Abstract
Background:
Intestinal Behçet’s syndrome is a major cause of morbidity and mortality in Behçet’s syndrome.
Objectives:
Current treatment challenges remain in refractory intestinal Behçet’s syndrome, when patients failed first and second-line therapies.
Design:
We reported the efficacy and safety profiles of tofacitinib in patients with moderate-severe intestinal Behçet’s syndrome in a retrospective single-center study.
Methods:
Treatment with glucocorticoids, immunosuppressors, or even anti-TNFα monoclonal antibodies (mAbs) had previously failed. Primary outcomes were clinical remission or low disease activity and endoscopic healing.
Results:
We included 13 patients; 11 were administered tofacitinib 5 mg twice daily, and 2 took tofacitinib 5 mg once daily. Nine patients achieved clinical remission after a mean treatment duration of 10.1 ± 7.0 months, and the other four had low disease activity. Follow-up endoscopy was available in 11 patients: 5 had achieved mucosal healing; the other 4 achieved marked mucosal improvement. Prednisone dosage was significantly reduced, from 30 (interquartile range: 20–30) mg/d to 2.5 (interquartile range: 0–12.5) mg/d (p < 0.001). No serious adverse event was observed.
Conclusion:
Tofacitinib could be an efficacious and generally well-tolerated option in patients with intestinal Behçet’s syndrome refractory to conventional agents, even anti-TNFα mAbs.
Introduction
Behçet’s syndrome is a complex disorder with heterogeneous clinical manifestations. 1 The prevalence of intestinal involvement in Behçet’s syndrome is high in China, featured by large and single ileocecal ulcers revealed by colonoscopy. 2 Higher disease activity at diagnosis, deep volcano-shaped intestinal ulcers, absence of mucosal healing, prior history of surgery, and lack of response to medical therapy increase the risk of perforation and bleeding. 3 The cumulative probabilities of surgery at 10 years were around 50% of patients after diagnosis. 4 Moreover, the recurrence after the initial operation was 46.1%; 50% of recurrence developed within 2 years.5,6 Therefore, intestinal involvement is one of the leading causes of mortality of Behçet’s syndrome.
Anti-TNFα monoclonal antibodies (mAbs), infliximab, and adalimumab, Tanida et al. 7 demonstrated their efficacy in intestinal Behçet’s syndrome, which has significantly improved the outcomes. Tanida et al. 7 and Iwata et al., 8 considered the standard therapy. 9 Recently, we reported the results from a cohort of 85 intestinal Behçet’s syndrome patients treated with infliximab. After 1 year of infliximab therapy, the non-mucosal healing rate was 34.12%, and no clinical remission rate was 23.53% of patients. 10 The refractory to anti-TNFα mAbs phenotype observed in our cohort was a common challenge in other tertiary academic medical centers. A Japanese fellow previously described the ulcer exacerbation rates were 24.5% (12 of 49 patients) in the patients treated with anti-TNFα mAbs. 11 Primary non-response or gradual loss of response to anti-TNFα mAbs remain high. 11 Accordingly, refractory cases remain a treatment dilemma, particularly non-responder to anti-TNFα mAbs.
Tofacitinib has proved its efficacy in inflammatory disorders like rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis.12–14 Compared to anti-TNFα mAbs directed against specific cytokines, JAKi target multiple pathways such as type l interferons (IFNs), interleukin (IL)-6, and IL-23, which have been implicated in the pathogenesis of Behçet’s syndrome. 15 Previously, Liu et al. 16 and our team 17 have reported tofacitinib might be effective in some phenotypes of Behçet’s syndrome, such as vascular, articular, and uveitis involvement. However, intestinal Behçet’s syndrome patients seemed to respond poorly to tofacitinib. 16 So far, no additional data have been reported on the efficacy of tofacitinib in treating intestinal Behçet’s syndrome. Here, we reported the efficacy and safety profiles of tofacitinib in 13 patients with moderate-severe intestinal Behçet’s syndrome who were refractory to conventional therapy and even anti-TNFα mAbs.
Patients and methods
Patients
We conducted a retrospective study on patients with intestinal Behçet’s syndrome treated with tofacitinib in the Division of Rheumatology and Immunology at Huadong Hospital, Fudan University, from November 2019 to January 2022. We selected the International Criteria for Behçet’s Disease 18 or the Korean criteria of intestinal Behçet’s syndrome 19 for inclusion. The latter criteria are based on the endoscopic features of ileocolonic ulcerations and systemic manifestations. Patients not fulfilling the criteria were clinically diagnosed with BS based on consensus from gastroenterologists, rheumatologists, pathologists, and endoscopists. This study conforms to the STROBE statement. 20 We have de-identified all patient details.
Inclusion criteria included (1) patients had moderate-to-severe disease severity defined by the disease activity index of intestinal Behçet’s disease (DAIBD). 21 (2) Subjects had previously failed to be treated with glucocorticoids, immunosuppressors, or even anti-TNFα mAbs.
Exclusion criteria included (1) patients who had been diagnosed with infection, tuberculosis, Crohn’s disease (CD), nonspecific colitis, and drug-induced disease (corticosteroids and nonsteroidal anti-inflammatory drugs); (2) patients with impaired cardiac, pulmonary, renal, or hepatic function, neurological disorders, and pregnant or breastfeeding women; and (3) patients with a history of malignancy.
Approach to treatment
Tofacitinib 5 mg was administered twice daily, concurrent treatments with moderate glucocorticoids were allowed, and immunosuppressants were withdrawn.
Clinical measurement
The primary outcomes were clinical remission and low disease activity, with a DAIBD score ⩽ 19, and endoscopic healing, defined as mucosal healing. Secondary outcomes included low-disease activity rate determined by DAIBD, endoscopic evaluations, 7 laboratory findings, and any serious adverse events (SAEs) between baseline and last follow-up.
Statistical analysis
Statistical analyses were performed using SPSS version 23.0 (IBM, Armonk, NY, USA). Values are expressed as means ± SD or medians with interquartile range (IQR), according to the data distribution. The student’s test or Wilcoxon signed-rank test was used when appropriate. A p value < 0.05 was considered significant.
Results
Baseline characteristics
Thirteen patients (3 males and 10 females) were analyzed; the mean age was 39.8 ± 4.2 years, and the mean disease duration was 14.9 ± 2.3 years. After enrollment, 11 patients were administered tofacitinib 5 mg twice daily, 2 took tofacitinib 5 mg once daily (according to their body weight), glucocorticoids, and immunosuppressants were withdrawn. The baseline demographic data are shown in Table 1. All patients were refractory to conventional agents; eight were further refractory to anti-TNFα mAbs with a mean number of 2 failed biologics; two patients had a history of surgical resections.
Characteristics and outcomes with tofacitinib therapy in patients with moderate-severe intestinal Behçet’s syndrome.
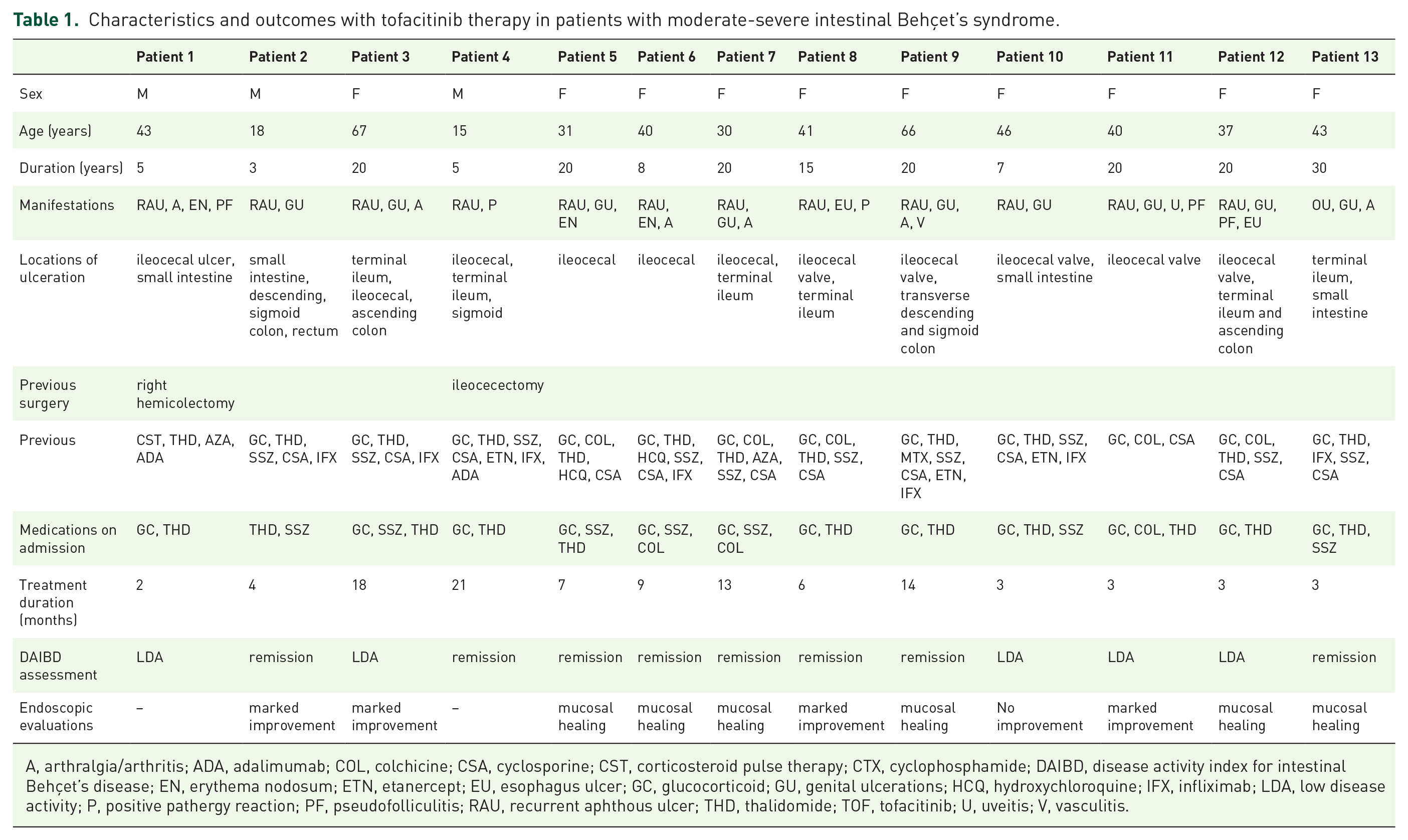
A, arthralgia/arthritis; ADA, adalimumab; COL, colchicine; CSA, cyclosporine; CST, corticosteroid pulse therapy; CTX, cyclophosphamide; DAIBD, disease activity index for intestinal Behçet’s disease; EN, erythema nodosum; ETN, etanercept; EU, esophagus ulcer; GC, glucocorticoid; GU, genital ulcerations; HCQ, hydroxychloroquine; IFX, infliximab; LDA, low disease activity; P, positive pathergy reaction; PF, pseudofolliculitis; RAU, recurrent aphthous ulcer; THD, thalidomide; TOF, tofacitinib; U, uveitis; V, vasculitis.
Efficacy of tofacitinib
Mean treatment duration 10.1 ± 1.9 months, DAIBD decreased from 90 (IQR: 75–100) to 10 (IQR: 0 – 25), p = 0.002, Figure 1. All patients experienced a rapid and sustained resolution of systemic symptoms, such as fatigue or fever (no. 5 and no. 7), and diminished or resolved abdominal symptoms, such as abdominal pain, constipation, or diarrhea (no. 1–4, 6). C-reactive protein (CRP) dropped from baseline 10.42 (IQR: 2.48–35.35) mg/l to last visit 0.80 (IQR: 0.50–1.28) mg/l, p = 0.003.

The improvement of DAIBD in the patients treated with tofacitinib intestinal Behçet’s syndrome.
Eight patients remained in remission at the last follow-up, and the others were in low disease activity. Follow-up colonoscopy was available in 11 patients; 6 patients (no. 5–7, 9, 12, and no. 13) have achieved mucosal healing; 4 (no. 2–3, 8, and no. 11) have achieved marked mucosal improvement, while no. 10 had no endoscopic improvement (Figure 2).
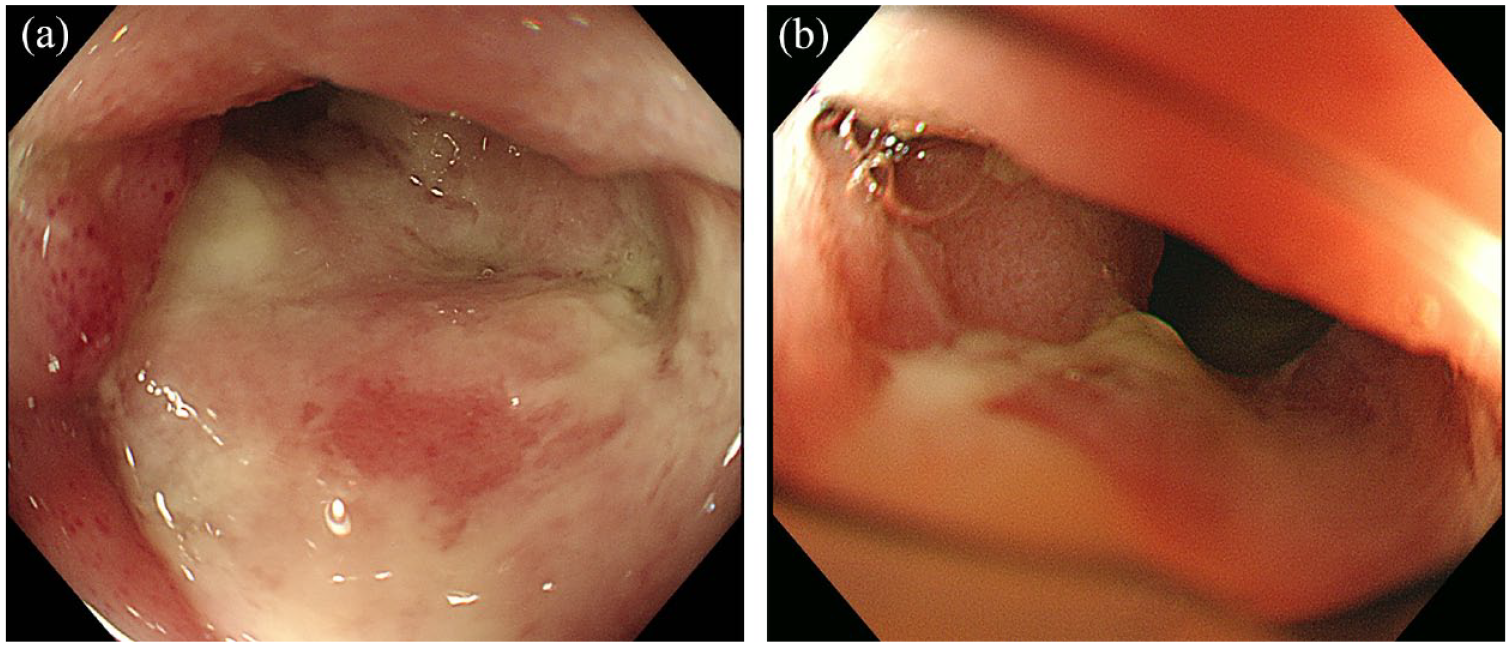
Panel (a) shows a 40-year-old female (no. 11) with ileocecal valve ulcer 5.0*4.0 cm at baseline. Panel (b) shows after 2 months’ treatment with Tofacitinib 5 mg twice daily, she achieved marked endoscopic improvement (ileocecal valve ulcer 1.0 cm in diameter).
Steroid-sparing effects
At the end of follow-up, tofacitinib 5 mg twice daily was maintained in nine patients, and it was successfully tapered down to 5 mg once daily in four cases. In five patients, corticosteroid was successfully withdrawn, and five other patients received a prednisone dosage of fewer than 10 mg daily. Prednisone dosage was significantly reduced from tofacitinib initiation 30 (IQR: 20–30) mg/d to the last follow-up 2.5 (IQR: 0–12.5) mg/d (p < 0.001).
Adverse events
Our study observed no SAEs during tofacitinib therapy, including no cases of herpes zoster, severe infection, cardiovascular event, venous thromboembolic event, and cause of mortality.
Discussion
Our study is the first report indicating the efficacy of tofacitinib in patients with intestinal Behçet’s syndrome, even refractory to anti-TNFα mAbs. We found that tofacitinib therapy was associated with significantly decreased disease activity scores. Within a mean treatment duration of 10 months, most patients (8/13) in our cohort were in remission. They achieved a mucosal healing rate of 55%. Its steroid-sparing effects were observed.
Recently, a meta-analysis of anti-TNFα mAbs in intestinal Behçet’s syndrome combined 13 studies with a total of 739 patients and estimated the pooled proportions of clinical remission at months 3 and 24 were 0.61 (95% CI: 0.48–0.78) and 0.38 (95% CI: 0.16–0.88), and mucosal healing at months 3 and 24 were 0.66 (95% CI: 0.50–0.86) and 0.69 (95% CI: 0.39–1.00), respectively. 22 Although anti-TNFα mAbs are the standard treatment for refractory diseases, the proportion of non-responders remains high. Therapeutic options in cases of failed anti-TNFα mAbs are limited. Our preliminary results imply the efficacy of tofacitinib is comparable to that of tumor necrosis factor inhibitor (TNFi) in intestinal Behçet’s syndrome. Considering most patients in our cohort were TNFi experienced, tofacitinib could have a superior benefit over TNFi.
The treatment options usually overlap between intestinal Behçet’s syndrome and CD. 11 The efficacy of tofacitinib in ulcerative colitis (UC) was not replicated in CD or a previous report with five intestinal Behçet’s syndrome cases. 16 From Liu et al. 16 report, they defined the clinical response as (1) improved: BS-related manifestations resolved and no newly present imaging/endoscopic findings observed and (2) unchanged or worsened: Behçet’s syndrome–related manifestations or abnormal laboratory parameters persisted or worsened after following up for 12–24 weeks. 16 As they mentioned, the intestinal ulceration was healed in one patient and persisted in the other five patients. At the end of the follow-up, four patients had a normal CRP, and two patients had an elevated CRP. However, the authors judged: that 1/6 patients improved, 1/6 worsened, and 4/6 patients were unchanged. Although we do not know the details of endoscopic response in Zhang’s study, we could find some conflicts between Behçet’s Disease Current Activity Form improvement, ESR, CRP levels, and the clinical response judgment. Besides, the high rates of fistula formation (3/6) and perforation (2/6) in Zhang’s study could lead to a poor clinical response.
In our cohort, most patients had features of poor prognosis, such as refractory to conventional treatments, a history of intestinal surgery, or large intestinal ulcers. Tofacitinib, as a pan Janus kinases (JAKs) inhibitor, provided a rapid and broad-spectrum cytokine-blocking effect in patients with intestinal Behçet’s syndrome. As a result, DAIBD scores and levels of CRP decreased substantially, correlated with endoscopic responses. Our results demonstrated the efficacy of tofacitinib in intestinal Behçet’s syndrome patients with large intestinal ulcers. No. 11 patient having a giant ileocecal valve ulcer (5 cm in diameter) achieved mucosal healing after 4 months of treatment.
The limitations of this study arise primarily from an uncontrolled observational design, a small sample size, and a short follow-up duration in several cases. It should be noted that we have more females than males in the cohort, which could lead to a selection bias.
Conclusions
In the present study, our preliminary analysis of tofacitinib administration to patients with refractory intestinal Behçet’s syndrome to conventional agents or even anti-TNFα mAbs demonstrated its high efficacy based on a significant decrease in disease activity index and endoscopy results and its safety profiles. Our data provided a possible application of tofacitinib as an option or rescue therapy in refractory intestinal Behçet’s syndrome to anti-TNFα mAbs. Our encouraging findings justify a more rigorous clinical trial to examine small molecule therapy in treating intestinal Behçet’s syndrome and other phenotypes of Behçet’s syndrome.
