Abstract
Introduction:
Rheumatic and musculoskeletal diseases (RMDs) have a significant impact on patients’ health-related quality of life (HRQoL) exacerbating disability, reducing independence and work capacity, among others. Predictors’ identification affecting HRQoL could help to place efforts that minimize the deleterious impact of these conditions on patients’ wellbeing. This study evaluates the influence of demographic and clinical predictors on the HRQoL of a cohort of RMD patients, measured using the Rosser classification index (RCI).
Methods:
We included patients attending the Hospital Clínico San Carlos (HCSC) rheumatology outpatient clinic from 1 April 2007 to 30 November 2017. The primary outcome was the HRQoL assessed in each of the patient’s visits using the RCI. Demographic and clinical variables extracted from a departmental electronic health record (EHR) were used as predictors: RMD diagnoses, treatments, comorbidities, and averaged HRQoL values from previous periods (for this last variable, values were imputed if no information was available). Association between predictors and HRQoL was analyzed using penalized generalized estimating equations (PGEEs). To account for imputation bias, the PGEE model was repeated excluding averaged HRQoL predictors, and common predictors were considered.
Discussion:
A total of 18,187 outpatients with 95,960 visits were included. From 410 initial predictors, 19 were independently associated with patients’ HRQoL in both PGEE models. Chronic kidney disease (CKD), an episode of prescription of third level analgesics, monoarthritis, and fibromyalgia diagnoses were associated with worse HRQoL. Conversely, the prescription in the previous visit of acid-lowering medication, colchicine, and third level analgesics was associated with better HRQoL.
Conclusion:
We have identified several diagnoses, treatments, and comorbidities independently associated with HRQoL in a cohort of outpatients attending a rheumatology clinic.
Keywords
Introduction
Health-related quality of life (HRQoL) assessment is a useful and well-extended way to measure, compare, and evaluate the overall (i.e. physical, social and mental) wellbeing of patients who suffer from chronic conditions, including those with rheumatic and musculoskeletal diseases (RMDs).1–3 Hence, HRQoL is a measure of the patient’s perception of his/her health status that comprises both subjective and objective components. 4
According to the 2017 Global Burden of Disease report, 5 together with mental health disorders, RMDs were the leading cause of disability worldwide, affecting all age segments, and having the greatest negative impact on patients’ HRQoL compared to other chronic disorders.6,7 The high impact on disability caused by RMDs can be explained due to their high prevalence and incidence, affecting around 25% of the overall EU population. 8 Besides disability, other characteristics of RMDs such as chronicity, pain, fatigue, motion dysfunction, and multimorbidity 9 have a deleterious effect on the patients’ HRQoL.
Different generic questionnaires for measuring the HRQoL have been employed in patients with chronic diseases, 10 as well as in RMD patients,11,12 such as the Short Form 36, 13 EuroQol5D, 14 or the Rosser classification index (RCI). 15 Other RMD-specific questionnaires have also been proposed to enhance patients’ characterization for specific disorders.11,16
Besides the increasing use of HRQoL in both clinical and observational studies as endpoints; 17 its relevance in the elaboration of medical guidelines and recommendations, 18 and in the design of medical interventions; attempts to identify predictors of HRQoL have also been made for different RMDs, such as Behçet’s disease, 19 fibromyalgia, 20 gout, 21 juvenile idiopathic arthritis, 22 rheumatoid arthritis, 23 systemic lupus erythematosus, 24 and systemic sclerosis. 25
However, most of the studies focus on finding HRQoL predictors in patients suffering a particular rheumatic disease, underestimating that the coexistence of other RMDs can have a synergistic effect on the subject’s HRQoL. 3
Therefore, we aimed to identify factors associated with the HRQoL of RMD patients attending an outpatient rheumatology clinic from a tertiary care center by considering the coexistence of multiple RMDs, the prescription of different medications and the presence of comorbidities, all of them potentially impairing the patient’s HRQoL.
Patients and methods
Patients
The Hospital Clínico San Carlos Musculoskeletal Cohort (HCSC-MSKC) 26 is a routine clinical practice cohort that includes subjects seen at the rheumatology outpatient clinic of our center, whose clinical and management information is stored using a departmental electronic health record (EHR). The HCSC-MSKC contains information from more than 35,000 patients attending our clinic from 1 April 2007 until 30 November 2017.
In every patient’s visit, information is collected both codified and as free text. The former includes RMD diagnoses, prescribed drugs, HRQoL (using the RCI), and the patient’s follow-up plan. The latter contains the clinical notes, the comorbidity, and the medication prescribed by other physicians.
In the present study, we have selected patients and patients’ visits from the HCSC-MSKC based on the following inclusion criteria:
Patients’ visits had to be spaced ⩽365 days apart from the previous and/or the following visit. Hence, the elapsed time between the first and the second visit and between the last and the previous visit had to be ⩽365 days. Isolated visits (i.e. visits separated by more than a year from the previous and the next visit) were excluded.
Patients had to be seen at least twice in our outpatient clinic (and both visits had to fulfill the previous criteria).
Cognitive impairment or communication limitations were not exclusion criteria.
After applying these criteria, 18,187 patients and 95,960 visits were studied (Figure 1).
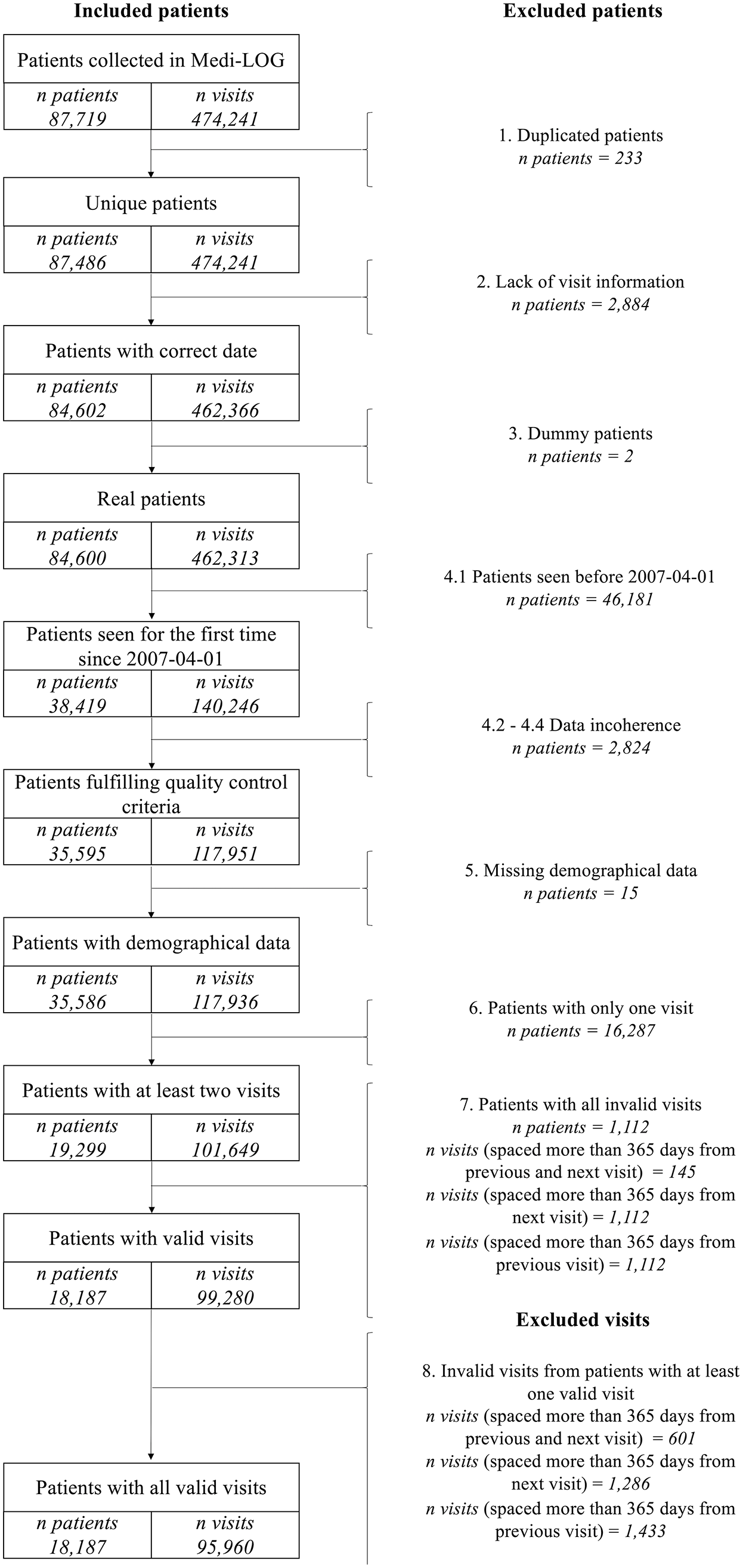
Flow chart of patients and visits included in the study after applying the inclusion and exclusion criteria.
Based on the 365 days cut-off point, we defined ‘episodes’ as the group of visits (at least two) separated by ⩽365 days among them (see Supplemental Figure 1).
The data were obtained during routine clinical practice with the informed consent of patients to be treated in a service that performs clinical care and research, hence there is not written informed consent. HCSC ethics review board approval was obtained as a retrospective study and waiver of informed consent was obtained for the use of de-identified clinical records. Furthermore, the study was conducted according to the Declaration of Helsinki.
Variables
Our primary outcome was the HRQoL, measured using the RCI, 27 and assessed in every patient’s visit. The RCI comprises two components, disability, and the distress levels (see Supplemental Table 1). The former covers the daily life activities of individuals (i.e. general mobility, self-care, usual activities, and social relationships) by considering eight ordinal levels ranging from no disability to unconsciousness. The latter is measured as emotional (i.e. mood) or physical (i.e. pain) distress and is defined as the worst measure between them. Distress has four different categories ranging from no distress/no pain to a high level of distress/pain. Up to 29 Rosser health states arouse by combining these two components. The key advantage of the RCI is its simplicity; in fact, a patient can be classified in less than 30 seconds, which overcomes the time-consumption drawback from other quality of life (QoL) measures.
As predictors, we included demographic and clinically related variables (diagnoses, treatments, comorbidities, and concomitant treatments) collected during the patients’ visits using our EHR.
Comorbidities and concomitant treatments included not only RMD-related variables, but also non-RMD variables. For instance, chronic kidney disease, neurological disease, asthma, anemia, psychiatric conditions, or hyperlipidemia are some examples of the comorbidities studied. Moreover, drugs such as insulin, tramadol, thyroid hormone, or surgery-related predictors such as thyroidectomy, appendicitis or articular prosthesis were also considered.
After a feature selection process (see Supplemental data ‘Predictors’), the number of categories related to diagnoses was 34, the number of categories related to treatments was 33, the number of comorbidities was 87, and the number of concomitant treatments was 34.
Based on our previous definition of episodes, we also considered ‘diagnoses and treatment episodes’. They were defined as consecutive follow-up periods (i.e. the end of one episode matches with the beginning of the next episode) for a particular diagnosis or treatment. These episodes begin in the visit when the patient receives that diagnosis or is prescribed with that treatment, and: (a) in the previous visit the patient had not received such diagnosis or treatment: in this case, the previous episode (if there was one) ends in the same visit as the new episode begins, or (b) the previous visit took place more than 365 days apart: in this case, the previous episode (if there was one) ends in that previous visit more than 365 days apart.
Based on the beginning and end of each episode, the duration from the first visit of the episode until each visit of the episode was calculated. In addition, the cumulative duration was calculated considering the total time that a patient has received a treatment or a diagnosis from the very first visit until each visit. For further details see Supplemental Table 2.
Episodes for each diagnosis and treatment were categorized based on their chronological order (1st episode, 2nd episode, 3rd episode, and so on). Furthermore, episodes with a reduced number of visits (<1% of 95,960) were grouped with those episodes taking place earlier to comply with the 1% criteria (see Supplemental Excel, DxT episodes).
For each visit, the patient’s RCI mean and median values of the previous visits in a period of 91, 182, 274, and 365 days were computed. If, for a particular visit, the patient had not attended our clinic in the time periods considered (or in the case of the first visit to our clinic); we assumed that their previous averaged HRQoL was the same as in the actual visit (we referred to this process as ‘imputation’).
A more detailed explanation can be found in Supplemental data ‘Quality of Life Imputation’ section and Supplemental Table 3.
Patients’ occupation or demographical variables such as age at the first visit or the total number of visits per patient were also considered.
Overall, 410 different variables were included in the analyses (see Supplemental data ‘Predictors’ section, Supplemental Excel file predictors and Supplemental Excel file categories: ‘Final diagnoses’ and ‘Final treatments’ sheet).
Statistical analyses
The association between demographic and clinical variables and repeated measures of HRQoL was analyzed using generalized estimating equations (GEEs) 28 and penalized generalized estimating equations (PGEEs). Three different working correlation structures were used in both the GEE and PGEE models: independence, exchangeable, and first-order autoregressive model AR(1). The variable selection process consisted of four sequential steps: GEE bivariable analyses, removal of highly correlated predictors, multivariate GEE analyses and PGEE analyses. For further details see Supplemental data ‘Statistical analyses’.
These steps were repeated for independence, exchangeable and AR(1) correlation structures. Based on the root mean square error (RMSE) values of the three different penalized models, the correlation structure with the lowest score was chosen.
Statistical analyses were performed using R statistical software version 3.3.2 29 and the ‘PGEE’ package. 30
Model explanation and interpretability
Clustering techniques were used to validate the results from the PGEE analyses, depending on the final selected predictors’ type, such as k-means in case all the predictors were continuous variables; 31 or the Kamila semi-parametric algorithm, in case a combination of continuous and categorical predictors were selected. 32 A summary of the variables selection and validation process can be found in Supplemental Figure 2.
Results
Patients’ description
After applying the selection criteria (see Figure 1), 18,187 patients with 95,960 observations (visits or contacts) were included. Table 1 and Supplemental Table 4 show the patients’ demographic and clinically related characteristics at the first valid visit.
Demographic and clinically related patient characteristics at the first valid visit, including the five most prevalent diagnoses and treatments given and prescribed by the rheumatologists, and comorbidities and concomitant treatments given by other physicians.

NSAIDs, non-steroidal anti-inflammatory drugs; Q1–Q3, first and third quartiles.
There were 16,139 (88.74%), 1825 (10.03%), 205 (1.13%) and 18 (0.1%) patients with up to one, two, three and four different follow-up episodes (i.e. group of visits spaced more than 365 days). From the 95,960 visits, 87,097 (90.76%), 8005 (8.34%), 803 (0.84%) and 55 (0.06%) belonged to the first, second, third and fourth follow-up episode, respectively. The percentage of imputed median and mean RCI values (excluding first visits) were 29.3% (91 day period), 10.7% (182 days), 4.7% (274 days) and 2.1% (365 days). All first visits (18.9%) were imputed due to the lack of previous health status values (see Supplemental Table 3).
The most prevalent diagnosis in the first valid visit is ‘no diagnoses’. This code is given when the precise diagnosis and/or the disease etiology is not clear and further tests such as laboratory or imaging are necessary to provide an accurate diagnosis. Considering all visits, 3594 patients (19.8%) received the ‘no diagnoses’ code at least once during their follow-up. Of them, 3115 patients (86.7%) receive this diagnosis in the first valid visit to our clinic. Furthermore, the ‘no diagnoses’ code is modified to a RMD diagnosis code in the next visit in 1560 patients (50.1%), and over the total course of the follow-up, in 2033 patients (65.3%). The most frequent RMD diagnoses given after a ‘no diagnoses’ code was back pain (9.5%), joint pain (8.0%), tendinitis (upper extremities: 7.8%), tendinitis (lower extremities: 6.4%), peripheral osteoarthritis (4.5%), knee osteoarthritis (4.5%), chronic polyarthritis (4.4%), peripheral neuropathy (4.1%), and rheumatoid arthritis (3.9%).
Model development
We present the results for the independent correlation structure, as it showed the lowest RMSE value (3.883, 4.033, and 3.902 for the independent, exchangeable, and AR(1) structures, respectively).
After carrying out the bivariate analyses, 292 predictors with a p-value < 0.15 were carried out to the next step. Afterwards, 49 predictors were removed due to their high pairwise correlation. From the remaining 243 predictors, 214 were combined into sets of clinically related terms (n = 66 different sets). After subsequent multivariate analyses, 125 predictors remained, and those without statistical significance were removed (n = 89). Predictors that could not be included in any set of clinically related terms were incorporated directly in the PGEE model (n = 28). Therefore, 153 predictors were included in the PGEE analysis; two of them were related to the patients’ averaged previous HRQoL before each particular visit. A full description of the removed predictors along these steps can be found in Figure 2 and in the Supplemental Excel file ‘Predictors selection independent’. In addition, in the Supplemental Excel files ‘Predictors selection exchangeable’ and ‘Predictors selection AR(1)’, the same process for the other two working correlation structures can be found.

Flow chart of the number of predictors during the selection process.
Two predictors related to the averaged HRQoL values from previous periods remained in the fourth step for the independent working correlation structure (mean RCI value over the last 91 days and median RCI value over the last year). Therefore, two grid searches for estimating the λ penalty tuning parameter were performed: one for the model that included the averaged previous HRQoL predictors and one for the model without those variables. Up to 29 different values were tested under k-fold =5; the minimum tested penalty value was 0.001, whereas the maximum penalty value was 1. The rest of the PGEE tuning parameters, such as the working correlation structure, the link and variance functions were defined as independence, Gaussian and identity, respectively. Additional details of the tested penalty values for the working correlation structures with and without averaged previous HRQoL predictors can be found in Supplemental Excel file ‘Penalty tuning parameter’ and in Supplemental Figures 3 and 4.
Selected predictors
The λ that yielded the smallest prediction error was λ = 0.04 for the model that included the averaged previous HRQoL predictors and 0.004 for the model without them.
For the first model, 53 predictors were selected after running the PGEE model and only 33 of them had statistical significance (p-value < 0.05). Table 2 and Supplemental Figures 5 and 6 show the demographic, diagnoses, treatments, comorbidities, and averaged previous HRQoL predictors negatively and positively associated with HRQoL.
Selected predictors after applying independent working correlation structure.

DM, demographic variable; DMARDs, disease-modifying anti-rheumatic drugs; Dx, diagnosis-related variable; DxE, diagnosis episode-related variable; HS, quality of life-related variable; NSAIDs, non-steroidal anti-inflammatory drugs; T, treatment variable; TE, treatment episode-related variable; XM, comorbidity variable.
For the model that did not include HRQoL-related predictors, 51 significant variables were finally selected. For this model, predictors negatively and positively associated with HRQoL can be found in Table 3 and Supplemental Figures 7 and 8.
Selected predictors after applying independent working correlation structure, without considering imputed HRQoL predictors (i.e. averaged and median RCI values).

DM, demographic variable; DMARDs, disease-modifying anti-rheumatic drugs; Dx, diagnosis-related variable; DxE, diagnosis episode-related variable; DxTFT, diagnosis total follow-up time; HS, quality of life-related variable; NSAIDs, non-steroidal anti-inflammatory drugs; ORL, otorhinolaryngology; T, treatment variable; TE, treatment episode-related variable; XM, comorbidity variable.
Table 4 shows the predictors shared between the two models. We observed that the effect’s direction was the same for both models, although its magnitude was lower when including the averaged previous HRQoL predictors. CKD, a first episode of third level analgesic treatment, a second episode of monoarthritis, and the diagnosis of fibromyalgia were the predictors with the negative effect on the HRQoL. Conversely, a first episode of anti-gout (or uric acid-lowering) medication and the prescription of third level analgesic treatment or colchicine in the previous visit showed the greatest positive effect on the HRQoL.
Shared predictors among the models with independent correlation structure.

DM, demographic variable; Dx, diagnosis-related variable; DxE, diagnosis episode-related variable; T, treatment variable; TE, treatment episode-related variable; XM, comorbidity variable.
Model validation
To validate our results, we performed clustering techniques using the PGEE models’ selected predictors as they were supposed to be closely related to the patients’ HRQoL. Clusters of patients with different mean RCI values were expected to be found. Because both models included continuous (such as the number of comorbidities) and categorical data (such as the presence or absence of a prescription or a diagnosis), the Kamila algorithm was employed for clustering. Clusters with four to six groups were calculated for both models.
For the model including averaged previous HRQoL predictors, observations from clusters were separated by their HRQoL, regardless of the number of groups. Conversely, for the model that did not include the HRQoL-related variables, observations were not split by the RCI value. A summary of the RCI median values by cluster in both models, depending on the number of clusters, can be found in Supplemental Table 5.
Discussion
We have studied the effect of demographic and clinically related variables in the HRQoL of patients with RMDs attending a rheumatology outpatient clinic. Models were built using PGEE, based on a set of predictors extracted from an EHR, which stores longitudinal patient data over up to 10 years.
After variable selection, 19 variables, including RMDs diagnoses, comorbidities, and treatments, showed a significant association with HRQoL, regardless of the inclusion of predictors related to the patient averaged HRQoL from previous visits.
The variable with the greatest negative influence on the HRQoL of patients affected with RMDs was the presence of CKD. This condition has previously been associated with lower HRQoL when compared with the general population.33,34 In our cohort, the presence of this comorbidity was significantly associated with the diagnoses of gout (17.4% versus 3.3% of patients, for those with and without CKD, respectively; p-value from χ2 test =8.4 × 10−40), which has previously been shown, 35 but also with the diagnoses of crystal arthropathies (8.2% versus 2.1%; p = 8.0 × 10−13), rheumatic polymyalgia (7.9% versus 3.6%; p = 6.9 × 10−5), and knee osteoarthritis (15.5% versus 1.4%; p = 0.03), among others (Supplemental Excel file CKD). Our results suggest that we should keep in mind the impact of this comorbidity in the care of patients with RMDs, as it could influence their HRQoL regardless of their diagnosis.
As for RMD diagnoses, monoarthritis had the greatest deleterious effect on HRQoL. To the best of our knowledge, no previous studies have analyzed the effect of this diagnosis on HRQoL. At least in our case, monoarthritis seems to be a temporary diagnosis, given that the etiology of this clinical presentation is still not clear: on the one hand, the proportion of visits in which this diagnosis was given diminished during follow-up (from 100% of first episods visits, to 62.9% in the second visits, 46.0% in the third, 37.4% in the fourth, 32.7% in the fifth, and 12.8% in the sixth and subsequent visits). Besides, only in 296 of the 1199 episodes in which monoarthritis was diagnosed (24.7%), no other RMD diagnosis was given. In the other episodes, other RMDs were also diagnosed, such as knee osteoarthritis (322 episodes, 26.9%), gout and crystal arthropathies (241 episodes, 20.1%), and lower limb tendinitis (88, 7.3%). However, we also observed differences when comparing the associated diagnoses given in the first versus the second and subsequent monoarthritis episodes: there was a decrease in the proportion of episodes when monoarthritis was the only diagnosis given (29.4% versus 7.1%), and an increase in the proportion of other diagnoses, such as knee osteoarthritis (24.4% versus 36.1%), or gout/crystal arthropathies (18.2% versus 27.4%). These changes in the patterns of diagnosis may contribute to the different HRQoL associated with monoarthritis episodes (Supplemental Excel file Monoarthritis).
On the other hand, fibromyalgia (see Table 2) has previously been associated with lower HRQoL compared with the general population,36,37 but also when compared with other RMDs, such as rheumatoid arthritis or spondyloarthritis. 38
Regarding treatments prescribed by the rheumatologists, we observed that uric acid-lowering medication was associated with a significant increase in HRQoL, once the patient received the first prescription (all subsequent episodes were combined due to the rule of 1%). We observed that in the 486 episodes of these medications, the mean (standard deviation) percentage of visits per episode in which uric acid-lowering drugs had been prescribed was 92.6% (18.7). Moreover, we observed that the higher the percentage the better the HRQoL in individual visits, even after adjusting for gender, age, diagnosis of gout in each visit, and prescription of uric acid-lowering drugs in the previous visit [coefficient 0.20 per 10% increase in the percentage of visits per episode in which uric acid-lowering drugs had been prescribed, 95% confidence interval (0.1–0.31), p = 2.0 × 10−4].
We have identified the following study limitations:
The misspecification of the working correlation structure can be an issue that could lead to a loss of efficiency in the estimation of the regression parameters. 39 Several approaches to optimize its selection have been developed.40–44 However, some authors suggest basing the working correlation structure decision on the mechanism in which the data are generated. 45 In our case, we decided to use the RMSE to base the correlation structure selection, meaning that we selected the fittest model for our data.
Regarding variable selection, we are aware that a bivariable selection step may introduce bias due to the lack of confounding control, which may result in the rejection or inclusion of inappropriate variables as studied by Sun et al. 46 In our study and due to the large number of predictors tested, we decided to increase the p-value threshold for deleting variables.
All predictors categorized as ‘treatments’ have a pharmacological component. Despite analyzing medical and surgical procedures (as comorbidities), we missed the inclusion of variables that measure the effect of non-pharmacological treatments, such as, physiotherapy 47 or occupational therapy, 48 which have been demonstrated to be associated with the HRQoL of RMD patients. The lack of these variables is explained by two factors: all patients received disease-specific exercise guidelines by their rheumatologist physician; and these variables are not codified in our EHR. However, non-pharmacological treatment data collected (e.g. assistive technology use) from a multidisciplinary team would potentially enrich the study.
Other disability-related predictors such as the patients’ degree of disability recognized by current legislation, and potential resources received according to that disability degree would be interesting to consider in future iterations.
Respecting the validation of our results using cluster analysis, only when including the variables related to the previous patient’s averaged HRQoL, could we discriminate groups of observations with different mean RCI values. These variables may introduce biases in the results as past values were imputed, as described in the Methods section: for the first patient’s visit since no previous health status information was available; and for those visits during follow-up with no previous data in the selected periods (91, 182, 274, and 365 days). Despite this issue, we were able to identify 19 predictors shared between the two models, see Table 4, that seem to influence the patients’ HRQoL, regardless of the presence or absence of averaged past RCI values.
Concerning the study strengths, we want to point out that GEE has been widely used in different fields, including rheumatology, 49 as these models allow us to take into account both the inter-subject and intra-subject variability, its robustness when the primary outcome variable (RCI) does not have a Gaussian distribution and when varying the working correlation structure, or its ability to use all of the available information even for unequal numbers of repeated measurements and unequal time intervals. 50 Besides, we have used a recent implementation of PGEE in R,30,51 addressing the analysis of longitudinal data with high-dimensional covariates. 52 This method can perform simultaneous estimation and variable selection by specifying a tuning penalty parameter, λ, which regulates the model complexity by shrinking the coefficients’ estimates to zero. A few studies have recently employed this package to study the working correlation structure selection 53 and the separation 54 issues in GEE.
In this study we tried to assess predictors governing the HRQoL of patients who attended an outpatient rheumatology setting, instead of finding predictors associated with concrete RMDs. In this scenario, in which the rheumatic population may be highly heterogeneous, we found that predictors which have a major impact on HRQoL are not diagnoses themselves – excepting fibromyalgia – but treatment requirement and comorbidities predictors. To the best of our knowledge this is the first study attempting to describe – in an outpatient setting – factors affecting the HRQoL of RMD patients. We believe that these results could be helpful for other rheumatic consultations because they reveal novelty factors that may determine the wellbeing of the patient.
Conclusion
In conclusion, we have shown several variables affecting the HRQoL of patients with RMDs attending a tertiary rheumatology outpatient clinic. The predictors with the greatest negative influence were the presence of CKD, monoarthritis, and fibromyalgia. Conversely, the use of uric acid-lowering drugs, third level analgesics and colchicine was associated with the greatest positive influence on HRQoL.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-docx-1-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-10-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-10-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-2-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-2-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-3-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-3-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-4-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-4-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-5-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-5-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-6-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-6-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-7-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-7-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-8-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-8-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xls-9-tab-10.1177_1759720X211034063 – Supplemental material for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis
Supplemental material, sj-xls-9-tab-10.1177_1759720X211034063 for Predictors of health-related quality of life in musculoskeletal disease patients: a longitudinal analysis by Alfredo Madrid-García, Leticia León-Mateos, Esperanza Pato, Juan A Jover, Benjamín Fernández-Gutiérrez, Lydia Abasolo, Ernestina Menasalvas and Luis Rodríguez-Rodríguez in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Authors’ contributions
LRR, LAA, and EM conceived and designed the study. LLM, EP, JAJ and BFG collected data. AMG, LRR, and EM performed the data analysis and interpreted the data. All authors were involved in the drafting and/or revising of the manuscript. All authors gave final approval of the version of the article to be published.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Instituto de Salud Carlos III (ISCIII), Ministry of Health, Spain (CPII17/00014; and RD16/0012/0014) and co-funded by el Fondo Europeo de Desarrollo Regional (FEDER). Funders had no role in the design, collection, management, analyses, interpretation of the data, preparation, review, approval or decision to submit the manuscript for publication.
Patient and public involvement
Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Patient consent for publication
Not required.
Ethics approval
The study was approved by the Hospital Clínico San Carlos Ethics Committee (approval number 20/268-E-BS). This study was conducted according to the principles of the Declaration of Helsinki.
Data availability statement
Data are available on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
