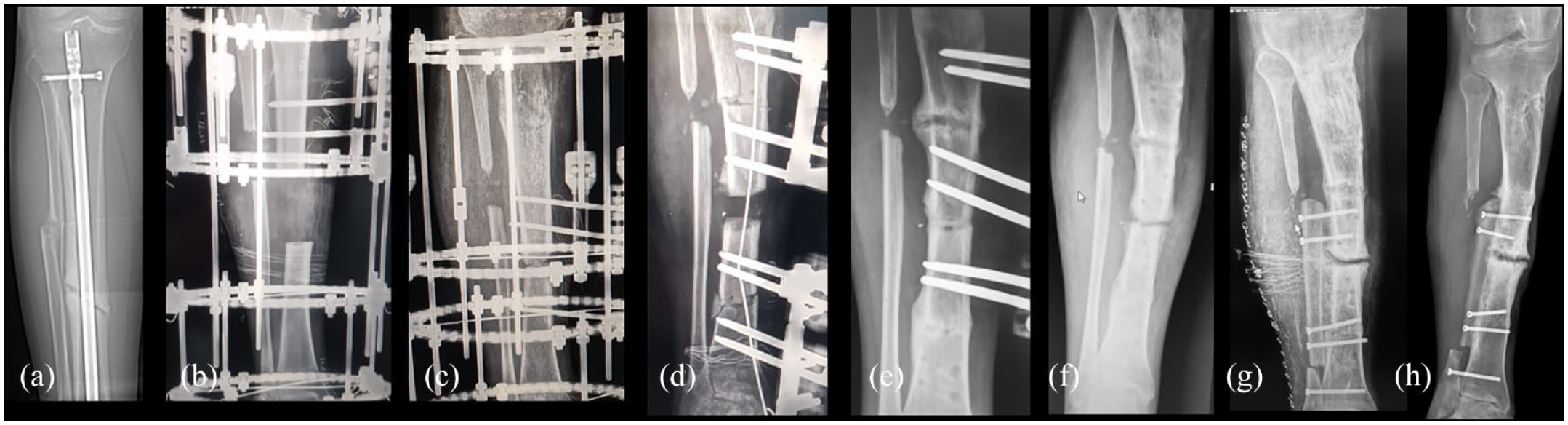
The efficacy of chipping corrective osteotomy (CCO) was recently reported in a small series in which a bone healing was achieved after a mean of 3.5 months. 63 The technique requires the use of a temporary external fixator, followed by a definitive osteosynthesis using a locking plate. CCO improves the bone healing probably thanks to a sort of biological chamber created by the chipping in fragments of the malunion site. Figure 2 shows a clinical case treated with chipping osteotomy.
A clinical case of a 38 years old male with a tibial varus malunion (a) after a road accident. Initially the malunion was treated using an exapodalic external fixator (b). In (c) the reoccurrence of varus deformity related to osteotomy partial non-union. In (d) intraoperative picture of the chipping osteotomy, leading to a good alignment and reliable healing; in (e) the post-operative X-ray; in (f) full length lower limb X-ray at 9 months after the surgery.
Fracture-Related Infections
Fracture-related infections (FRI) are a challenging and demanding complication with a severe impact on the healthcare systems, 64 considering the length of hospitalization, rehospitalization rate, 65 and the infection recurrence that may lead to a subsequent limb amputation in 3–5% of cases. 66
The reported rate of FRI for closed fractures was 1–2%, but it grew up to 30% in cases of open fractures.67,68
Theoretically, to reduce FRI occurrence, the treatment of an open fracture should be conducted in less than 6 h from the trauma, 69 and should be based on debridement, wound irrigation and antibiotic therapy. 69 However, current evidence reduced the impact of the 6-h rule on the infection rate, underlying, instead, the more relevant role of a careful surgical debridement and appropriate antibiotic treatement.70–73
In 2018, the International Consensus Meeting held in Philadelphia, proposed some criteria to define FRI, differentiating them in ‘confirmatory’ and ‘suggestive’ (see Table 3). 74
International Consensus Meeting (ICM) 2018 criteria for the diagnosis of FRI. 74
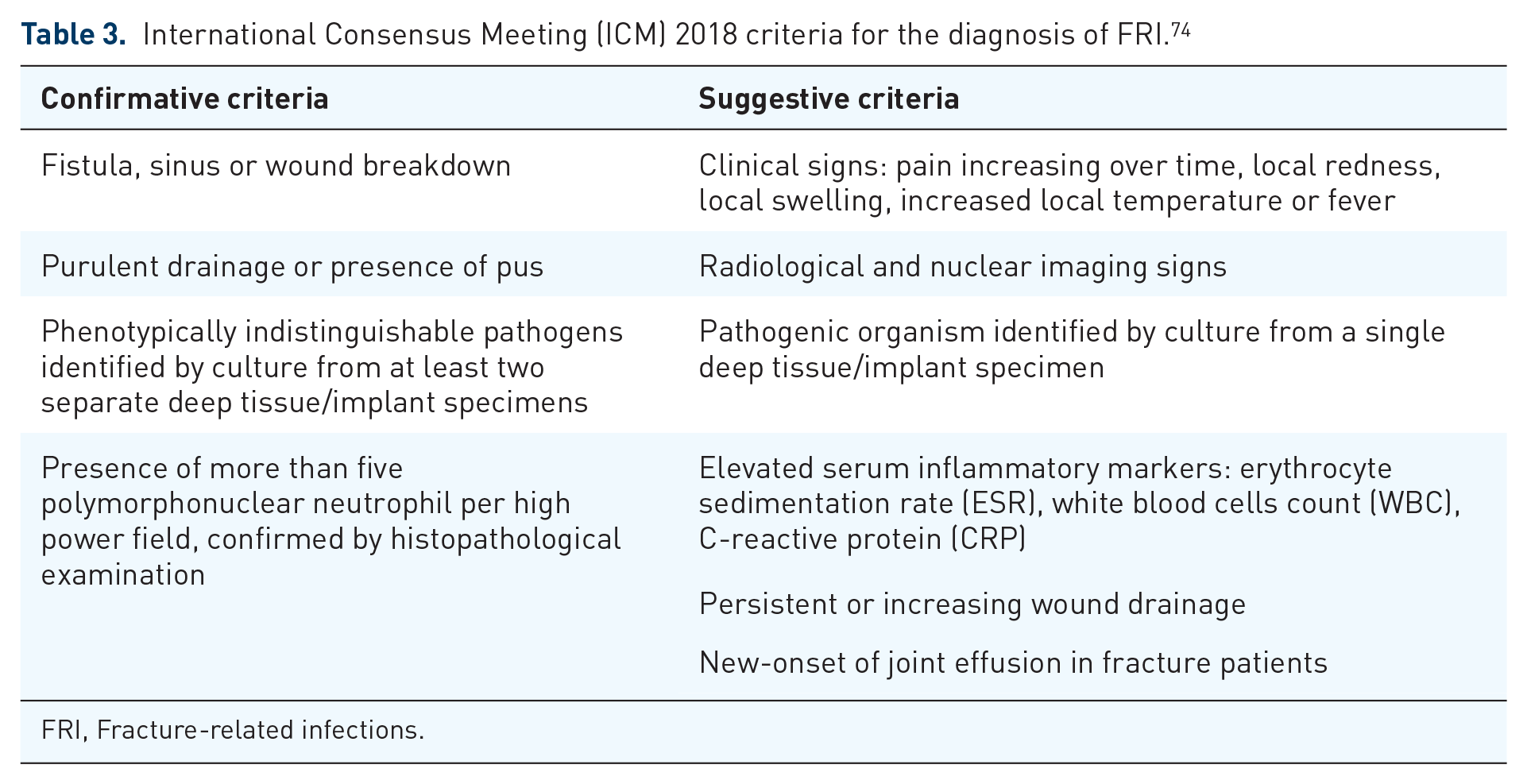
FRI, Fracture-related infections.
The treatment of FRI may be extremely difficult and frustrating for both the patient and the surgeon. One of the main aspects that may explain the poor outcomes of FRI is the formation of the bacterial biofilm on the foreign material represented by the fixation devices. 75 The biofilm is a polymeric matrix mainly consisting of bacterial products that protects them from harmful environmental conditions, including the host immune responses and antimicrobial agents.76–79 Moreover, the formation of a biofilm may lead also to improper diagnosis since bacteria in the biofilm are in a somewhat anergic state. A recent Systematic Review proposed that sonication of fluid culture might improve diagnostic abilities, especially in those patients already treated with antibiotic therapy. 80
Very often, to effectively treat FRI, the biofilm must be surgically excised. In this perspective, the use of an antiseptic/antibiofilm wound lavage may be of aid, as recently underlined by Whitely et al. 81 in their delayed debridement animal model of open fractures.
However, the timely identification of a FRI remains relevant to appropriately guide the surgeon in the treatment decision-making.74,82 The relevance of timing is underlined by one of the most widely used classification of FRI, based on the time elapsed between the fracture fixation and the onset of the infection. In an early onset FRI (between 0 and 1 week after surgery) the fracture is supposed to be not healed yet and the surgeon should try to retain the fixation implant. For this reason, in most cases surgical debridement with deep samples for cultures, and subsequent specific antibiotic therapy is often the treatment of choice [debridement, antimicrobial therapy and implant retention (DAIR)]. The antibiotic therapy may be prolonged until the bone healing occurred and then the implant should be removed. 83 In case of delayed onsets FRI (between 2 and 10 weeks after surgery), and in late/chronic onsets the removal of the hardware is always necessary, followed by surgical debridement and antibiotic therapy. There is no clear recommendation for the timing of an effective DAIR, but the rate of success decreases constantly with time. In fact, clinical studies reported a success rate of 90% when applied within 3 weeks,84,85 70% within 6 weeks,86,87 51–67% over 10 weeks after surgery.65,85,88 However, time, fracture healing status and implant stability are not the unique factors to consider when treating a FRI. In this context the Cierny’s classification may be of aid in the treatment decision-making, especially in case of late/chronic infections (see Tables 4 and 5). 89
Cierny’s classification.

Source: Adapted from Cierny et al. 89
Proposed intervention according to the Cierny’s classification.

Source: Adapted from Wassif et al. 90
After a surgical debridement and especially in case of chronic/late osteomyelitis or in case of infected non-union, one of the main critical issues to face up might be the reconstruction of the subsequent bone loss. Several techniques are available, including the use of autologous or heterologous bone graft, bone substitute or bone transport with external fixator.17,20,37,91
The ideal bone substitute should present osteoinduction (the process by which osteogenesis is induced), osteoconduction (the process through which the bone grows on a surface) and osseointegration (the stable anchorage of an implant achieved by direct bone-to-implant contact) properties. Only autografts (bone graft obtained from the affected patient) present all these three characteristics, but their availability is very limited. 92 The non-biological materials, such as ceramics, metals, alloys, polymers, composites, and hydrogels are generally referred as alloplasts and synthetic materials. 92 Considering their wide availability, synthetic bone substitute (i.e. calcium sulfate, calcium phosphate, hydroxyapatite) are generally used with the purpose to achieve bone loss reconstruction exploiting their reported osteoconduction and osteointegration properties. 93 However, both may be severely impaired by the persistence of infection. Therefore, several antibiotic-loaded bone substitutes had been proposed with encouraging results. 94
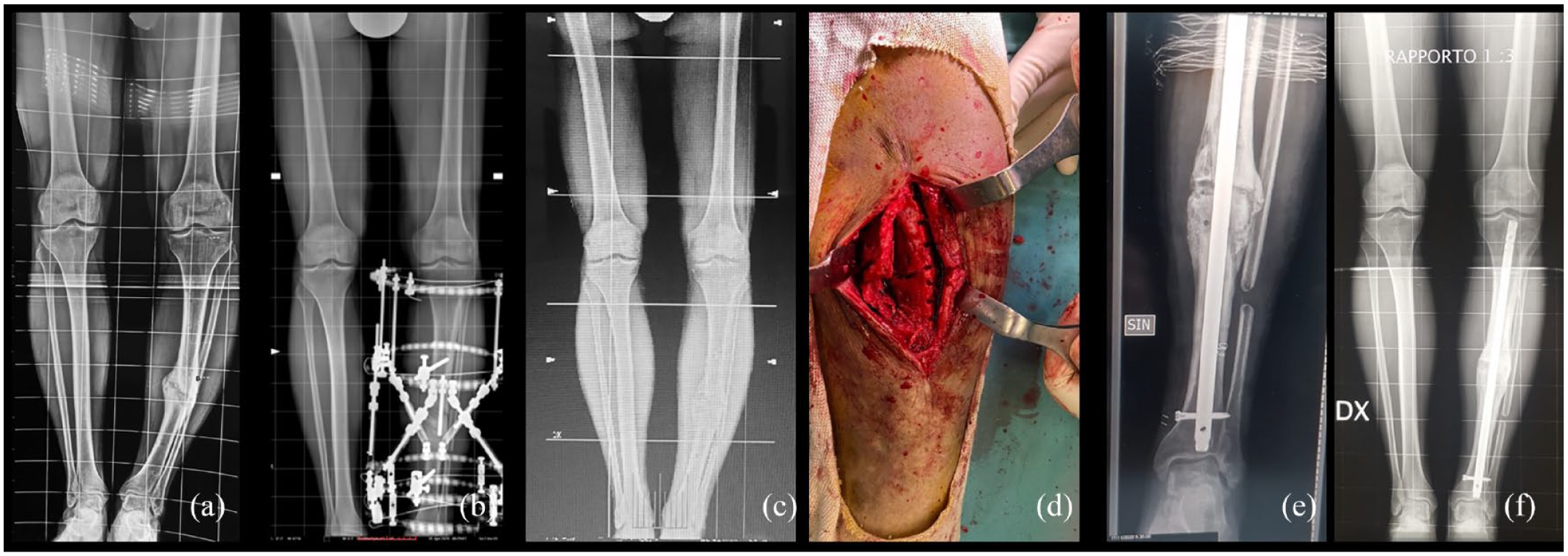
A case series of 100 patients with chronic osteomyelitis treated though a single-stage debridement and the application of an absorbable, gentamicin-loaded, calcium suphate/hydroxyapatite biocomposite, reported a healing rate of 96%, with rare adverse events. 95 Figure 3 shows a case of a chronic tibial osteomyelitis treated using an antibiotic-loaded bone substitute.

A clinical case of a 61 years old female with a tibial chronic osteomyelitis following a road accident occurred 40 years before our observation. In (a) and (b) clinics and antero-posterior and latero-lateral X-rays at the time of our observation. The patient was treated through a sequestrectomy and application of an antibiotic-loaded bone substitute. In (c) the intraoperative fluoroscopy before (left) and after (right) the application of the bone substitute. In (d) antero-posterior and latero-lateral X-rays at 1 month after the surgery. Note the partial bone substitute reabsorption. In (e) and (f) X-rays and clinics at 4 months after the surgery.
Anyway, also in the case of using antibiotic-loaded bone substitutes, prolonged antimicrobial therapy is recommended (at least 6 weeks after implant removal or 12 weeks in case of implant retention).96,97 One issue that limits the antibiotic use for FRI is also the poor bone penetration of most antibiotics. Currently there is a relevant effort to achieve a high bone antibiotic concentration using also local delivery agents.98–100 However a satisfactory kinetics of these local antibiotic-loaded carriers is not reported yet.
Conclusions
HEMTs are often associated with severe complications, especially non-union, malunion and FRI, that may lead to limb amputation. Although good outcomes were reported with limb amputation, this treatment still presents high long-term costs and disputable functional results. The limb preservation surgery for HEMTs-related complications may be frustrating and difficult, and often requires multiple/staged procedures. The orthopedic surgeon must use several techniques to tailorize-to-the-patient the treatment. In order to improve patients’ outcomes while reducing costs, an appropriate limb salvage technique selection is mandatory, even combining them. Moreover, the surgeon must keep in mind that bone biology is as essential as fracture stability and bone stock preservation/restoration. Finally, in our opinion the pivotal factor to effectively treat the HEMTs-related complications is the engagement of a close cooperative relationship between the patient and the surgeon.
