Abstract
Objective:
Autologous hematopoietic stem cell transplantation (AHSCT) is a therapeutic option for patients with severe and progressive systemic sclerosis (SSc). Here, we aimed to investigate how AHSCT affects the vasculopathy of SSc patients.
Methods:
Twenty-seven SSc patients were retrospectively assessed, before and after AHSCT, for vessel morphology (nailfold capillaroscopy), skin expression of endothelial markers and serum levels of markers of inflammation, angiogenesis and endothelial activation. Skin biopsies were analyzed by immunohistochemistry (IHC) for expression of CD31, VE-cadherin, E-selectin, angiopoietin-1 (Ang1), angiopoietin-2 (Ang2), Tie-2, vascular endothelial growth factor A (VEGFA), vascular endothelial growth factor receptor 2 (VEGFR2), and endothelin-1 before and 12 months post-AHSCT. Serum samples from SSc patients were assessed before and up to 36 months after AHSCT for IL-6, von Willebrand factor (vWF), CXC Motif Chemokine Ligand 8 (CXCL8), Endothelin-1, epidermal growth factor (EGF), VEGFA, Pentraxin-3, Intercellular Adhesion Molecule 1 (ICAM-1), E-selectin, P-selectin, Thrombomodulin and IL-18 levels, and compared to healthy control samples.
Results:
On nailfold capillaroscopy, the number of capillaries increased at 1 year, while giant capillaries decreased at 6 months and 1 year after AHSCT. In the skin biopsies, expression of E-selectin notably decreased and Ang1 increased after AHSCT. At baseline, all vascular markers evaluated in the serum were significantly higher in SSc patients when compared to healthy controls, except for ICAM-1. When compared at different time points after AHSCT, Thrombomodulin, Pentraxin-3, vWF, and IL-18 levels remained generally stable at high levels until 36 months after AHSCT.
Conclusion:
Our results suggest that AHSCT contributes to improvements of the vessel morphology and dermal microvasculopathy, but does not normalize elevated levels of serum vascular markers in SSc patients. Additional vascular therapeutic approaches might contribute to more effectively treat the endothelial injury.
Introduction
Systemic sclerosis (SSc) is a complex autoimmune disease, characterized by micro and macro vasculopathy, immunological abnormalities, and fibroblast activation that lead to excessive accumulation of connective tissue and result in fibrosis of the skin and internal organs. 1 In the past 20 years, autologous hematopoietic stem cell transplantation (AHSCT) has emerged as a therapeutic option for patients with severe and progressive SSc. 2 Randomized studies have shown long-lasting clinical remission, longer survival and better quality of life in SSc patients treated with AHSCT, when compared to those treated with conventional immunosuppressive therapies.3–5 Mechanistic investigations show that AHSCT modulates fibrosis and immune activation,6–9 but little is known about the impact of AHSCT on microvascular morphology, endothelial injury and vascular repair.10–12
Although the etiology of SSc is still unknown, injury and apoptosis of endothelial cells stand as initial events in the proposed pathogenesis of the disease. 13 Raynaud’s phenomenon is an early manifestation that reflects abnormal functioning of thermoregulatory vessels and usually precedes skin fibrosis in months to years. 14 Physiologically, several changes occur in the endothelial environment of SSc patients, including an imbalance between pro- and anti-angiogenic mediators, increased expression of cell adhesion molecules, platelet activation, deregulation of vascular tonus, impaired compensatory vasculogenesis and angiogenesis, and production of reactive oxygen species (ROS).15,16 Morphological abnormalities in the peripheral microcirculation can be assessed by nailfold capillaroscopy. The characteristic ‘scleroderma pattern’ includes sequential and dynamic capillaroscopic findings of widened capillaries, avascularity, hemorrhages and distortion of the normal capillary architecture. 17 The scleroderma pattern has been subclassified into early, active and late stages. 18 In the early stage, there are isolated giant capillaries and microhemorrhages, but with preserved architecture and no evident loss of capillaries. In the active stage, there are frequent giant capillaries and microhemorrhages, while the late stage is characterized by extensive avascular areas. 19
Several studies have investigated potential vascular biomarkers that may reflect endothelial damage. Critical biomarkers include endothelial activation and adhesion molecules, angiogenic mediators, and vasoconstrictors.20–23 To better understand the efficacy of AHSCT on SSc-associated vasculopathy, we evaluated serum, histological and nailfold capillaroscopy markers before and after transplantation.
Methods
Study design
In this retrospective study, SSc patients were evaluated for clinical, serum and histological parameters, before and after AHSCT. Clinical data were retrieved from patient medical records, serum samples were available from baseline (pre-transplantation) and from 6, 12, 24 and 36 months post-transplantation, and skin biopsies were available from baseline and 12 months post-transplantation. Baseline assessments were performed shortly before the procedure, and were completed at a median (range) interval of 6 (2–19) days prior to the start of the hematopoietic progenitor cell mobilization regimen. Routine baseline clinical evaluations included Rodnan’s skin score (mRSS), high resolution computed tomography, pulmonary function test, echocardiography, 24-h electrocardiography monitoring (Holter test), esophageal manometry and biochemical tests for kidney and liver functions, as well as complete blood counts, serum C-reactive protein dosage and anti-nuclear antibody and anti-SCl70 titers. Ongoing immunosuppressive treatment was stopped immediately before the start of the evaluations. Data from baseline were compared to those from subsequent time points. A group of healthy controls was included to compare serum markers.
Once considered suitable for transplantation, patients signed informed consent forms agreeing to undergo transplantation. Additional written informed consents for blood and skin biopsy collection were obtained from all patients upon enrolment for transplantation. The local ethics board (Comitê de Ética em Pesquisa do Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto, Ribeirão Preto, Brazil) approved the study protocol (number 71204717.6.0000.5440) on December 13, 2017. Due to the retrospective design of the study, patient-signed consent for publication was waived by the review board, and patient details were de-identified. The reporting of this study complies with the STROBE statement. 24
Patients and transplant procedure
Systemic sclerosis patients fulfilling the American College of Rheumatology/European League Against Rheumatism (EULAR) 2013 criteria, and who underwent AHSCT from November 2011 to September 2016, were retrospectively considered for this study. Indications for AHSCT included diffuse skin involvement and/or interstitial lung disease, worsening despite conventional immunosuppressive treatment. Detailed eligibility criteria for AHSCT and the transplant procedure have been previously reported. 25 Briefly, the autologous hematopoietic progenitor cells were mobilized from the bone marrow to the peripheral blood using a combination of 2 g/m2 intravenous cyclophosphamide plus subcutaneous injections of granulocyte-colony stimulating factor (G-CSF). Progenitor cells were harvested by apheresis and immediately cryopreserved, unmanipulated. Later, patients received the conditioning regimen, consisting of 200 mg/kg intravenous cyclophosphamide plus rabbit anti-thymocyte globulin, divided in five consecutive days of infusions. Three patients received an alternative regimen of 120 mg/m2 Fludarabine and 120 mg/m2 Melphalan plus rabbit anti-thymocyte globulin due to baseline cardiac involvement. Subsequently, the autologous progenitor cells were thawed at the bedside and infused intravenously. Patients remained in isolated protection until leukocyte engraftment (approximately 9 days after cell infusion) and were discharged from the hospital thereafter. Patients were then followed at the outpatient clinic at predefined intervals until 5 years after AHSCT. Patients eligible for this study had serum samples and skin biopsies available from baseline, and from at least one post-transplantation time point, as well as clinical data registered in medical records. Thirty-nine consecutive adult SSc patients that underwent AHSCT during the study period were initially considered for the study. However, one subject died shortly after AHSCT (transplant-related death) and 11 did not have samples collected before and after the procedure. Therefore, 27 patients fulfilled the eligibility criteria for the present study and were included.
Modified Rodnan’s skin score
Skin thickening was evaluated by mRSS, as previously described, for a final score ranging from 0 to 51. 26 All patients were evaluated, at all time points, by the same certified investigator, non-blinded to patient status.
Skin biopsies
Skin samples were obtained from patient forearms, through incisions with a ‘punch’ cutting tool, under local anesthesia with 2% lidocaine. Subsequent biopsies were performed in areas adjacent to the first, on the same limb. After collection, the material was fixed in 10% buffered formalin and subsequently included in paraffin blocks. For all patients, skin biopsies were available from baseline and from the closest period to the 12-month time point, corresponding to a mean (standard deviation) time of 11.5 (2.4) months after transplantation.
Serum evaluations
Approximately 5 mL of peripheral blood were collected in tubes not containing anticoagulants. Samples were centrifuged at 1500 rpm, for 10 min at room temperature. Then, 1 mL of the serum was fractionated in 250 µL aliquots and stored at -80° for later analysis. Serum levels of IL-6, vWFA, CXCL8, EGF, VEGFA, pentraxin-3, E-selectin, trombomodulin, P-selectin, V-CAM1, I-CAM1, endothelin-1, and IL-18 were determined by immune-enzymatic method using commercially available multiplex assay (Magnetic Luminex Assay, R&D System Inc., Minnesota, USA).
Immunostaining
Immunohistochemistry
Four-micrometer paraffin sections were placed on previously treated glass slides. The slides were then incubated at (20°C) for 60 min with monoclonal antibodies for anti-CD31 (1:100; Abcam, UK), anti-VE-cadherin (1:50; Novus Biologicals, USA), anti-E-selectin (1:100; Novus Biologicals, USA), anti-Ang1 (1:100; Abcam, UK), anti-Ang2 (1:100; Abcam, UK), anti-Tie-2 (1:100; Novus Biologicals, USA), anti-VEGFA (1:100; Abnova, USA), anti-VEGFR2 (1:100; Novus Biologicals) and anti-Endothelin-1 (1:200; Novus Biologicals, USA), diluted in BSA 0.1% (bovine serum albumin). Antibody binding was revealed using chromogen NOVARED™ (Vector, SK-4800, USA). Counterstaining was performed with hematoxylin.
Hematoxylin and eosin
The 5 µm paraffin sections were deposited on glass slides. Then, the sections were incubated with hematoxylin for 1 min and washed under running water. Ammonia was added to obtain bluish color and incubated with eosin for 1 to 2 min. Sections were then hydrated in increasing gradients of alcohol and diaphanized in xylol. Slides were mounted with Permount (Electron Microscopy Sciences, USA).
Histological analyses
A Carl Zeiss LSM710 Observer Confocal System was used for microscopic observation. Images were analyzed by Image J software (1.53 h version; National Institutes of Health, USA), with a plug-in package developed by McMaster University (Ontario, Canada).
Nailfold widefield capillaroscopy
Patients underwent widefield nailfold capillaroscopy before and at 6 and 12 months after AHSCT, using a SZ2 Olympus stereomicroscope (Olympus Corporation, Japan). All fingers, except the thumbs, were inspected under 10 to 40X magnification with real-time evaluation of the full nailfold region of each finger, for the following variables: number of enlarged (over four times the normal width of capillary loops) and giant (over 10 times wider than normal width of capillary loops), avascular score, number of microhemorrhages, and capillary loops. 27 A single certified researcher evaluated all patients at all time points.
Statistical analysis
Descriptive data were defined as percentages, mean (standard deviation) or median (range or interquartile range, IQR). Data were initially evaluated for normal distribution by Shapiro-Wilk test, and parametric or non-parametric tests chosen accordingly. Way-ANOVA or Kruskal–Wallis tests followed by Tukey’s or Dunn’s multiple comparisons tests were used to analyze differences between means of three or more groups. To evaluate how time is associated with clinical and laboratory data, mixed effects linear regression models were used, where time was the fixed event. Orthogonal contrasts approach was used to compare data from different time points. Wilcoxon or paired t test was used for differences between means of two groups. Correlations between mRSS values and histological expression of markers were tested using Spearman’s test. A p value ⩽ 0.05 was considered statistically significant. GraphPad 8 (8.3.1 version; GraphPad Software In., USA) was used for analysis and construction of graphs.
Results
Patient characteristics and clinical outcomes
All 27 patients included in the study had the diffuse subtype of SSc. Detailed patient characteristics are shown in Table 1. Median (range) disease duration was 2 (0.5–14) years, age at transplantation was 33 (21–59) years and 74% of patients were female. Esophageal motility and lung parenchyma were affected in 92% and 70% of patients, respectively. Twenty-five (94%) patients had positive antinuclear antibodies, and 18 (66.7%) had positive anti-topoisomerase I (anti-Scl70) antibody titers. Twenty-five patients presented clinical evidence of vasculopathy before transplantation, with Raynaud’s phenomenon, digital ulcers, and/or telangiectasias. Clinical evaluation of skin fibrosis, assessed by mRSS, improved from mean (SD) 20.9 (8.0) at baseline to 14.1 (6.9) at 6 months (p < 0.0001), and from baseline to 12.4 (7.1) at 12 months (p < 0.0001) after AHSCT (Table 2).
Baseline characteristics of patients.

AHSCT, autologous hematopoietic stem cell transplantation; ATG, anti-thymocyte globulin; n, number of patients; SD, standard deviation.
Patient characteristics before and after stem cell transplantation.
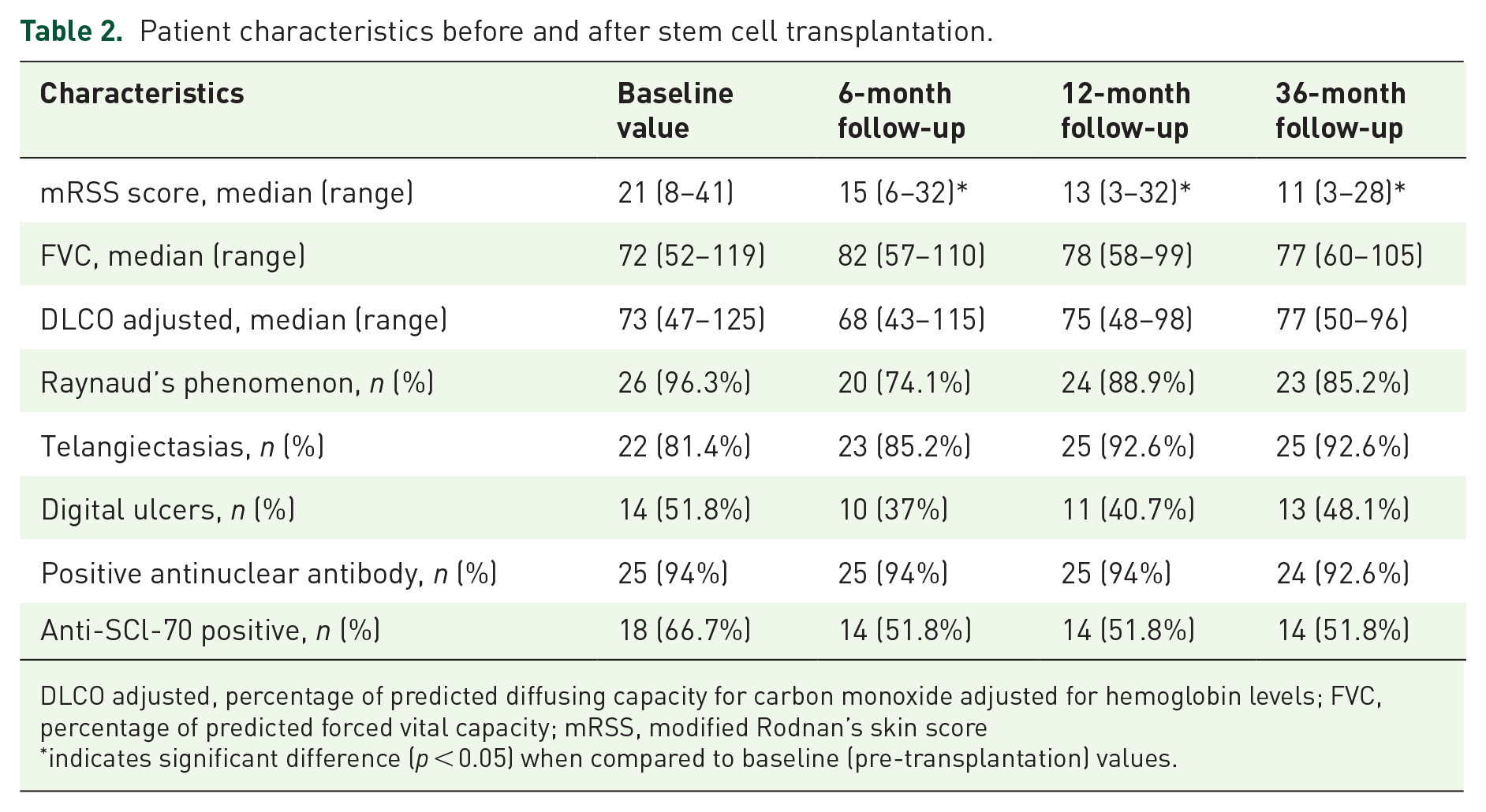
DLCO adjusted, percentage of predicted diffusing capacity for carbon monoxide adjusted for hemoglobin levels; FVC, percentage of predicted forced vital capacity; mRSS, modified Rodnan’s skin score
indicates significant difference (p < 0.05) when compared to baseline (pre-transplantation) values.
Nailfold capillaroscopy assessment post-transplantation
At baseline, all patients presented a scleroderma pattern under nailfold capillaroscopy. The number of capillaries increased from median (IQR) 5.8 (5.2–6.4) at baseline to 7.6 (7.4–8.4) at 12 months after AHSCT, (p < 0.05); (Figure 1(a)). There was also a reduction in the number of giant capillaries from 0.8 (0.1–1.5) at baseline to 0.15 (0.0–0.4) at 6 months (p < 0.05) and 0.2 (0.0–0.6) at 12 months after AHSCT (p < 0.05) (Figure 1(c)). No differences were observed in the other variables, such as capillary loss (deletions) (p = 0.177); (Figure 1(b)), enlarged (p = 0.489); (Figure 1(d)) and ramified capillaries (p = 0.489); (Figure 1(e)), and microhemorrhages (p = 0.662); (Figure 1(f)).
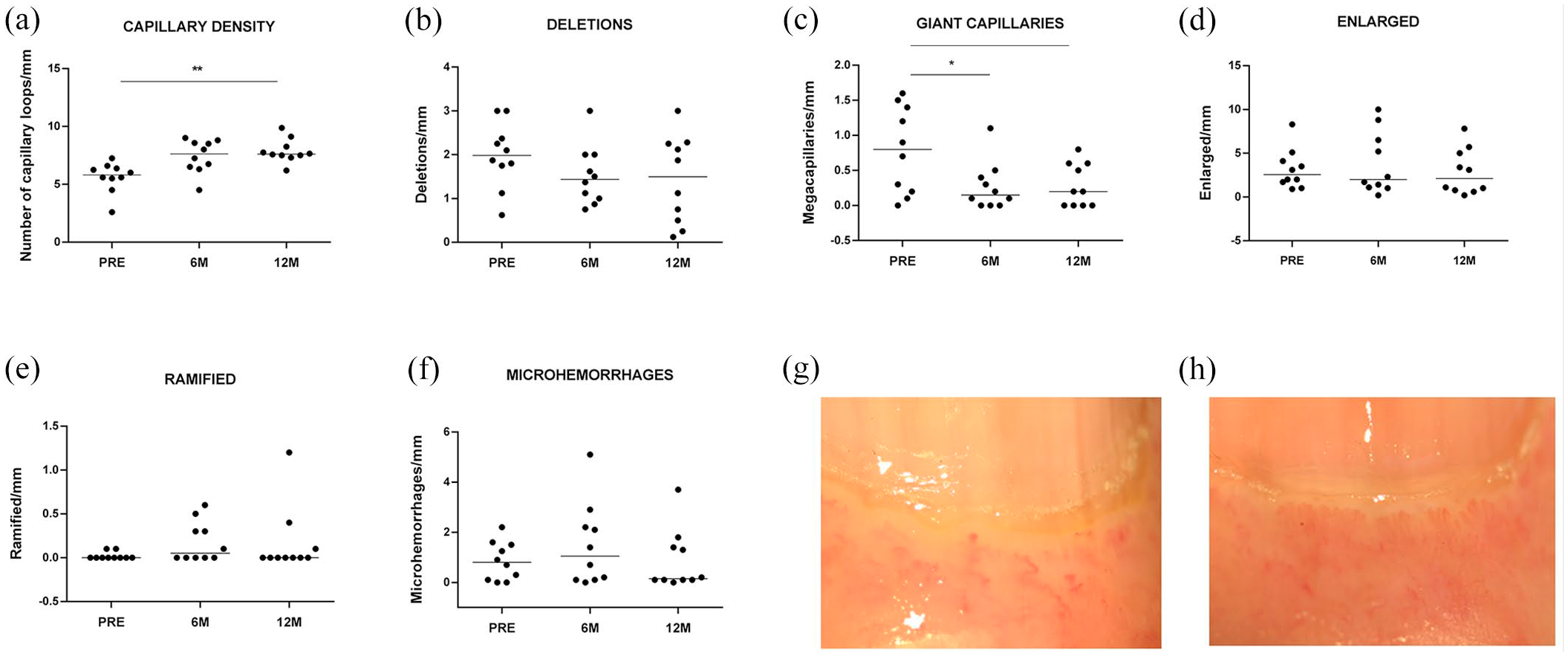
Nailfold capillaroscopy assessment before and after transplantation. Median of (a) Number of capillaries/mm. (b) Deletions/mm (c) Giant capillaries/mm. (d) Enlarged capillaries/mm. (e) Ramified/mm (f) Number of microhemorrhages. Nailfold capillaroscopy images from the fourth right finger of a representative patient showing late scleroderma pattern before (g) and an increase in the number of capillary loops at 12 months after (h) transplantation *p < 0.05, **p < 0.01.
Analyses of vascular biomarkers in skin biopsies post-transplantation
In skin biopsies, there was a decrease in the percentage of marked area of E-selectin from median (IQR) 0.78 (0.12–2.75) at baseline to 0.04 (0.0–0.33) post-AHSCT, (p < 0.0001) and Ang1 from 1.52 (0.33–6.53) at baseline to 0.11 (0.01–0.45) post-AHSCT, (p < 0.001); (Figure 2(c) and (e) and Supplemental Table S1). No difference in the expression of CD31 (p = 0.1893), VE-cadherin (p = 0.7676), Endothelin-1 (p = 0.9578), Ang2 (p = 0.2235), Tie2 (p = 0.6710), VEGFA (p = 0.5477), and VEGFR2 (p = 0.1309) were observed after AHSCT (Figure 2(a), (b), (d), (f)–(h), (i) and Supplemental Table S1). Figure 3 shows immunostaining images, before and after AHSCT, for E-selectin (Figure 3(a) and (b)) and Ang1 (Figure 3(c) and (d)). There was a positive correlation between expression of E-selectin in skin biopsies and mRSS (r = 0.5024; p < 0.001) (Figure 4). No other significant correlations were detected between mRSS and histological markers.

Analyses of vascular biomarkers in skin biopsies post-transplantation. Expression of markers (a) CD31, (b) VE-cadherin, (c) E-selectin, (d) Endothelin-1, (e) Ang1, (f) Ang2, (g) Tie2, (h) VEGFA, (i) VEGFR2 in skin biopsies of patients with SSc. Highly significant differences were found for E-selectin after AHSCT compared to baseline (p < 0.0001). Ang1 was decreased after AHSCT (p < 0.001). Each symbol indicates a SSc patient. Error bars indicate the median (interquartile range). ***p < 0.001 and ****p < 0.0001.

Skin biopsy immunostaining before and after transplantation in systemic sclerosis patients. All photographs were taken under 20× magnification. (a) anti-E-selectin staining before and (b) 12 months post-transplantation. (c) anti-Ang1 staining before and (d) 12 months post-transplantation.

Endothelial activation in dermal capillaries correlates with modified Rodnan’s score. The percentage of marked area of E-selectin immunostaining positively correlates with the clinical score for skin involvement (mRSS).
Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy
At baseline, all vascular markers evaluated in the serum were significantly higher in SSc patients when compared to healthy controls (p < 0.05), except for ICAM-1 (p > 0.05); (Figure 5(m) and Supplemental Table S2). At all post-transplantation time points, serum levels of IL-6, vWF-A2, CXCL8, VEGF-A, E-selectin, P-selectin, VCAM-1 and Endothelin-1 remained higher than healthy controls (p < 0.05); (Figure 5(a)–(c), (e), (g), (i)–(k) and Supplemental Table S2). When we compared levels of each serum marker at different time points, before and after transplantation, there was a decrease of vWF-A2 levels at 12 months (p < 0.05), followed by subsequent increase at 30 months (p < 0.01); (Figure 5(b) and Supplemental Table S3), a decrease of Pentraxin-3 levels at 30 months (p < 0.05); (Figure 5(f) and Supplemental Table S3), an increase of Thrombomodulin-1 at 6 months (p < 0.01), decreasing at 30 months (p < 0.05); (Figure 5(h) and Supplemental Table S3). IL-18 increased from 6 to 24 months (p < 0.01) and 36 months (p < 0.05); (Figure 5(l)).

Assessment of serum vascular biomarkers post-transplantation. Mean levels (± SD) of serum cytokines (a) IL-6, (b) vWF-A2, (c) CXCL8, (d) EGF, (e) VEGF-A, (f) Pentraxin-3, (g) E-selectin, (h) Thrombomdulin, (i) P-selectin, (j) VCAM-1 (k) Endothelin-1, (l) IL-18, (m) ICAM-1 in SSc patients before and after AHSCT were assessed by Magnetic Luminex Assay. For all biomarkers, n = 10 controls, n = 22 at pre and 12 months, n = 17 at 6 and 24 months, n = 20 at 18 months, n = 8 at 30 months and n = 10 at 36 months. *p < 0.05; **p < 0.01 ***p < 0.001 ****p < 0.0001 . Error bars indicate mean (standard error of the mean).
Discussion
The goal of this study was to assess the impact of AHSCT on vascular aspects associated with SSc. We detected improvements in microvascular morphology followed by a decrease in the degree of endothelial activation in the histopathological analysis of the dermis. However, the serum markers of endothelial activation and injury remained at high levels on long-term follow-up after AHSCT, indicating persistence of vascular damage. These findings are in accordance with our clinical vascular outcomes, as the frequency of Raynaud’s phenomenon, telangiectasias and digital ulcers remained mostly unaffected after AHSCT.
The patients included in this study had diffuse SSc and most presented gastrointestinal and pulmonary involvement, and positive anti-SCl70 antibodies, characterizing a severe disease profile.28,29 Capillaroscopy evaluations showed that after the transplantation there was vascular remodeling with an increase in the number of capillary loops and a reduction in the number of dilated capillaries (giant capillaries). A previous study has shown that AHSCT induces a change from late to active capillaroscopy pattern, while no modification was detected in conventionally-treated patients. 12 A similar trend was seen in our patients, also suggesting a shift from late toward early/active pattern. These results indicate a positive effect of AHSCT on vascular remodeling, improving microvessel architecture.
In order to more comprehensively evaluate the vascular status of our patients after AHSCT, we included histopathological and serum analyses. On dermal biopsies, we did not observe changes in the expression of adherens junction proteins (CD31 and VE-cadherin) at 12 months post-transplantation, when compared to baseline, in accordance with other studies.10,11 We conjecture that patients were transplanted at advanced stages of microvascular involvement, precluding the reconstitution of junctional proteins. Nevertheless, we were able to detect changes in the capillaroscopy pattern, possibly reflecting a transitional stage of vascular remodeling. Future studies, with longer follow-up and evaluations at later time points after AHSCT should answer these questions.
Unlike other adhesion molecules, E-selectin is only expressed in endothelial cells activated by inflammatory cytokines. 30 In our patients, we observed that expression of E-selectin decreased in the dermis after transplantation. Furthermore, we also found a positive correlation between E-selectin expression on the skin and mRSS values. We consider this correlation interesting because vascular injury and immune dysregulation in SSc lead to chronic tissue injury resulting in fibrosis. 31 Serum E-selectin levels have been previously associated with clinical manifestations of pulmonary fibrosis32,33 and decreased DLCO and FVC values, 34 suggesting an association with fibrotic mechanisms. E-selectin levels are also high in patients with late-scleroderma pattern in capillaroscopy. 35 It is therefore possible that the decline in E-selectin levels may contribute to improvements in fibrosis.
In addition to local damage, we assessed circulating markers of vascular injury.
Most serum markers were high at baseline, when compared to healthy controls, none returning to normal levels until at least 3 years after AHSCT. These data denote how the endothelial environment is compromised in SSc patients when compared to healthy individuals, with an active endothelium status, chronic overexpression of pro-angiogenic and vasoconstriction factors, and platelet activation.
Serum levels of inflammatory cytokines, such as IL-18 and IL-6, and levels of cell adhesion molecules, P-selectin, ICAM-1, and E-selectin, involved in leukocyte recruitment, remained elevated, not returning to healthy control levels after transplantation. Previous studies have also detected elevated serum levels of adhesion molecules and cytokines associated with inflammation in SSc patients.36,37 Collectively, these data indicate that systemic inflammatory stimuli persist after AHSCT. In the short term, we believe that transplant-related events may contribute, such as toxicity of the conditioning regimen, infections and transfusions of blood products. On a long follow-up of 36 months, however, we must acknowledge that pathogenic mechanisms associated with SSc, leading to endothelial damage, possibly persist after AHSCT.
Circulating levels of angiogenic factors, VEGF, VEGFA and CXCL8 were high at baseline, when compared to healthy controls and did not change post-transplant, suggesting persistence of mechanisms associated with aberrant angiogenic expression. It is still to be investigated if these markers remain elevated due to the persistent endothelial injury or if there is a baseline disorder that perpetuates their exaggerated expression. Future molecular investigations may contribute to unveil the exact mechanisms.
In SSc-associated vasculopathy there is also dysregulation of the vascular tone due to high levels of vasoconstrictor factors. 38 Serum levels of ET-1 remain increased at all time points after transplantation. This indicates persistence of the pathological vasoconstrictor profile after treatment, thus not achieving a protective vasodilator state. Similarly, thrombomodulin and vWF were increased in the serum of our SSc patients before AHSCT, compared to controls. After transplantation, the levels remained high, with significant reduction, albeit non-sustained, at early time points. Thrombomodulin and vWF have critical roles in endothelial hemostasis, involved in platelet aggregation and inflammation, further contributing to vasculopathy and fibrosis.39,40
This study is limited by its retrospective design and small sample size. Only 27 of the 39 patients initially evaluated had available samples for analysis. For the immunohistochemistry data, we cannot exclude the possibility of a type 2 statistical error, due to the small number of samples and high variability of results. In addition, the long-term assessment was limited to serum biomarkers, as skin biopsies and capillaroscopic analyses were only available in the first year after transplantation. Despite these limitations, we were able to include a large number of biomarkers and standardize sample collection times and areas for collection of skin biopsies. In addition, except for one transplant-related death, the excluded patients did not present a different clinical profile from those included (data not shown), indicating that the evaluated subpopulation represents the total group of transplanted patients. Collectively, our results show that AHSCT improves small vessel morphology even in short follow-up of 1 year, reflecting beneficial effects of AHSCT on SSc-associated vasculopathy. We were also able to show that, as recovery of the vascular damage was not fully achieved after AHSCT, there may still be room for further improvements, to be developed in future studies.
In conclusion, we demonstrate that autologous transplantation to some extent promotes vascular remodeling and improves local dermis inflammation. It is possible that AHSCT has distinct therapeutic effects on different aspects of vascular disease, which would explain a few of the contrasting results observed between systemic and local evaluations. While the cutaneous niche may benefit the most, systemic inflammation may perpetuate endothelial activation. Future studies are necessary to clarify transplant-associated mechanisms on SSc vasculopathy and to guide additional therapies aiming to improve patient outcomes.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221084845 – Supplemental material for Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy
Supplemental material, sj-docx-1-tab-10.1177_1759720X221084845 for Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy by Maynara Santana-Gonçalves, Djúlio Zanin-Silva, Álvaro Henrique-Neto, Daniela A. Moraes, Marianna Y. Kawashima- Vasconcelos, João R. Lima-Júnior, Juliana B. E. Dias, Vinícius Bragagnollo, Júlia T. C. de Azevedo, Dimas T. Covas, Kelen C. R. Malmegrim, Leandra Ramalho and Maria Carolina Oliveira in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X221084845 – Supplemental material for Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy
Supplemental material, sj-docx-2-tab-10.1177_1759720X221084845 for Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy by Maynara Santana-Gonçalves, Djúlio Zanin-Silva, Álvaro Henrique-Neto, Daniela A. Moraes, Marianna Y. Kawashima- Vasconcelos, João R. Lima-Júnior, Juliana B. E. Dias, Vinícius Bragagnollo, Júlia T. C. de Azevedo, Dimas T. Covas, Kelen C. R. Malmegrim, Leandra Ramalho and Maria Carolina Oliveira in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-3-tab-10.1177_1759720X221084845 – Supplemental material for Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy
Supplemental material, sj-docx-3-tab-10.1177_1759720X221084845 for Autologous hematopoietic stem cell transplantation modifies specific aspects of systemic sclerosis-related microvasculopathy by Maynara Santana-Gonçalves, Djúlio Zanin-Silva, Álvaro Henrique-Neto, Daniela A. Moraes, Marianna Y. Kawashima- Vasconcelos, João R. Lima-Júnior, Juliana B. E. Dias, Vinícius Bragagnollo, Júlia T. C. de Azevedo, Dimas T. Covas, Kelen C. R. Malmegrim, Leandra Ramalho and Maria Carolina Oliveira in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors thank the contributors of the Cellular and Molecular Pathology Laboratory, Department of Pathology and Forensic Medicine, University of São Paulo, especially Ms. Deisy Mara da Silva, for the histological processing. We also thank the staff from the Hemotherapy and Regional Blood Center and from the Bone Marrow Unit, University Hospital of the Ribeirão Preto Medical School. Finally, we thank Lucas Santana for his contribution in editing the images.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the São Paulo Research Foundation, Center for Cell-based Therapy, FAPESP. Nº [n°2013/08135-2]; by the Higher Education Personnel Improvement Coordination (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES), finance code 001, [nº 88882.328420/2019-01]; by the Brazilian Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq) [nº 134198/2018-2.] and by the Fundação de Apoio ao Ensino, Pesquisa e Assistência do Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto (FAEPA).
Data availability statement
Data cannot be shared for ethical/privacy reasons.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
