Abstract
Introduction:
Viscosupplementation with hyaluronic acid (HA) is indicated for non-responders to non-pharmacological therapy, to analgesics or when non-steroidal anti-inflammatory drugs (NSAIDs) are contraindicated. The aim of this study is to compare the efficacy, safety and costs of three different HA treatments (Sinovial® Forte, sinovial one and hyalgan).
Patients and methods:
Ninety patients with grade I/II Kellgren–Lawrence knee osteoarthritis were included in three groups, the first was treated with hyalgan (weekly for 5 weeks), the second with Sinovial® Forte (weekly for 3 weeks) and the third group with a single injection of sinovial one.
Results:
All three treatments were effective, with an average reduction in the Western Ontario and McMaster Universities osteoarthritis index (WOMAC) score of 18.9 points for hyalgan, 18.04 points for Sinovial® Forte and 17.92 points for sinovial one. The comparison of the three groups did not show any statistical difference in terms of efficacy. National health system (NHS) and social costs are, respectively, €419.12 and €853.43 for hyalgan, €338.64 and €599.22 for Sinovial® Forte, €221.56 and €308.42 for sinovial one.
Conclusion:
All three treatments were equally effective with no statistically significant differences; thus, the treatment with sinovial one may be considered as clinically effective as the other two regimens, but with a very efficient cost profile in early symptomatic knee osteoarthritis.
Introduction
Osteoarthritis is the most common joint disease which affects the cartilage involving the whole joint, including the subchondral bone, ligaments, capsule, synovial membrane and the periarticular muscles. The disease leads to cartilage degeneration with fibrillation, fissures, ulcerations and full thickness loss of the joint surface.1,2 The age, the sedentary lifestyle and the increasing trend of overweight and obesity are strongly related with increasing osteoarthritis prevalence. 3 It is expected that the burden of osteoarthritis on the healthcare system and overall economy will continue to increase in the near future. 4 The knee is one of the most frequently affected joints associated with activity limitations, the need for walking devices and increased use of analgesic and non-steroidal anti-inflammatory drugs (NSAIDs). Moreover, symptomatic knee osteoarthritis is one of the leading causes of disability. 5
Hyaluronic acid (HA) is a natural glycosaminoglycan and a component of the synovial fluid and cartilage matrix that is synthesized by synovial cells, fibroblasts and chondrocytes and is secreted into the joint.
HA has a good safety profile characterized by no known drug interactions, limited local side effects and nearly absent systemic effects.6–8 In fact, when HA is compared to NSAIDs, burdened by the risk of gastrointestinal, renal and cardiovascular side effects, it results in a very safe treatment of knee osteoarthritis.
Viscosupplementation with HA is indicated for non-responders to non-pharmacological therapy, to analgesics (including acetaminophen) or when NSAIDs are contraindicated.9,10 There are now many formulations of HA, with treatment regimens that vary from single injection to the most common 3-weekly or 5-weekly courses every 6 months. Moreover, the choice of the HA formulation is wider, from low molecular weight (range 500,000–730,000 Dalton) to intermediate (range 800,000–1,200,000) and high molecular weight (>1,500,000 Dalton) up to the cross-linked and polymerized with very high molecular weight (average 6,000,000 Dalton).
Despite the efficacy of viscosupplementation being well demonstrated in most of the trials and reviews,6–8,11 there is a considerable heterogeneity in methodology employed in trials, with strong discrepancy between the inclusion and exclusion criteria, the severity of knee osteoarthritis, the HA molecular weight and treatment schedule. These problems make the comparison of data derived from different studies difficult. The fact that most of the published studies (63%) are industry funded and that in none of these was an unfavorable conclusion about the efficacy of HA reported, may suggest a potential conflict of interest and/or a potential low objectivity of the data presented. 12 A meta-analysis has shown that the high and ultra-high molecular weight HAs are not superior to lower molecular weight formulations; 13 however, the only advantage of high/ultra-high molecular weight HA seems to be a better control of pain over time compared to corticosteroids. 14
The aim of this real life retrospective observational study in a knee osteoarthritis population was to compare the efficacy and safety of three different HA intra-articular formulations (low and intermediate molecular weight concentrations) over a period of 6 months. Furthermore, a cost-effectiveness analysis with direct and indirect costs for each treatment protocol was also performed.
Materials and methods
Patients
Out of 200 knee osteoarthritis patients, the charts of 90 adult patients with knee osteoarthritis grade I/II according to Kellgren–Lawrence were selected and included in the study. All patients gave informed consent to the intra-articular injection; ethics board approval was not required for a labelled procedure in accordance with the policy of our institution. Patient exclusion criteria were intra-articular injection of any HA or corticosteroids in the previous 6 months, previous knee surgery, treatment with sysadoas initiated within the past 3 months, active skin disease or infection at the injection site; systemic inflammatory conditions, autoimmune diseases, connective tissue diseases, bleeding diathesis or use of anticoagulants (except aspirin <325 mg/day); and any other medical condition that might make the patient unsuitable for the retrospective evaluation in this study or the absence of the longitudinal clinical data in the charts (Figure 1).

Patient selection and inclusion/exclusion criteria.
Treatments
After selection, the 90 charts of the patients were divided into three groups. The first group was treated with a weekly injection of hyalgan for 5 weeks (Fidia Farmaceutici SpA, Italy), a sodium hyaluronate derived from rooster combs with a concentration of 10 mg/ml in 2 ml. The second group was treated with a weekly injection of Sinovial®Forte 1.6% for 3 weeks (IBSA Group, Switzerland, Switzerland), a fermentative derived HA with a concentration of 32 mg/2 ml. The third group was treated with a single injection of sinovial one 2.0% (IBSA Group, Switzerland), a fermentative derived HA with a concentration of 50 mg/2.5 ml.
Assessment and safety
Patients were routinely assessed by the physician who also assisted them to fulfill the Western Ontario and McMaster Universities osteoarthritis index (WOMAC) score 15 at baseline (T0) and after 6 months (T2). Adverse events (AEs) were recorded with particular attention to local pain reaction at injection site or post-injection reactions such as effusion or pseudoseptic arthritis.
Cost-efficacy analyses
The determination of the direct health costs was performed according to the data present in the charts and included baseline evaluation (rheumatology clinic visit, X-ray of the knee, articular ultrasound, laboratory analysis), injection of the product to the dosage indicated in the data sheet and a follow-up evaluation of 6 months after the first injection. Unit costs of the activities listed above are shown in Table 1. The costs have been estimated on the basis of national tariff nomenclature of outpatient services (Nomenclature 2012, D. MdS 18 October 2012). The direct costs of treatment were calculated at 6 months of treatment and include the cost of follow-up. The cost of drugs is related to the price indicated on the packaging containing the same number of injections as indicated in the data sheet. The evaluation of non-medical direct costs included the costs of transport for the patient (assuming 30 km as the average distance and €0.29/km as reported by the Italian Automobile Club, March 2016). For the calculation of indirect costs, using the approach of human capital and assuming that each patient loses half a day for the treatment administration and does not need a care giver, we calculated that the average value of the hourly productivity loss amounted to €138.64 (average revenue per working day in 2014, Bank of Italy 2015).
Unit costs.

Statistical analyses
The Shapiro–Wilk test was performed to assess the normality of the distribution of each continuous variable and the folded F-test was used to evaluate the equality of the variance of the differences between T0 and T1. In order to evaluate the efficacy (difference between T0 and T1 in the WOMAC scale) the difference between the treatment protocols was used in the T test.
Results
Out of 200 evaluated charts, 90 patients were selected after applying the exclusion criteria: three patients for previous knee surgery, 29 for previous HA injection, 23 for <325 mg/day, one for loss to follow-up previous corticosteroid injection, 10 for sysadoas initiated within the past 3 months, five for the use of anticoagulants (except aspirin).
The unit costs of treatment and the patients’ baseline characteristics are presented in Tables 1 and 2, respectively. Gender, mean age and Kellgren–Lawrence knee osteoarthritis grade were equally distributed between the groups. The baseline mean WOMAC score was 51 ± 2.566 in the first group (hyalgan), 50 ± 2.242 in the second group (Sinovial® Forte) and 51 ± 2.706 in the third group (sinovial one). All three treatments were effective, with an average reduction in the WOMAC score of 18.9 points (37.5%) for hyalgan, 18.04 points (36.08%) for Sinovial® Forte and 17.92 points (35.14%) for synovial one.
Patients’ baseline characteristics.

WOMAC, Western Ontario and McMaster Universities osteoarthritis index.
The comparison of the three groups did not show any statistical difference in terms of efficacy (mean reduction in the WOMAC scale) (hyalgan versus Sinovial® Forte p = 0.22; hyalgan versus sinovial one p = 0.17; Sinovial® Forte versus sinovial one p = 0.88). The results are shown in Table 3.
Results.
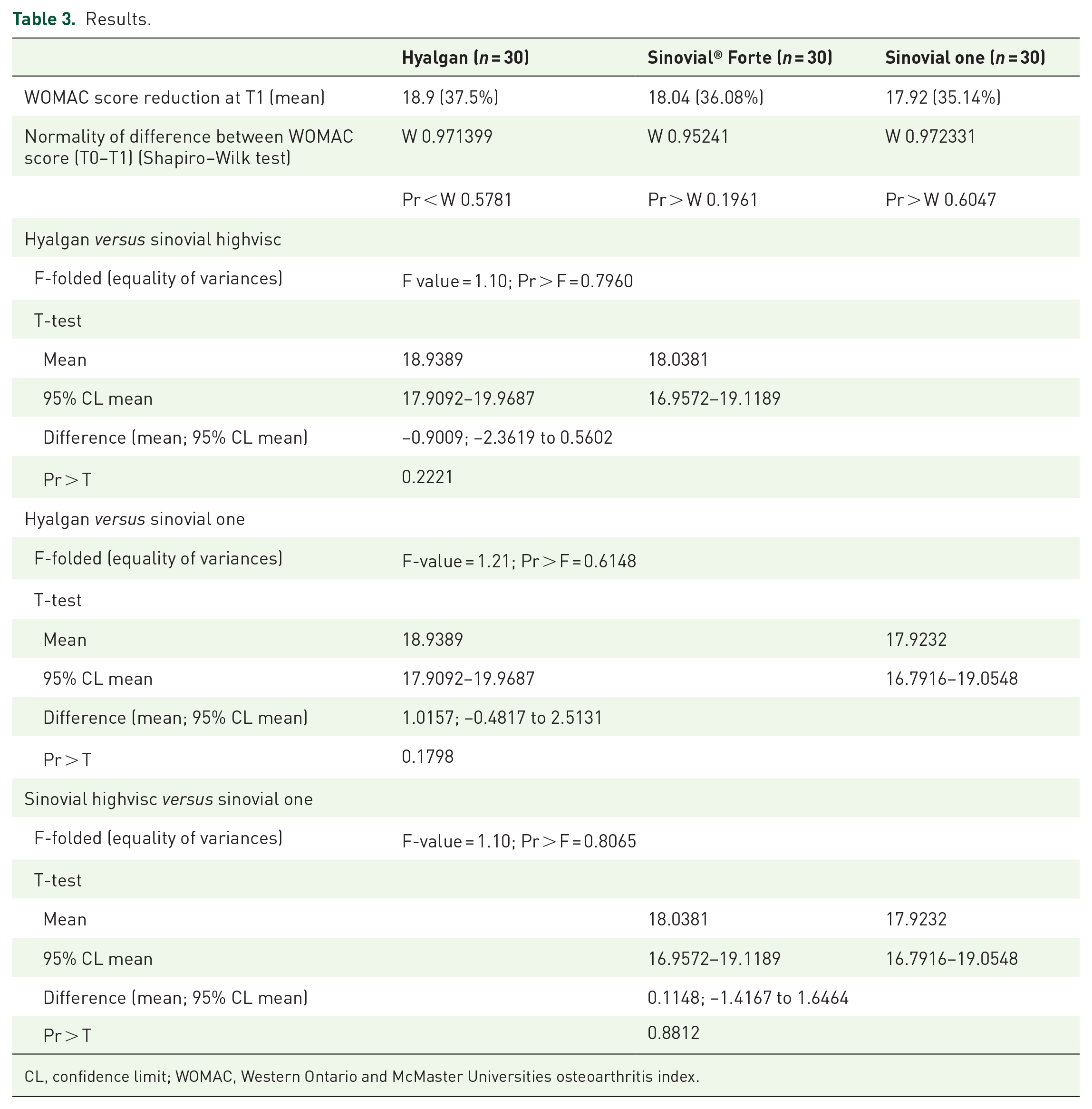
CL, confidence limit; WOMAC, Western Ontario and McMaster Universities osteoarthritis index.
No major AEs were observed; only five patients reported local pain in the injection site (two in the hyalgan group, two in the synovial highvisc group and one in the synovial one group) successfully treated with local ice application. None of them needed pain killers or NSAIDs.
At 6 months, direct costs of treatment amounted to €19.12 for group 1 (hyalgan), €338.64 for group 2 (Sinovial® Forte), and €221.56 for group 3 (synovial one 2.0%). Non-medical direct costs were €87.72 for group 1 (hyalgan), €52.63 for group 2 (Sinovial® Forte), and €17.54 for group 3 (synovial one 2.0%) (Table 4). The estimated indirect costs were €346.59 for group 1 (hyalgan), €207.95 for group 2 (Sinovial® Forte), and €69.32 for group 3 (synovial one 2.0%) (Table 3). For the national health system (NHS), the treatment of patients with sinovial one 2.0% seems the most convenient, with a cost of €221.56, lower by 34.57% when compared to Sinovial® Forte (€338.64) and by 47.14% when compared to hyalgan (€419.12). From the societal perspective, sinovial one 2.0% remains the main treatment at a total cost of €308.42, lower by 48.53% compared to Sinovial® Forte (€599.22) and by 63.86% compared to hyalgan (€853.43) (Table 4). Finally, for the health system every point of reduction in the WOMAC score corresponds to a cost of €22.18 for hyalgan, €19.87 for Sinovial® Forte and €12.36 for sinovial one 2.0%. For what concerns the social perspective the cost is estimated at €45.15 for hyalgan, €35.17 for Sinovial® Forte and €17.21 for sinovial one 2.0% (Table 4).
Cost-effectiveness analysis.
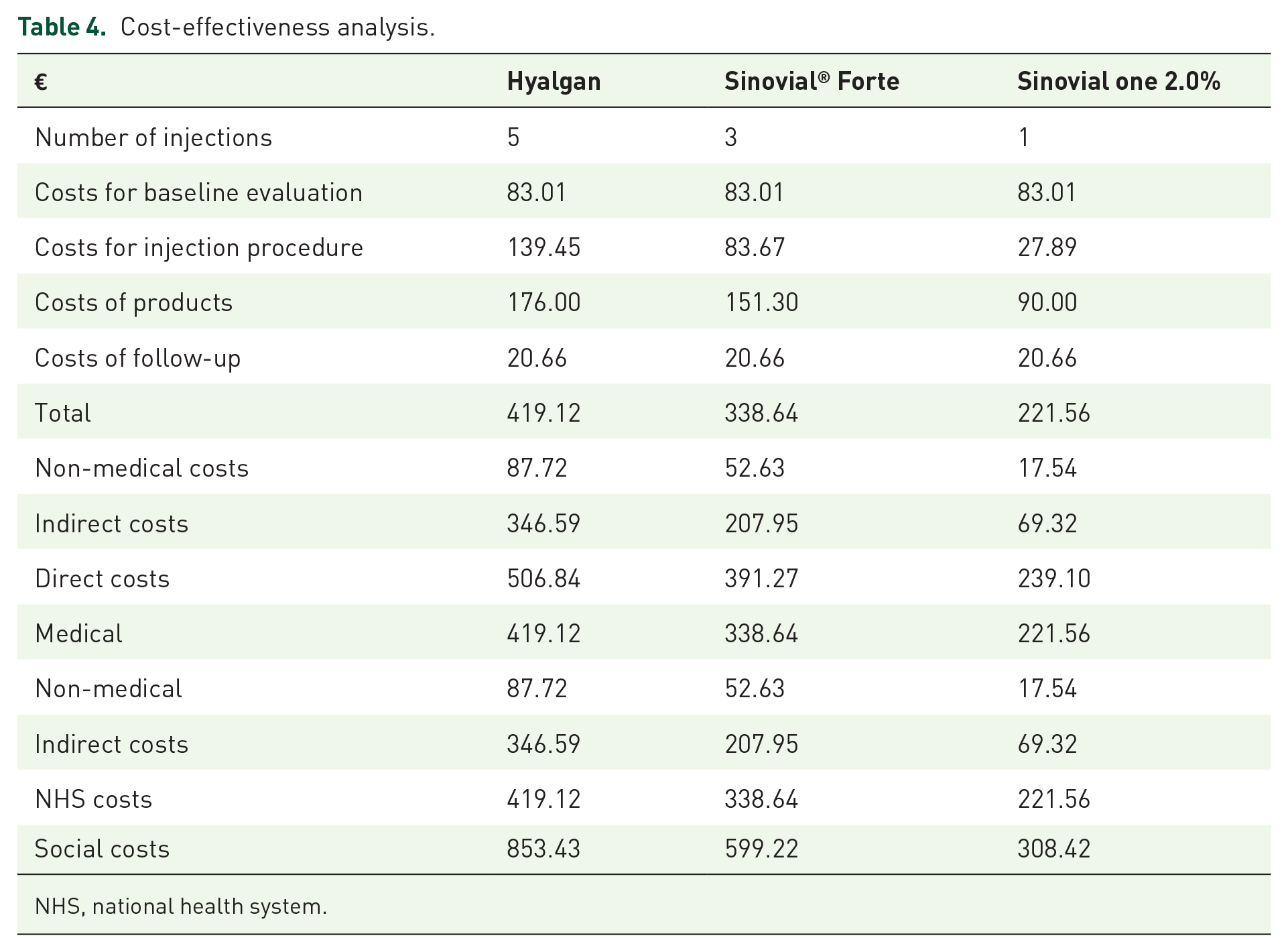
NHS, national health system.
Discussion
Viscosupplementation with HA is a well-established treatment for symptomatic knee osteoarthritis. Although there are several studies in the literature reporting results on the efficacy of HA in knee osteoarthritis, comparative studies on different HA formulations are scarce. In particular, there is little evidence on alternative treatment protocols.
When efficacy is analyzed, our retrospective data demonstrate the non-superiority one on each other of the three different intra-articular regimens with low to intermediate molecular weight HA. In fact, they were equally effective in early symptomatic knee osteoarthritis as there are no statistically significant differences among the three treatment protocols tested. The WOMAC score detected in the three regimens after 6 months was on average similar, thus testifying to the efficacy of all three HAs used in early knee osteoarthtis. These results are in agreement with the previous studies published in the literature.
In the past decade, the use of HA in the treatment of osteoarthritis has been questioned several times. In 2013, the American Academy of Orthopaedic Surgeons (AAOS) 2013 guidelines for knee osteoarthritis did not recommend the use of HA. 16 This decision has been criticized, 17 in particular because there is evidence showing positive results characterized by a reduction of pain and recovery of the function, thus supporting the use of HA in knee osteoarthritis. 18 However, it is also true that the use of HA is fraught with heterogeneous trials with conflicting conclusions. 18 Probably, this problem has recently prompted the American College of Rheumatology (ACR) to recommend against the use of HA, suggesting instead the use of non-steroidal inflammatory drugs. 19 In osteoarthritis, the use of HA is characterized by rare AEs and long-term safety. In knee osteoarthritis HA may allow the reduction as much as possible of the use of NSAIDs and their side effects (cardiovascular, gastrointestinal, renal etc.), in particular in elderly patients.
The interest in HA is also due to the cost-effectiveness profile. In our patients, it should be observed that the assessment comes down to a cost-minimization analysis because no significative differences among the treatments have been highlighted both in terms of effectiveness and safety. The assessments were conducted assuming both the perspective of the NHS and of the society. Direct healthcare costs taken into consideration were those related to the pharmacological treatment and its administration. Adopting the society perspective, non-medical direct costs (essentially travel expenses for reaching the healthcare facility) and the patient’s loss of productivity due to treatment (indirect costs) were then added. Regarding the quantification of direct healthcare costs, in the Italian clinical practice the injection procedure is the same for all the treatments and calls for the following diagnostic tests prior to beginning the cycle of treatment: visit with a rheumatologist, knee X-ray, ultrasound of the articulations, lab tests (blood count, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), creatinine, transaminases, protidogram and RA test); subsequently the product is injected according to the dosage indicated in the data sheet, with a follow-up at 6 months. Although the efficacy data are similar for the three different treatment regimens, we believe that this evidence may have a pivotal significance in the management of knee osteoarthritis. In fact, the data support the choice of a treatment protocol tailored not only on the evidence of efficacy, but also on the expectations and social needs of the patient as well as on his compliance with treatment. Moreover, therapies for early knee osteoarthritis are generally long lasting so it is crucial that they are effective, safe, well accepted by the patient, easily accessible and finally at the lowest cost to the healthcare system. Therefore, the treatment with a single injection of a fermentative derived HA with a concentration of 50 mg/2.5 ml (sinovial one 2.0%) may be considered as clinically effective as the other two regimens but with a very efficient cost profile which may suggest its use in everyday practice.
In conclusion, the treatment with sinovial one 2.0% is useful both in the NHS and societal perspectives and envisages an efficient use of resources. This approach may in fact not only reduce the costs for the health system but also contain the indirect cost to which the patients and their families are usually submitted when this regimen is chosen to fight early knee osteoarthritis. It should also be remarked that sinovial one 2.0%, in addition to being the cheapest alternative, through the administration of only one injection per cycle of treatment represents a further benefit in terms of patients’ quality of life. Further works are needed to confirm these preliminary results.
Key messages
• Viscosupplementation is a safe and well-established treatment for symptomatic knee osteoarthritis.
• Early osteoarthritis treatments are long lasting so it is crucial that they are at the lowest costs.
• Comparison of treatment protocols and alternative regimens are welcome.
