Abstract
The circumventricular organs (CVOs) are unique areas within the central nervous system. They serve as a portal for the rest of the body and, as such, lack a blood-brain barrier. Microglia are the primary resident immune cells of the brain parenchyma. Within the CVOs, microglial cells find themselves continuously challenged and stimulated by local and systemic stimuli, even under steady-state conditions. Therefore, CVO microglia in their typical state often resemble the activated microglial forms found elsewhere in the brain as they are responding to pathological conditions or other stressors. In this review, I focus on the dynamics of CVO microglia, using the pineal gland as a specific CVO example. Data related to microglia heterogeneity in both homeostatic and unhealthy environments are presented and discussed, including those recently generated by using advanced single-cell and single-nucleus technology. Finally, perspectives in the CVO microglia field are also included.
Microglia in circumventricular organs (CVOs) continuously adapt to react differentially to the diverse challenges they face. Herein, I discuss microglia heterogeneity in CVOs, including pineal gland. Further studies are needed to better understand microglia dynamics in these unique brain areas.
Keywords
Introduction
Microglia are long-lived parenchymal phagocytic cells that reside in the central nervous system (CNS). Pio del Rio-Ortega pioneered their description almost a century ago (Sierra et al., 2016). Since then, our understanding of microglia biology has expanded greatly (Prinz et al., 2019; Stratoulias et al., 2019). This is due in part to state-of-the-art experimental models and technologies. Nowadays, microglia are considered to be a highly complex cell population that functions as both a primary residential “warrior” against local and systemic adversities, and as a very active participant in CNS development, plasticity, homeostasis, and functionality (Prinz et al., 2019). Microglia heterogeneity extends well beyond their morphological characteristics and also includes their brain region distribution and abundance, their phenotypes, and their response capacity to specific brain conditions and insults (Grabert et al., 2016; Tay et al., 2017; Stratoulias et al., 2019). Microglial precursor cells originate from the embryonic yolk sac to colonize the developing brain, where they continue their differentiation into microglial cells (Bennett and Bennett, 2020; Prinz et al., 2017; Prinz et al., 2019). In addition, established microglia mitotically self-renew themselves throughout life.
The circumventricular organs (CVOs) have emerged as well-regulated ports in the CNS, where microglial cells find themselves in a dynamic and challenging environment. It is expected, therefore, that CVO microglia must function differently than in the brain at large (Takagi et al., 2019). Herein, I focus on the dynamics of CVO microglia, using the pineal gland (PG) as a specific CVO example.
Circumventricular Organs
The CVOs are distinctive neuroepithelial structures that are found across vertebrate species, from fish to humans (Duvernoy and Risold, 2007; Gross and Weindl, 1987; Kiecker, 2018; Miyata, 2015). CVOs are distributed along the midline of the brain, close to the third and fourth ventricles (Figure 1A). They are comprised of three sensory regions: area postrema (AP), organum vasculosum of the lamina terminalis (OVLT), and subfornical organ (SFO), and four secretory areas: median eminence (ME), neurohypophysis (NH), subcommissural organ (SCO), and PG. In humans, the main CVOs (ME, NH, and PG) are well known and appreciated on brain imaging but is unlikely to visualize the CVOs AP, OVLT, SFO, and SCO (Horsburgh and Massoud, 2013). When the PG is imaged using advanced techniques, calcification within the organ often facilitates image contrast. PG calcification progresses with ageing and is associated with lower production of the hormone melatonin (MEL), and pathological conditions related to neurodegenerative diseases and circadian disorders (Tan et al., 2018).
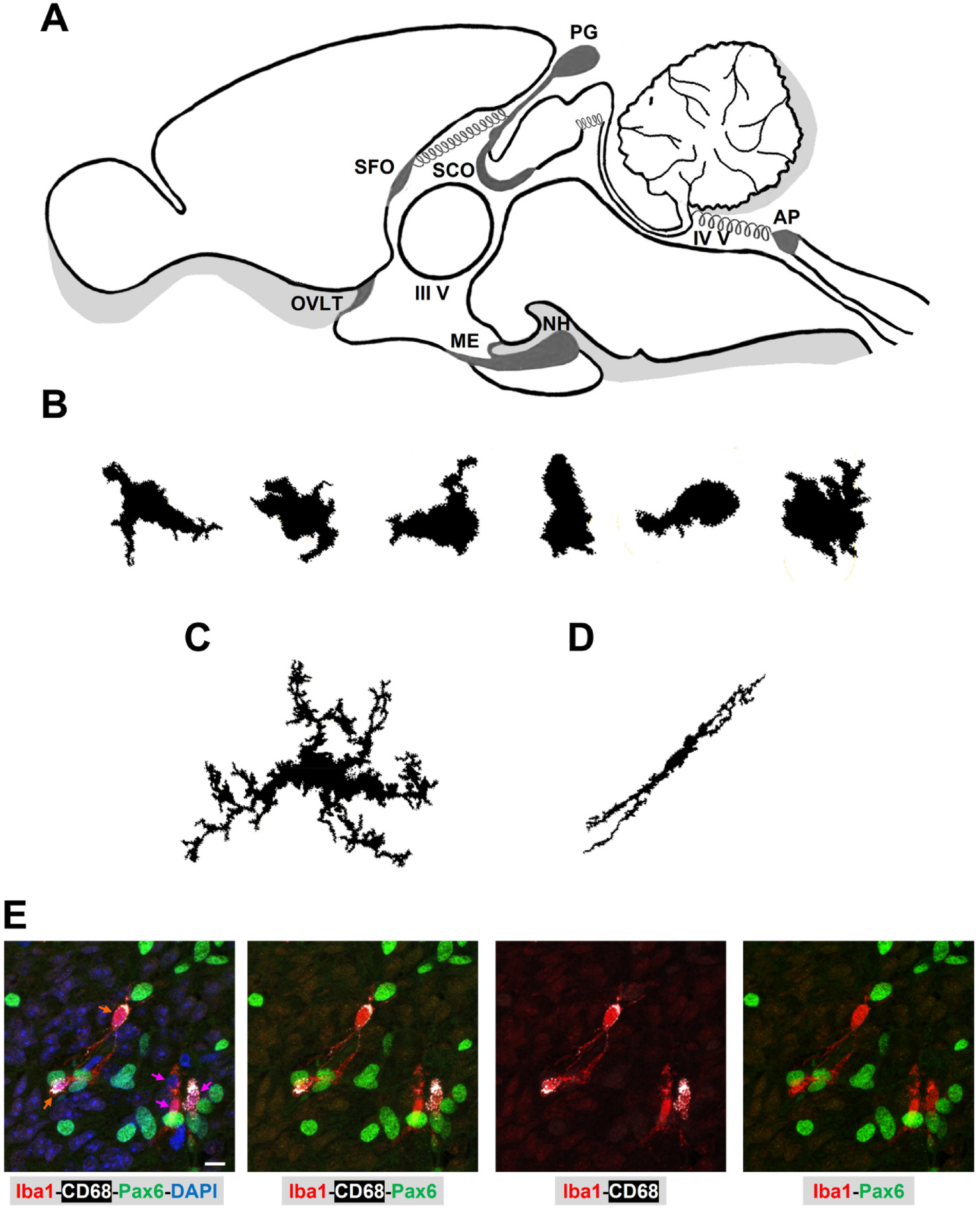
Microglia morphotypes in the rodent circumventricular organs. A: Schematic representation of a midsagittal section of the rodent brain illustrating the anatomical location of the circumventricular organs (CVOs), close to the third and the fourth ventricles (III V and IV V, respectively). The CVOs compromise three sensory regions: area postrema (AP), organum vasculosum of the lamina terminalis (OVLT), and subfornical organ (SFO), and four secretory areas: median eminence (ME), pineal gland (PG), neurohypophysis (NH), and subcommissural organ (SCO). B, C and D: Drawings of F4/80-immunoreactive microglia identified in CVOs (compact cells with a few short and thick projections, or no projections at all; B), in hypothalamus (ramified cell; C), and in corpus callosum (longitudinally branched cell; D) of the normal adult mouse brain. See Lawson et al. (Lawson et al., 1990) and Gordon et al. (Gordon et al., 2014) for further details. E: Phagocytic microglia in the normal adult rat PG. Microglial cells immunoreactive for Iba1 (Microglia/macrophage marker ionized calcium-binding adaptor protein 1; red) and CD68 (Cluster of differentiation 68; also known as LAMP4: lysosomal/endosomal-associated membrane glycoprotein 4; white) interact with interstitial cells positive for the transcription factor Pax6 (Paired box 6; green). Fuchsia arrows: compact microglia with a few cellular processes. Orange arrows: longitudinally branched microglia. Confocal microscopy; objective: 60X, scale bar: 10 μm. DAPI: Nuclear marker 4′,6-diamidino-2-phenylindole; blue. See Ibañez et al. (Ibanez Rodriguez et al., 2016) for further details about animal procedures and immunolabeling protocols. Microglial cells represent a highly heterogenous population in terms of morphology, with variations that are linked to their microenvironment.
CVOs lack a complete blood-brain barrier (BBB), while other regions of the healthy CNS are protected by a highly regulated BBB, formed by microvascular endothelial cells (ECs) that establish impermeable tight junctions (TJs) (Benz and Liebner, 2020; Ben-Zvi and Liebner, 2021; Iadecola, 2017; Kugler et al., 2021; O'Brown et al., 2018; Wilhelm et al., 2016). ECs (and TJs) are supported and influenced by other cellular elements, including neurons, astrocytes, pericytes, and microglia, which collectively make up the neurovascular unit (NVU) to selectively control vascular permeability and proper function (Iadecola, 2017; Kugler et al., 2021). A comparative and current look at the BBB across organisms, and also across brain regions, was analyzed by O’Brown and colleagues (O'Brown et al., 2018). The authors concluded that even though substantial progress has been made in the field, the nature and properties of the human BBB remain largely unexplained.
The CVOs are known as “the windows of the brain” (Gross and Weindl, 1987). They are, therefore, essential points of communication between the brain and the rest of the body, and also between different areas of the brain itself. At the CVOs, blood, cerebrospinal fluid (CSF) from the choroid plexus (CP), and specialized and nonspecialized cells, gather in a tightly interactive landscape that influences brain stability, functionality, and response to pathogens and other insults. In fact, CVOs have been involved in body fluid and thermal homeostasis, energy balance, chemoreception of blood-derived messengers, and neuroinflammation, among other functions (Miyata, 2015). Like the CP, CVOs generally possess a high density of leaky and/or fenestrated blood vessels, which facilitates their sensory and/or secretory functions (Ben-Zvi and Liebner, 2021). The molecular mechanisms involved in the establishment of this permeable vessel phenotype are still far from being understood, and they may be found to vary significantly between CVOs and among species. Inhibition of BBB formation induced by the Wnt/β-catenin signaling pathway in the murine AP, and endothelial fenestration driven by VEGF (vascular endothelial-derived growth factor) and TGFβ (transforming growth factor beta) in the developing zebrafish neurohypophysis (NH), have been associated with high capillary permeability (Anbalagan et al., 2018; Wang et al., 2019). In the zebrafish model, a dual mechanism was proposed that involves a third pituicyte factor, Cyp26b (Cytochrome P450, family 26, subfamily B; enzyme that catabolizes retinoic acid and modulates its local gradients (Hernandez et al., 2007)), which represses the non-permeable BBB endothelial fate, concomitantly with the fenestrae induction by VEGF and TGFβ (Anbalagan et al., 2018).
Endothelial barrier properties do vary among the CVOs. Furthermore, regional barrier heterogeneity has been found within sensory CVOs (Benz and Liebner, 2020; Wilhelm et al., 2016). This diversity occurs on a single-cell level, and it is closely related to a coordinated spatial distribution of TJ proteins such as Cldn5 (Claudin 5), Ocln (Occludin), and ZO-1 (Zonula occludens protein 1), and other endothelial markers including the permeability signal Meca32/Plvap (plasmalemma vesicle associated protein). In addition, other extra-endothelial barriers, such as those formed by pericytes and astrocytes, also play key roles to finally control what is exchanged with the brain parenchyma through the CVOs (Benz and Liebner, 2020; Miyata, 2015; Wilhelm et al., 2016). Structural reconstruction within at least the adult sensory CVOs has been documented to occur by angiogenesis and/or neurogliogenesis, contributing also to CVO plasticity (Furube et al., 2020; Miyata, 2015; Morita et al., 2015).
Humoral messengers are key mediators of the CVOs. Circulating substances that do not cross the BBB, such as hormones (insulin, leptin, irisin, and ghrelin, among others) and cytokines (e.g., IL-1β: interleukin-1 beta and TNF-α: tumor necrosis factor-alpha), may find that CVOs serve as entry points to allow them to modulate brain structure and function (Goncalves and De Felice, 2021). In a recent review that discussed modes of action of extracellular vesicles (EVs) at the CNS barriers, the author speculated about the exchange of messengers carried by EVs through the CVOs, and the need of further studies on this particular way of brain-periphery interaction (Kramer-Albers, 2022). On the other hand, neural pathways that integrate the specialized CVOs also play key roles in processing information between the brain and the rest of the body (Jimenez et al., 2001; McKinley et al., 2019; Moller and Baeres, 2002). However, analysis of CVO neural networks is mostly beyond the scope of this review, with the exception of the neural connections to the PG and their potential influence on pineal microglia.
Pineal Gland: A Secretory Circumventricular Organ
The pineal gland (PG) is an unpaired organ located in the roof of the diencephalon, associated with the third ventricle (Kiecker, 2018; Moller and Baeres, 2002). This secretory CVO is the primary source of melatonin (MEL), which is a time-cue hormone that is conserved across species, including humans (Arendt, 2003; Farias Altamirano et al., 2019; Maronde and Stehle, 2007). The PG synthesizes MEL in its main cell type, the pinealocytes, and releases it into the blood and CSF at night, which synchronizes physiology and behavior to the prevailing environmental light-dark conditions (Lee et al., 2020; Mays et al., 2018; Simonneaux and Ribelayga, 2003; Tan et al., 2016; Yu et al., 2016).
Among CVOs, the developmental processes of the PG are one of the best characterized, and its evolutionary origins have also been substantially investigated (Ekstrom and Meissl, 2003; Kiecker, 2018; Klein, 2006; Korzh and Kondrychyn, 2020; Mano and Fukada, 2007; Maronde and Stehle, 2007). Although, it is still not well understood how the mammalian PG lost its own photoreception capacity to directly respond to the light-dark cycles, and instead evolved its current mechanisms to receive the synchronizing information indirectly from the hypothalamic suprachiasmatic nuclei (SCN) (Ekstrom and Meissl, 2003; Falcon et al., 2009; Mano and Fukada, 2007). Furthermore, some of the secretory features of the PG have not been extensively explored. This includes the establishment and dynamics of the vascular niche and the glial cells, and their impact on pineal structure and function.
The PG shares similar hallmark characteristics with other individual CVOs, and also with other neural tube-derived structures, across species (Kiecker, 2018; Moller and Baeres, 2002). However, a common master signature has not yet been identified between PG and the other CVOs, especially the secretory ones. The overlapping hallmarks include a plethora of transcription factors (TFs) that orchestrate the definition and maintenance of pineal phenotype in a tightly regulated spatial-temporal manner (Kiecker, 2018; Maronde and Stehle, 2007; Rath et al., 2013). Some of these TFs are key regulators of pineal ontogeny and function, while others play important but compensable roles in, for example, framing the MEL circadian rhythm. Pax6 (Paired box 6), Otx2 (Orthodenticle homeobox 2), Lhx9 (LIM homeobox protein 9), and CREB1 (Cyclic AMP-responsive element binding protein 1) are examples of essential pineal TFs, while Lhx4 and NeuroD1 (Neurogenic differentiation factor 1) fit well in the second TF category (Castro et al., 2015; Farias Altamirano et al., 2019; Farias Altamirano et al., 2022; Hertz et al., 2019; Munoz et al., 2007; Ochocinska et al., 2012; Rath et al., 2013; Yamazaki et al., 2015). For example, PAX6 mutations in humans result in pineal aplasia (Abouzeid et al., 2009; Mitchell et al., 2003)
The pineal blood vascular architecture is another overlapping feature with the rest of the CVOs, and its organization has also been shown to be species- and region-dependent (Duvernoy and Risold, 2007; Moller and Baeres, 2002). In rat, a rich vascular network of freely anastomosing fenestrated capillaries and wide negative-charged pericapillary spaces is fed by the posterior cerebral arteries; the capillaries then drain mainly into the great cerebral vein via several efferent vessels (Hogendorf et al., 2001; Murakami et al., 1988; Moller and Baeres, 2002; Taguchi et al., 2000). However, in many other species, including humans, pineal capillary permeability appears to be highly dependent on the region within the organ, particularly core areas versus periphery (Duvernoy et al., 2000; Moller and Baeres, 2002). Also, in these species, overall capillary permeability is substantially reduced, as compared to rodents. For the most part, the PG's vascular development steps, and the underlying cellular and molecular mechanisms that participate in it, have not yet been well defined. Therefore, vascularization comparisons between the PG and the CNS, particularly the retina, may yield some clues, since both these organs are phylogenetically related, and retina vascularization is well characterized (Farias Altamirano et al., 2019; Maronde and Stehle, 2007; Rattner et al., 2019; Selvam et al., 2018; Tata et al., 2015; Vieira et al., 2020). VEGF family and its receptors have emerged as one of the main driving forces that regulates retina vasculature during development and in response to ischemia/hypoxia (Apte et al., 2019; Rattner et al., 2019; Uemura et al., 2021). Elevated levels of astrocytic VEGF, together with HIF-1α (hypoxia-inducible factor-1 alpha), nitric oxide synthases (NOSs) and NO, were found in adult rat PG after hypoxia (Kaur et al., 2007). This imbalance was accompanied by increased vascular leakage and lower local MEL. Administration of exogeneous MEL reverted hypoxia-induced VEGF and NO production, and its consequences in pineal vascular homeostasis.
Pineal vascular remodeling was proposed by Taylor and colleagues (Taylor et al., 1988), based on the elevated levels of ESAF (endothelial cell-stimulating angiogenesis factor) found in bovine and human PGs. ESAF levels in the bovine PG were substantially higher than in other neuronal areas such as cerebral cortex and retina, and higher than in other vascular tissues such as liver and kidney. Pineal angiogenic capacity was confirmed morphologically and functionally (but not mechanistically) in solid autografts implanted intracerebrally in adult rats (Villegas, 2004). The fenestrated vascular phenotype within the pineal graft was donor-determined, while a continuous host-derived endothelium with an intact BBB was observed in the graft-CNS interface. Pineal graft revascularization and reperfusion facilitated implant survival and enhanced pinealocyte secretory function.
Regarding the cells associated with pineal vasculature, a recent single-cell RNA sequencing (scRNA-seq) analysis of the rat pineal transcriptome identified endothelial cells (ECs) and vascular and leptomeningeal cells (VLMCs), among the non-pinealocyte cell types (Chang et al., 2020; Coon et al., 2019; Mays et al., 2018). ECs and VLMCs were transcriptomically distinguished from two MEL-producing pinealocytes (α and β), three astrocytes (α, β, and γ) and two microglial subtypes (α and β) (Mays et al., 2018). More recently, scRNA-seq was applied to unravel cellular heterogeneity in a different secretory CVO, the mouse NH (Chen et al., 2020). NH is the site where the hypothalamic hormones oxytocin (OXT) and arginine-vasopressin (AVP) are released to mediate osmotic equilibrium, blood pressure, reproduction, and lactation. In the murine NH, ECs, and VLMCs were also distinguished from three other cell types, of which one was the main astrocyte-like pituicyte. In both studies, fenestrated ECs were identified by the markers Emcn (Endomucin), Flt1 (FMS-like tyrosine kinase 1 that encodes VEGFR1: VEGF receptor 1), and Plvap, among other common and unshared markers (Chen et al., 2020; Mays et al., 2018). Both studies introduced Lum (Lumican)+/Dcn (Decorin)+ VLMCs for the first time in the PG and NH research, which reasonably suggests that VLMCs may function as multipotent stem cells in these two CVOs (Nakagomi and Matsuyama, 2017). In NH, a close spatial association between Lum+ cells and fenestrated Plvap+ capillaries was shown (Chen et al., 2020). Both single-cell transcriptomic analyses point to promising strategies for further investigation of the cellular and molecular mechanisms within the PG and NH CVOs. For instance, new studies may reveal novel intercellular interactions and crosstalk mechanisms that occur between various cell types, including microglia.
Microglia Within the Circumventricular Organs
Microglia have crucial roles in the normal brain activities, in ageing, and also in pathological conditions with spatially restricted patterns, such as some neurodegenerative diseases (Scheiblich et al., 2020). Nowadays, it is well accepted that microglia represent a highly heterogenous resident cell population (Gordon et al., 2014; Walker and Lue, 2015). This heterogeneity is in accordance with the microglia capacity to sense and to respond as both phagocytes and as immune cells, to their local microenvironment. A deeper understanding of the diverse nature of microglia heterogeneity may lead to new diagnostic and therapeutic targets. In particular, “omics” data analyses from many sources have come to shed light about microglia diversity across species, including humans (Alsema et al., 2020; Dowsett et al., 2021; Dubbelaar et al., 2021; Dumas et al., 2021; Grabert et al., 2016; H et al., 2017; Kracht et al., 2020; Lopez-Atalaya et al., 2018; Matcovitch-Natan et al., 2016; Mays et al., 2018; Noristani et al., 2017; Prinz et al., 2019; Thion et al., 2018). Furthermore, the biology and dynamics of non-parenchymal macrophages, known as CAMs (CNS-associated macrophages), are emerging to become a hot research field (Croese et al., 2021; Prinz et al., 2017; Prinz et al., 2021; Ransohoff et al., 2003; Wohleb et al., 2013; Zhu et al., 2022). CAMs are found at all CNS interfaces, in both healthy and diseased individuals. CAMs include meningeal, perivascular, and CP macrophages, as well as infiltrating monocytes that result from insults. These macrophages have been characterized based on their origins, fates, morphotypes, surface epitope expression, functional cell states, and interactomes. Interestingly, CVOs have been proposed as gateways for trafficking peripheral leukocytes and bone marrow-derived myeloid lineage cells, into the CNS (Kelly et al., 1993; Markus et al., 2018; Murayama et al., 2019; Ransohoff et al., 2003; Shimada and Hasegawa-Ishii, 2017; Vargas-Caraveo et al., 2015). However, most CAMs share with microglia their longevity, the lack of replacement by peripheral myeloid cells, and a low self-renewal rate in homeostatic conditions (Prinz et al., 2017; Prinz et al., 2021). Microglia, and potentially CAMs, were found to host to a high number of genetic variations associated with multiple pathologies, from rare monogenetic diseases to Parkinson's disease, Alzheimer's disease, frontotemporal dementia, schizophrenia, and multiple sclerosis (Prinz et al., 2021). It follows then, that these cells may be useful targets for modulating the CNS. Although, their scarcity and a lack of cell-type specific technology may limit progress in these areas.
Within the CVOs, phenotypic diversity of microglial cells and their non-parenchymal siblings remains a poorly investigated area. In animal models, there is consensus, however, that microglia exhibit a distinct morphology in all the CVOs that consists of compact round or slightly elongated somas with just a few or no cellular processes, as was evidenced in the normal mouse brain by the expression of the monocyte/macrophage F4/80 antigen (Figure 1B) (Gordon et al., 2014; Lawson et al., 1990; Waddell et al., 2018), and the expression of Iba1 (ionized calcium-binding adaptor protein 1, a marker for microglia and macrophages), CD11b (a cluster of differentiation receptor also known as ITGAM: integrin subunit alpha M), and CX3CR1 (fractalkine receptor) (Jurga et al., 2020; Takagi et al., 2019). These morphotypes differ from the surveilling microglia present in the rest of the homeostatic CNS, which are typically characterized by small somas and highly ramified and motile projections (Figure 1C and 1D) (Augusto-Oliveira et al., 2022; Gordon et al., 2014; Lawson et al., 1990). However, CVO microglia do resemble the activated microglial forms found in the rest of the CNS under unhealthy and stimulating conditions (Leyh et al., 2021). In the CVOs as compared to other brain regions, to the best of my knowledge, a fine characterization of microglia morphotypes has not been done yet for both humans and non-human primates (Ambrose et al., 2020; Barger et al., 2019; Cooper et al., 2018; Morgan et al., 2014; Rezaie and Male, 1999; Salamanca et al., 2019).
In terms of phenotypes, a recent study showed that M1 and M2 polarized microglial stages coexist in the normal murine AP, ME, SFO, and OVLT, although with certain variations among these four CVOs (Takagi et al., 2019). The M1 phenotype co-expressed CX3CR1 with either CD16/32 (low-affinity Fcγ III/II receptors that mediate phagocytosis and cytotoxicity) or CD86 [type I membrane protein that belongs to the immunoglobulin superfamily and is involved in antigen presentation; ligand for the receptors CD28 and CTLA-4 (cytotoxic T-lymphocyte associated protein 4) on T cell membranes (Halliday et al., 2020)]. The M2 phenotype was identified by expression of CX3CR1 with either CD206 [a pattern recognition receptor (PRR) and a mannose receptor that mediates endocytosis of pathogens, for example] or Ym1 (Chil3: Chitinase-like protein 3 without chitinase activity that mediates inflammation and pathogenesis (Zhu et al., 2020)). Although the application of M1/M2 terminology in the microglia field has been dismissed (Ransohoff, 2016), the coexistence of pro-inflammatory and anti-inflammatory microglial states within CVOs suggests that responses may vary from one microglial cell to another. This is a key example of functional diversity among activated microglia in theses organs. A single-nucleus RNA sequencing analysis (NucSeq) of the hindbrain, containing AP and the nucleus tractus solitarius (NTS), in mice under eating and fasting regimes, was recently released (Dowsett et al., 2021). In this study, a cluster of microglia marked by the expression of Tmem119 (encodes the Transmembrane protein 119 (Uff et al., 2022; Vankriekelsvenne et al., 2022)), was discriminated from several clusters of neurons and oligodendrocytes, among others. Around fifteen genes were found to be differentially expressed in the hindbrain microglia cluster after an overnight fast. Although, a correlation between gene profiles and microglial cell diversity has not been established.
Besides the poorly explored microglia heterogeneity within CVOs, little is known about how these resident phagocytes become integrated spatially and functionally into their local cell network. The NH of the Cx3Cr1-GFP (green fluorescence protein) microglia/macrophage transgenic reporter mouse (Jung et al., 2000), provides an example where GFP+ cells were found in close proximity to both Srebf1 (Sterol regulatory element binding transcription factor 1)+ pituicytes and Cyp2f2 (Cytochrome P450, family 2, subfamily f, polypeptide 2)+ epithelial-like cells (Chen et al., 2020). This suggests a possible crosstalk between pituitary cells and the phagocytes, which might impact axon shape and function in this CVO. Cell-cell communication most likely will be found to be mediated by several mechanisms, including those driven by receptors and their ligands (e.g., CX3CR1/Fractalkine, purinergic receptors/ATP, ADP, or adenosine, and MT1 and MT2 MEL receptors/MEL (Hardeland, 2021; Illes et al., 2020; Lin et al., 2020; Pawelec et al., 2020)).
Microglia Within the Pineal Gland
Migrating microglial precursor cells that express Iba1, reach, and colonize the developing rat PG from both the brain surface and the ventricular cavity (Figure 2) (Farias Altamirano et al., 2019; Ibanez Rodriguez et al., 2016; Munoz, 2018). This invasion takes place very early in the formation of the pineal primordium (around embryonic day 15 in rat), and it disrupts the otherwise homogeneous cell population of the incipient gland. Precisely, the pineal anlage is formed by radial Pax6+/Vimentin+ neuroepithelial cells that proliferate, differentiate, and spatially reorganize themselves and their progeny, to first give rise to pinealocytes and later to astrocytes (Ibanez Rodriguez et al., 2016; Munoz, 2018; Rath et al., 2013).
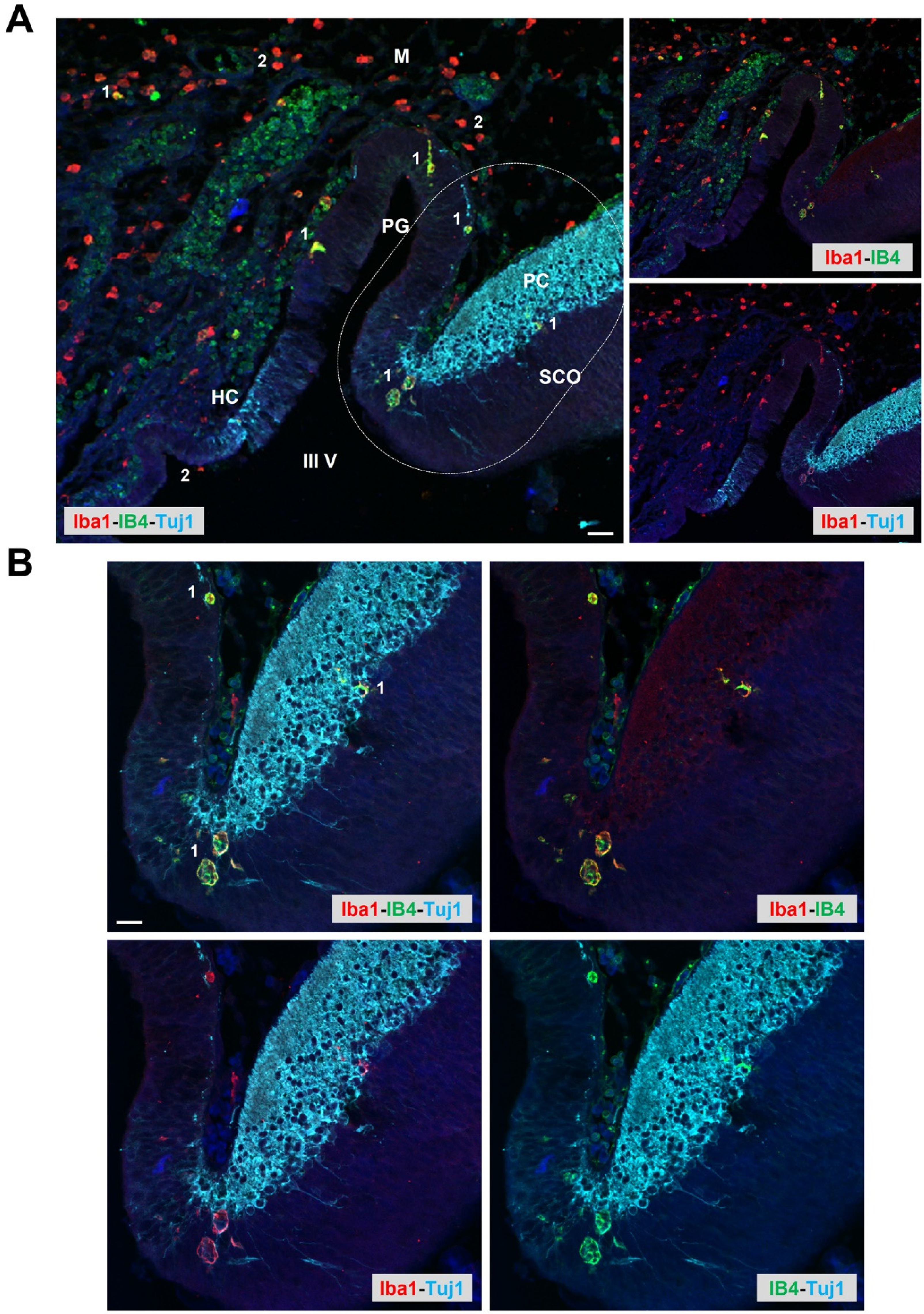
Cellular heterogeneity during microglia colonization and establishment in the developing rat pineal gland. Potential coupling with vascularization and innervation. Sagittal section of an embryonic (E16) rat brain, immunolabeled for Iba1 (red) and Tuj1 (neuronal marker β-tubulin III; cyan) and stained with IB4 (α-D-galactosyl-specific isolectin B4 from Griffonia simplicifolia; green). Two microglial subpopulations are clearly distinguished: one is positive for both Iba1 and IB4 (1), and the second is reactive only for Iba1 (2). While the double-labeled cells are observed in the pineal primordium (PG) and neighboring brain regions, the second type is predominant in the meninges (M) and in the ventricular cavity (III V: third ventricle). Iba1+/IB4+ cells are seen in proximity to developing Tuj1+ elements and IB4+ blood vessels. A: Confocal microscopy; objective: 20X, scale bar: 30 μm. HC: Habenular commissure. PC: Posterior commissure. SCO: Subcommissural organ. B: Higher magnification of the inset shown in A; objective: 40X, scale bar: 20 μm. See Ibañez et al. (Ibanez Rodriguez et al., 2016) for further details about animal procedures and immunolabeling protocols.
From the primordium stage and throughout the whole ontogeny of the rat PG, microglial cells generally resemble those found in other CVOs, with compact round or oval somas and having just a few or no projections, while exhibiting high uptake and phagocytic capacity (Figure 1E) (Ibanez Rodriguez et al., 2016; Kaur et al., 1997; Moller and Baeres, 2002; Moller et al., 2006). However, microglial cells must plastically adapt themselves, in order to engage with other elements within the developing and adult PG, and to respond to local and systemic stimuli (Ibanez Rodriguez et al., 2016; Ibanez Rodriguez et al., 2018; Jiang-Shieh et al., 2005; Tsai and McNulty, 1999). Microglia responsiveness within the rat PG is facilitated by their proliferative potential, and their self-renewal capacity. As shown in the developing cerebral cortex of prenatal and postnatal macaques and rats (Cunningham et al., 2013), pineal microglia immunoreactive for Iba1+ and CD68 (Cluster of differentiation 68; also known as LAMP4: lysosomal/endosomal-associated membrane glycoprotein 4), have some preference to contact, surround, and eventually phagocytize precursor-like cells (Figure 1E) (Ibanez Rodriguez et al., 2016; Munoz, 2018). Phagocytosis of Pax6+ cells continues into adulthood, even after their number has reached its minimum. No progress has yet been made in understanding the mechanisms that are behind this phenomenon.
Ibañez and colleagues also showed that pineal microglia participate in remodeling blood vessels and nerve fibers (Figure 2) (Ibanez Rodriguez et al., 2016). However, the dynamics and mechanisms that guide microglia during the vascularization and innervation of the developing PG have not yet been established. For an example that offers some insight, IL34 (Interleukin 34)/CSF-R (Colony stimulating factor-receptor) signaling was shown to drive blood vessel-mediated microglia colonization of the optic cup in the developing zebrafish retina (Ranawat and Masai, 2021). In this model, microglia establishment was also found to be influenced by local neurogenic states. On the other hand, in the developing murine retina, colonizing microglia have been shown to control vascular patterning, based on their paracrine interplay with blood vessels, and under the influence of localized tissue mechanics (Dudiki et al., 2020). This interaction involves Kindlin3-dependent and integrins-independent microglia bipolarization in response to stiff extracellular substrates, and the inhibition of the proangiogenic TGFβ1 signaling.
For another perspective, microglia have been shown to network with MEL-secreting pinealocytes, both structurally and mechanistically. Co-culturing pinealocytes and microglial cells, in the presence of proliferating astrocyte-like cells from 1-day-old Sprague-Dawley rats, showed a reduction of pinealocyte neurite length and an increase of the MEL precursor serotonin in the culture medium after 7 days; the total number of pinealocytes did not change significantly (Tsai and McNulty, 1997). In vitro analysis of cytokines of interest, such as IFN-γ (interferon-gamma), IL-1β, TNF-α, and TGFβ1, showed variable impact on the neonatal cell network of pinealocytes, microglia, and astrocytes (Tsai et al., 2001). In addition, microglia were identified in proximity to serotonin-immunoreactive pinealocyte projections within the postnatal PG of Wistar rats, although phagocytosis events of these elements were rarely observed (Ibanez Rodriguez et al., 2016).
Regarding heterogeneity of microglia and macrophages within the PG, several studies have shown diversity in their morphotypes and in their phenotypes, as well as diversity in the regional distribution of these phagocytes (Ibanez Rodriguez et al., 2016; Jiang-Shieh et al., 2003; Mays et al., 2018). In the developing rat PG, two microglial subpopulations were distinguished based on Iba1 immunolabeling and staining with IB4 (α-D-galactosyl-specific isolectin B4 from Griffonia simplicifolia) (Figure 2) (Ibanez Rodriguez et al., 2016). This evidence suggests that IB4+ vascularization of the developing PG may be coupled with microglia colonization. Even though individual (and rarely clustered) microglia/macrophages are widely distributed in the interstitial and perivascular compartments of the adult rat PG, cell subpopulations exhibited differing immunoreactive profiles and presented distinctive topographic patterns (Jiang-Shieh et al., 2003). This suggests that pineal microglia respond on a single-cell level, as has also been proposed for the microglia of other CVOs, as well.
Phenotypically, two microglial subtypes, designated as α and β, were distinguished by their single-cell transcriptomic profiles in the adult rat PG (Figure 3) (Coon et al., 2019; Mays et al., 2018). They represented 64% and 36%, respectively, of all microglial cells that were profiled. Both subpopulations showed high expression of Aif1 (Allograft inflammatory factor 1; encode Iba1) and Lyz2 (Lysozyme 2; encode the bacteriolytic enzyme Lysozyme C-2). They differed, however, in the levels of complement component transcripts (C1qa, C1qb, and C1qc higher in α-cells), and MHC (Major histocompatibility complex) Class II transcripts [RT1 Class II locus Da (RT1-Da), RT1-Db1, RT1-Ba, RT1-Bb, and RT1-DOa higher in β-cells]. Based on their differential antigen-presenting capability, α-microglia might be related to anti-inflammatory phenotypes, whereas β-microglia might pertain to pro-inflammatory ones (Abellanas et al., 2019; Schetters et al., 2017). Furthermore, both microglial subtypes differed in their day-night rhythmic nature. This might be explained by the differential expression of catecholamine receptor genes, including Adrb2 (Adrenoceptor beta 2) and Adrb1 (Adrenoceptor beta 1), which were both expressed at higher levels in α-microglia, as compared to β-microglia. In fact, very few transcripts within the α-cell subpopulation were found to be up-regulated during daytime, and some of these transcripts overlapped with other cell types. For example, Vldlr (Very low-density lipoprotein receptor) was differentially expressed in α-microglia, and also in both of the pinealocyte subtypes, as well. Other rhythmic genes in α-cells were Mt-nd3 (NADH dehydrogenase 3, mitochondrial), Evi2a/2b (Ecotropic viral integration site 2A and 2B), LOC100362149 (Ribosomal protein S20-like), Sirpa (Signal-regulatory protein alpha), and Qprt (Quinolinate phosphoribosyltransferase). Interestingly, several clock genes were found not to be rhythmic in both microglial subpopulations. Microglia subtypes were also distinguished by their purinergic receptor gene profiles, with P2ry6 (Pyrimidinergic receptor P2Y6) and P2rx4 (Purinergic receptor P2X 4) at higher levels in α-microglia, and with P2ry10 (P2Y receptor family member 10) only found in β-microglia. Expression of Chrne (Cholinergic receptor nicotinic epsilon subunit) occurred exclusively in β-microglia, which suggests that only this subtype responds to cholinergic inputs. Additionally, some transcripts related to the TNF-α ligand/receptor system were also differentially expressed in both microglial subpopulations. α-Microglia predominantly expressed Tnfaip8l2 (TNF alpha induced protein 8 like 2), whereas β-microglia predominantly expressed Tnfaip8 (TNF alpha induced protein 8). Furthermore, α-microglia also expressed higher levels of both Alox5ap (Arachidonate 5-lipoxygenase activating protein) and Pla2g15 (Phospholipase A2, group XV) genes, which are linked to redox and lipid metabolism. scRNA-seq, as applied by Mays and colleagues in the normal adult rat PG (Mays et al., 2018), unraveled many potential molecular pathways related to microglia dynamics. Some of these may be shared with other areas of the CNS, such as signaling cascades triggered by adrenergic and purinergic receptors (Gyoneva and Traynelis, 2013; Illes et al., 2020; Lin et al., 2020; Liu et al., 2019; Stowell et al., 2019). Undoubtably, “omics” technology has advanced our knowledge about pineal microglia biology. However, cautionary validation is needed to rule out signal contamination from more dominant cell types within the PG (e.g., pinealocytes and astrocytes), and also from the impact of cell dispersion over microglia morphotypes and phenotypes (Cadiz et al., 2022; Mays et al., 2018).
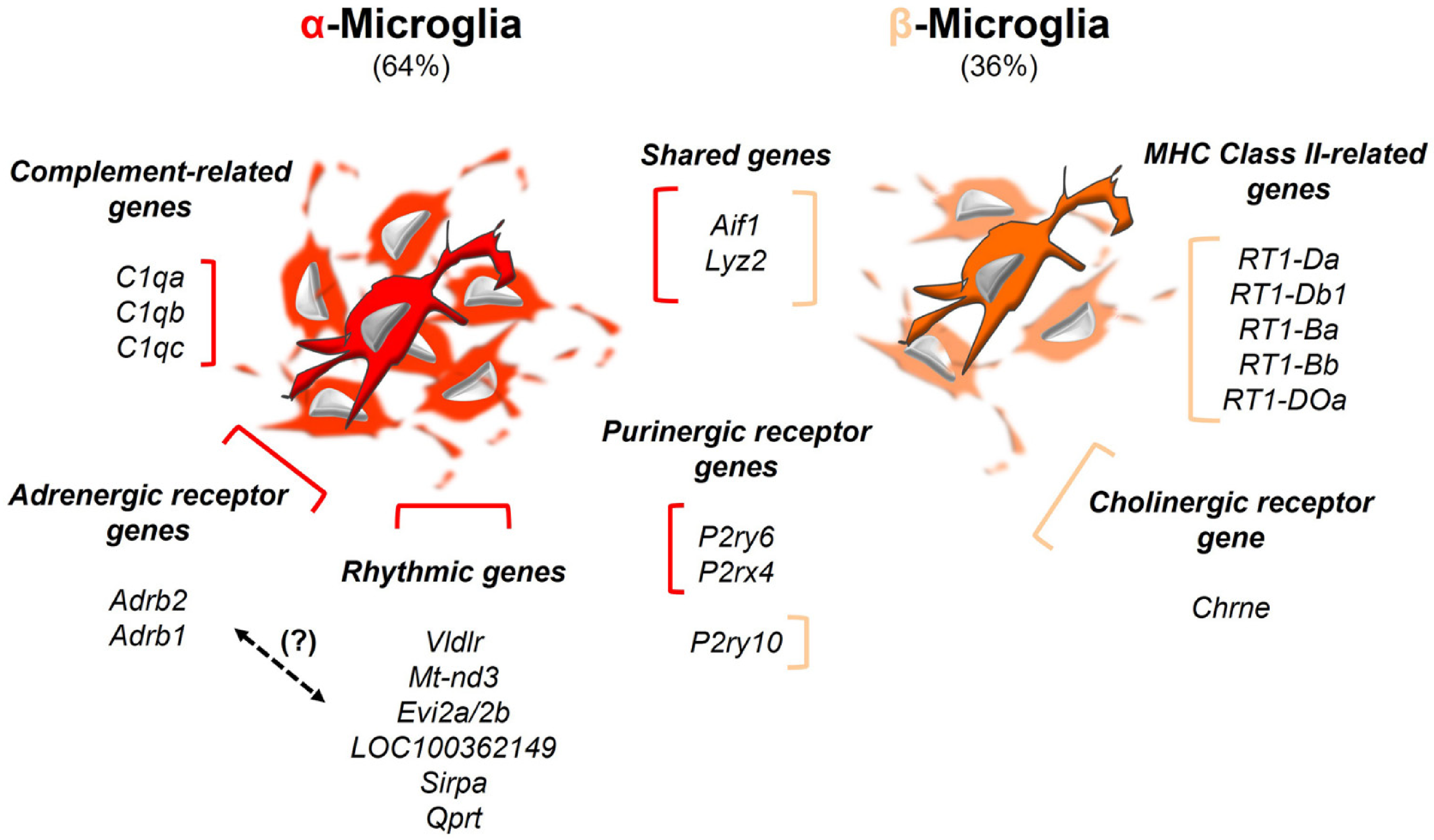
Diversity of microglial phenotypes in the normal adult rat pineal gland. α-Microglia (red) and β-microglia (orange) were distinguished from other pineal cell types by their unique transcriptomic signatures. They represented 64% and 36%, respectively, of all microglial cells that were profiled. Only 1% of total sequenced pineal cells corresponded to these phagocytes. Even though their overall proportion was low, the two Aif1+/Lyz2+ microglial phenotypes were distinctly identified based on the levels of transcripts related to complement components (C1qa, C1qb, and C1qc); MHC Class II proteins (RT1-Da, RT1-Db1, RT1-Ba, RT1-Bb, and RT1-DOa); adrenergic (Adrb2 and Adrb1), purinergic (P2ry6, P2rx4, and P2ry10), and cholinergic (Chrne) receptors, and rhythmically expressed genes (Vldlr, Mt-nd3, Evi2a/2b, LOC100362149, Sirpa, and Qprt), among others. (?): The rhythmic nature of α-microglia might be related to the expression of adrenergic receptors, which differentiates them from β-microglia. See Mays et al. (Mays et al., 2018) for further details.
Microglia Diversity Under Non-Homeostatic Conditions in the Pineal Gland and in Other Circumventricular Organs
As mentioned above, CVOs are constantly activated by exposure to local and systemic stimuli, including cytokines, toxic factors, and pathogens. CVO microglia have been shown to be highly reactive to inflammation induced by lipopolysaccharides (LPSs), which are major components of the outer membrane of gram-negative bacteria. A transient, but robust, proliferation of Tmem119+ microglial cells in mice was reported in some CVOs (AP, ME, OVLT, and SFO), and also in neighboring brain regions, following a single low-dose injection of LPSs derived from Escherichia coli and Salmonella enterica (100 µg/kg of body weight) (Furube et al., 2018). A higher LPS dose (1mg/kg) was observed to cause substantially wider microglia induction that extended into many other areas of the mouse brain. Interestingly, locomotor activity—an indicator of behavior and sickness—was marginally reduced only during the first night following low-dose LPS, while high-dose LPS robustly and persistently affected this activity. This evidence supports the CVOs’ capacity to sense and to respond to mild stimuli before consequences manifest themselves. Ninety minutes following a single non-endotoxemic LPS dose (0.5 mg/kg), microglia morphotypes, and their responsiveness in rat CVOs were shown to vary with respect to the origin of the LPS itself and their potency as both agonists or antagonist of TLR4 (Toll-like receptor 4), and as NF-κB (Nuclear factor-kappa B) inducers (Vargas-Caraveo et al., 2020). This study included the pro-inflammatory LPSs from Escherichia coli and Porphyromonas gingivalis, and also the anti-inflammatory and TLR4-antagonist LPS extracted from the cyanobacteria Rhodobacter sphaeroides.
Regarding the PG, several studies have analyzed how the structure and function of this gland is impacted by cell wall components derived from both gram-negative and gram-positive bacteria (da Silveira Cruz-Machado et al., 2010; da Silveira Cruz-Machado et al., 2012; Fernandes et al., 2017; Ibanez Rodriguez et al., 2018; Jiang-Shieh et al., 2005; Moravcova et al., 2021; Spisska et al., 2020; Tsai and McNulty, 1999). In the rat PG, and 24 h after administering either LPS from Escherichia coli (50 µg/kg) or LTA (lipoteichoic acid) from the gram-positive Staphylococcus aureus (20 mg/kg), microglial cells were found to be activated based on their immunoreactivity for CD11b (OX-42) and MHC Class II antigen (OX-6) (Jiang-Shieh et al., 2005). Additional effects included inducement of GFAP+ (Glial fibrillary acidic protein) astrocytes, vacuolation of pinealocytes, and increased serotonin levels. Interestingly, the diversity found in microglial phenotypes was tracked by the expression levels of CD68, which was a marker induced by LPS, but not by LTA, under the conditions described above. In addition, plasma MEL was reduced only by LPS, but not by LTA, which suggests that differential crosstalk occurs among pinealocytes, microglia, and astrocytes, in these environments. Further understanding is needed of interactions between microglia and astrocytes within the PG and within other CVOs as well, and under both healthy and inflammatory conditions (Clayton et al., 2022; Murayama et al., 2019; Shimada and Hasegawa-Ishii, 2017). Evidence has emerged from experimental models, although less from humans, of how microglia and astrocytes influence and coordinate each other to finally contribute to CNS homeostasis, immune response, BBB maintenance, and synaptic support, among other functions (Garland et al., 2022; Villarreal et al., 2021). Pineal astrocytes represent a highly heterogenous cell population in terms of morphotypes, phenotypes, and regional distribution within the organ (Ibanez Rodriguez et al., 2016; Mays et al., 2018; Moller and Baeres, 2002). This astrocyte heterogeneity might impact microglia responsiveness on a single-cell level. Interestingly, astrocytes, especially γ-astrocytes, continue expressing the essential TF Pax6 in the adult rat PG (Mays et al., 2018). This astrocyte subpopulation might be the preferred cell target for the phagocytic microglia that are present in the mature gland (Farias Altamirano et al., 2019; Ibanez Rodriguez et al., 2016; Ibanez Rodriguez et al., 2018; Munoz, 2018).
Significant advances have been made to understand the molecular and cellular mechanisms behind the regulation of MEL production resulting from an innate immune response (Barbosa Lima et al., 2019; Carvalho-Sousa et al., 2011; da Silveira Cruz-Machado et al., 2010; da Silveira Cruz-Machado et al., 2012; Fernandes et al., 2017; Markus et al., 2018). In the presence of LPS, interaction between pinealocytes and microglia in the rat PG involved the activation of TLR4 and TNF/TNFR1 (TNF receptor 1), the translocation of NF-κB dimers into the cell nuclei, the induction of Tnf/Tnfr1, and the repression of aa-nat (encode for AA-NAT: Arylalkylamine-N-acetyltransferase, penultimate enzyme in the MEL biosynthetic pathway). In vivo inhibition of pineal MEL synthesis by 0.5 mg/kg LPS was reversed by antagonizing either the glucocorticoid receptors or the α1-adrenoreceptors, or also by full adrenalectomy (Fernandes et al., 2017). These results demonstrate the interplay between glucocorticoids and the sympathetic tonus during inflammation. It has been proposed that part of the NF-κB-related mechanisms triggered by local and general inflammation, involves a temporary shift in the source of MEL production from pinealocytes to non-pinealocyte cell types (e.g., activated microglia and macrophages), either within the PG or outside of it (Markus et al., 2018; Pontes et al., 2006). During the resolution phase, the task of MEL production reverts to the pinealocytes, as is normal. Glia-pinealocyte interactions under endotoxin conditions have not yet been fully characterized, including the morphological and phenotypic diversity that may be manifested among microglial cells and their non-parenchymal siblings, and also the precise mechanisms that astrocytes use in their involvement. However, formation of phagocyte clusters has been shown to be a feature of LPS administration in the rat PG, which suggests the proliferation of local microglia and/or the recruitment of monocytes and other macrophages (Ibanez Rodriguez et al., 2018; Jiang-Shieh et al., 2005). Indeed, LPS-induced clustered microglia were found to be highly phagocytic and diverse, based on the expression levels of CD68 and PCNA (Proliferating cell nuclear antigen) (Ibanez Rodriguez et al., 2018).
Heterogeneity at a single-cell level was recently documented in pineal microglia in a neonatal HIBD (hypoxic-ischemic brain damage) rat model (Ding et al., 2022). Perinatal HIBD causes a wide range of neurological symptoms, including motor and cognitive impairment (Benitez et al., 2014; Rice et al., 1981; van der Kooij et al., 2010). Sleep and circadian disruptions in children with HIBD were found to correlate with the occurrence of pineal cysts, a sign of inflammation and cell death (Ding et al., 2016). These processes were also present in the perinatal rat PG, mainly at 72 h post HIBD surgery (Ding et al., 2022). As previously reported in the normal adult rat PG (Mays et al., 2018), five pineal cell types and their subtypes, in both the HIBD and sham operated pups, were identified by scRNA-seq at 24 and 72 h after surgeries (Ding et al., 2022). Among them, pinealocytes, astrocytes and microglia were the most affected by HIBD, which might explain why this model profoundly impairs MEL synthesis (Yang et al., 2017). The two microglial subpopulations, α and β, were mainly distinguished by their differences in expressing genes related to complement components (C1qa, C1qb and C1qc) and MHC Class II proteins (RT1-Da, RT1-Db1 and RT1-Ba), respectively (Ding et al., 2022). Both microglial subtypes did react to the neonatal procedure, but with different temporal transcriptomic signatures. While Ccl3 (C-C motif chemokine ligand 3) and Ccl4 (C-C motif chemokine ligand 4) were upregulated in α-microglia at both time points, β-microglia showed a delayed activation of Ccl3, but not of Ccl4, at 72 h post HIBD. In addition, overall α- and β-microglial cell populations decreased at 72 h post HIBD. This was mainly caused by pyroptosis, which is a kind of inflammation-induced programmed cell death (PCD), that is driven by signaling from NLRP3 (NOD-like receptor family, pyrin domain containing 3), Caspase-1, and GSDMD (Gasdermin D) (Matsudaira and Prinz, 2022). HIBD-induced pyroptosis was confirmed by the authors at both transcriptional and protein levels, and via colocalización of the microglial marker Iba1 with Caspase-1 in pineal sections (Ding et al., 2022).
Interestingly, pineal microglia were found to react differentially to two surgical procedures that disrupt the sympathetic regulation of the gland, which consequently abolishes the MEL circadian rhythm, as well (Ibanez Rodriguez et al., 2018). One of these was the complete excision of both superior cervical ganglia (SCG) known as bilateral SCGx (Superior cervical ganglionectomy) (Madhani et al., 2022; Savastano et al., 2010). The second procedure was bilateral SCGd (Superior cervical ganglia decentralization), which removes 2–3 mm of the sympathetic trunk to each SCG but leaves the efferent nerve connections to the PG intact (Hartley et al., 2015; Madhani et al., 2022). Both surgical models induced transient microgliosis within the PG (Figure 4) (Ibanez Rodriguez et al., 2018). Four days after either SCGx or SCGd, microglial cells, especially those that participate in clusters, were found to be highly phagocytic (high CD68) and enriched in PCNA (Figure 4D and 4E). However, the density of interstitial Pax6+ cells and the interaction between these cells and the Iba1+ microglia were substantially affected in the SCGx PG, but not in the SCGd PG. (Ibanez Rodriguez et al., 2018). This might be related to the local Wallerian degeneration of the sympathetic nerve fibers immunoreactive for Tuj1 (neuronal marker β-tubulin III), that occurs only after the bilateral SCGx (Figure 4C). The effect of these surgical models on phenotypic diversity of pineal microglia has not yet been sufficiently studied.
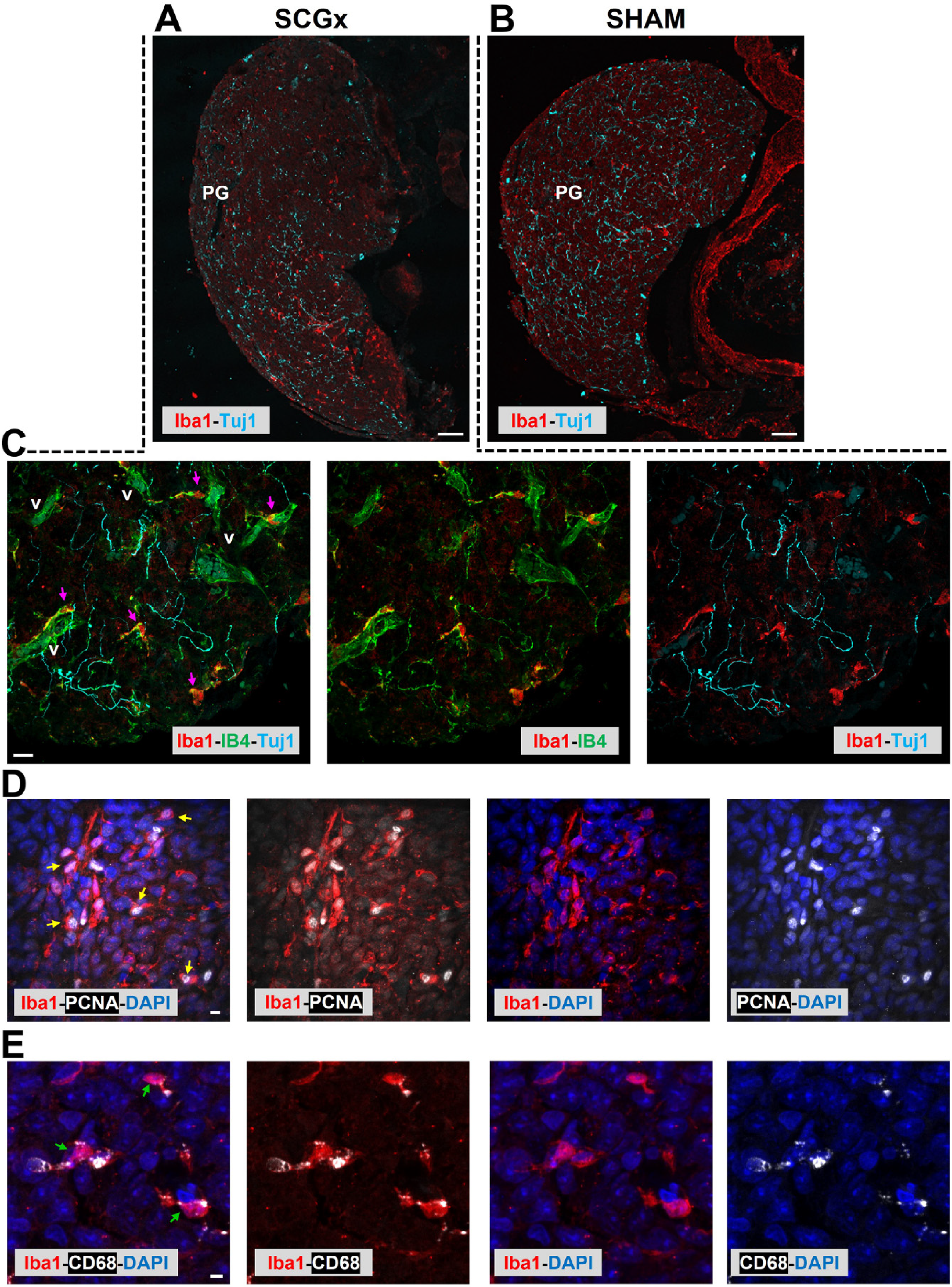
Constantly activated microglia within the pineal gland sense and respond to local inflammation. Pineal gland (PG) sections from adult SCGx and sham rats immunolabeled for Iba1 (red), Tuj1 (cyan), CD68 (white), and PCNA (Proliferating cell nuclear antigen; white). IB4 (green) was used to identify both blood vessels and microglial cells, and DAPI (blue) for staining cell nuclei. A and B: The excision of both superior cervical ganglia (SCGx: bilateral superior cervical ganglionectomy) induces Wallerian degeneration of the Tuj1+ sympathetic nerve fibers within the PG and concomitantly, a transient Iba1+ microgliosis, as compared with sham operated rats. Confocal microscopy of PGs collected 6 days after surgeries; objective: 10X, scale bar: 50 μm. C: Compact Iba1+ microglia (fuchsia arrows) with a few cellular projections, associated with blood vessels (v), interact with degenerated Tuj1+ nerve fibers in the SCGx PG at 6 days after surgery. Objective: 60X, scale bar: 10 μm. D: Microglial cluster in the SCGx PG at 6 days after surgery. Most Iba1+ cells appear immunoreactive for PCNA (yellow arrows). Objective: 100X, scale bar: 5 μm. E: Highly phagocytic Iba1+/CD68+ microglial cells (green arrows) are present in the SCGx PG at 4 days after surgery. Objective: 40X, zoom: 2X, scale bar: 5 μm. Modified from Ibañez et al. (Ibanez Rodriguez et al., 2018), where further details about animal procedures and immunolabeling protocols can be found. The reproduction of this copyrighted material was authorized by the publisher John Wiley & Sons, Inc.
Conclusion and Future Perspectives
In the unique brain structures that are the CVOs, microglia are in a continuous state of activation and are poised to perform urgent tasks as they arise. To dynamically adapt, CVO microglia may exhibit a diverse set of morphotypes and phenotypes. This diversity may vary between regions within the same CVO, and among different CVOs, and across species. Herein, I have reviewed microglia heterogeneity in some CVOs, including the pineal gland (PG), under both normal and pathological conditions. Some description is also included about how non-parenchymal macrophages or CAMSs participate. However, further studies are needed to extend our understanding of this dynamism, especially during ageing and under specific neurodegenerative states. Little is known, for example, about the dynamics of pineal microglia during ageing and age-related diseases, such as Alzheimer's and Parkinson's diseases. Understanding the functional roles of pineal microglia may explain the disruption of the MEL circadian rhythm, as reported in these conditions (Proserpio et al., 2018; Spinedi and Cardinali, 2019; Vallee et al., 2020). Impaired MEL synthesis and secretion might be related to a dysfunctional cell network of pinealocytes, microglia and astrocytes, within the PG. In addition, further studies may clarify microglia involvement in the organogenesis, vascularization, and innervation of the PG, and also in the establishment and functionality of the other pineal cell elements. Interestingly, two independent sc-RNA-seq analyses unraveled at least two distinctive microglial phenotypes, designated as α and β, which were found in both the neonatal and the adult rat PG (Ding et al., 2022; Mays et al., 2018). This suggests that transcriptomic diversity of pineal microglia is established early postnatally, which is consistent with the perinatal definition of the global transcriptomic identity of the PG, as reported in rat (Hartley et al., 2015). It is expected that advanced experimental models, and single-cell and single-nucleus technology will continue to shed light into microglia heterogeneity in the CVOs, and in the CNS at large. Improved understanding in these areas may lead to novel strategies to target and manipulate microglia and macrophages for diagnostic and therapeutic purposes.
Footnotes
Acknowledgments
We thank Raymond D. Astrue for editing the manuscript.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from CONICET (Argentina; PIP-CONICET 112-201101-00247 and PUE 2017; http://www.conicet.gov.ar), ANPCyT (Argentina; PICT 2012-174; PICT 2017-499; ![]() ), and NIH-CONICET (Argentina and USA; F65096).
), and NIH-CONICET (Argentina and USA; F65096).
