Abstract
Background:
The antifracture efficacy of ibandronate at vertebral and nonvertebral sites was assessed.
Methods:
A literature review of randomized phase III clinical trials, meta-analyses or observational studies that reported fracture endpoints or surrogate markers, and compared ibandronate with placebo or an active comparator.
Results:
In a phase III study, 2.5 mg daily oral ibandronate reduced the incidence of new vertebral fractures versus placebo and the relative risk reduction (RRR) was sustained over 3 years (62%; p = 0.0001). In two bridging studies, oral ibandronate 150 mg once monthly and 3 mg quarterly intravenous (i.v.) were superior to oral 2.5 mg daily in producing bone mineral density (BMD) increases at all sites over 2 years (p < 0.05). These improvements were sustained over 5 years. In meta-analyses of pivotal ibandronate studies, doses equivalent to annual cumulative exposure (ACE) ≥ 10.8 mg (including 150 mg once monthly and 3 mg quarterly i.v.) significantly reduced the incidence of nonvertebral fractures versus placebo or ACE 5.5 mg (2.5 mg daily) (RRR 29.9% and 38%, respectively; p < 0.05). Therefore, prevention of nonvertebral fractures was found in all patients with the commercially available highest doses, and not only in high-risk patients as observed in randomized clinical trials with lower doses. Finally, a 12-month, observational study of claims databases reported comparable rates of nonvertebral fractures and a statistically significantly lower rate of vertebral fractures (p < 0.01) with ibandronate versus weekly bisphosphonates.
Conclusion:
A large body of evidence suggests that ibandronate has sustained vertebral and nonvertebral antifracture efficacy in women with postmenopausal osteoporosis.
Keywords
Introduction
Fractures are the key clinical feature of postmenopausal osteoporosis and their prevention in the treatment of the disease is of vital importance due to the negative clinical, social and economic implications osteoporotic fractures have for patients and healthcare systems [Johnell and Kanis, 2006]. Clinically, osteoporotic fractures are associated with increased morbidity and mortality [Bliuc et al. 2009; Melton, 2003; Ismail et al. 1998; Browner et al. 1996; Chrischilles et al. 1991]. In a study by Johnell and Kanis, an estimated 740,000 deaths globally were associated with hip fracture and 1.75 million quality-adjusted life years globally were estimated to have been lost due to the condition [Johnell and Kanis, 2004]. Vertebral and hip fractures are associated with a number of medical conditions that impact negatively on patient health and wellbeing, including kyphosis, height loss, back pain and reduced ability to walk [Cummings and Melton, 2002]. Furthermore, the occurrence of a vertebral fracture substantially increases the likelihood and severity of further fractures [Johnell and Kanis, 2005]. This can create a ‘fracture cascade’, whereby patients who suffer an initial vertebral fracture then continue to suffer from further fractures thereafter, although the full mechanisms underlying this phenomenon are still not fully understood [Briggs et al. 2007]. Time from fracture is an important predictor of future fracture risk. In a study by Giangregorio and Leslie, major and minor fractures both showed a time-dependent decline in importance as risk factors [Giangregorio and Leslie, 2010]. Disabilities caused by osteoporotic fracture, such as loss of mobility or back pain, can also impact upon a patient's health-related quality of life [Rabenda et al. 2007; Tosteson et al. 2001; Oleksik et al. 2000]. Therefore, maintaining fracture protection over the long term is crucial to maintain lifestyle.
As well as the clinical issues associated with osteoporotic fractures, their increasing occurrence brings with it economic implications. With the global ageing population, the incidence of osteoporotic fractures is expected to increase further in the future. In addition, many patients with postmenopausal osteoporosis are relatively young and are increasingly treated with antifracture therapy for more than 5 years [Geusens, 2009]. As a result, increased financial burdens will be placed upon healthcare systems to treat, care for and rehabilitate patients with such fractures [Melton et al. 2004].
The nitrogen-containing bisphosphonates have proven antifracture efficacy for the treatment of postmenopausal osteoporosis [Black et al. 2007, 1996; Chesnut et al. 2004; Harris et al. 1999]. As such, they are the preferred treatment option for this disease [Sambrook and Cooper, 2006]. Here, we review the antifracture efficacy of ibandronate over time, which at marketed doses of 150 mg once monthly oral and 3 mg quarterly intravenous (i.v.), is licensed for the treatment of postmenopausal osteoporosis. Four different levels of supportive evidence from trials are utilized in this review: information from randomized clinical trials, from meta-analyses of clinical trials, clinical trials using surrogate markers and observational studies. The most reliable way of gathering evidence of antifracture efficacy is directly from randomized clinical trials, which contain large patient cohorts and are designed specifically to control for potential biases. However, it is becoming ethically unacceptable to perform randomized controlled trials versus placebo in populations at high risk for fracture [Brody et al. 2003; Temple, 2003]. This is particularly true for the incidence of nonvertebral fractures, where the therapeutic effect is masked by a greater proportion of true traumatic fractures than for vertebral fractures. Therefore, it is becoming increasingly important to include further sources of evidence, e.g. from meta-analyses of randomized controlled trials and indirect evidence from surrogate endpoints for fracture. Finally, observational database studies comparing ibandronate to other nitrogen-containing bisphosphonates are reviewed.
A statistically significant linear relationship between bone mineral density (BMD) changes at the spine and hip and relative risk reduction (RRR) in nonvertebral fracture was shown in a meta-analysis of controlled trials [Hochberg et al. 2002]; as BMD values increased at the spine and hip the risk of fractures decreased (p = 0.02 and p = 0.006, for spine and hip, respectively). Individual trials have also demonstrated the correlation between BMD changes and antifracture efficacy. Analysis from the Fracture Intervention Trial (FIT) showed that patients who had ⩾3% increases in total hip BMD after 12 months of alendronate therapy were at the lowest risk of new vertebral fracture (3.2% versus 6.3% of patients whose BMD levels remained stable or declined) [Hochberg et al. 1999]. Similar results were reported after 24 months and for spine BMD at the same time points. Analysis of data from two studies of ibandronate showed similar results [Adami et al. 2004]. There is also evidence that patients treated with antiresorptive therapies with a loss or no change in BMD may still achieve a reduction in fracture risk versus placebo [Cummings et al. 2002; Wasnich and Miller, 2000]. However, patients who gain BMD with antiresorptive therapy achieve greater reductions in vertebral fracture risk than in those who have a decrease in BMD.
Reductions in bone turnover markers (BTMs) are also regarded as valid surrogate endpoints for antifracture efficacy. A relationship between reductions in BTMs and fracture incidence has also been reported [Hochberg et al. 2002]: as BTM decreases, fracture incidence decreases [Bauer et al. 2004; Eastell et al. 2003]. Overall, a 70% reduction in bone resorption markers and a 50% reduction in bone formation markers significantly reduced nonvertebral fracture risk by 40% (p = 0.047) and 44% (p = 0.009), respectively [Hochberg et al. 2002].
In this review, the long-term antifracture protection provided by ibandronate treatment for postmenopausal osteoporosis was assessed by reviewing direct (fracture endpoints) and indirect (surrogate marker endpoints) evidence of anti-fracture efficacy from multiple sources.
Methods
A literature search was carried out using the PubMed online scientific citation database of published, peer-reviewed manuscripts up to and including 12 February 2010. The keywords used for the literature search were ibandronate, post-menopausal osteoporosis, fracture and BMD.
To meet the inclusion criteria manuscripts had to be primary, peer-reviewed publications, in English, which reported either fracture endpoints or surrogate marker endpoints comparing ibandronate (oral or i.v.) with placebo or an active comparator. The results were then further assessed to include only phase III clinical trials, meta-analyses or observational studies. The literature search results were supplemented with any study data that was known to the authors.
Results
A manual review of the search results identified 14 papers that met the prespecified criteria for inclusion; 11 papers reporting data from five randomized clinical trials and two long-term extension (LTE) studies [Eisman et al. 2008; Stakkestad et al. 2008; Delmas et al. 2006, 2004; Reginster et al. 2006; Chesnut et al. 2005, 2004; Felsenberg et al. 2005; Miller et al. 2005; Adami et al. 2004; Recker et al. 2004]; two reporting data from meta-analyses [Cranney et al. 2009; Harris et al. 2008]; and one reporting data from an observational study [Harris et al. 2009]. Three abstracts reporting data from the two LTE studies were also identified by the authors [Felsenberg et al. 2010, 2009; Bianchi et al. 2009]. Details of these studies are summarized in Table 1.
Overview of reviewed randomized controlled trials, meta-analyses and an observational study with ibandronate.

50 mg on 2 consecutive days
in the MOBILE extension study a subset of patients from MOBILE either continued or were re-randomized to receive 100 mg or 150 mg once-monthly ibandronate for an additional 3 years
in the DIVA extension study patients from DIVA either continued or were reassigned to receive 2 mg bi-monthly or 3 mg quarterly i.v. ibandronate for an additional 3 years.
BONE, oral iBandronate Osteoporosis vertebral fracture trial in North America and Europe; MOBILE, Monthly Oral iBandronate In LadiEs; DIVA, Dosing IntraVenous Administration; VIBE, eValuation of IBandronate Efficacy.
Evidence from antifracture randomized controlled clinical trials
Two randomized, controlled, antifracture trials have been carried out for ibandronate: the BONE (oral iBandronate Osteoporosis vertebral fracture trial in North America and Europe) study of an oral ibandronate regimen administered daily (2.5 mg) or intermittently (20 mg every other day for 12 doses every 3 months) for 3 years [Chesnut et al. 2004]; and the IV fracture prevention study of an i.v. ibandronate regimen (0.5 mg and 1.0 mg every 3 months) for 3 years (Table 1) [Recker et al. 2004].
In the IV fracture prevention study, the cumulative incidence of new vertebral fractures with both 0.5 mg and 1 mg i.v. ibandronate once every 3 months was lower than with placebo at 3 years (8.7%, 9.2% and 10.7%, for the intention-to-treat [ITT] population, respectively), but the magnitude of the RRR did not reach statistical significance (Table 2) [Recker et al. 2004]. In the per-protocol (PP) population, the RRR with 1 mg i.v. ibandronate (26%) approached statistical significance (p = 0.0549). The Intermittent Regimen Intravenous Ibandronate Study (IRIS) showed that a higher i.v. dose of 2 mg ibandronate once every 3 months provided more substantial increases in BMD than 1 mg ibandronate once every 3 months (increase in lumbar spine BMD from baseline of 5.0% versus 2.8%, respectively) [Adami et al. 2004]. Therefore, the authors concluded that the dose of ibandronate used in the IV fracture prevention study (0.5 mg and 1.0 mg every 3 months) was suboptimal [Recker et al. 2004].
Overview of vertebral antifracture and surrogate marker efficacy for reviewed randomized controlled trials with ibandronate.

PP population.
BONE, oral iBandronate Osteoporosis vertebral fracture trial in North America and Europe; CI, confidence interval; DIVA, Dosing IntraVenous Administration; ITT, intention to treat; LTE, long-term extension; MOBILE, Monthly Oral iBandronate In LadiEs; PP, per-protocol.
The sustained efficacy of a daily oral ibandronate regimen to reduce the incidence of vertebral fractures was demonstrated in BONE, a phase III clinical trial in which vertebral fracture risk reduction was the primary endpoint [Chesnut et al. 2005, 2004; Delmas et al. 2004]. After the first year of treatment the RRR of a new vertebral fracture was 58% (95% confidence interval [CI]: −2% to 83%; p = 0.0561; ITT population) with 2.5 mg daily oral ibandronate compared with placebo (Table 2) [Chesnut et al. 2004]. This RRR was sustained over 3 years of treatment (year 2: 61% [95% CI: 33–77%], p = 0.0006; year 3: 62% [95% CI: 41–75%], p = 0.0001) (Figure 1). Interestingly, this was also the first trial to demonstrate vertebral antifracture efficacy of an intermittent regimen (20 mg every other day for 12 doses every 3 months), with a RRR of new vertebral fracture of 56% (95% CI: 27–74%; p = 0.0017) and 50% (95% CI: 26–66%; p = 0.0006) over 2 and 3 years, respectively (Figure 2). At 1 year a RRR was not observed with the intermittent regimen. A post hoc analysis of data from the BONE study showed that daily ibandronate also has a significant and sustained effect on the incidence of new moderate or severe vertebral fractures [Felsenberg et al. 2005]. The relative risk was reduced by 59% at years 1, 2 and 3.

Relative risk reduction (RRR) of new vertebral fracture with oral daily ibandronate versus placebo in BONE over 3 years.

Incidence (%) of new vertebral fracture with oral daily (2.5 mg) and intermittent ibandronate (20 mg) in BONE over 3 years. Relative risk reduction (RRR) versus placebo is indicated. (Adapted from Chesnut et al. [2004]).
In BONE, nonvertebral fracture risk reduction was demonstrated in a post hoc subgroup analysis of high-risk patients with a baseline femoral neck BMD T-score < −3.0, with a RRR of 69% (p = 0.013) with the daily regimen versus placebo [Chesnut et al. 2004]. A further subgroup analysis in high-risk patients with a baseline femoral neck BMD T-score < −2.5 and a history of clinical fracture in the past 5 years showed a non-vertebral fracture RRR of 60% (p = 0.037) with the daily regimen versus placebo [Epstein et al. 2009].
In the overall BONE study population there was no statistically significant reduction in the risk of nonvertebral fracture between treatments, but this was not the primary endpoint of the trial and the study was not designed or powered to demonstrate an effect at nonvertebral sites. The patient population in the BONE study was at relatively low risk for new nonvertebral fractures, with a relatively high mean BMD at the total hip at baseline (mean T-score = −1.73). Nonsignificant reductions in nonvertebral fracture have also been reported in other randomized controlled trials of the treatment of postmenopausal osteoporosis with the oral bisphosphonates alendronate [Cummings et al. 1998; Black et al. 1996] and risedronate [Reginster et al. 2000], in which incident nonvertebral fracture was also a secondary endpoint. Unlike vertebral fractures, nonvertebral fractures generally occur as a result of external trauma. Therefore, the nonvertebral fracture risk of the patient cannot be considered as the only predictor of the osteoporotic condition [Rossini et al. 2009]. External factors are unlikely to be modified by treatment and may become more important if the patient population recruited into a study is not at a high risk of fracture [Sebba, 2009].
Surrogate marker evidence from randomized controlled clinical trials
BONE [Chesnut et al. 2005] and the IV fracture prevention study [Recker et al. 2004] demonstrate the relationship between changes in BMD and BTM levels and antifracture efficacy with ibandronate. These studies suggest that achieving significant reductions in fracture risk requires a correlation with BMD and reductions in BTM levels.
In BONE, as discussed previously, 2.5 mg daily oral ibandronate was associated with a significant reduction in vertebral fracture risk versus placebo after 3 years. Over the same time period, mean lumbar spine BMD increased from baseline by 6.5% and 5.7% with daily ibandronate and intermittent ibandronate, respectively (both p < 0.0001 versus placebo; PP population) [Delmas et al. 2004]. Both the daily and intermittent ibandronate regimens provided a pronounced reduction in median urinary c-telopeptide of type I collagen (CTX) levels relative to baseline within 3 months of administration (58.9% and 49.2%, respectively). These median reductions from baseline were sustained throughout the 3-year study (65.3% and 52.7%, respectively; p < 0.0001 for both regimens versus placebo).
In the IV fracture prevention study the incidence of new vertebral fractures with both 0.5 mg and 1 mg i.v. ibandronate was lower than placebo at 3 years, but this did not reach statistical significance [Recker et al. 2004]. This related to small increases in mean lumbar spine BMD (0.5 mg, 3.9%; 1 mg, 4.9%) and decreases in median urinary CTX (0.5 mg, −41.4%; 1 mg, −45.0%) relative to baseline.
Bridging trials
Where a drug has already demonstrated antifracture efficacy in a randomized, controlled trial, authorities such as the European Medicines Agency (EMEA) and the US Food and Drug Administration (FDA) suggest the use of ‘bridging studies’ using surrogate endpoints for fracture for the approval of new dosing regimens or routes of administration [European Medicines Agency, 2005]. Bridging studies, with BMD changes as their primary endpoint and BTM changes as their secondary endpoint, have led to the approval of 70 mg weekly alendronate and 35 mg weekly risedronate regimens [Brown et al. 2002; Rizzoli et al. 2002]. The monthly dosing of 75 mg risedronate on 2 consecutive days a month (5 mg daily × 30 days = 150 mg) was also approved in 2008 based on data from a bridging trial [Delmas et al. 2008]. Changes in BMD and BTMs have been widely used as surrogate markers for antifracture efficacy with ibandronate [Eisman et al. 2008; Adami et al. 2007; Delmas et al. 2006; Reginster et al. 2006; Miller et al. 2005]. Ibandronate at doses of 150 mg oral once monthly (double the equivalent of the daily dose, 2.5 mg daily × 30 = 75 mg) and 3 mg quarterly i.v. were also approved based on results from two bridging studies, the MOBILE (Monthly Oral iBandronate In LadiEs) study [Reginster et al. 2006; Miller et al. 2005] and the Dosing IntraVenous Administration (DIVA) study (Table 1) [Eisman et al. 2008; Delmas et al. 2006].
MOBILE and DIVA
For the purposes of this review, only the marketed doses of ibandronate (150 mg once monthly and 3 mg quarterly i.v.) used in these studies will be discussed. Both 150 mg once monthly ibandronate and 3 mg quarterly i.v. ibandronate provide dose levels that are approximately twice as high as oral daily dosing (annual cumulative exposure [ACE] dose levels: daily, 5.5 mg; once monthly, 10.8 mg; quarterly i.v., 12 mg). The MOBILE and DIVA studies demonstrate that both ibandronate once monthly and quarterly i.v. regimens are able to increase BMD and suppress BTMs to levels that are consistent with antifracture efficacy at vertebral and nonvertebral sites.
In the MOBILE study, ibandronate 150 mg once monthly was superior to 2.5 mg daily treatment in terms of mean lumbar spine BMD gains at all sites after 1 year [Miller et al. 2005] and 2 years (Table 2 and Figure 3) [Reginster et al. 2006]. After 1 year, lumbar spine BMD increased from baseline by 4.9% with 150 mg once-monthly ibandronate versus 3.9% with daily treatment (p < 0.001; PP population) [Miller et al. 2005]. This increase was sustained at 2 years, with an increase in lumbar spine BMD from baseline of 6.6% (95% CI: 6.0–7.1%) with 150 mg once-monthly ibandronate versus 5.0% (95% CI: 4.4–5.5%) with daily treatment (p < 0.001; PP population) [Reginster et al. 2006]. The ITT analysis confirmed these findings. At 2 years, significant BMD increases at all sites (total hip, femoral neck and trochanter) with 150 mg ibandronate were also observed (p < 0.05 versus daily treatment).
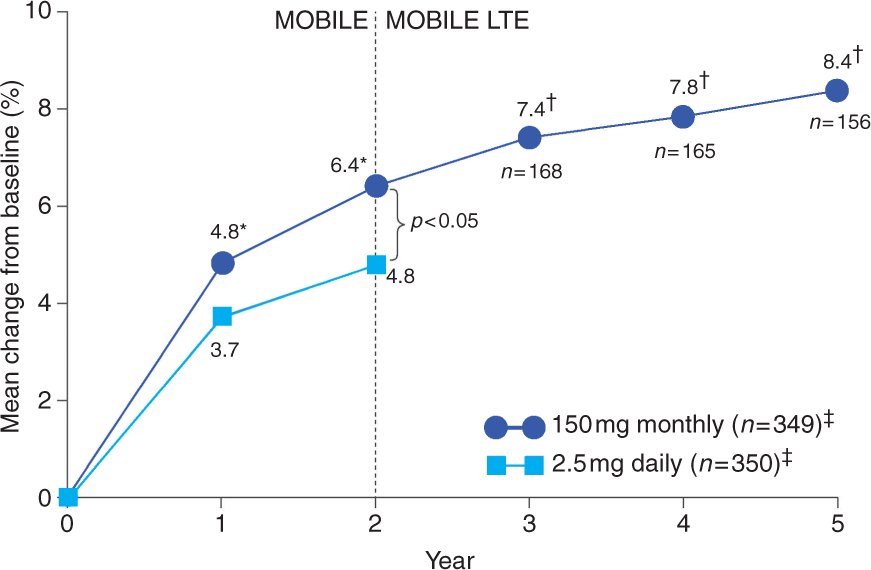
Mean change from baseline in lumbar spine BMD over time with oral ibandronate 150 mg once monthly versus 2.5 mg daily in the MOBILE LTE study (ITT population). *p < 0.05 versus 2.5 mg daily; † p < 0.001 versus MOBILE baseline; ‡at 2 years [Felsenberg et al. 2009, 2010; Reginster et al. 2006; Miller et al. 2005].
Similarly, ibandronate 3 mg quarterly i.v. was shown to increase BMD significantly more than 2.5 mg oral daily after 1 and 2 years (p < 0.05) in the DIVA study (Table 2 and Figure 4) [Eisman et al. 2008; Delmas et al. 2006]. After 1 year, lumbar spine BMD increased from baseline by 4.8% (95% CI: 4.5–5.2%) with 3 mg quarterly i.v. ibandronate versus 3.8% (95% CI: 3.4–4.2%) with daily oral treatment (p < 0.001; PP population) [Delmas et al. 2006]. This increase was sustained at 2 years, with an increase in lumbar spine BMD from baseline of 6.3% (95% CI: 5.7–6.8%) with 3 mg quarterly i.v. ibandronate versus 4.8% (95% CI: 4.3–5.4%) with daily treatment (p < 0.001; PP population) [Eisman et al. 2008]. At 2 years, significant BMD increases at total hip and trochanter with 3 mg quarterly i.v. ibandronate were seen (p < 0.001 versus daily treatment).

Mean change from baseline in lumbar spine BMD over time with i.v. ibandronate 3 mg every 3 months versus oral ibandronate 2.5 mg daily in the DIVA LTE study (ITT population). *p < 0.001 versus 2.5 mg daily; † p < 0.001 versus MOBILE baseline; †at 2 years [Bianchi et al. 2009; Eisman et al. 2008; Delmas et al. 2006].
Serum CTX (sCTX) was markedly reduced from baseline after 3 months (>50% change from baseline within 3–6 months) and this change was sustained by > 50% throughout the 2-year studies with ibandronate 150 mg once monthly and 3 mg quarterly i.v. [Eisman et al. 2008; Delmas et al. 2006; Reginster et al. 2006; Miller et al. 2005]. Furthermore, the proportion of patients reporting a ⩾50% reduction in sCTX from baseline was significantly higher with the once-monthly dose versus daily dosing after 2 years (p = 0.002).
Long-term extension studies: MOBILE and DIVA
In the MOBILE LTE study, patients from MOBILE who received either ibandronate 100 mg monthly or 150 mg monthly continued on the same treatment. Those who did not receive this treatment in MOBILE were re-randomized to receive 100 mg or 150 mg once-monthly ibandronate. An analysis of the ITT population who received 5 years' continuous treatment with ibandronate 150 mg once monthly (2 years during MOBILE, 3 years during MOBILE LTE) showed sustained efficacy in lumbar spine BMD during the LTE with an increase from baseline of 7.8% (95% CI: 7.0–8.7%) at 4 years to 8.4% (95% CI: 7.5–9.4%) at 5 years (Figure 3 and Table 2) [Epstein et al. 2009; Felsenberg et al. 2009]. BMD gains at the total hip, femoral neck and trochanter, achieved during the 2-year MOBILE study, were generally maintained during the extension study [Felsenberg et al. 2010, 2009]. BMD increases at the total hip (3.5%), femoral neck (3.2%) and trochanter (6.0%) were reported after 5 years' continuous monthly ibandronate treatment compared with MOBILE baseline.
The early suppression of BTMs sCTX and sP1NP achieved in MOBILE was sustained over 5 years of 150 mg once monthly ibandronate therapy [Felsenberg et al. 2010]. A rapid and pronounced decrease in median trough sCTX from baseline was seen during the first 3 months of treatment: −66% (95% CI: −70% to −60%; n = 140). The magnitude of suppression was generally sustained at year 5, but decreased slightly: −57% (95% CI: −65% to −30%; n = 19). Similarly, a pronounced decrease in median trough sP1NP from baseline was seen at month 12: −72% (95% CI: −79% to −67%; n = 23). These decreases in sP1NP were sustained at a similar magnitude at year 5: −61% (95% CI: −75% to −56%; n = 19). In terms of absolute changes in BTM, in contrast with the results of the study with suboptimal doses of ibandronate [Recker et al. 2004], the BTM decreases described in MOBILE show evidence for reductions to the ‘premenopausal’ normal reference range [Adami et al. 2008].
In the DIVA LTE study, patients from DIVA who received either i.v. ibandronate 2 mg every 2 months or 3 mg every 3 months continued on the same treatment. Those who did not receive these doses in DIVA were re-assigned to receive i.v. ibandronate 2 mg every 2 months or 3 mg every 3 months in DIVA LTE. An analysis of the ITT population who received 5 years' continuous treatment with i.v. ibandronate 3 mg every 3 months showed sustained efficacy in lumbar spine BMD during the LTE, with increases of 7.7% at 4 years and 8.1% at 5 years (Figure 4) [Bianchi et al. 2009]. BMD increases at the total hip, femoral neck and trochanter were generally sustained after 5 years' continuous i.v. ibandronate treatment [Bianchi et al. 2009].
Evidence through meta-analyses
Two recent meta-analyses of ibandronate trials have demonstrated nonvertebral fracture protection with different ibandronate regimens [Cranney et al. 2009; Harris et al. 2008]. Of note, these were analyses of individual patient data rather than the mean values reported in each study, thus increasing the validity of the analyses. This was achieved by merging patient data into ACE groups, based on a mean absorption rate of 0.6% for oral dosing [Barrett et al. 2004], and 100% for i.v. dosing. ACE is calculated according to the equation: ACE = single dose × number of doses/year × absorption factor. Overall, licensed ibandronate doses were grouped into ACE categories equal to 5.5 mg (2.5 mg daily) and ⩾ 10.8 mg (including 150 mg once monthly, 3 mg quarterly i.v. and 2 mg every 2 months i.v.). In the meta-analysis by Harris and colleagues a placebo group (ACE= 0 mg) was included for comparison purposes [Harris et al. 2008].
Both meta-analyses were limited to trials with at least 2 years of follow up, the duration of the bridging studies. In the larger of the two meta-analyses, ibandronate doses from four studies (BONE, IV fracture prevention study, MOBILE and DIVA, already discussed individually above), were grouped by ACE (Table 1) [Harris et al. 2008]. Overall, including data from all available study years, the higher and less-frequently administered ibandronate doses (ACE ⩾ 10.8 mg) significantly reduced the relative risk of all nonvertebral fractures (RRR: 29.9%; 95% CI: 0.50–0.99%; p = 0.041) as well as of nonvertebral fractures at six key sites (RRR: 34.4%; 95% CI: 0.45–0.96%; p =0.032) and clinical fracture (RRR: 28.8%; 95% CI: 0.55–0.92%; p = 0.010) versus placebo (Figure 5). However, a limitation of this meta-analysis was that not all of the studies included were placebo-controlled. Therefore, the two groups of patients (i.e. those receiving placebo and those receiving once-monthly/quarterly i.v. ibandronate) were from different studies. Patients were pooled within dose groups and fracture rates across all four studies trials were compared with placebo (derived from the two 3-year studies; BONE and IV fracture prevention study).

The reduction in risk of clinical fracture, nonvertebral fracture and nonvertebral fracture at key sites with oral ibandronate doses (ACE ⩾10.8 mg) versus placebo from a meta-analysis of four studies (BONE, IV fracture prevention study, MOBILE and DIVA. Relative risk reduction (RRR) versus placebo is indicated.
In the meta-analysis by Cranney and colleagues, individual patient data from eight randomized controlled studies (n = 9753) were evaluated (Table 1) [Cranney et al. 2009]. Ibandronate doses from MOBILE and DIVA (results already discussed individually above) were grouped in order to assess nonvertebral fracture incidence [Cranney et al. 2009]. Overall, ibandronate doses equivalent to an ACE ⩾ 10.8 mg (150 mg once monthly, 3 mg quarterly i.v. and 2 mg every 2 months i.v.) were associated with a significant reduction in the incidence of nonvertebral fracture (RRR 38%; 95% CI: 0.40–0.97%; p = 0.038) versus an ACE of 5.5 mg (2.5 mg daily). The advantage of this meta-analysis was that randomization was maintained. This meant that comparisons between groups were only made with common doses of ibandronate, thus underlining the robustness of this particular meta-analysis.
Meta-analyses are associated with limitations, as reported for the studies involving ibandronate. Although a significant risk reduction in nonvertebral fracture with ibandronate at ACE doses ⩾10.8 mg versus ACE doses of 5.5 mg [Cranney et al. 2009] and placebo [Harris et al. 2008] was shown, this was not a primary endpoint in any of the individual studies included in the analyses.
Observational study antifracture evidence
A number of nitrogen-containing bisphosphonates have been registered for the treatment of postmenopausal osteoporosis on the basis of placebo-controlled, clinical trials with evidence of antifracture efficacy [Black et al. 2007; Chesnut et al. 2004], thus posing the question of their comparative effectiveness. Owing to the large sample sizes required, any direct comparative clinical trial of antifracture efficacy is unrealistic. Observational studies offer one possibility of overcoming this hurdle.
Data regarding the use of osteoporotic drugs and the incidence of clinical fractures can be collected and analysed from medical claims databases, derived from insurance companies. They offer a real-world clinical perspective into the effects of different bisphosphonate regimens, outside the confines of a controlled clinical trial setting [Silverman, 2010]. However, due to lack of randomization, observational studies are subject to potential bias and confounding, which can be reduced but not fully eliminated by appropriate statistical methods.
Silverman and colleagues were the first to report a comparison between two bisphosphonate regimens: risedronate and alendronate [Silverman et al. 2007]. A similar 12-month observational study, VIBE (The eValuation of IBandronate Efficacy database fracture study), comparing the risk of fracture in women treated with monthly oral ibandronate or weekly bisphosphonates (alendronate and risedronate), was carried out in patients from two US-based databases (Table 1) [Harris et al. 2009]. In more than 64,000 patients, the incidence of any clinical fracture was significantly lower (p = 0.052) in patients receiving ibandronate therapy compared with patients on weekly bisphosphonates (adjusted relative risk: 0.82, 95% CI: 0.66–1.00%). The incidence of vertebral fracture was significantly lower in patients receiving ibandronate compared with patients on weekly bisphosphonates (adjusted relative risk: 0.36, 95% CI: 0.18–0.75%; p = 0.006). The RRR in nonvertebral and hip fractures was comparable among the monthly and weekly bisphosphonates.
Observational studies using fracture databases are subject to some restrictions. Unmeasured differences in patient characteristics (e.g. in baseline BMD) between different therapy groups may bias efficacy rates reported [Silverman, 2010; Grimes and Schultz, 2002]. Also, as these are data from databases, there is no information on whether the patients actually took the medication. Therefore, caution must be taken when interpreting results from observational studies and further validation may be needed for results that show statistically significant differences between therapies.
Conclusions
In the evaluation of any treatment, the whole body of evidence should be considered. A large amount of data from randomized, controlled fracture trials, bridging studies using surrogate endpoints, meta-analyses and observational studies based in a clinical setting is available to suggest that ibandronate has sustained vertebral and nonvertebral antifracture efficacy in women with postmenopausal osteoporosis.
BMD increases and BTM suppression are well-established surrogate markers for antifracture efficacy, and are recognized endpoints for fracture in bridging trials of new bisphosphonate regimens. Their association is supported by several analyses [Hochberg et al. 2002, 1999; Wasnich and Miller, 2000]. However, according to these analyses, 20–60% of fracture risk reduction is explained by the respective surrogate marker and regulatory agencies still require studies with specific fracture endpoints for initial registration.
In the BONE trial, ibandronate demonstrated vertebral antifracture efficacy with 2.5 mg daily ibandronate and the intermittent regimen [Chesnut et al. 2004]. In this fracture trial ibandronate (daily and intermittent dose) only demonstrated nonvertebral antifracture efficacy in a post hoc subgroup analysis of patients at higher risk [Chesnut et al. 2004]. Therefore, there has been some debate as to whether ibandronate provides antifracture efficacy at nonvertebral as well as vertebral sites. In addition to the pivotal fracture trial data in BONE, the sustained antifracture efficacy of ibandronate at varying doses has been demonstrated in the MOBILE and DIVA studies assessing surrogate endpoints (i.e. BMD and BTMs) [Felsenberg et al. 2010, 2009; Bianchi et al. 2009; Eisman et al. 2008; Delmas et al. 2006; Reginster et al. 2006; Miller et al. 2005]. The efficacy of ibandronate has also been demonstrated by meta-analyses [Cranney et al. 2009; Harris et al. 2008] and an observational study [Harris et al. 2009]. Thus, the efficacy of ibandronate from the clinical trial setting to the real-world setting of an observational study has been demonstrated to be comparable. However, as with any study, one should acknowledge the limitations of these data.
Ibandronate 150 mg once monthly oral and 3 mg quarterly i.v. are clearly superior to ibandronate 2.5 mg daily in terms of BMD and BTM changes as shown in the MOBILE and DIVA bridging trials, respectively [Delmas et al. 2006; Reginster et al. 2006]. These data suggest that the BMD and BTM changes obtained with ibandronate are associated with both vertebral and nonvertebral fracture efficacy. This has been reported in other studies that assessed the relationship of BMD and BTM levels to fracture risk [Hochberg et al. 2002; Wasnich and Miller, 2000].
Two meta-analyses of ibandronate studies have confirmed that ibandronate once monthly oral and quarterly i.v. are also superior to daily dosing in terms of nonvertebral fracture risk reduction, the only analyses to date to demonstrate this with an intermittent oral bisphosphonate regimen [Cranney et al. 2009; Harris et al. 2008].
Furthermore, an observational study indicates that once-monthly oral ibandronate is at least as effective as weekly alendronate and risedronate in preventing clinical fractures [Harris et al. 2009].
Footnotes
Acknowledgements
The authors would like to acknowledge medical writing assistance provided by Dr Louise Profit (Gardiner-Caldwell Communications) in the preparation of this manuscript, funding for which was provided by F. Hoffmann-La Roche and GlaxoSmithKline. The authors, who are guarantors of the manuscript content and who provided final sign-off, conceived the manuscript. No funding was given to any author for the preparation of this manuscript.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare that they have no conflicts of interest.
