Abstract
Osteoporosis is a significant public health concern, particularly for postmenopausal women. Current treatment options may not be appropriate for all women. Selective estrogen-receptor modulators (SERMs) are a class of molecules with tissue-selective activity. Bazedoxifene is currently in clinical development for the prevention and treatment of postmenopausal osteoporosis. In a 2-year, phase III, osteoporosis prevention study (N = 1583), bazedoxifene 10, 20, and 40 mg was shown to preserve bone mineral density and decrease biochemical markers of bone turnover compared with placebo in postmenopausal women at risk for osteoporosis. In a pivotal 3-year, phase III, osteoporosis treatment study (N = 7492), bazedoxifene 20 and 40 mg significantly reduced the incidence of new vertebral fractures compared with placebo (p < 0.05 for both) in postmenopausal women with osteoporosis. In a post hoc subgroup analysis of women at higher risk for fracture (n = 1772), bazedoxifene 20 mg significantly reduced the risk of nonvertebral fractures versus placebo (p = 0.02) and raloxifene 60 mg (p = 0.05). Bazedoxifene 20 mg has demonstrated sustained efficacy in reducing the risk of vertebral fractures over 5 and 7 years. Overall, bazedoxifene was generally safe and well tolerated, with favorable endometrial and breast safety profiles. As with other SERMs, the rate of deep vein thrombosis was higher in the bazedoxifene groups compared with placebo at 3 and 5 years. Considering its demonstrated efficacy and safety, bazedoxifene may be an appropriate osteoporosis therapy for women who cannot take or are unwilling to take bisphosphonates because of safety or tolerability issues. Bazedoxifene may also be appropriate for younger women at increased fracture risk who are concerned about the effects of long-term bisphosphonate therapy. This article reviews the results of key clinical trials of bazedoxifene for the prevention and treatment of postmenopausal osteoporosis and describes its role in clinical practice.
Keywords
Introduction
Osteoporosis-related fractures are a significant public health concern that contribute to considerable costs both economically and in terms of morbidity and mortality [National Osteoporosis Foundation, 2010]. It has been estimated that 9 million new osteoporotic fractures occurred globally each year [Johnell and Kanis, 2006]. Postmenopausal women are at increased risk of osteoporosis due to a decline in the production of endogenous estrogens [Gambacciani and Vacca, 2004], which may lead to increased bone remodeling, disordered bone architecture, and an increased risk of fractures [National Osteoporosis Foundation, 2010]. Currently approved pharmacologic prevention and/or treatment options for osteoporosis include bisphosphonates, estrogens, selective estrogen-receptor modulators (SERMs), parathyroid hormone, calcitonin, strontium ranelate (outside of North America), and denosumab [Amgen, 2010a, 2010b; North American Menopause Society, 2010]. Despite the availability of numerous therapies, many women at risk for or diagnosed with osteoporosis are not receiving appropriate therapy [National Osteoporosis Foundation, 2010].
SERMs are a class of molecules that exhibit estrogen receptor (ER) agonist or antagonist activity depending on the tissue they are targeting. An ‘ideal’ SERM would act as an ER agonist by exerting a protective effect on bone and improving lipid parameters, while also acting as an ER antagonist by maintaining breast and endometrial safety [Komm and Lyttle, 2001]. Bazedoxifene (BZA) is a novel SERM that is currently being developed for the prevention and treatment of postmenopausal osteoporosis; it was recently approved in the European Union for the treatment of osteoporosis in postmenopausal women at increased risk of fracture [Pfizer, 2011]. BZA was initially chosen for development following a rigorous preclinical selection process designed to identify candidate molecules based on the criteria for an ‘ideal’ SERM described above [Komm et al. 2001]. BZA is chemically distinct from other SERMs [Lewiecki, 2007] and has demonstrated a unique profile in preclinical evaluations [Komm et al. 2005]. Ovariectomized rats treated with BZA showed significant increases in bone mineral density (BMD) and compressive bone strength compared with controls [Komm et al. 2005]. In morphine-addicted rats, a well accepted preclinical model used to evaluate vasomotor instability [Merchenthaler et al. 1998], BZA did not have a vasomotor effect as assessed by changes in the rat’s tail temperature. In an immature rat model, BZA did not result in significant increases in uterine wet weight or any histological changes, suggesting that BZA did not have stimulatory estrogenic effects on the uterus. Finally, BZA did not stimulate Michigan Cancer Foundation-7 (MCF-7) cells, a well established ER-positive human breast cancer cells line, and also inhibited stimulation of these cells by 17 β-estradiol [Komm et al. 2005].
The objective of this article is to review the key findings from the global clinical studies of BZA, including the two extensions of the pivotal osteoporosis treatment study to 5 and 7 years. We also describe clinical findings of BZA partnered with conjugated estrogens (CEs), a new menopausal therapy known as a tissue selective estrogen complex (TSEC), and outline the potential role of BZA in clinical practice.
Evidence from clinical trials of bazedoxifene
Phase II studies
Early clinical findings in healthy, postmenopausal women (N = 494) who were treated with BZA 5 mg for 3 months [Ronkin and Clarke, 2001] demonstrated significant reductions in markers of bone remodeling. In a 6-month study (N = 497), all doses of BZA evaluated (2.5, 5.0, 10, 20, 30, and 40 mg) were shown to be safe and well tolerated in healthy postmenopausal women [Ronkin et al. 2005]. BZA 2.5, 5, 10, and 20 mg were not associated with changes in endometrial thickness from baseline compared with placebo; women treated with BZA 30 or 40 mg had smaller changes from baseline in endometrial thickness than those given placebo. In a 2-year study of postmenopausal Japanese women with osteoporosis (N = 429), daily treatment with BZA 20 and 40 mg led to significant improvements in BMD at the lumbar spine, total hip, femoral neck, and greater trochanter compared with placebo [Itabashi et al. 2011]. BZA 20 and 40 mg were also associated with significant changes from baseline in markers of bone turnover, and both doses were well tolerated.
Phase III osteoporosis prevention study
BZA has been evaluated in two global, randomized, double-blind, placebo- and active-controlled, phase III studies (Table 1). The 2-year osteoporosis prevention study was conducted in healthy postmenopausal women (N = 1583; mean age, 57.6 ± 6.5 years) who were at risk for osteoporosis, defined as BMD T-score at the lumbar spine or femoral neck between −1.0 and −2.5 or other clinical risk factors [Miller et al. 2008]. Women were randomly assigned to daily oral doses of BZA 10, 20, or 40 mg, raloxifene (RLX) 60 mg, or placebo. All women were supplemented daily with 600 mg elemental calcium. Among the intent-to-treat (ITT) population (n = 1434), all doses of BZA and RLX 60 mg were associated with maintenance of BMD, while placebo showed significant bone loss at all skeletal sites. At 2 years, the mean percent change from baseline in lumbar spine BMD compared with placebo was 1.08 ± 0.28% for BZA 10 mg, 1.41 ± 0.28% for BZA 20 mg, 1.49 ± 0.28% for BZA 40 mg, and 1.49 ± 0.28% for RLX 60 mg (p < 0.001 versus placebo for all). Similar improvements in BMD versus placebo were observed at 2 years with all doses of BZA at the total hip (p < 0.001) and with BZA 20 and 40 mg at the femoral neck (p < 0.01) and femoral trochanter (p < 0.001) [Miller et al. 2008].
Study designs for phase III clinical trials of bazedoxifene.

BMD, bone mineral density; BZA, bazedoxifene; CTx, C-telopeptide; DXA, dual-energy X-ray absorptiometry; OC, osteocalcin; RLX, raloxifene.
BZA 10, 20, and 40 mg were also associated with significantly greater reductions from baseline in serum osteocalcin and C-telopeptide (CTx) levels compared with placebo (p < 0.001) at all time points evaluated through 2 years of treatment. Reductions from baseline in serum osteocalcin at 2 years with BZA 10, 20, and 40 mg and RLX 60 mg (−21%, −22%, −22%, and −27%, respectively) were significantly lower compared with placebo (−6%; p <0.001 for all). Similarly, reductions from baseline in the bone resorption marker serum CTx at 2 years with BZA 10, 20, and 40 mg and RLX 60 mg (−25%, −24%, −22%, and −32%, respectively) were significantly lower compared with placebo (−13%; p < 0.001 for all).
BZA also showed improvements in lipid parameters compared with placebo [Miller et al. 2008]. Compared with placebo, total cholesterol significantly decreased with BZA 10 and 40 mg (p < 0.05), low-density lipoprotein (LDL) cholesterol significantly decreased with all doses of BZA (p < 0.05), and high-density lipoprotein (HDL) cholesterol significantly increased with BZA 10 and 20 mg (p < 0.05) at 2 years. Triglyceride levels significantly increased from baseline in the BZA 20 and 40 mg groups (p < 0.001 versus baseline for both), but this was also the case in the placebo group (p < 0.01 versus baseline), and all values remained within the normal treatment range [Miller et al. 2008].
BZA 10, 20, and 40 mg were generally safe and well tolerated. The incidences of adverse events (AEs), serious AEs, and withdrawals due to AEs were similar among women treated with BZA, RLX, or placebo. The incidence of hot flushes in the BZA groups (20–24%) and the RLX 60 mg group (19%) was higher than that in the placebo group (14%; overall p < 0.05). There were no cardiovascular safety concerns noted in this study. The incidence of breast cancer (n = 1 for BZA 10 mg; n = 2 for BZA 20 mg; n = 0 for BZA 40 mg; n = 1 for RLX 60 mg; n = 2 for placebo) was low and similar among treatment groups. None of the BZA doses were associated with significant changes from baseline in mean endometrial thickness at 2 years, and there were no consensus diagnoses of endometrial hyperplasia or carcinoma in any treatment group [Pinkerton et al. 2009a; Miller et al. 2008].
Phase III osteoporosis treatment study
The pivotal phase III treatment study of BZA was conducted in healthy postmenopausal women (N = 7492; mean age, 66.4 ± 6.7 years) with osteoporosis, defined as BMD T-score at lumbar spine or femoral neck between −2.5 and −4.0 without prevalent vertebral fracture or greater than −4.0 with prevalent vertebral fracture (Table 1) [Silverman et al. 2008]. Women were randomly assigned to daily oral doses of BZA 20 or 40 mg, RLX 60 mg, or placebo. All women were supplemented with daily oral calcium (up to 1200 mg) and vitamin D (400–800 IU). Among 6847 women in the modified ITT population, the percentage of women with new vertebral fractures at 3 years (Kaplan–Meier estimates) was significantly lower with BZA 20 mg (2.3%), BZA 40 mg (2.5%), and RLX 60 mg (2.3%) compared with placebo (4.1%; p < 0.05; Table 2 and Figure 1). These incidences corresponded to risk reductions relative to placebo of 42% [hazard ratio (HR) 0.58; 95% confidence interval (CI) 0.38–0.89] for BZA 20 mg, 37% (HR 0.63; 95% CI 0.42–0.96) for BZA 40 mg, and 42% (HR 0.58; 95% CI 0.38–0.89) for RLX 60 mg. A subgroup analysis showed that the treatment effect was similar (p = 0.89) among women with or without prevalent fracture [Silverman et al. 2008].
Efficacy results of the phase III treatment study [Silverman et al. 2008].

BMD, bone mineral density; BZA, bazedoxifene; CTx, C-telopeptide; NR, not reported; OC, osteocalcin; RLX, raloxifene; RRR, relative risk reduction; SE, standard error.
Incidence of new vertebral fractures, Kaplan–Meier estimate (RRR versus placebo) at 5 years [Silverman et al. 2011]: BZA 20 mg, 4.5%$ (35%); BZA 40/20 mg, 3.9%§ (40%); placebo, 6.8%.
p < 0.05 versus placebo.
p < 0.001 versus placebo.
p < 0.01 versus placebo.

Incidence of new vertebral fractures at 3 years in the phase III osteoporosis treatment study. Kaplan–Meier fracture rates (%) were determined at 3 years for the incidence of new vertebral fractures in the intent-to-treat population (n = 6847). Adapted from [Silverman et al. 2008], ©2008 American Society for Bone and Mineral Research, with permission from John Wiley & Sons, Inc. BZA, bazedoxifene; CI, confidence interval; HR, hazard ratio; RLX, raloxifene; RRR, relative risk reduction. *p < 0.05 versus placebo. $p = 0.89 for treatment by prevalent fracture status interaction.
There was no significant difference among treatment groups in the overall incidence of nonvertebral fractures after 3 years, with incidences of 5.7%, 5.6%, 5.9%, and 6.3% for BZA 20 and 40 mg, RLX 60 mg, and placebo, respectively (Table 2). Nonvertebral fractures were further evaluated in a post hoc analysis of a subgroup of women at higher risk for fracture (n = 1772), defined as femoral neck T-score of up to −3.0 and/or at least one moderate or severe vertebral fracture or at least two mild vertebral fractures at baseline. In this higher-risk subgroup, BZA 20 mg was associated with a 50% reduction in nonvertebral fracture risk relative to placebo (HR 0.50; 95% CI 0.28–0.90; p = 0.02) and 44% relative to RLX 60 mg (HR 0.56; 95% CI 0.31–1.01; p = 0.05; Figure 2). When the data for both BZA treatment groups were combined, the incidence of nonvertebral fractures was 40% lower compared with placebo (HR 0.60; 95% CI 0.37–0.95; p = 0.03) [Silverman et al. 2008].

Incidence of nonvertebral fractures among women at higher risk for fracture at 3 years in the phase III osteoporosis treatment study. Kaplan–Meier fracture rates (%) at 3 years were determined for the incidence of nonvertebral fractures in a subgroup of women at higher risk for fracture (n = 1772; defined as a femoral neck T-score ≤ 3.0 and/or ≥1 moderate or severe vertebral fracture or ≥2 mild fractures). Adapted from [Silverman et al. 2008], ©2008 American Society for Bone and Mineral Research, with permission from John Wiley & Sons, Inc. BZA, bazedoxifene; RLX, raloxifene; RRR, relative risk reduction. *Relative to placebo.$Relative to RLX 60 mg.
Because the reduction in nonvertebral fracture risk for higher-risk women with BZA was shown using a post hoc analysis, there are limitations to the interpretation of these results. The efficacy of BZA on fractures in the pivotal phase III study was independently re-evaluated using the Fracture Risk Assessment Tool (FRAX) [Kanis et al. 2009; McCloskey et al. 2009], an algorithm that calculates the 10-year probability of fracture based on easily obtained clinical risk factors with or without femoral neck BMD [Kanis et al. 2008]. These analyses evaluated the effect of BZA on fracture incidence as a function of fracture risk using combined BZA data (20 and 40 mg doses) from the overall study population [Kanis et al. 2009; McCloskey et al. 2009]. Results showed that the efficacy of BZA increased with increasing fracture probability. The risk of all clinical fractures was significantly reduced with BZA for women with 10-year fracture probabilities at or above 16% (80th percentile of risk); for morphometric vertebral fractures, BZA significantly reduced the risk in women with 10-year fracture probabilities at or above 6.9% (41st percentile of risk) [Kanis et al. 2009]. In a similar analysis of nonvertebral fractures based on FRAX, BZA was shown to effectively decrease the risk of nonvertebral fractures in women with a 20% or higher 10-year probability of fracture [McCloskey et al. 2009]. These findings provide support for the results of the post hoc analysis in the phase III treatment study that showed a significant reduction in nonvertebral fractures in higher-risk women treated with BZA [Silverman et al. 2008].
Changes in BMD and bone turnover were also evaluated in the phase III treatment study (Table 2). The mean percent changes from baseline in lumbar spine BMD at 3 years were significantly greater for BZA 20 mg (2.21 ± 0.16%), BZA 40 mg (2.38 ± 0.16%), and RLX 60 mg (2.96 ± 0.16%) compared with placebo (0.88 ± 0.16%; p < 0.001 for all). Responses at the total hip were also significantly greater for BZA 20 mg (0.27 ± 0.12%), BZA 40 mg (0.50 ± 0.12%), and RLX 60 mg (0.90 ± 0.12%) compared with placebo (−0.83 ± 0.12%; p < 0.001 for all). Markers of bone turnover were significantly reduced in women treated with BZA or RLX versus placebo (p < 0.001). The median percent reductions from baseline in serum osteocalcin at 1 year for BZA 20 and 40 mg and RLX 60 mg (−37%, −39%, and −41%, respectively) were significantly lower compared with placebo (−21%; p < 0.001 for all). The median percent reductions from baseline in serum CTx for BZA 20 and 40 mg and RLX 60 mg (−46%, −49%, and −55%, respectively) were also significantly lower compared with placebo (−27%; p < 0.001 for all) [Silverman et al. 2008].
BZA was associated with lipid profile improvements over 3 years [Christiansen et al. 2010]. Total cholesterol and LDL cholesterol were significantly decreased from baseline with BZA 20 and 40 mg and RLX 60 mg (p < 0.05 versus placebo), and HDL cholesterol was significantly increased with BZA 20 and 40 mg and RLX 60 mg (p < 0.05 versus placebo). Although triglyceride levels significantly increased from baseline in all treatment groups (p < 0.001 versus baseline), there were no significant differences among groups.
BZA 20 and 40 mg were shown to be generally safe and well tolerated over 3 years of treatment (Table 3) [Christiansen et al. 2010; Archer et al. 2009b; Silverman et al. 2008]. The overall incidences of AEs, serious AEs, and withdrawals due to AEs were similar among treatment groups. As in the prevention study, the incidence of hot flushes was significantly greater for women treated with BZA 20 mg (12.6%), BZA 40 mg (13.0%), and RLX 60 mg (12.0%) compared with placebo (6.3%; overall p < 0.001). The incidence of cardiovascular AEs was low and similar among groups. There was no difference among groups in the incidence of stroke (Table 4). There was a significantly higher incidence of deep vein thrombosis (DVT) in the BZA and RLX groups [n = 8 (0.4%) for BZA 20 mg, n = 10 (0.5%) for BZA 40 mg, and n = 8 (0.4%) for RLX 60 mg] compared with the placebo group [n = 1 (0.1%); overall p < 0.05]. There were numerically fewer breast cancer cases in the BZA groups [n = 5 (0.3%) for BZA 20 mg and n = 4 (0.2%) for BZA 40 mg] than in the placebo group [n = 8 (0.4%)]; this study was not powered to evaluate statistical significance among these events. The incidences of endometrial carcinoma and endometrial hyperplasia were low and similar among treatment groups [Christiansen et al. 2010; Archer et al. 2009b; Silverman et al. 2008].
Safety results of the phase III treatment study.

AE, adverse event; BZA, bazedoxifene; RLX, raloxifene.
Overall p < 0.001 at both 3 and 5 years (chi-square test).
Overall p < 0.01 at both 3 and 5 years (chi-square test).
Includes one case with breast lesion present at baseline and diagnosed later on in the study.
Overall p = 0.05 at 5 years (chi-square test).
Rate of cerebrovascular and venous thromboembolic events in the phase III treatment study.*
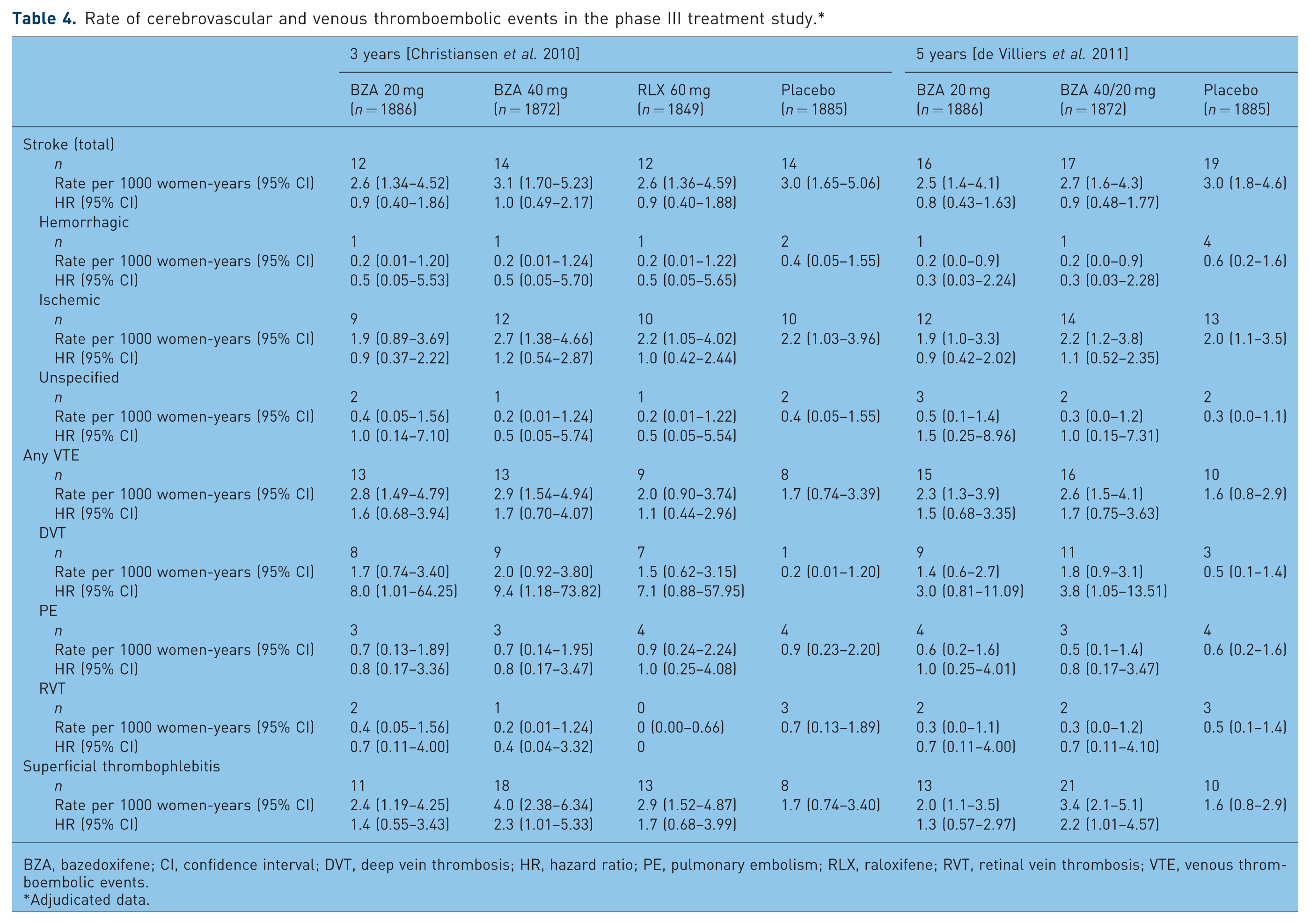
BZA, bazedoxifene; CI, confidence interval; DVT, deep vein thrombosis; HR, hazard ratio; PE, pulmonary embolism; RLX, raloxifene; RVT, retinal vein thrombosis; VTE, venous thromboembolic events.
Adjudicated data.
Two consecutive 2-year extensions of the 3-year core treatment study have been completed [de Villiers et al. 2011; Palacios et al. 2011; Silverman et al. 2011]. A total of 4216 women were enrolled in the first 2-year extension (years 4–5) [Silverman et al. 2011], and 1732 women were enrolled in the subsequent 2-year extension (years 6–7). During the first extension, women receiving BZA 40 mg were transitioned to BZA 20 mg (BZA 40/20 mg group), and women receiving RLX 60 mg were discontinued after 4 years. During the second extension, women who previously received BZA 40 mg in the core study continued to receive BZA 20 mg. Supplementation with elemental calcium (1000–1200 mg/day) and vitamin D (400–800 IU/day) was continued for all women. The significant reduction in new vertebral fractures with BZA 20 mg was sustained through 5 and 7 years. Reductions in the risk of new vertebral fracture with BZA 20 mg versus placebo, based on Kaplan–Meier estimates, were 35% (p = 0.014) and 30% (p = 0.022) at 5 and 7 years, respectively. Consistent with the results at 3 years, the incidence of nonvertebral fractures at 5 and 7 years was similar among treatment groups in the overall population. Nonvertebral fracture results at 5 years were further analyzed in a subgroup of women at higher risk of fracture (n = 1324) using the same criteria as for 3 years (baseline femoral neck T-score of up to −3.0 and/or at least one moderate or severe vertebral fracture or at least two mild vertebral fractures at baseline). In this higher-risk subgroup, there was a 37% reduction in the risk of nonvertebral fracture with BZA 20 mg (p = 0.06); combined BZA data showed a 34% reduction versus placebo (p = 0.05) [Silverman et al. 2011].
The safety and tolerability profile of BZA at 5 and 7 years was similar to that at 3 years (Table 3) [de Villiers et al. 2011; Palacios et al. 2011; Christiansen et al. 2010]. The overall incidence of AEs, serious AEs, and discontinuations due to AEs was similar among treatment groups at 5 and 7 years. Consistent with what was seen at 3 years, the incidence of hot flushes was significantly greater for women treated with BZA than with placebo at 5 and 7 years (p < 0.001). The risk of venous thromboembolic events associated with BZA treatment at 5 and 7 years was similar to the risk at 3 years (Table 4). The rate of DVTs per 1000 women-years at 5 years was significantly higher for women treated with BZA 20 mg (1.4; 95% CI 0.6–2.7) and BZA 40/20 mg (1.8; 95% CI 0.9–3.1) than placebo (0.5; 95% CI 0.1–1.4). BZA was associated with a favorable breast and endometrial safety profile. There was no difference in the incidence of breast cancer among the BZA and placebo groups at 5 years. At 5 years, there was a lower incidence of endometrial carcinoma with BZA 20 mg (n = 0) and BZA 40/20 mg (n = 3) than with placebo (n = 6; overall p = 0.05). The change from baseline in endometrial thickness at 5 years was similar for BZA 20 mg (−0.04 ± 0.13 mm), BZA 40/20 mg (0.07 ± 0.13 mm), and placebo (−0.19 ± 0.13 mm).
Other phase III studies
Changes in mammographic breast density were evaluated at 2 years in a subset of women enrolled in the phase III osteoporosis treatment study (N = 444) [Harvey et al. 2009]. Women eligible for participation in this study were up to 62 years old, had completed 2 years of treatment with BZA 20 or 40 mg, RLX 60 mg, or placebo, and had mammogram pairs taken at baseline and 2 years. At 2 years, the mean changes from baseline in mammographic breast density for BZA 20 mg (−1.2%), BZA 40 mg (−0.4%), RLX 60 mg (−0.5%), and placebo (−0.2%) were small and not significantly different among treatment groups.
BZA has also been evaluated in a 6-month, randomized, double-blind, placebo-controlled, phase III study of postmenopausal Asian women at risk of osteoporosis (N = 486; mean age 57.2 years) in China, Korea, and Taiwan [Xu et al. 2011]. Women were randomly assigned to daily oral doses of BZA 20 mg or placebo and supplemented with calcium carbonate 600 mg daily. BZA 20 mg was associated with significantly greater lumbar spine BMD (0.41%) compared with placebo (−0.32%; p < 0.01) at 6 months. Similar results were observed at the femoral neck (−0.08% versus −0.69%; p = 0.014), femoral trochanter (0.50% versus −0.23%; p = 0.010), and total hip (−0.03% versus −0.77%; p < 0.001). At 6 months, the median percent reductions from baseline in serum osteocalcin and C-telopeptide in women treated with BZA 20 mg (−19.2% and −30.6%, respectively) were significantly lower than those in women given placebo (−6.3%, p < 0.001 and −8.8%, p < 0.001, respectively). The overall incidence of AEs was similar among the BZA and placebo groups.
The tissue selective estrogen complex
The TSEC is a new menopausal therapy that combines a SERM with one or more estrogens, the goal of which is to optimize the tissue−selective activity of each component. An ideal TSEC would treat menopausal symptoms (e.g. hot flushes, vulvar/vaginal atrophy), prevent osteoporosis, and improve lipid parameters, while maintaining breast and endometrial safety [Komm, 2008]. The first TSEC in clinical development partners BZA with CE. The phase III Selective estrogens, Menopause, And Response to Therapy (SMART)-1 trial evaluated various doses of BZA/CE in generally healthy, postmenopausal women with a uterus [Archer et al. 2009a; Lindsay et al. 2009; Lobo et al. 2009; Pickar et al. 2009]. At 2 years, all doses of BZA/CE (BZA 10, 20, or 40 mg/CE 0.45 or 0.625 mg) were associated with significant improvements in BMD and decreases in bone turnover compared with placebo in women at risk for osteoporosis [Lindsay et al. 2009]. Also, treatment with BZA 20 mg/CE 0.45 and 0.625 mg significantly reduced the number and severity of hot flushes at 12 weeks and improved measures of vaginal atrophy at 2 years in subsets of symptomatic women. Lipid parameters were also improved with all doses of BZA/CE [Lobo et al. 2009]. In addition to demonstrating efficacy in treating menopausal symptoms and protecting against bone loss, BZA 20 mg/CE 0.45 and 0.625 mg were associated with a low incidence of endometrial hyperplasia and high rates of cumulative amenorrhea, similar to those with placebo [Archer et al. 2009a; Pickar et al. 2009].
BZA/CE has also been evaluated in other phase III studies (SMART-2, SMART-3 trials) in larger populations of symptomatic postmenopausal women. In the SMART-2 trial (N = 332) of postmenopausal women with moderate to severe vasomotor symptoms, BZA 20 mg/CE 0.45 and 0.625 mg significantly reduced the number (p < 0.001) and severity (p < 0.001) of hot flushes compared with placebo at 12 weeks. More women in the BZA 20 mg/CE 0.45 mg (61%) and BZA 20 mg/CE 0.625 mg (73%) groups than in the placebo group (27%; p < 0.001) experienced at least a 75% reduction in the number of moderate to severe hot flushes at 12 weeks. BZA/CE was also associated with significantly greater treatment satisfaction (p < 0.001 versus placebo) and improvements in sleep parameters (time to fall asleep, sleep adequacy, sleep disturbance, and sleep problem indices I and II; p < 0.001 versus placebo) [Pinkerton et al. 2009b]. In the SMART-3 trial (N = 664) of postmenopausal women with moderate to severe vulvar/vaginal atrophy, BZA 20 mg/CE 0.45 and 0.625 mg significantly increased the percentage of superficial cells (p < 0.01), significantly decreased the percentage of parabasal cells (p < 0.01), significantly reduced vaginal pH, and significantly improved vaginal dryness and reports of most bothersome vaginal symptom compared with placebo (p < 0.05). These findings for the four co-primary endpoints of the SMART-3 trial all indicate improvement in the symptoms of vulvar/vaginal atrophy with BZA/CE [Kagan et al. 2010]. Women treated with BZA/CE also had significant improvements from baseline in measures of sexual function and quality of life compared with placebo, and were more satisfied with treatment than with placebo [Bachmann et al. 2010].
The role of bazedoxifene in clinical practice
The first-line treatment choice for postmenopausal osteoporosis is generally considered to be a bisphosphonate [North American Menopause Society, 2010]. The efficacy of bisphosphonates in reducing the risk of vertebral and nonvertebral fractures has been clearly demonstrated [Hochberg, 2008], and although head-to-head trials have not been undertaken, analyses conducted using observational databases suggest that the common bisphosphonates (alendronate, risedronate, ibandronate, zoledronic acid) generally display similar vertebral antifracture efficacies, though differences have been observed at other skeletal sites (e.g. ibandronate has not been shown to be effective against hip fractures) [Harris et al. 2009; Pazianas et al. 2009; Silverman et al. 2007; Liberman et al. 2006]. Bisphosphonates may be administered orally based on several different dosing schedules or intravenously every 3 months or yearly.
Some concerns associated with bisphosphonates, however, may limit their utility for certain women. Oral dosing can be complex, requiring both a pre- and post-dose fast for appropriate absorption, with the post-dose fast in an upright position [Cole et al. 2008]. The complex dosing schedules contribute to low rates of compliance and persistence [Silverman and Gold 2008]. Bisphosphonate use may also present significant safety and tolerability concerns for many women. Gastrointestinal intolerability, ranging from mild acid reflux to serious esophageal ulcers, is a common concern [MacLean et al. 2008]. Intravenous administration of bisphosphonates may be accompanied by acute phase reaction symptoms, although they are generally mild to moderate in severity and become less frequent with repeated intravenous infusions [Black et al. 2007; Bobba et al. 2006]. Bisphosphonates have also been associated with reports of acute renal failure [Strampel et al. 2007; Bobba et al. 2006]. Long-term safety considerations with bisphosphonates include a possible increased risk of atypical fractures [Dell et al. 2010; Shane et al. 2010; Schneider, 2009] and a possible risk of osteonecrosis of the jaw, particularly among patients with cancer receiving high-dose bisphosphonates for bone metastases [Silverman and Landesberg 2009; Khosla et al. 2007; Strampel et al. 2007].
SERMs are an appropriate alternative for women who cannot take or choose not to take bisphosphonates because of the considerations described above. Because women who experience gastrointestinal intolerability with one bisphosphonate are likely to experience similar issues with other bisphosphonates [Ideguchi et al. 2008], SERMs such as BZA may be an option for these women. Women who cannot take bisphosphonates due to renal insufficiency may be candidates for SERMs. SERMs may also be appropriate for women who cannot adhere to the dosing requirements for bisphosphonates.
Clinical data to date indicate that BZA, which has demonstrated a favorable efficacy and safety profile over 7 years, may be an appropriate pharmacological therapy for certain women. Based on its efficacy on nonvertebral fractures as seen in subgroup analyses, BZA may be considered a first-choice treatment in younger women at increased risk of fracture, particularly those who are likely to remain on therapy for many years and may be concerned about the long-term safety of bisphosphonates. Findings from the post hoc analysis of nonvertebral fracture data from the phase III osteoporosis treatment study [Silverman et al. 2008] are consistent with the results of the independent reanalyses based on FRAX [Kanis et al. 2009; McCloskey et al. 2009]. Together, these data provide support for the efficacy of BZA in women who are at higher risk of fracture based on known skeletal risk factors.
Conclusions
Osteoporosis presents a significant public health challenge. As the population of postmenopausal women increases, the development of new treatment options for osteoporosis is becoming increasingly important. BZA is a promising therapy for postmenopausal osteoporosis based on its favorable safety and tolerability profile and efficacy in preventing bone loss and fractures. SERMs such as BZA may be an appropriate therapy for women who cannot or choose not to take bisphosphonates due to gastrointestinal intolerability, safety concerns, or contraindications. SERMs may also be appropriate for younger women at increased risk of fractures who expect to remain on therapy for many years. BZA has also been paired with CE (TSEC) for the prevention of osteoporosis and treatment of menopausal symptoms. Considering its favorable safety and tolerability profile with demonstrated efficacy in preventing fractures, the role of BZA among currently available osteoporosis treatments is emerging.
Footnotes
Acknowledgements
Medical writing support for this manuscript was provided by Katie Gersh, PhD, of MedErgy. The authors retained full editorial control over the content of the article.
Funding for editorial assistance was provided by Pfizer Inc, Collegeville, PA.
Drs Komm and Chines are employees of Pfizer Inc.
