Abstract
Biologics are substances synthetized from biological sources used in the prevention and treatment of several diseases. Rheumatologists have many years of experience with biologics for the treatment of immune-mediated diseases and osteoporosis. Randomized clinical trials and postmarketing studies have demonstrated that treatment with biologics can result, albeit infrequently, in serious adverse events. To date, several risk mitigation strategies have been identified and implemented. The objective of the present perspective review is to examine the risk mitigation strategies of biologic treatments, with special focus on anti-tumor necrosis factors and denosumab.
Introduction
Biologic treatments have revolutionized the treatment of patients with serious inflammatory autoimmune diseases and, more recently, with severe osteoporosis. Biologics are defined as substances produced by living organisms used in the prevention, diagnosis, or treatment of cancer, immune-mediated diseases, and other diseases. Biological drugs include antibodies and interleukins. These substances with specific targets are also called biologic agents, biological response modifiers, or biologic disease-modifying antirheumatic drugs (bDMARDs) for rheumatic disease treatment. 1
In chronic inflammatory rheumatic, skin and gastrointestinal (GI) diseases, the benefits of biologics have been largely demonstrated in patients with severe disease. Biologics have been demonstrated in randomized controlled trials (RCTs) and in daily practice to significantly attenuate disease progression, by reducing pain and swelling, joint damage, skin and GI lesions, and by improving health-related quality of life of arthritis, skin disease and GI inflammation.2,3 The list of biologics beyond anti-tumor necrosis factor (TNF) in these conditions is rapidly increasing, with the development of new antibodies directed against interleukins (ILs), such as antibodies against IL-6, IL-17 and IL-23 in inflammatory rheumatic diseases. Biologics used to treat rheumatic chronic arthritis disorders may also have primarily cellular targets, for example, abatacept interferes with the activation of T cells and rituximab binds to the protein CD20 on the surface of B cells inducing/triggering B cell death.
In osteoporosis, monoclonal antibodies have been developed against the receptor activator of the nuclear factor kappa-B ligand (RANKL) 4 and recently against sclerostin to reduce the risk of vertebral and nonvertebral fractures. 5
Rheumatologists have been utilizing biologics for use in inflammatory rheumatic disease for over 20 years, starting with anti-TNFs, and with denosumab in osteoporosis for nearly 10 years.
However, in RCTs and postmarketing surveys, it has become clear that such potent biologic treatments can also result in serious adverse events, more commonly than placebo and standard nonbiologic treatments.
Therefore, risk minimization strategies have been implemented allowing patients to receive the greatest benefits from biologic drugs, despite their potential risks. The purpose of our review is to advise clinicians on how to consider and integrate evidence on the benefit–risk ratio of biologics in daily practice.
We have selected the example of anti-TNFs because they are the first and most frequently prescribed biologics in inflammatory rheumatic diseases. In osteoporosis, we also have reviewed denosumab, the most commonly prescribed biologic for fracture prevention.
The example of anti-TNFs in inflammatory rheumatic, skin and GI diseases
In addition to their tremendous efficacy, anti-TNFs have potential side effects, which are listed in Table 1 together with potential approaches to risk minimization.
Potential adverse events of anti-TNFs.
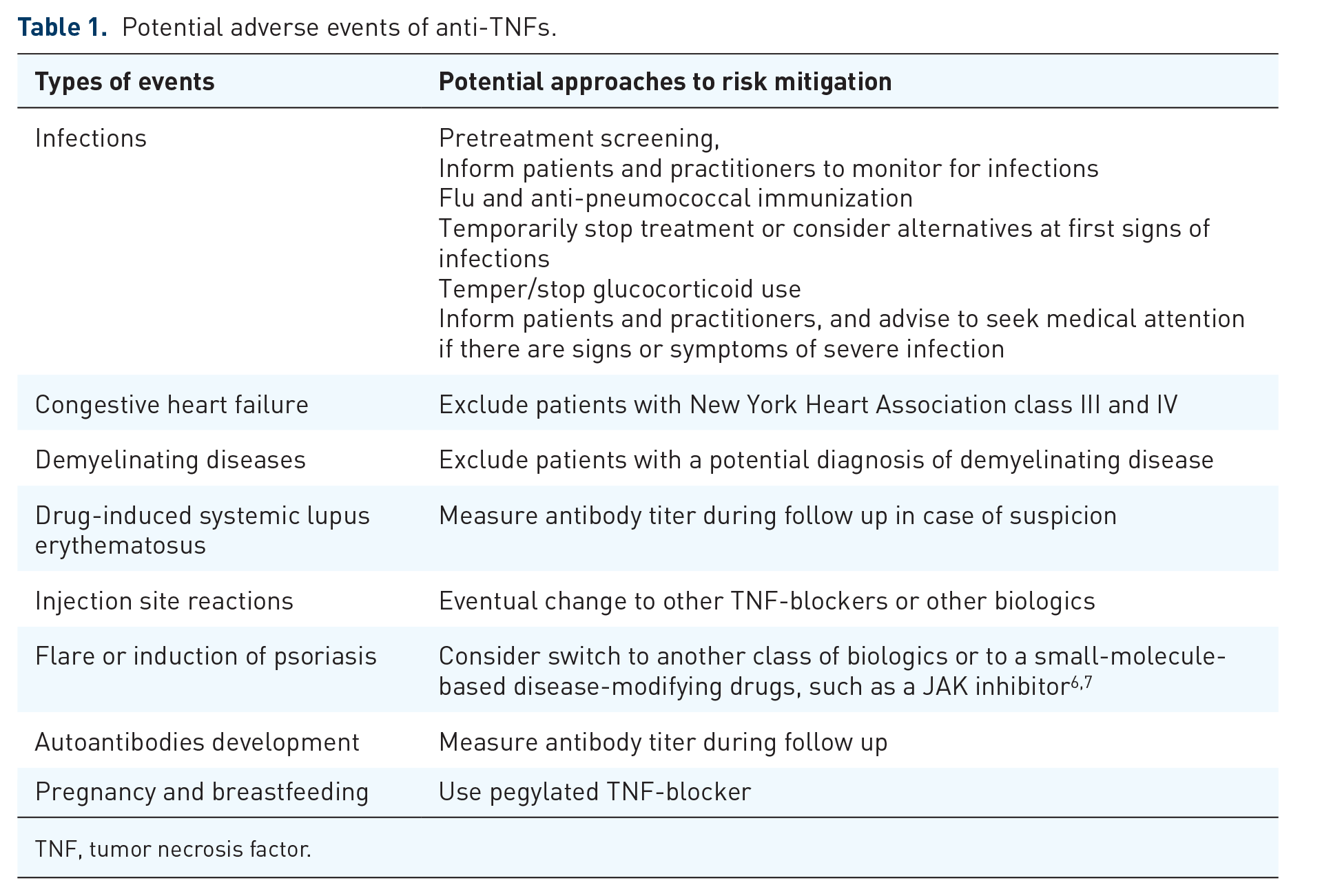
TNF, tumor necrosis factor.
One of the first examples comes from the anti-TNF infliximab. Immediately after its introduction, an increased incidence of tuberculosis was first detected. 8 The introduction of rigorous measures to screen patients for latent tuberculosis or disease in all patients before starting anti-TNF has decreased the incidence of tuberculosis in rheumatic patients. 9 This is a striking example of a serious side effect that can be mitigated effectively by physicians. In high-risk patients, risks and benefits should be reviewed very carefully. For example, in daily practice, anti-TNFs are not prescribed in patients with grade 3 or 4 congestive heart failure, and in line with that, congestive heart failure is very seldom observed in our patients.
Clinicians have made progress in preventing, monitoring for, and managing the major adverse events associated with anti-TNFs and other biologics. Fundamental to this strategy of risk minimization has been a refinement of our knowledge on the pathophysiology of inflammatory cytokines. 10
Most of the available data related to the safety of anti-TNFs are derived from clinical trials. RCTs provide the best quality evidence for efficacy and are required for market approval; however, they are often too small, too short in duration, and performed in patients who are too healthy (healthy subject bias) 11 to adequately define the full risk of such drugs. Observational studies, which are conducted on large populations, can help ascertain the safety of medications. Sources such as the United States Food and Drugs Administration (US FDA) MedWatch and disease registries (e.g. the National Database Registry of Rheumatoid Arthritis and CORRONA) have contributed significantly to identify important safety issues with anti-TNFs. Nevertheless, methodological limitations (confounding by indication, patient dropout, switching therapies, and limited control populations) must be considered when interpreting observational data of therapeutic safety. Clinicians must individualize the infection risk assessment not only on the basis of the specific anti-TNFs used or the expected duration of therapy, but also by taking into account the baseline risk susceptibility of a given patient. 12
In patients with autoimmune inflammatory rheumatic diseases, immunosuppressive therapy increases the risk of infections. Several recommendations for minimizing the risk of infections have been proposed. 13 Screening for latent tuberculosis infection is recommended and includes medical history for risk factors, interferon-γ release assay, tuberculin skin test and baseline chest radiograph. 9 In addition to screening for latent tuberculosis and infection followed by anti-tuberculosis therapy if appropriate,10,14,15 screening for chronic hepatitis B virus infection and HIV is also recommended. 16 Antiviral prophylaxis may be warranted for individuals who are positive for hepatitis B surface antigen. 17 Vaccinations, ideally, should be administered during the remission phase of diseases and, in the best case scenario, prior to the initiation of immunosuppression. Live-virus vaccines (e.g. varicella-zoster vaccine or measles-mumps-rubella combined vaccine) may be contraindicated in people receiving anti-TNFs, although additional data are needed before definitive recommendations can be made. In contrast, nonlive vaccines (e.g. influenza and pneumococcal, tetanus toxoid, human papilloma virus) can be safely administered and should be strongly recommended with anti-TNF therapy. Vaccinations in high-risk and pediatric populations should be administered on a case-by-case basis. Caregivers of patients on anti-TNFs should follow national vaccination schedules, with the exception of the oral poliomyelitis vaccine. 13
Anti-TNFs were traditionally contraindicated during pregnancy and lactation, mostly because of a lack of safety data; however, in the last few years, encouraging data support the use of some anti-TNFs in pregnant women with arthritis, leading to the change of the label in some countries. Indeed, a pharmacokinetic study demonstrated the lack of placental transfer of the molecule in pregnant women.18,19 These findings reassured the scientific community and opened a new perspective for pregnant women that, so far, were not treated adequately.
With the introduction of new non-anti-TNFs, new adverse effects have been reported. For example, IL-6 inhibition increases the risk of GI perforation. 20 IL-17 inhibition is burdened by an increased rate of mucocutaneous candidiasis, 21 can exacerbate inflammatory bowel diseases 22 and a higher incidence of suicide has been reported with the use of brodalumab. 23 Another example comes from IL12/IL23 inhibition that likely increases the risk of reversible posterior leukoencephalopathy syndrome. 24
The example of denosumab in osteoporosis
Denosumab was the first biologic used for the prevention of fractures in high-risk patients.
It increases bone mineral density (BMD) and decreases the risk of vertebral and nonvertebral fractures, including hip fractures. A large population-based cohort study showed that denosumab and zoledronic acid have comparable clinical safety and effectiveness with regard to the risk of serious infection, cardiovascular (CV) diseases, and osteoporosis fractures within 365 days after initiation of these medications. 25
In the past decade we have seen an increasing fear of rare adverse events related to antiresorptive drugs, namely osteonecrosis of the jaw and atypical femoral fractures.26,27
In Table 2 we show the potential adverse events related to denosumab treatment and potential approaches to risk mitigation.
Potential adverse events of denosumab.

A new biologic that has been recently approved in the US, South Korea, Canada, Australia and Japan, but not yet in Europe, for the treatment of osteoporosis is romosozumab, a potent bone anabolic agent with some antiresorptive properties. 5 Romosozumab has profound effects on BMD and has been shown to lower vertebral and clinical fracture risk compared with placebo, and even with the active comparator alendronate. 28 This was shown in patients who are at particularly high risk for subsequent fractures, because of the combination of low bone mass (reflected by low BMD by dual-energy X-ray absorptiometry) and low bone quality (reflected by the presence of vertebral fractures). 29 In contrast to the placebo-controlled study, in a head-to-head comparison with alendronate, there was a 0.5% risk difference in myocardial infarction and stroke. 28 With regulatory approval of this new drug for patients with high-risk fractures, there is now a need for new risk minimization strategies around these potential adverse events. For example, excluding those patients with a recent CV event, past major ischemic events, and possibly those with a very high Framingham CV risk score might be appropriate with this drug; however, the CV risk remained low with the use of romosozumab and, as noted, was not different from that of placebo.
It is very useful that regulatory agencies try to optimize the approval of new drugs, based on their efficacy and safety; however, the patient voice is also critical. We are living in the days of shared decision-making, and we can imagine that making treatment decisions around a slightly higher, but relevant CV risk might be different in patients with one or more painful and disabling fractures than in patients with only a low T score.
Conclusion
After more than a decade of experience with biologics in inflammatory rheumatic, skin, and GI diseases and osteoporosis, the benefit–risk ratio of many biologics is generally considered favorable for most of patients with serious inflammatory arthritis or with severe osteoporosis at high fracture risk. Importantly, we have learned how to reduce the risk with the use of our most commonly used biologics. For newer biologics in osteoporosis, such as romosozumab, additional risk mitigation strategies are proposed that could offer similarly favorable benefit–risk ratios for those at the highest risk of fractures.
Footnotes
Acknowledgements
This review does not contain any original data collection or intervention with human participants or animals performed by any of the authors. For this type of review article, a formal consent and ethical board approval is not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Giovanni Adami declares that he has no conflict of interest.
Kenneth G. Saag declares a research grant from Amgen and Merck and a consultant fee from Amgen, Lilly, Merck and Radius and Roche.
Roland D. Chapurlat declares a research grant from Amgen, Merck, Chugai, UCB and a speaker/consultant fee from Amgen, Lilly, UCB, BMS, Abbvie, Pfizer, Arrow, Ultragenyx and Chugai.
Nuria Guañabens declares a speaker/consultant fee from Alexion, Amgen, Eli Lilly and UCB.
Glenn Haugeberg declares a research grant, speaker fees, and/or consultant fees from Amgen Pfizer, UCB, Abbott, Lilly, Novartis, Roche, and Biogen.
Willem F. Lems declares a speaker/consultant fee from Pfizer, Amgen, Eli Lilly and UCB.
Matijevic Radmila declares that she has no conflict of interest.
Nicola Peel declares that she has no conflict of interest.
Denis Poddubnyy declares a research grant from Abbvie, Merck, Novartis, Pfizer and a speaker/consultant fee from Abbvie, BMS, Boehringer, Celgene, Janssen, Lilly, MSD, Novartis, Pfizer, Roche, UCB.
Piet Geusens declares a research grant from Amgen, Pfizer, Merck, UCB, Abbott, Lilly, BMS, Novartis, Roche, Will Pharma and a speaker/consultant fee from Amgen and Lilly.
