Abstract
For several thousand years (~4000) Boswellia serrata and Curcuma longa have been used in Aryuvedic medicine for treatment of various illnesses, including asthma, peptic ulcers, and rheumatoid arthritis, all of which are mediated through pathways associated with inflammation and pain. Although the in vivo pharmacology of both these natural ingredients is difficult to study because of poor bioavailability, in vitro data suggest that both influence gene expression mediated through nuclear factor kappa B (NF-κB). Therefore, the activity of pathways associated with inflammation (including NF-κB and lipoxygenase- and cyclooxygenase-mediated reduction in leukotrienes/prostaglandins) and those involved in matrix degradation and apoptosis are reduced, resulting in a reduction in pain. Additive activity of boswellic acids and curcumin was observed in preclinical models and synergism was suggested in clinical trials for the management of osteoarthritis (OA) pain. Overall, studies of these natural ingredients, alone or in combination, revealed that these extracts relieved pain from OA and other inflammatory conditions. This may present an opportunity to improve patient care by offering alternatives for patients and physicians, and potentially reducing nonsteroidal anti-inflammatory or other pharmacologic agent use. Additional research is needed on the effects of curcumin on the microbiome and the influence of intestinal metabolism on the activity of curcuminoids to further enhance formulations to ensure sufficient anti-inflammatory and antinociceptive activity. This narrative review includes evidence from in vitro and preclinical studies, and clinical trials that have evaluated the mechanism of action, pharmacokinetics, efficacy, and safety of curcumin and boswellic acids individually and in combination for the management of OA pain.
Keywords
Introduction
Osteoarthritis (OA) is a leading cause of disability in older adults, with over 500 million cases globally in 2019.1,2 Management of musculoskeletal pain and OA often includes pharmacologic agents, such as nonsteroidal anti-inflammatory drugs (NSAIDs). NSAIDs are broadly effective but are associated with safety concerns, including gastrointestinal, hepatic, and cardiovascular adverse events.3–5 Most patients with OA also have other comorbidities, such as diabetes, hypertension, and dyslipidemia, which complicates the identification of suitable treatment options and can further increase inflammation, leading to disease progression.6,7 Therefore, additional safe and effective alternative pain relief options are needed.
Patients and physicians are beginning to explore complementary and prophylactic therapies that may offer both symptom relief and a favorable safety profile.8,9 In addition, patients and physicians want interventions that (1) eventually become prophylactic options to help treat the root cause of the condition, (2) decrease inflammation and pain, which subsequently reduces damage, (3) have more favorable adverse-effect profiles, and (4) can be taken long term. Natural ingredients have the potential to fill these needs and could minimize NSAID exposure and associated adverse events.10–13 Furthermore, in vitro bioassays suggest that the use of natural ingredients in combination with NSAIDs could provide a more favorable safety/risk–benefit profile; 14 molecular studies investigating the combination at the chemical level are ongoing. Some natural ingredients can be part of the solution in the approach to personalized pain medicine; however, many studies described in the literature did not combine NSAIDs and natural ingredients in the comparator arm of their trials.15–17
Two natural ingredients, Curcuma longa and Boswellia serrata, have been used for several thousand years (~4000) for the treatment of inflammatory, pain-related, and degenerative disorders.18–20 Synergy of the beneficial anti-inflammatory and antinociceptive effects from both curcumin and boswellic acids has been suggested both in vitro and in vivo, and pharmacokinetic and clinical studies in humans reported no major safety concerns when these two natural ingredients are combined.21–24 Therefore, identifying the appropriate dose and formulation for the combination of these natural ingredients for complementary and/or synergistic targeting of OA and musculoskeletal pain is of interest. In addition, identification of the appropriate dosing in combination with NSAIDs can increase the number of pain treatment options for patients and provide an improved risk profile.
The objective of this narrative review was to examine the efficacy and safety of curcumin and boswellic acids alone to support their use in combination to treat and manage musculoskeletal pain in patients with OA. These two natural ingredients were recently investigated for many different biological conditions, such as OA and cancer, and those findings are relevant for identifying the affected molecular pathways. In addition, individual complementary and prophylactic therapies were found to be non-inferior to NSAIDs in clinical trials.10,12,13,25–27 The combination of natural ingredients may prove to be as effective as, or more effective than, pharmacologic agents due to their unique mode of action and complementary and/or synergistic effects.
Curcumin mechanism of action
Curcumin, the main component of polyphenolic compounds called curcuminoids, is extracted from turmeric, a product of the Curcuma longa plant; additional curcuminoid components include demethoxycurcumin and bisdemethoxycurcumin (Figure 1). 28 The evidence for the mechanism of action of curcumin is derived from in vitro and in vivo studies, including cell lines representative of synovial cells and chondrocytes, rat and mouse models, and clinical samples from patients with OA and healthy volunteers. Curcumin downregulates the cyclooxygenase-2 (COX-2) pathway, reducing the production of prostaglandins associated with inflammation (Figure 2).29,30 Curcumin also downregulates and directly inhibits lipoxygenase (LOX) and downregulates inducible nitric oxide synthase, mitogen-activated protein kinases, and Janus kinases, which are associated with inflammatory processes.29,31 Downstream effects of curcumin through these pathways include inhibition of nuclear factor kappa-light-chain-enhancer of activated B cell (NF-κB)-mediated gene expression of cytokines, including reduced production of tumor necrosis factor-alpha (TNF-α), interleukins (ILs-1, -2, -6, -8, and -12), monocyte chemoattractant protein (MCP), migration inhibitory protein, prostaglandin E2 (PGE2), matrix metalloproteinase (MMP)-2,-3,-9, inflammasome NLRP3, and reactive oxygen species.28,32–38 IL-10, a cytokine associated with reducing inflammation, is increased by curcumin supplementation in various inflammatory diseases, and the anti-inflammatory activity of IL-10 is enhanced through blocking pathways associated with inflammation. 39 For pain, curcumin’s antinociceptive effect is mediated by IL-10 via augmentation of Nrf2 and Cu/Zn superoxide dismutase. 39 Nrf2/ARE is a key pathway in curcumin-mediated protection against inflammation and oxidative stress in chondrocytes. 40

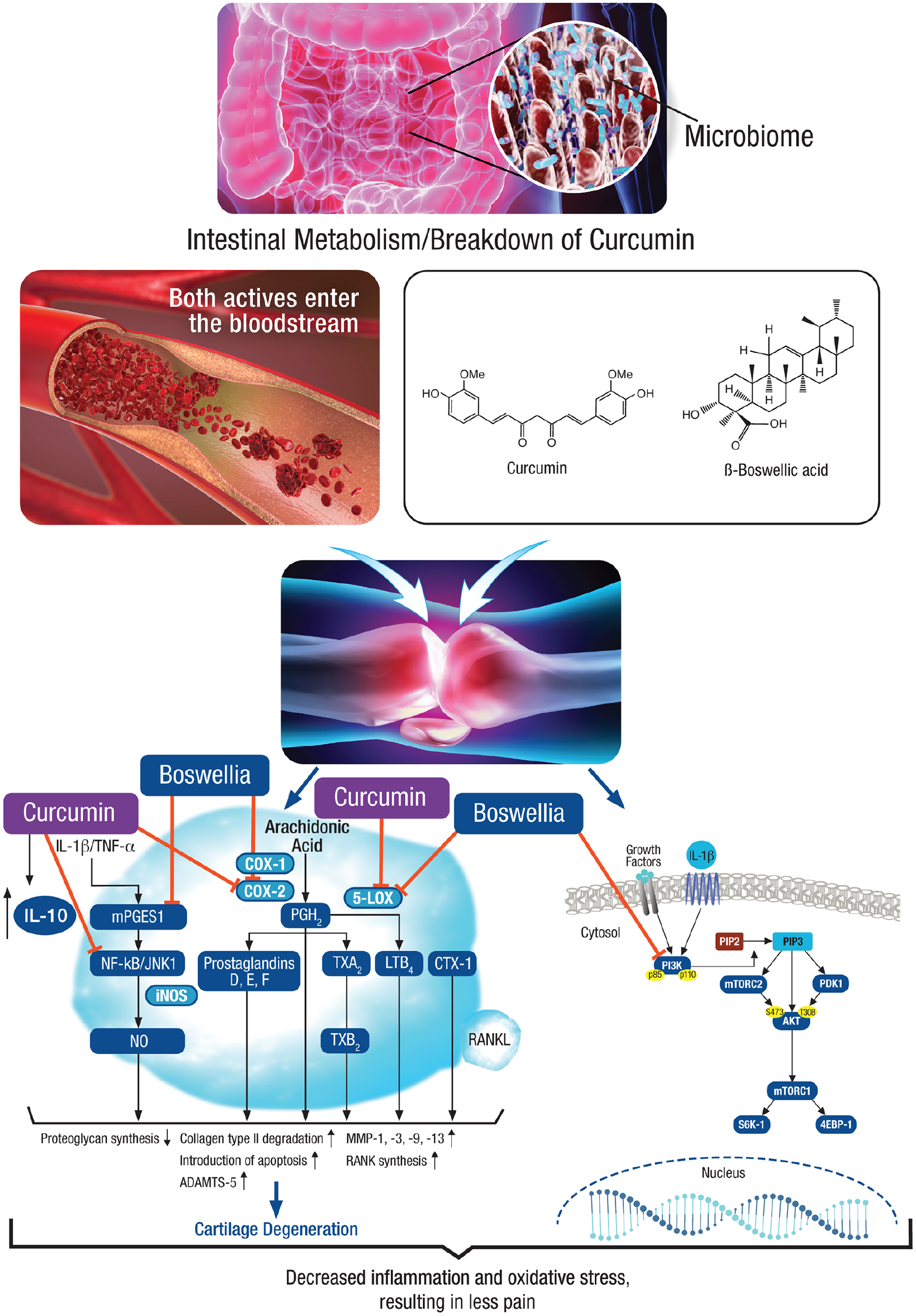
Mechanism of action of curcumin and boswellic acids in osteoarthritis based on in vitro study data. 4EBP-1, eukaryotic translation initiation factor 4E-binding protein 1; 5-LOX, 5-lipoxygenase; ADAMTS, a disintegrin and metalloproteinase with thrombospondin motifs; AKT, protein kinase B; COX, cyclooxygenase; CTX, carboxy-terminal cross-linking telopeptide of type I collagen; IL, interleukin; iNOS, inducible nitrous oxide synthase; JNK, c-Jun N-terminal kinase; LTB, lymphotoxin-beta; MMP, matrix metalloproteinase; mPGES-1, microsomal prostaglandin E synthase-1; mTORC, mammalian target of rapamycin complex; NF-κB, nuclear factor-kappa B; NO, nitrous oxide; PGH2, prostaglandin H2; PI3 K, phosphatidylinositol 3-kinase; PIP3, phosphatidylinositol 3,4,5 trisphosphate; RANKL, receptor activator of NFκB ligand; S6 K-1, ribosomal protein S6 kinase beta-1; TNF-α, tumor necrosis factor–alpha; TXA2, thromboxane A2; TXB2, thromboxane B2.
Curcumin also was shown to protect human temporomandibular joint chondrocytes from matrix degradation. 40 Curcumin has potential effects on Toll-like receptor 4 (TLR4) in models of OA.41,42 A study of intra-articular administration of curcumin in the right knee of a rat with OA (classical model of OA induced by anterior cruciate ligament transection), showed that curcumin repressed lipopolysaccharide-induced IL-1β and TNF-α secretion from the synovium and inhibited lipopolysaccharide-induced overexpression of TLR4 and downstream NF-κB in cartilage and synovial tissues. 41 Thus, curcumin effectively reduces activity in inflammatory pathways known to be involved with OA pathogenesis. 32 Through reductions in inflammation, OA pain is reduced as well.
Curcuminoids have demonstrated other activities in preclinical studies, including antineoplastic, antifungal, analgesic, antimicrobial, antioxidant, hepatoprotective, hypoglycemic, immunostimulant, antiasthmatic, and hypercholesterolemic effects.28,45 Although most of these observations are from in vitro studies, the authors hypothesized that the potential universal effect of the curcuminoids on inflammation/oxidative stress occurs through an effect on redox imbalance, which could suggest that curcumin may be a prodrug because it appears to be active in numerous pathways. 46
This hypothesis also is supported by curcumin’s poor bioavailability in humans. Furthermore, the prodrug theory could support the variability observed in different preparations of curcumin; bioactive intermediates from oxidative metabolism of curcumin account for at least some of the observed activity. 46 The biologic effects of curcumin in the gut also may explain its overall anti-inflammatory effects, as CurA from Escherichia coli in the human intestine has been shown to produce tetrahydrocurcumin, a metabolite of curcumin that has been shown to be biologically active.47,48 If the microbial products of curcumin (metabolites), which have yet to be identified, are responsible for the beneficial effects of curcumin, then limited bioavailability or absorption in the small intestine may not be a significant issue. For example, there have been various studies in rats in which tissue or serum malondialdehyde concentrations were lowered by 12–30% when either 100 or 200 mg/kg curcumin was administered before intestinal ischemia reperfusion.47,49–52 Curcumin has effects on microbiota, intestinal permeability, gut inflammation, oxidative response, anaphylactic reactions to oral allergic food exposure, and bacterial, parasitic, and fungal infections, 47 and it is believed that positive effects on the microbiome may have effects on extra-intestinal disease. 47 Concentrated extracts of curcumin through solvent extraction of turmeric, such as BCM-95 and C3, 53 provide another mechanism for increasing bioavailability. The efficacy of this approach is illustrated by one pharmacokinetic study in healthy volunteers that showed an approximate seven-fold increase in bioavailability with BCM-95 compared with standard curcumin. 54 Overall, the mechanism of action and clinical pharmacology of curcumin in vivo need additional research in humans to confirm the in vitro data and to address how to achieve effective concentrations of active curcumin metabolites.
Boswellic acids mechanism of action
Boswellia is derived from the gum resin extracts of the Indian olibanum tree, Boswellia serrata, which contains a mixture of triterpene acids known as boswellic acids (Figure 1).45,55
Two boswellic acids, 11-keto-β-boswellic acid (KBA) and acetyl-11-keto-β-boswellic acid (AKBA), were proposed as the main transducers of pharmacologic effects. 55 Boswellic acids inhibit the 5-LOX pathway, reducing the production of proinflammatory leukotrienes.56,57 However, no clear correlation has been established between KBA/AKBA plasma concentrations and the effective concentrations for inhibiting 5-LOX, which may be due to the poor absorption of AKBA after oral administration. 55 One approach to increase AKBA bioavailability has been to use concentrated extracts, such as ApresFlex and 5-Loxin, which have between 20% and 30% AKBA, respectively. 58 In a preclinical rat model, ApresFlex showed greater absorption versus the same dose of 5-Loxin. 59 Further research into concentrated extracts could provide more insight into the effective concentrations needed to inhibit 5-LOX. Boswellic acids also inhibit COX-2 in the arachidonic acid pathway, metabolites of which mediate pain and inflammation. 57 COX-2 inhibition decreases prostaglandin H2 and subsequently PGE2, which promotes inflammation.55,57 β-Boswellic acid has shown higher steady-state plasma concentrations in humans than AKBA, which aligns with the in vitro IC50 values for cathepsin G (catG) and microsomal prostaglandin E synthase-1 (mPGES-1), suggesting an alternative pathway for anti-inflammatory effects. 55 mPGES-1 is likely to be involved in cartilage/matrix homeostasis through mechanosensitive gene induction, and is a potential therapeutic target in OA. 60
Another pathway implicated in the anti-inflammatory effects of AKBA is NF-κB. In a rat model of osteoporosis, administration of 35 mg/kg AKBA for 42 days resulted in significant improvement in calcium content and bone mineral density (p < 0.01 for both) and downregulation of NF-κB and NF-κB-regulated gene expression, suggesting that AKBA has a role in management of postmenopausal osteoporosis through inhibition of osteoclastogenesis. 61
The PI3K/AKT pathway regulates target genes involved in cancer and OA, and in vitro studies with AKBA in these different disease states suggests that there may be a common mechanism of action. Potential anticancer effects of AKBA have been observed in human gastric cancer cell lines through induction of apoptosis, cell cycle arrest, autophagy suppression, and inhibition of cell migration and the PI3K/AKT pathway.62–65 Anticancer effects were studied in a variety of cell lines and cancer types and have demonstrated dose-dependent responses, the potential to use AKBA as an adjuvant to chemotherapy, and as a promising therapy for non-small cell lung cancer.62,63,65 Recent in vitro and in vivo studies have suggested a potential new role of the PI3K/AKT pathway in OA, where inhibition of PI3K/AKT prevents expression of target genes (e.g. TNF-α, IL-6, MMPs, and COX-2) that increase the inflammatory responses and cartilage matrix degradation that lead to OA development.66–68 Boswellic acids therefore may downregulate inflammation and oxidative stress in OA, thereby reducing cartilage damage and joint pain.
Comorbidities, such as diabetes and obesity, can amplify mechanical and inflammatory pressure on the joints in OA. Boswellic acids act on inflammation in diabetes and other comorbid conditions that make joint pain worse in OA, and may prevent types 1 and 2 diabetes through suppression of the expression of proinflammatory cytokines.69,70 Additional research is needed to confirm the pharmacodynamics of boswellic acids in vivo, similar to curcumin. The safety of boswellic acids has been studied preclinically in primates and rats, with no toxic side effects observed. 45 One of the side effects reported in humans is skin reactions or rashes, but thus far, no serious safety issues have been documented. 71
Antinociceptive effects of curcumin and boswellic acids
Preclinical OA animal models in mice and rats showed antinociceptive effects of curcumin and boswellic acids via topical and oral administration, respectively.72,73 In the mouse OA model, topical curcumin nanoparticles reduced tactile hypersensitivity in the von Frey test, increased distance traveled, and increased rearing compared with vehicle-treated animals that also had destabilization of the medial meniscus (all p < 0.05). 72 This suggests curcumin is effective in relieving pain associated with OA. Additional investigation showed that oral and topical curcumin slowed the progression of OA in the mouse model through decreased cartilage erosion and proteoglycan loss, reduced synovitis and subchondral plate thickness, reduced degradation of type II collagen and aggrecan, and lowered expression of MMP-13 and ADAMTS5 compared with vehicle controls. 72 In the rat OA model, rats treated with boswellic acids improved weight-bearing function measured via the von Frey test at day 14 and latency of paw withdrawal at day 15 compared with monoiodoacetate control rats. 73 In vitro studies of boswellic acids showed dose-dependent inhibition of 5-LOX activity and strongly inhibited PGE2 production in lipopolysaccharide-induced peripheral blood mononuclear cells, indicative of inhibition of the COX pathway for inflammatory pain. 73 Furthermore, boswellic acids helped block the harmful effects of proinflammatory cytokines on human chondrocytes in culture. 73 Therefore, there is the potential for both curcumin and boswellic acids to be protective for chondrocytes in OA and for additive benefit in reducing pain in OA. Additional research in humans is needed to confirm the exact mechanism of these natural ingredients in reducing pain in OA.
Curcumin for OA
Data from clinical trials evaluating the efficacy of curcumin for managing OA (Table 1) support that curcumin is effective at reducing musculoskeletal pain and inflammation in patients with OA, thereby increasing function and quality of life.
Clinical trials of curcumin for OA.

ESR, erythrocyte sedimentation rate; hsCRP, high-sensitivity C-reactive protein; IL, interleukin; KOOS, knee injury and osteoarthritis score outcome; LPFI, Lequesne pain functional index; KPS, Karnofsky performance scale; KPSI, Karnofsky performance scale index; mfr, manufacturer; NA, not applicable; NSAIDs, nonsteroidal anti-inflammatory drugs; OA, osteoarthritis; PGADA, patient global assessment of disease activity; PGE2, prostaglandin E2; sVCAM-1, soluble vascular cell adhesion molecule; TNF, tumor necrosis factor; VAS, visual analogue scale; WOMAC, Western Ontario and McMaster Universities score.
A meta-analysis of five studies with 599 patients with OA concluded that curcumin improved the Western Ontario and McMaster Universities (WOMAC) score and visual analog scale (VAS) score compared with placebo (both p < 0.01). 89 There was no statistical difference between curcumin and placebo for side effect rate nor was there a difference between curcumin and ibuprofen. 89 Another systematic review and meta-analysis focused on 32 randomized controlled trials (N = 2038 participants) that evaluated the anti-inflammatory effects of oral curcumin through blood concentrations of inflammatory markers. 90 Large reductions in TNF-α, MCP-1, IL-8, IL-6, and C-reactive protein (CRP) concentrations were observed in participants treated with curcumin versus those in the control group (all p < 0.01), further supporting in vitro and preclinical model findings. 90
Furthermore, a systematic review and meta-analysis of 11 randomized controlled trials comparing the efficacy of turmeric extracts versus placebo and five trials comparing efficacy versus NSAIDs in 1810 adults with knee OA showed turmeric significantly reduced knee pain and improved physical function compared with placebo but had effects similar to NSAIDs. 91 Although biomarkers of inflammation were evaluated in multiple trials, there were no significant between-group differences reported. 91 Rates of adverse events were lower in the turmeric extract groups versus those for NSAIDs; however, rates were similar between groups treated with turmeric extract and placebo. 91 An additional study suggests that long-term use of curcumin supplements for the management of OA is preferred to improve pain and reduce stiffness. 92 Therefore, curcumin at doses between 160 and 2000 mg/day was efficacious in the management of OA and demonstrated similar efficacy to NSAIDs, including diclofenac (100 mg day) and ibuprofen (400–1200 mg/day).80,81,89,91
Combining curcumin with other natural ingredient supplements for management of OA pain has also been investigated in clinical trials. Notably, two clinical trials showed significant benefit from the combination of curcumin and glucosamine as measured by improvement in walking (p < 0.001) and pain (reduced VAS score; p < 0.05 for both studies compared with control group), suggesting that combining curcumin with other natural product supplements may be beneficial in OA.76,82
Boswellic acids for OA
Clinical trials of boswellic acids in OA suggest that formulation and composition of the oral capsules/tablets influences efficacy in reducing pain and increasing function (Table 2).11,93–95 The majority of clinical trials had a smaller number of participants, but two randomized trials had more than 100 participants (N = 120 and N = 440) and showed that boswellic acids demonstrated equal effectiveness in the management of OA pain compared with glucosamine.13,96 In the smaller of these two prospective randomized trials, patients with knee OA (N = 120) received either boswellic acids or glucosamine sulfate for 60 days. VAS and Lequesne total scores were assessed during follow-up visits at 2 and 6 months. 13 At baseline and 2 months, patients treated with boswellic acids had higher mean VAS and Lequesne total scores versus patients treated with glucosamine; however, these scores were lower at 6 months with boswellic acids versus glucosamine treatment (p = 0.08 and p = 0.02, respectively). Patients treated with glucosamine showed early reduction of mean scores at 2 months, but scores remained generally unchanged from 2 to 6 months. 13 Biomarkers of inflammatory pathways were not reported in the clinical trials evaluating boswellic acids for OA pain management; therefore, further research is needed to confirm in vitro and preclinical model findings. Boswellic acids have a well-defined safety profile with few adverse events reported during clinical trials.
Clinical trials of boswellic acids for OA.

AKSS, American Knee Society Score; BBA, β-boswellic acid; GI, gastrointestinal; hsCRP, high-sensitivity C-reactive protein; LPFI, Lequesne pain functional index; NA, not applicable; OA, osteoarthritis; PGA, patient global assessment; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities score.
Combination of curcumin and boswellic acids
Several studies have investigated the combination of curcumin and boswellic acids as management for OA pain (Table 3). One primary study (N = 201) showed that the combination of curcumin and boswellic acids reduced WOMAC total score (p < 0.001) from baseline to visit 3 at 84 days and compared with placebo (p < 0.05) after 3 months of continuous treatment. 24 The treatments were well tolerated and no serious adverse events were observed. 24 Because there are a limited number of clinical studies, additional studies are needed to fully understand the potential synergy of curcumin and boswellic acids for the management of OA pain. Investigation of curcumin and boswellic acids alone and in combination allows for the identification of synergistic pharmacologic effects. 22 The synergistic activity of these two natural ingredients could reduce the dosage and frequency of consumption of acetaminophen and NSAIDs, as both individually have been shown to reduce NSAID use, which would be advantageous for patients with OA through decreased NSAID exposure and associated adverse events.10,12,13,25–27 A 2-by-2 factorial randomized controlled trial is needed to provide evidence for the synergistic effects.
Studies of curcumin and boswellic acids in combination for OA pain.
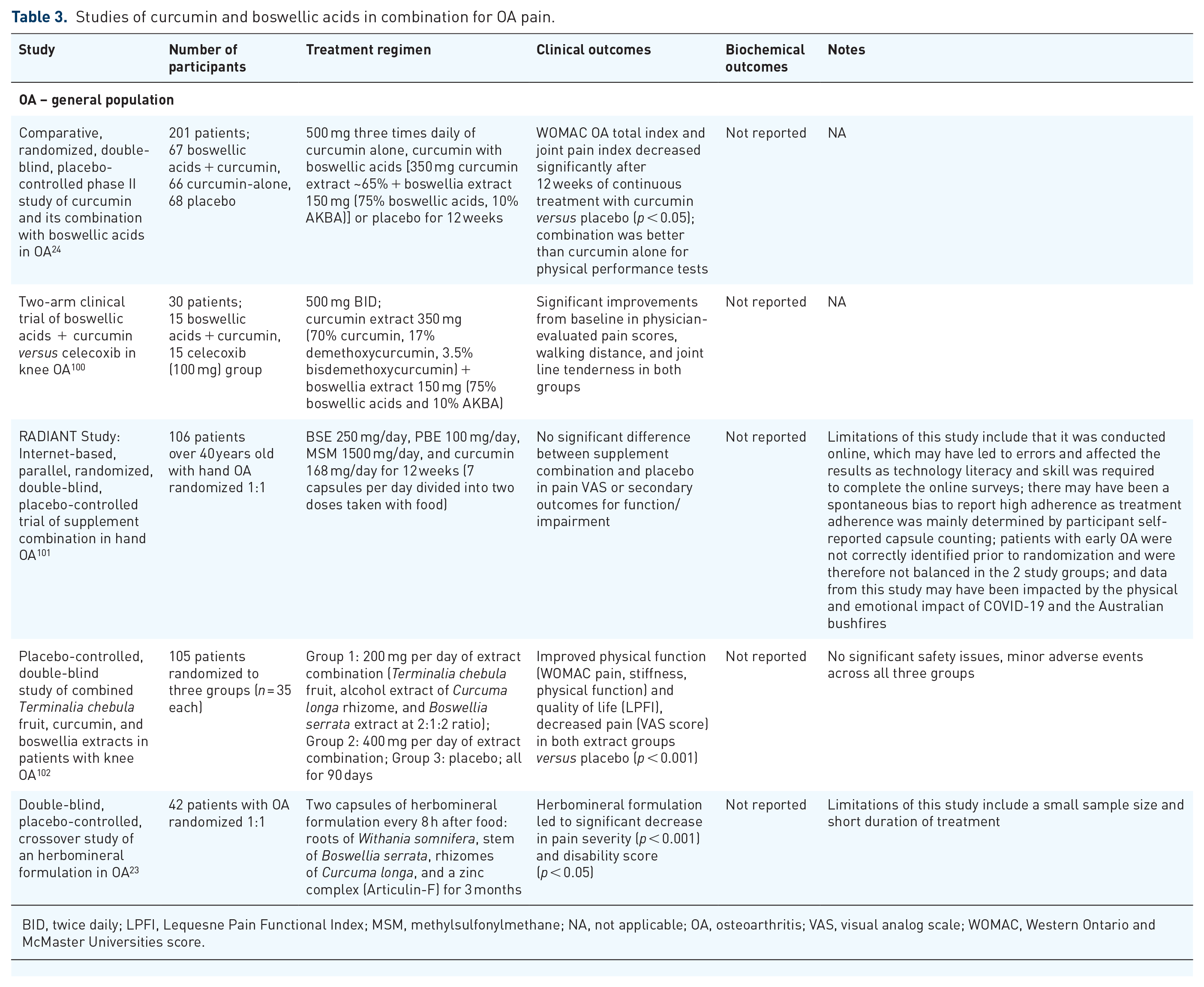
BID, twice daily; LPFI, Lequesne Pain Functional Index; MSM, methylsulfonylmethane; NA, not applicable; OA, osteoarthritis; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities score.
In addition to the number of clinical trials evaluating the combination, further support comes from numerous animal studies that evaluated the combination of curcumin and boswellic acids in other disease states, including rheumatoid arthritis and cancer. Preclinical data on the combination of solubilized micellar curcumin and micellar Boswellia in the treatment of rheumatoid arthritis in a rat model showed anti-inflammatory effects comparable to NSAIDs even at much lower doses. 103 In addition, preclinical data suggest a possible role for the combination of curcumin and boswellic acids as chemoprevention of colorectal cancer and as an antiviral agent.104–106 Antiproliferative and proapoptotic effects were observed simultaneously, as well as the promotion of epithelial turnover in the adenomatous polyposis coli (Apc) mouse model for intestinal carcinogenesis in animals receiving an enriched diet with curcumin, AKBA, and silymarin for 110 days. 105 Although the animals with the enriched diet still developed small bowel carcinomas at a similar rate to those animals receiving the standard diet (85% versus 100%), the enriched diet led to a significant (p < 0.001) decrease in the number and size of polypoid lesions. 105 In further support of the chemopreventive effects of curcumin and AKBA, a mouse xenograft model demonstrated that each product alone suppressed tumor growth, but when used in combination, synergistic tumor suppression was observed. 104
Curcumin and boswellic acids (as well as other constituents, such as maritime pine) were studied in combination for other indications in clinical trials in humans, including acute musculoskeletal pain, chronic kidney disease, benign thyroid nodules, diverticulitis, tendinopathy, and Gulf War syndrome.107–112 In healthy adults with acute musculoskeletal pain, treatment with the combination of curcumin and boswellic acids or with acetaminophen for 7 days reduced pain intensity at a similar rate and to a similar level. 113 The only difference observed between the two groups was improved reduction in the affective domain of the McGill Pain Questionnaire in the curcumin and boswellic acids treatment group (8.57 times better, p = 0.027). 113 In patients with early-stage chronic kidney disease, treatment with curcumin and boswellic acids for 8 weeks increased PGE2 levels to a lesser extent than placebo. 109 Spirulina–curcumin–boswellia (400–50–50 mg per capsule, twice daily for 6 weeks) was effective in reducing the size of benign thyroid nodules in 29 of 34 patients; although the authors of the study did not specify a mechanism, they did propose that this reduction may be related to the anti-inflammatory effects of the natural ingredients. 108 An investigation in healthy adult males who were master athletes that followed the Mediterranean diet and took a curcumin and Boswellia serrata supplement for 3 months showed significant decreases in advanced glycation end products and malondialdehyde compared with those who only followed the Mediterranean diet (p < 0.05), suggesting that supplementation could improve muscle performance. 114
Pharmacokinetics of boswellic acids and curcumin
A phase I, crossover study showed that there were no pharmacokinetic interactions between curcumin and boswellic acids when administered in combination, and no adverse events were reported when either supplement was taken alone or in combination. 21
Safety
Both curcumin and boswellic acids ingredients have been ‘generally recognized as safe’ by the World Health Organization,115,116 and there is a long history of their use. However, for both curcumin and boswellic acids, some allergic dermatitis was reported for individuals who frequently handle the natural product.115,116 Overall, curcumin and boswellic acids in combination are associated with few/infrequent adverse events.
Limitations
The conclusions of this review are based on our review of the literature and should be interpreted with caution as we did not perform an independent critical appraisal of selected trials. This was beyond the scope of this manuscript as this is a narrative literature review and not a systematic literature review. Another limitation is that no definitive conclusion could be made regarding the synergism of using curcumin and boswellic acids together for the management of inflammation and pain in OA. While a few studies have compared the effects of the combination of curcumin and boswellic acids, these studies have compared the effects of the combination treatment with placebo and not with each treatment individually. Therefore, additional studies are required to conclusively demonstrate the synergistic effect of these treatments.
Conclusion
This review article integrates all available data on curcumin and boswellic acids for the management of inflammation and pain in OA and provides the rationale for the combined use of these ingredients. Further metabolomic studies to determine if microbial metabolites of curcumin and boswellic acids are related to their clinical efficacy would be helpful for dosing recommendations. Additional research on absorption, as well as determination of bioavailability and the relationship with clinical efficacy is needed for curcumin to establish a dose-effect. 22 There may also be potential in using concentrated extracts of curcumin and boswellic acids to overcome issues with bioavailability.54,59 The anti-inflammatory effects of boswellic acids and curcumin have translated to the improvement of symptoms of OA and joint pain as shown in numerous clinical trials. Antinociceptive effects were demonstrated in preclinical models of OA for both boswellic acids and curcumin. The combination of these two anti-inflammatory natural ingredients suggests potential synergistic activity, which supports their use to further improve OA symptoms and pain. This combination could also improve patients’ quality of life and reduce the dosage and frequency of consumption of acetaminophen and NSAIDs.10,12,13,25–27 Other potential mechanisms have been recognized in recent years (eg, antioxidant effects and effects on microbiota) and there are key molecular targets identified from research on cancer that are relevant to OA (i.e. PI3K or AKT enzymes). 22 There is support for the use of curcumin and boswellic acids in combination to reduce inflammation and pain in patients with OA.
