Abstract
Osteoporosis in children differs from adults in terms of definition, diagnosis, monitoring and treatment options. Primary osteoporosis comprises primarily of osteogenesis imperfecta (OI), but there are significant other causes of bone fragility in children that require treatment. Secondary osteoporosis can be a result of muscle disuse, iatrogenic causes, such as steroids, chronic inflammation, delayed or arrested puberty and thalassaemia major. Investigations involve bone biochemistry, dual-energy X-ray absorptiometry scan for bone densitometry and vertebral fracture assessment, radiographic assessment of the spine and, in some cases, quantitative computed tomography (QCT) or peripheral QCT. It is important that bone mineral density (BMD) results are adjusted based on age, gender and height, in order to reflect size corrections in children. Genetics are being used increasingly for the diagnosis and classification of various cases of primary osteoporosis. Bone turnover markers are used less frequently in children, but can be helpful in monitoring treatment and transiliac bone biopsy can assist in the diagnosis of atypical cases of osteoporosis. The management of children with osteoporosis requires a multidisciplinary team of health professionals with expertise in paediatric bone disease. The prevention and treatment of fragility fractures and improvement of the quality of life of patients are important aims of a specialised service. The drugs used most commonly in children are bisphosphonates, that, with timely treatment, can give good results in improving BMD and reshaping vertebral fractures. The data regarding their effect on reducing long bone fractures are equivocal. Denosumab is being used increasingly for various conditions with mixed results. There are more drugs trialled in adults, but these are not yet licenced for children. Increasing awareness of risk factors for paediatric osteoporosis, screening and referral to a specialist team for appropriate management can lead to early detection and treatment of asymptomatic fractures and prevention of further bone damage.
Introduction
Long bone fractures in childhood are common, 1 but early identification of pathological causes of bone fragility is important for the identification of any hidden vertebral fractures, prevention of future fractures and screening for associated complications. Understanding of normal growth, puberty and the developing skeleton is needed to accurately make this assessment.
Definition of paediatric osteoporosis
In 2013, the task force convened by the International Society of Clinical Densitometry (ISCD) produced Paediatric Positions that define paediatric osteoporosis.2,3 Based on these positions, osteoporosis in children is defined by the presence of a clinically significant fracture [i.e. vertebral fracture (VF)] or a significant fracture history and a low bone mineral density (BMD). These positions emphasise the importance of recognising VFs that can be asymptomatic, or can present even with a normal bone density. Below are the official position statements for the definition of osteoporosis:
The finding of one or more vertebral compression fractures is indicative of osteoporosis in the absence of local disease or high energy trauma regardless of the BMD z-score.
In the absence of VFs, the diagnosis of osteoporosis is indicated by both the presence of a clinically significant fracture and BMD z-score ⩽−2. Clinically significant fractures are either:
- Two or more long bone fractures by age 10 years;
- Three or more long bone fractures at any age up to 19 years.
Recently, the strict use of ISCD criteria to define osteoporosis in children has been challenged. A more contemporary view suggests taking into account further characteristics, including underlying condition and fracture risk, use of glucocorticoids, signs of genetic disorder, clinical features of fracture including site, BMD trajectory, etc. 4 These are nuances of osteoporosis diagnosis encountered in clinical care that are not definitively tackled by the current ISCD criteria.
This article gives an overview of the definition of primary and secondary osteoporosis in children/young persons, and the diagnosis, management and follow up of children/young persons with osteoporosis.
Pathophysiology
In childhood, bone mass, and to a large extent, strength, is determined by accrual of new bone (growth) and by remodelling of existing bone. Three key specialised cells enable the skeleton to establish and maintain bone strength: bone forming osteoblasts, bone resorbing osteoclasts and regulatory osteocytes that sense and feedback regulatory signals. Osteoblasts and osteoclasts are coupled tightly in the form of remodelling units, which renew and repair the bone constantly. Signalling between all three units is important in skeletal integrity. Compromise of this signalling can result in altered bone strength but can also be a target for pharmacological manipulation. A key signalling pathway is the RANK (receptor activator of nuclear factor K beta), RANKL (RANK ligand) and OPG (osteoprotegerin) pathway. RANKL is released from osteoblasts and osteocytes and, upon activation of RANK, induces osteoclast differentiation. Osteoblasts also express OPG, which is a soluble inhibitor to RANKL. Thus, the balance of RANKL and OPG determines the differentiation of osteoclasts and subsequent resorption. 5 In children, there is a net increase in bone mass with deposition of bone by osteoblasts being greater than resorption by osteoclasts. 6 In osteoporosis, this balance is disrupted due either to primary genetic causes or the impact of secondary causes, resulting in insufficient bone mass and suboptimal resilience to withstand mechanical stress.
Primary osteoporosis
Primary osteoporosis in children covers a range of bone fragility conditions that have a genetic origin. With an incidence of 1:15–20,000 births, the most common form of primary osteoporosis in children is osteogenesis imperfecta (OI), 7 which comprises a group of disorders characterised by abnormalities in type I collagen synthesis or processing. However, it is now well known that the bone fragility in OI and other primary osteoporosis derives not only from abnormalities in type 1 collagen, but also from multiple other factors including high bone turnover, osteoblast dysfunction, matrix abnormalities and abnormal mineralisation. 8 The diagnosis of OI is made on clinical history and examination, and is supported by positive family history and other investigations, such as imaging techniques, genetic analysis and histology. Genetic analysis for known genes is helpful but not diagnostic.
OI ranges from mild to severe, based on the degree and frequency of fractures, deformities and growth retardation. The Sillence initial classification from 1979 of four types of OI (I–IV) based on severity of disease has now been expanded to include novel forms based on the underlying mutations that are listed on the Online Mendelian Inheritance of Man (OMIM; http://www.ncbi.nlm.nih.gov/omim/) database.9,10 However classification, mixing gene-based OI types with phenotypically defined types has been controversial. 11
Other causes of primary osteoporosis include homocystinuria, osteoporosis-pseudoglioma syndrome (OPPG), primary osteoporosis due to mutations in WNT1 and PLS3 genes, geroderma osteodysplasticum, RAPADILINO syndrome, gnathodiaphyseal dysplasia, spondylo-ocular syndrome, cleidocranial dysplasia, Ehlers–Danlos Syndrome and Hajdu–Cheney syndrome (HCS). 8 A more inclusive list is given in Table 1.
Genetic primary osteoporotic conditions (excluding OI).
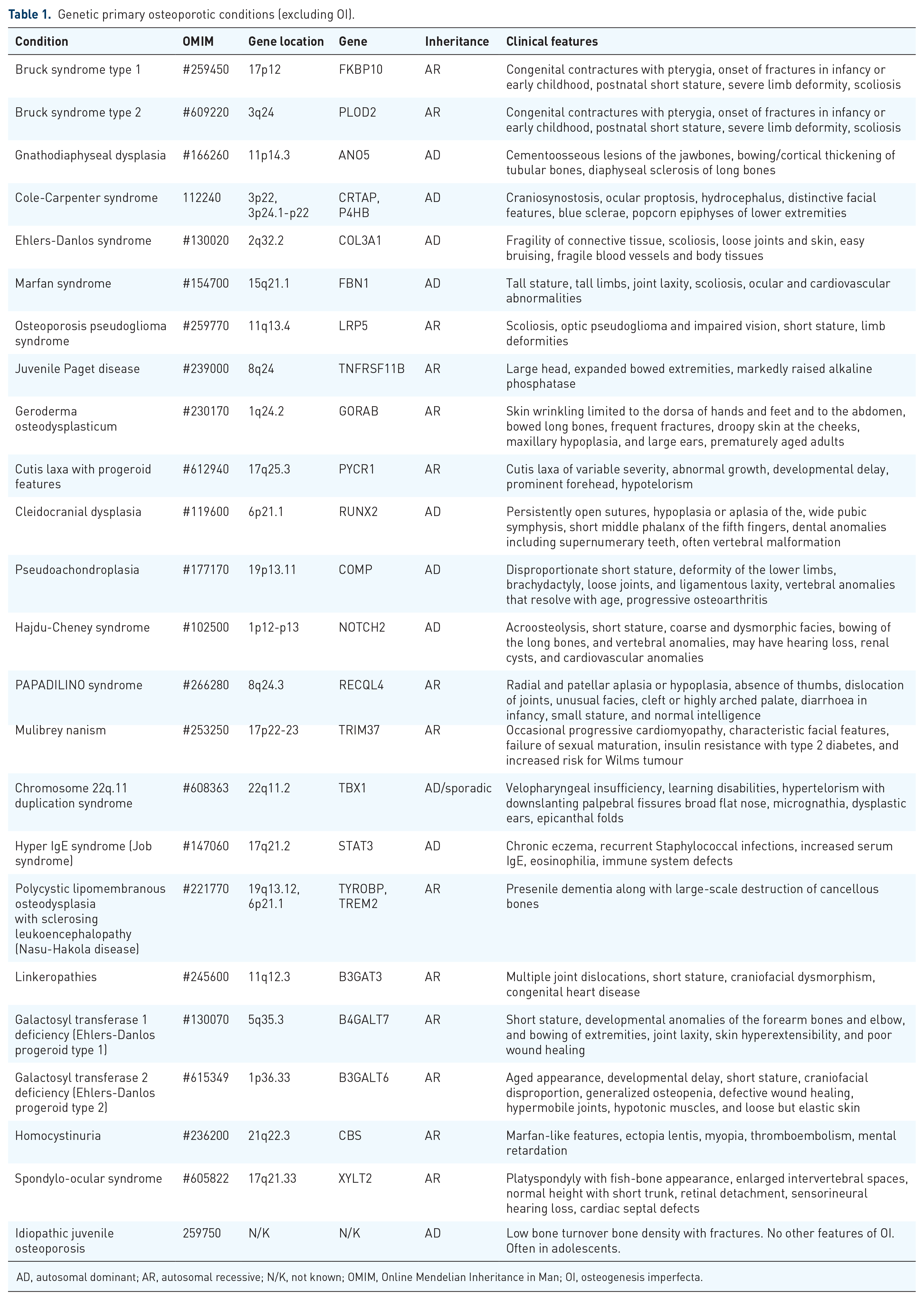
AD, autosomal dominant; AR, autosomal recessive; N/K, not known; OMIM, Online Mendelian Inheritance in Man; OI, osteogenesis imperfecta.
Idiopathic juvenile osteoporosis (IJO) is a diagnosis of exclusion. 12 It classically presents with vertebral and metaphyseal fractures in the prepubertal period. Patients have reduced BMD and reduced trabecular bone density measured on dual-energy X-ray absorptiometry (DXA), quantitative computed tomography (QCT) and peripheral QCT (pQCT).13,14 Bone biopsies of these children classically show a low turnover osteoporosis with reduced bone formation and increased bone resorption, suggesting decreased ability of cancellous bone to adapt to the increasing mechanical needs during growth.13–15 The location of the fractures is likely to reflect the areas in which growth is occurring. Growth is not compromised but, with decreased osteoblast function, bone mineralisation, and thus strength, is. With ongoing secondary mineralisation, which is a slower process, 16 the bones are able to then mineralise, which may explain the phenotype. In most cases, there is spontaneous remission after puberty, but some might remain with residual deformities. Genetic exclusion of other causes of idiopathic osteoporosis is important, as children with heterozygous loss-of-function mutations in LRP5 were previously thought to have IJO. Identification of the mutation in the children also led to the diagnosis of osteoporosis in their parents. 17
Secondary osteoporosis
Secondary osteoporosis is the result of chronic conditions or their treatment, 18 for example with corticosteroids, anticonvulsants and proton pump inhibitors (PPI), that have been found to affect the bone mineral density and/or cause fractures.19–22 With the advances of medicine, the life expectancy of patients with chronic conditions has been extended significantly. Therefore, the longer term effects of morbidity have become more important, especially since there has been an increase in the number of children presenting with secondary osteoporosis. The most common causes are immobility, inflammatory conditions treated with steroids, Duchenne muscular dystrophy (DMD) and other myopathies, leukaemia and other cancers, hypogonadism and thalassaemia.
Disuse osteoporosis
The Mechanostat Theory, initially proposed by Frost, 23 provides the basis on how development of bone strength in childhood is driven by mechanical loads. Loss of these loads leads to loss of bone tissue strain and results in reductions in bone mass and, in some cases, disuse osteoporosis. Nonambulation causes significant decrease of BMD. 24 It also significantly impacts bone geometry, causing decreased periosteal circumference, 25 which is the main reason these children continue to fracture despite BMD-altering treatments like bisphosphonates. The most common form of disuse osteoporosis is among children with cerebral palsy (CP). The prevalence of fractures in CP ranges between 6% and 50%, and incidence increases with severity. 26 The typical site of these fractures is in the distal femur and proximal tibia. Not all children with low BMD will require treatment, and prophylactic bisphosphonate treatment is difficult to justify, but can be considered if they are due orthopaedic surgery that requires better bone quality. Based on the latest consensus statement, they should be treated if they sustain two low trauma long bone fractures (not including hand, feet) or VF. Duration of treatment varies between centres with a minimum of 12 months. Bisphosphonates should be used only after optimising vitamin D status, calcium intake, physical therapies to maximise mobility, and gonadal hormone treatment of absent, delayed or arrested puberty or late-presenting hypogonadism. 27
Effect of steroids
The Steroid Induced Osteoporosis in the Paediatric Population (STOPP) Consortium – a longitudinal prospective study from Canada that followed children with chronic diseases treated with glucocorticoids – demonstrated the effect of steroids on the skeleton for various different conditions.28–30 The follow up of 186 children treated for acute lymphoblastic leukaemia (ALL) showed that 77.3% had complete reshaping of vertebral fractures without any treatment with bisphosphonates. Vertebral fractures at baseline (regardless of severity) and glucocorticoid exposure were the strongest predictors of both VF and non-VF risk in childhood ALL. Importantly, there was a 32.5% cumulative incidence of VF from baseline to 6 years’ follow up, of which 39% were asymptomatic. Older children and those with more severe vertebral collapse at diagnosis were more likely to have persistent vertebral deformity following VF. Therefore, the authors concluded that treatment should focus on these children. 28 The same consortium followed children treated with steroids for rheumatoid conditions and showed that increase in average daily glucocorticoid (prednisone equivalents) dose was associated with an increased VF risk. Almost one-half of the children were asymptomatic, and therefore would have been undiagnosed in the absence of radiographic monitoring, stressing the importance of surveillance of these populations with DXAs and vertebral fracture assessments (VFAs—see later section) or lateral spinal radiographs. 29
Children with DMD and other myopathies requiring high dose steroid treatment are also at risk of developing osteoporosis that can often present with asymptomatic VF or long bone fractures.31,32 The occurrence of VF ranges from 15% to 75% after a mean duration of 6.3 years on treatment, and the incidence of long bone fractures is 24% in treated and 26% in untreated patients. 31 This is due to the combination of long-term high-dose steroid use, immobility and delayed puberty. If VF are left untreated, they can lead to a ‘vertebral fracture cascade’ that impacts significantly on their quality of life. 33 DMD Care Considerations Working Group published guidance in 2018 advising for early monitoring of bone health with DXA and either lateral spinal radiographs or VFA and treatment with bisphosphonates when indicated. 32
Effect of proton pump inhibitors
A large population-based study in the United States (US) showed that proton pump inhibitors (PPIs) are associated with increased fracture risk in young adults when they are given at higher intensity or when total exposure to them is increased, but did not manage to show any dose-response effect to children or higher fracture risk. 34 However, another US study and a nationwide register-based cohort study from Sweden showed that PPI use was associated with a small but significant increased risk of any fracture in children; the effect was greater after 1 month and even greater after 1 year of use. 35 The most common sites for fractures were lower extremity, rib and spinal fractures. 35
Inflammatory bowel disease
Paediatric inflammatory bowel disease (IBD), especially Crohn’s disease (CD), is commonly associated with poor skeletal health, due to the direct effects of chronic inflammation, prolonged use of glucocorticoid (GC), poor nutrition, delayed puberty and low muscle mass.36,37 At diagnosis, children with IBD had lower BMD z-score compared with controls, 38 and those with CD had also reduced trabecular volumetric BMD, thin cortices of the diaphysis and on pQCT. 39 Despite that, the incidence of VF was low but not absent,36,39,40 especially when patients are treated with corticosteroids, 41 so care of all paediatric patients with IBD should include an evaluation of BMD. Regarding long bone fractures, there is no evidence of increased risk in children with IBD.36,42,43
Puberty
The onset of puberty is associated with a significant increase in bone mass. The maximal accumulation of aerial BMD (aBMD) at the lumbar spine and at the femoral neck or mid-shaft occurs in the last stages of puberty in both sexes, but especially in boys. 6 Sex steroids can act on the skeleton through direct stimulation of their receptor in bones and indirect activation of the GH-IGF-I axis by oestrogens, leading to triggering of longitudinal bone growth. Beside their bone-anabolic actions, androgens and oestrogens both exert anti-resorptive effects. The anti-resorptive action of oestrogens appears to be mediated by the downregulation of cytokines, which are also involved in osteoclast formation. 44 A study by Ferrari et al. showed that girls with fractures had decreased peak bone mass gain in the axial and appendicular skeleton and reduced vertebral bone size, indicating that childhood fractures may be markers for low peak bone mass and persistent bone fragility. 45 The incidence of fracture at the distal end of the radius or ulna is higher in children between the ages of 10 and 14 years than in prepubertal children or young adults, underlying the effect of puberty – a period of rapid growth – on bones. 46 The asynchrony between BMD accumulation and height gain is likely to play a role, as skeletal volume is increasing at a faster rate than the mineralization process. Exposure to oestrogens and testosterone has a protective effect on BMD and is associated with a reduced incidence of atraumatic fractures. Delayed puberty as part of a chronic condition like DMD, IBD, thalassaemia or in cases of primary or secondary pubertal arrest (e.g. Turner syndrome, hypogonadotrophic hypogonadism, ovarian failure, Klinefelter syndrome, anorexia nervosa) can result in reduced bone mineral density and subsequent increased rate of fractures. 47
Thalassaemia major
The pathogenesis of osteoporosis in thalassaemia major is polyfactorial and involves bone marrow expansion due to increased erythropoiesis and endocrine dysfunction secondary to excessive iron loading, including delayed puberty and hypoparathyroidism, this results in increased osteoclastic activity and insufficient osteoblastic bone formation.48–50 The reported frequency of osteoporosis, even in well treated patients with thalassaemia major varies from 13.6% to 50%. The recommendations of the International Network on Growth Disorders and Endocrine Complications in Thalassaemia (I-CET) for diagnosis and management of osteoporosis in thalassaemia major need to be followed. 51 An adult study showed that vertebral bone density measurements by DXA are influenced by hepatic iron overload in non-compliant with chelation therapy patients, particularly in patients with a liver iron concentration >5000 μg Fe/g wet tissue, so that must be taken into account when interpreting DEXA results. 52
Investigations
Bone biochemistry
After careful history and examination, baseline investigations for calcium, phosphate, magnesium, ALP, PTH, Vitamin D, urinary calcium to creatinine ratio should be strongly considered. In most cases these markers will be normal, but primary disorders of bone metabolism must be excluded and bone health should be optimised in children with fragility fractures. Furthermore, normal calcium and vitamin D, as well as normal bone profile, is essential before initiating treatment for osteoporosis. There are limitations in the use of calcium to creatinine ratio. Levels can alter depending on whether the urine are dilute or concentrated. Also, creatinine levels may be raised secondary to various factors, such as renal impairment, destruction of muscle, high dietary intake of meat, hypothyroidism, testosterone use, or reduced secondary to reduced muscle mass, malnutrition, vegetarian diet, giving falsely high or low ratio levels.
Dual-energy X-ray absorptiometry
At the 2013 update of the position paper of the ISCD the committee concluded that DXA is the preferred method for assessment of areal bone mineral content (BMC) and density (aBMD) and that estimating aBMD should be part of the overall assessment for children at elevated risk of a clinically significant fracture. 2 Measurements of total body less head (TBLH) and/or posterior-anterior lumbar spine aBMD or BMC are recommended. 53 The 2019 ISCD position added the use of proximal femur, distal lateral femur and 55% radius DXA measurements, if reference data are available. Proximal femur DXA measurements can be used for assessing children with reduced weight bearing, or in children at risk for bone fragility who would benefit from continuity of DXA measurements through the transition into adulthood. DXA measurements at the 33% radius may be used clinically in ambulatory children who cannot be scanned at other skeletal sites. Lateral distal femur (LDF) DXA measurements, correlate well with increased lower extremity fragility fracture risk in non-ambulatory children and can be used in the presence of non-removable artefacts, scoliosis, and positioning difficulties. 54 The advantages of DXA are low radiation dose, short scan time and widespread availability. DXA is a two dimensional measurement, so it can underestimate BMD in children with short stature or growth delay and overestimate BMD in tall children. Therefore, the measurements should be size-corrected using the appropriate methods and bone mineral apparent density (BMAD) for i-DEXA Lunar scanners or height adjusted Z-score (HAZ) for Hologic scanners are the preferred methods to correct for short stature.55–58 Efforts have been made to create reference range data for children.59,60 DXA is generally not used in children less than 5 years due to movement artifact and lack of reference data.
Lateral spine radiograph
Lateral spine radiographs can diagnose VF, but can sometimes be of limited quality, depending on breathing technique and quality of machine, especially at the thoracic spine, due to low BMD and overlying ribs and lungs. The main drawback, though, is increased radiation exposure. Given that VF are a main manifestation of osteoporosis in primary and some secondary osteoporotic conditions, and that routine screening is necessary, the use of a low-radiation method to detect VF is pressing. These facts contribute to the need for an effective, low-radiation technique. EOS imaging is a low-dose, X-ray technology that can simultaneously take full-body, frontal and lateral images of the skeletal system of a patient, using significantly less radiation than traditional X-rays or CT scans. It is not used widely yet, but could also be a potential diagnostic method.
Vertebral fracture assessment
The addition of lateral spine imaging technology to the densitometer, for VFA, represented a major advancement in the ability to diagnose VF and osteoporosis. 61 VFA with DXA may reach a high level of accuracy for diagnosing VF.62,63 The latter can cause significant pain, but can often be asymptomatic and remain undiagnosed; therefore, VFA can be a very useful screening tool for VF. VF are evaluated using the modified Genant semiquantitative method. 64 The automated vertebral morphometry software can be inaccurate, especially in very low bone density and, whilst this may be useful in a research setting, the clinical reporting of VF should be done by a specialist with significant experience in VFA reporting. A recent study assessed the use of DXA VFA in children and compared that against spinal radiographic assessment (RA) for the identification of VF in children. It demonstrated that VFA is as good as RA for detecting moderate and severe VF. 65 The ability to obtain the image at the same time the bone density is done, with significantly lower radiation exposure than with spine radiography, better image quality (Figure 1) and at a lower cost makes VFA a preferred method for the detection of VF in children,61,65 especially since there is an increased number of conditions like OI, DMD and thalassaemia that require routine screening regularly for asymptomatic VF. Based on the 2019 update of the ISCD position statement, following VFA, additional spine imaging should be considered in the following circumstances: (a) vertebrae that are technically unevaluable by VFA (i.e. not sufficiently visible, movement artifact and scoliosis), provided the detection of a VF would change clinical management; (b) assessment of a single, Genant Grade 1 VF, if confirmation of a Grade 1 VF alone would change clinical management; (c) radiographic findings that are not typical for an osteoporotic VF (e.g. suspected destructive inflammatory or malignant processes, congenital malformations, acquired misalignments or dislocations). 54

Plain spine radiograph (a) and VFA view on Lunar i-DXA (b) on the same patient.
Quantitative computed tomography and peripheral QCT
QCT and pQCT can describe the geometry of both trabecular and cortical bone, but also measures the BMD (and muscle density), along with muscle geometry [diaphyseal and cortical cross-sectional area (CSA)]. It is specifically useful for children with spinal deformities, contractures or metallic implants, where DXA imaging can prove challenging. pQCT has much smaller radiation compared with QCT and is, therefore, preferred. 66 pQCT measured parameters such as volumetric BMD, cortical area and strength–strain index have been related to fractures in children. 67 High-resolution scanners (HRpQCT) have low radiation dose, give clear images and can safely assess important elements of bone microstructure in adolescents and children.68,69 An attempt to create reference values by different groups has been partially successful, though current reference data should be applied only in the centers where the data have been developed. 70 Both pQCT and HRpQCT are used in a few specialised centres around the world, mostly as part of research, and need highly qualified personnel to interpret their data. 66
Panoramic mandibular X-ray
There seems to be a causal relationship between osteoporosis and periodontitis that has not been clarified. It has been reported that osteoporosis and mandibular cortical index are associated with horizontal alveolar bone loss (periodontitis). The mandibular cortical width has the best specificity, whereas all indices had variable sensitivity reported by different studies. Though, currently, panoramic radiograph is not a recognised tool for diagnosing osteoporosis and is used only as part of research, its reliability in screening/detecting osteoporotic patients has been frequently investigated and cited. 71
Trans-iliac bone biopsy with tetracycline labelling
Transiliac bone biopsy gives qualitative and quantitative data of bone samples. This is the only site for which paediatric histomorphometric reference data have been published. 72 Dynamic parameters of bone cell function can be measured when patients have received tetracycline labelling. A bone biopsy is helpful in understanding the underlying histological process for bone disease, when diagnosis in not straightforward or in differentiating types of osteoporosis. Examples are cases of frequent low impact fractures with normal bone density, differentiation between various types of OI or in unexplained mechanism of fractures. Since trans-iliac bone biopsies are invasive, and require a short but general anaesthesia, they are not recommended for monitoring of treatment outside of highly specialized centres, and research protocols.
Bone turnover markers (bone formation and bone resorption markers)
Bone turnover markers (BTMs) have been used frequently in the adult populations in order to assist with the diagnosis and management of osteoporosis. However, their use in the paediatric population is limited. Children have significantly elevated bone marker levels due to high skeletal growth velocity and rapid bone turnover during childhood growth.73 –75 Osteocalcin and β-crosslaps (β-CTx), have been used in assessing bone metabolism in children with OI and were reduced after treatment with pamidronate. 76 Delayed puberty was also found to affect levels of BTMs, as it affects growth. 77 OPG and total s-RANKL are thought to be indicative of lower osteoblast and increased osteoclast signalling. 78 In children with clinical bone fragility, BTMs correlated with each other, but not with lumbar BMD Z-score. While they were not reliable predictors of degree of low BMD, the bone markers showed suppression during bisphosphonate therapy and may be helpful in monitoring the response to therapy. 79 Sensitivities and predictive values of single markers are still poor.74,80 Paediatric reference ranges need to be used.81,82
Genetics
With the advances in genetic testing, there is enhanced availability and understanding of the multiple genes associated with bone fragility. Obtaining a definite genetic diagnosis can help confirm clinical suspicion and direct management of osteoporosis. It also enables cascade screening of other family members, genetic counselling and early detection of condition, leading to preventive treatment. 83 It is particularly helpful in children with frequent fractures, but no other extra-skeletal features of OI or of other primary osteoporosis syndromes; however, the likelihood of finding a mutation is relatively small (28%). 84 It can also be performed in cases where non-accidental injury is being investigated and when diagnosis is not straightforward. Autosomal dominant mutations to COL1A1 and COL1A2 genes should be sought first, as they cover 97% of patients with OI type 1 and 77% of moderate to severe OI. As a next step a panel of genes associated with primary osteoporosis should be used.84,85 Whole exome sequencing can be used to detect rarer genes. 86 A rapidly increasing number of whole-genome sequencing association studies are being conducted to identify rare variants associated with complex traits and diseases, so this might be the future of genetics of rare diseases.
Management
General approach
Management of osteoporosis requires a patient-centred multidisciplinary approach. The team should include doctors (including geneticists), nurses, physiotherapists (PT), occupational therapists (OT), psychologists and social workers, but there should also be access to dentists, audiologists, orthopaedic and spinal surgeons, neurosurgeons and a pain management team. PT and OT are of outmost importance for optimising independence in mobility and daily living. Needs vary depending on the age and severity of each case. In severe neonatal cases, respiratory support might be necessary, as rib fractures can compromise breathing. It is also important to support the head and spine until babies have adequate muscle tone, and encourage prone positioning and sitting in inclined seats in order to prevent scoliosis and brachycephaly. In severe cases parents should receive instructions on safe handling of the baby soon after diagnosis in order to prevent future fractures. Various aids from splints and insoles to wheelchairs need to be provided, as well as pencil holders and wrist splints to help with hypermobility. Encouragement of early mobility as soon as possible after a fracture is of outmost importance. Children with dentinogenesis imperfecta need a 6-monthly dental review and good dental hygiene should be promoted.
Patients with osteoporosis should also be advised to avoid contact or high impact sports and activities that can cause or deteriorate VF, such as horse-riding and trampolining.
It is important to maintain sufficient levels of vitamin D and calcium in accordance with the current guidelines, 87 as insufficiency can impair the response to bisphosphonate therapy and increase the risk of hypocalcaemia after treatment. The serum 25-hydroxyvitamin D concentrations should be maintained above 50 nmol/l.
Secondary prevention of fractures
One of the earliest signs of bone fragility is the presence of VF, which in children can often be asymptomatic. Therefore, spine fracture surveillance in high risk groups might detect VF at an early stage and lead to early intervention and prevent non-reversible damage. Predictors of VF are the long term use of corticosteroid, immobility, delayed puberty or pubertal arrest, chronic inflammation, back pain, reduction in spine BMD z-score, thalassaemia with frequent transfusion in children older than 10 years. Children with these risk factors should undergo regular lateral spine imaging commencing early on their disease course.
When assessing treatment or the potential for recovery without treatment there are three aspects to be considered 20 :
- The severity of bone morbidity, a greater number of fractures and greater degree of compression, the greater the need to start treatment urgently.
- The remaining growth potential, as being closer to final height reduces the potential of vertebral remodelling.
- The likelihood of persistent risk factors for bone fragility. Children who have resolution or treatment of their primary disease (such as transplant with ALL or diagnosed coeliac) may allow healing to occur without intervention.
Although a low BMD can assist in diagnosis of an osteoporotic condition, the presence of fractures and continuing risk are indications for treatment and not the BMD value. BMD is, however, used primarily for monitoring treatment with bisphosphonates.
Medication
The main difference between children and adults with osteoporosis is the potential in children not only to recover BMD, but also to reshape previous fractured vertebral bodies,88–90 sometimes even without specific osteoporosis treatment.28,91,92 Taking advantage of this period of rapid growth can lead to optimal results and prevent future comorbidities of osteoporosis.
Antiresorptives
Bisphosphonates
Bisphosphonates remain the main medical treatment, but new drugs are evolving and various ongoing clinical trials on children with osteoporosis might broaden our therapeutic choices over the coming years. They are off-label worldwide, with the exception of neridronate in OI, in Italy.
Bisphosphonates are currently the only recommended medical treatment and the standard of care for osteoporosis in children in the United Kingdom (UK). They have been used both for primary and secondary osteoporosis. 93 Bisphosphonates are antiresorptive agents that inhibit osteoclast activity and suppression of bone turnover. 94 Recent studies have demonstrated that some beneficial effects of bisphosphonates on the skeleton could be due to the prevention of osteoblast and osteocyte apoptosis. 95
Intravenous pamidronate, neridronate or zoledronate are the standard of care in children with moderate to severe OI, while bisphosphonate treatment remains under consideration in children with mild forms of OI and depends on various factors, like bone density, number of long bone or VF, and remaining growth. A systematic Cochrane review of 14 randomised studies on bisphosphonates in children concluded that oral or intravenous bisphosphonates significantly increase bone mineral density in children and adults with this condition with similar ability. 96 It is unclear whether oral or intravenous bisphosphonate treatment consistently decreases fractures, though multiple studies report this independently,76,97,98 and no studies of intravenous bisphosphonates report an increased fracture rate with treatment. Other studies show no decrease in fracture rate despite increase in BMD.99–101 The authors also commented that the majority of trials analysed were small and not powered to show a statistically significant difference in many outcome measures, such as mobility, fracture rate and quality of life. 96 Furthermore, studies on the effect of intravenous bisphosphonates on vertebral compression fractures, showed that reshaping, which is growth dependent, is better the earlier they are started.88,89,102 Unfortunately, large-scale, placebo-controlled long-term trials on bisphosphonate would be extremely difficult to conduct, as it would be unethical to leave children untreated, so clinicians need to make decisions based on existing evidence.
Risedronate is the most potent oral bisphosphonate, and a large trial showed that it increased lumbar BMD and decreased the risk of long bone fractures, but there was no significant improvement in vertebral shape and in some cases there was deterioration. 103 A different group showed that despite increase in BMD, risedronate treatment did not result in histomorphometric differences in cortical or trabecular indices. 100 In another large controlled trial on children with glucocorticoid-induced osteopaenia in juvenile rheumatic disease, risedronate increased BMD, but there the number of VF increased compared with the control group and BTMs remained unchanged. 104
Zoledronate is the most potent bisphosphonate treatment, can be given over 30 min and the infusion is repeated 6 monthly. Saraff et al. compared the efficacy and treatment costs between zoledronate and pamidronate, and showed that zoledronate is a considerably cheaper alternative to pamidronate with comparable efficacy, and a more convenient option for patients due to fewer hospital visits. 105 It is therefore preferred, even though many centres still administer pamidronate due to lack of experience with zoledronate. There is no consensus regarding the dose, frequency and duration of bisphosphonate treatment. Originally pamidronate was given in a dose of 0.5–1 mg/kg per day administered over 3 days every 3 months, 106 but since then shorter and lower dose protocols have been adapted.107,108 Zoledronate was given 3 monthly initially, 109 but then was found to have similar efficacy when given 6 monthly, 110 which is the common practice. The dose ranges from 0.025 mg/kg/day to 0.05 mg/kg/day.105,109–112 Neridronate is also a potent IV bisphosphonate that is given 3 monthly at a dose of 2 mg/kg.113–115
Approximately 18–27% of patients receiving first doses of IV bisphosphonate experience an acute phase reaction (fever, headache, myalgia, arthralgia, malaise) or, rarely, vomiting occurring up to 72 h after the infusion. Transient hypocalcaemia, hypophosphatemia and a rise in C-reactive protein can be observed but are rarely of clinical significance. Acute phase reactions are more likely to occur in patients with secondary osteoporosis (13% of all infusions) – a group who may require closer monitoring. 112 The incidence is reduced approximately 50% by acetaminophen (before and for 24–48 h after infusion) and decreases with subsequent infusions. 116 Hypocalcaemia can be more easily counter-regulated when levels of vitamin D and daily calcium intake before infusion are sufficient. In case of hypocalcaemia or when history of previous hypocalcaemia after the infusion exists, calcium supplementations is required for the next few days after the infusion. In order to prevent the acute phase reactions many centres administer half the dose for the first infusion. 117 Bisphosphonate use in adult patients has been linked to osteonecrosis of the jaw. However, no paediatric cases have been reported so far.118,119
In bisphosphonate therapy, metaphyseal sclerotic lines of long bones, called zebra lines, are seen on X-rays.73–75 These represent periods of the drug inhibiting osteoclastic activity and increased bone and calcified cartilage with low remodelling activity.73,120 In adults, there is a documented risk of atypical femoral subtrochanteric insufficiency fractures (stress fractures) with long-term bisphosphonate therapy.121–123 However, Vuorimies et al. looked at 127 femoral fractures in 24 patients with OI and compared the group that was naïve to treatment with the ones during bisphosphonate treatment and those after discontinuation. The pattern of femoral fractures was similar in all groups and was not related to bisphosphonate dose, but more to the severity of OI. 124 Trejo et al. came to the same conclusion, showing atypical appearance in about a quarter of nondeformed femur fractures that occurred in children with OI, which was related to severity of OI rather than bisphosphonate treatment. 125 Pamidronate therapy has been also associated with delayed healing of osteotomy sites after intramedullary rodding procedure, but not delayed fracture healing. 126 Maintaining a bisphosphonate infusion-free interval of some months after the osteotomy, and ensuring that there is adequate callous formation before the next dose, reduces the chances of delayed healing. 127 A detailed table on bisphosphonate therapy in OI can be found at the 2016 Cochrane review. 96 The authors’ approach of Zoledronate use for paediatric osteoporosis is shown in Table 2.
Authors’ approach for use of zoledronate in children with osteoporosis.

ALL, acute lymphoblastic leukaemia; BMAD, bone mineral apparent density; BMD, bone mineral density; CMD, congenital muscular dystrophy; CP, cerebral palsy; DMD, Duchenne muscular dystrophy; IBD, inflammatory bowel disease; OI, osteogenesis imperfecta; SDS, standard deviations.
Denosumab
Denosumab is a far more potent inhibitor of bone resorption, but its effects on the growing skeleton are not yet well described. It is an inhibitor of RANKL that has emerged as an important novel therapy for skeletal disorders. It binds with high affinity and specificity to RANKL, mimicking the inhibitory effects of OPG and resulting in rapid suppression of bone resorption. 128 The FREEDOM trial studied the effects of Denosumab on adults, 129 but a large scale trial of Denosumab on children with OI is still ongoing. Data on children are isolated to case series. A large scale phase III randomised, double-blind, placebo-controlled, parallel-group study to evaluate the safety and efficacy of denosumab in paediatric subjects with glucocorticoid-induced osteoporosis is ongoing [ClinicalTrials.gov identifier: NCT03164928], assessing the change of lumbar spine BMD Z-score from baseline to 12 months after administering denosumab 1 mg/kg BW (up to a maximum of 60 mg) SC every 6 months. Another multicentre, single-arm study to evaluate the efficacy, safety and pharmacokinetics of denosumab in children with OI has stopped recruiting, but is still ongoing [ClinicalTrials.gov identifier: NCT02352753]. It is studying the effect of burosumab on BMD after administering denosumab 1 mg/kg (up to a maximum of 60 mg) subcutaneously every 3 months for a minimum of 12 months, and all subjects will receive appropriate calcium and vitamin D. There is also an extension study of the previous one on children with OI [ClinicalTrials.gov identifier: NCT03638128].
Denosumab has been used in children with OI, giant cell tumour, Paget disease, DMD and fibrous dysplasia, and was found to cause modest increase in BMD and suppress BTMs.130–137 Nevertheless, its action is of shorter duration compared with adults. 138 The follow up for 2 years of four children with OI type VI that were treated with Denosumab showed an increase in BMD, a normalisation of vertebral shape, an increase in mobility, and a reduced fracture rate. 133 The same group followed 10 children diagnosed with type I, III and IV OI and reported that denosumab significantly improved BMD by week 48. 134 Follow up of this trial cohort used longer intervals between injections based on urinary bone turnover markers. Despite stable laboratory findings for bone resorption markers, patients presented with a significant reduction of areal BMD during the follow-up year, but there were no new VF and vertebral shape further improved. 139 However, a 23-month-old boy with type 6 OI who was treated with denosumab had a persistent decline in BMD and continued to fracture over a 12-month treatment period. 140
In contrast to bisphosphonates, denosumab’s effect on bone turnover is rapidly reversible. 128 Also, discontinuation of denosumab can lead to rebound increased bone turnover and severe hypercalcaemia in children, as soon as 4 weeks after the injection.136,141 This might be related to the high baseline bone turnover in children, especially in conditions like OI. Uday et al. reported the first case of jaw osteonecrosis in a child treated with Denosumab for giant cell tumour of bone. 136 Denosumab did not seem to adversely affect growth plates in a growing child, 142 though another case report showed sclerotic epiphyseal bands that faded with time. 143
Anabolics
Various forms of osteoporosis in childhood are characterized by low bone turnover (e.g. osteoporosis due to neuromuscular disorders and glucocorticoid exposure). In these cases antiresorptive therapy causes further reduction of bone turnover with potential risks, which has led to the increased interest in anabolic therapies.
Growth hormone
Growth hormone (GH) has been shown to increase bone mineral content, mainly through increasing muscle area and strength–strain index and by changing bone geometry in children with GH deficiency.144,145 However, there is no clear benefit in terms of reduced fracture rates and improvement in bone mass compared with bisphosphonate treatment in children with OI, given that GH increased lumbar spine BMD by only 5–7% over the first 6 months of therapy. 146 Treatment with GH should be limited to indicated current conditions as the current evidence does not support its isolated use for osteoporosis.
Testosterone
Bone mineralisation increases with age, height and weight through childhood, with a significant gain during pubertal development. Serum testosterone concentration at the age of 12 years is correlated positively with a subsequent increase in BMAD by the age of 18 years. 147 Delayed puberty is a common feature of boys with DMD and other steroid treated conditions, such as IBD, or even in thalassaemia. It has been shown that hypogonadal patients increase their BMD when treated with sex steroids in adulthood, but normal adult BMD values are not reached, while if treatment starts at an appropriate age, they will acquire a better increase in BMD. 148 Therefore, induction of puberty with testosterone must not be forgotten and postponed in boys with delayed puberty. In healthy boys, circulating oestradiol (that can be aromatised from testosterone) is associated positively with bone maturation and aBMD and vBMD, and negatively with endosteal circumference, whereas testosterone is associated positively with lean mass, lumbar and whole-body bone area, trabecular and cortical area, and periosteal circumference at the radius. 149 Therefore, testosterone administration in boys with delayed puberty will increase their BMD, but also muscle strength, and subsequently reduce the risk for fractures. In boys with DMD, testosterone is unlikely to increase muscle mass and strength due to the nature of the condition. A recent study investigating the effect on testosterone treatment on DMD confirmed that there is no change in lean body mass index, but interestingly motor functions scores in non-ambulatory boys improved and there was statistically significant increase in median BMD of the lumbar spine. 150
Oxandrolone, a non-aromatizable synthetic derivate of 5-alpha dihydrotestosterone, may be a preferred anabolic agent in children, as it prevents conversion to oestrogen and earlier closure of epiphyseal growth plates. It is not commonly used though for induction of puberty.
Parathyroid hormone
Teriparatide, recombinant human parathyroid hormone (PTH) has been used as treatment of osteoporosis in adults, but it is not used in children with osteoporosis due to the potential risk of osteosarcoma whilst their growth plates are still open. 151 This is based on studies in cancer-prone rats treated with high doses of PTH and there were no malignant bone changes with lower doses.152,153
Anti-sclerostin antibodies
The glycoprotein sclerostin is secreted by osteocytes and interacts with LRP5 and LRP6 receptors, inhibiting Wnt signalling and bone formation. Anti-sclerostin antibodies have been found to increase bone formation in mice and postmenopausal women, but this effect decreases with time and there is bone loss after therapy discontinuation.154,155 This can be prevented with the use of anti-resorptive therapy after discontinuation of anti-sclerostin antibodies. 156 In a phase IIa randomised trial Glorieux et al. showed that BPS804 anti-sclerostin antibody stimulates bone formation, reduces bone resorption and increases aBMD in adults with moderate OI. 157 A paediatric trial is ongoing, but the results are still not available.
Anti-tumour growth factor beta therapy
Tumour growth factor beta (TGFβ) is a multifactorial growth factor. Modulation of TGF-β signalling has proven effective pre-clinically in the treatment of multiple pathologies. 158 Data from murine models suggest that injections with anti-TGF-β may increase bone mass in OI mice. 159 The effect of anti-TGFβ antibodies in humans is yet to be investigated. Losartan is an angiotensin II type 1 receptor blocker that also downregulates TFG-β signalling and there are ongoing trials on its potential use as a treatment for osteoporosis. 160
Whole body vibration therapy
Several small randomised controlled trials and observational studies in children with CP and other paediatric disabling conditions have demonstrated a beneficial effect of whole body vibration (WBV) therapy on walking speed, muscle strength, spasticity and balance. Based on the mechanostat theory, 23 attempts have been made to implement WBV therapy to the management of osteoporosis. A trial on children with Crohn’s disease showed that ‘low-magnitude’ vibration is much less effective than bisphosphonate therapy, at least when bone density is considered the main outcome measure. 161 Also, in children with OI, WBV resulted increase in lean mass without changes in muscle function or bone mass, suggesting reduced biomechanical responsiveness of the muscle-bone unit. 162
Conclusion
Osteoporosis in children is quite distinct from the pathology in adults, as it has a different definition, different methods of diagnosis and monitoring and different treatment options. Primary osteoporosis comprises mainly of OI, but there are other forms that also cause reduced bone density and may require treatment. Secondary osteoporosis can be a result of immobility and muscle disuse, iatrogenic, with high dose steroids as the most common contributor, chronic inflammation, such as in inflammatory bowel disease, delayed or arrested puberty and thalassaemia major. Investigations involve bone biochemistry to exclude other causes of metabolic bone disease, DXA scan for bone densitometry and VFA, radiographic assessment of the spine and in some cases QCT or pQCT. It is important that BMD results are adjusted based on age and gender, but also on height for lumbar spine, in order to reflect size corrections. Genetics are increasingly being used for the confirmation of diagnosis and classification of various cases of primary osteoporosis. Bone turnover markers are used less frequently in children, but can be helpful in monitoring treatment, and bone biopsy is another tool that can assist in the diagnosis of atypical cases of osteoporosis.
The management of children with osteoporosis requires a multidisciplinary team of health professionals with expertise on paediatric bone disease. It aims for the prevention and treatment of long bone and VF and improvement of the quality of life of patients. The drugs used most commonly are bisphosphonates, which have been used for over 30 years with good results in improving BMD and reshaping VF when started early. The data regarding their effect on reducing long bone fractures are equivocal. The unlicenced use of denosumab is increasingly being reported for various conditions with mixed results. There are many more drugs trialled in adults with osteoporosis and some on children with OI, but as yet none has been licenced for use in children. Finally, WBV therapy has been used in osteoporosis, but with less effect on bone structure or fracture risk. Increasing awareness of the various forms and risks of paediatric osteoporosis and referral to a specialist team for appropriate management can lead to early detection and treatment of asymptomatic fractures and prevention of further bone damage.
