Abstract
The cuprizone (CPZ) model allows the study of the biochemical processes underlying nonautoimmune-mediated demyelination, remyelination, and chronic white matter disease progression. CPZ is a copper (Cu) chelator that chiefly causes oligodendrocyte apoptosis in the corpus callosum and cerebellum when administered in the mouse diet. While disruption of Cu homeostasis is known to cause neurodegeneration (as is observed in Wilson’s and Menkes disease), no consensus exists to date as to CPZ’s mechanism of action. We sought to determine whether CPZ-induced pathology is due to Cu depletion as is generally believed. Cu supplementation in chow, in stoichiometric excess to the added CPZ, did not reduce CPZ-induced demyelination in C57Bl/6 mice. Moreover, equivalent doses of other known Cu chelators neocuproine and D-penicillamine (D-Pen) failed to induce central nervous system (CNS) demyelination. Since administration of D-Pen in the treatment of Wilson’s disease can induce hypocupremia, we next sought to recreate penicillamine-induced Cu deficiency to compare with purported CPZ-induced Cu deficiency. The resulting clinical phenotype and histopathology were unlike that of CPZ. D-Pen-treated mice exhibited digit paralysis, tail flaccidity, subcutaneous hemorrhaging, and optic and sciatic neuropathy, all of which were prevented with Cu supplementation. No demyelination of the corpus callosum or cerebellum was observed, even with D-Pen doses tenfold higher than CPZ. Intriguingly, addition of D-Pen to the CPZ diet paradoxically prevented demyelination in a dose-dependent manner.
Summary Statement
The demyelinating effects of CPZ are not due to Cu deficiency but are instead consistent with acute toxicity of a CPZ + Cu complex.
Introduction
Cuprizone (CPZ), a synthetic chelating compound originally used to detect trace Cu (Nilsson, 1950; Peterson & Bollier, 1955; Wetlesen, 1957), became of interest in biomedical research when it was discovered to exert toxic effects in the CNS of laboratory mice (Carlton, 1966). A 0.2-0.5% (w/w) CPZ dose added to normal rodent chow for several weeks causes oligodendrocyte death, gliosis and subsequent demyelination in the corpus callosum, cerebellum, and several other myelinated brain regions (Hiremath et al., 1998; Matsushima & Morell, 2001; Koutsudaki et al., 2008; Gudi et al., 2009; Pott et al., 2009; Nyamoya et al., 2017). The CPZ model is now widely applied in multiple sclerosis (MS) research as an experimental tool to investigate the mechanisms underlying CNS demyelination and remyelination (Kipp et al., 2016), and has more recently been employed to examine how primary myelin degeneration can entrain a secondary immune response into the brain (Scheld et al., 2016; Caprariello et al., 2018; Almuslehi et al., 2020; Kaddatz et al., 2021).
Despite its use in the study of white matter injury and repair for over 50 years, the mechanism of CPZ-induced demyelination is still incompletely understood (reviewed by Kipp et al., 2009; Praet et al., 2014; Zirngibl et al., 2022). Since CPZ chelates Cu, it is widely presumed that CNS damage is a result of Cu dyshomeostasis, as is observed in Menkes and Wilson’s disease, and acquired Cu deficiency in the human (Prodan et al., 2002; Kumar, 2006). As Cu is an essential trace element for a number of metalloenzymes involved in cellular respiration, the prevailing hypothesis suggests that CPZ induces a state of Cu deprivation, which most prominently affects oligodendrocytes due to their elevated metabolic demands associated with myelination.
To explore this hypothesis, published studies aimed at determining the status of Cu in the brains of CPZ-treated mice have yielded discrepant results. An initial report by Venturini (1973) employed a colorimetric assay based on CPZ’s proclivity to turn blue when bound to trace amounts of Cu in solution (Peterson & Bollier, 1955). Brains from CPZ-fed mice, desiccated and reconstituted with dissolved CPZ, had lower absorbance values than control mouse brains, suggestive of a reduced Cu content. However, more recent experiments using inductively-coupled plasma spectroscopy and X-ray fluorescence reported normal brain Cu content (Jeyasingham et al., 1998; Moldovan et al., 2015). Disconcertingly, additional studies using atomic absorption spectrophotometry (Zatta et al., 2005) and commercial colorimetric Cu assay kits (Tezuka et al., 2013) reported increased brain Cu levels with CPZ treatment. To add to this ambiguity, several attempts by Carlton (1966, 1967) to supplement the CPZ diet with copper sulfate failed to prevent CNS abnormalities. However, the experiments used weanling mice, which are highly susceptible to CPZ toxicity, and used a maximum supplement of only 260 ppm copper sulfate. CPZ binds Cu with a stoichiometry of 2 CPZ:1 Cu (Zatta et al., 2005; Messori et al., 2007), therefore addition of 260 ppm Cu salts was greatly sub-stoichiometric given the 0.2–0.5% (w/w) CPZ administered to the animals in these studies. Based on the stoichiometry we reasoned that a >0.5 molar excess of Cu over CPZ would have been required to bind all the CPZ, leaving some available free Cu to prevent toxicity in Carlton’s experiments.
In this study, we first attempted to prevent 0.2% CPZ-induced demyelination in 8-week-old C57Bl/6 mice by supplementing with a slight stoichiometric excess of Cu. We then attempted to induce CNS demyelination via dietary administration of other well-established Cu chelators by way of comparison to CPZ. Taken together, our results indicate that CPZ’s toxicity is not due to Cu depletion, but instead is consistent with a gain-of-toxicity due to an unusual CPZ:Cu complex.
Materials and Methods
Animal experiments and tissue collection
All animal experiments were carried out in accordance with guidelines set out by the Canadian Council on Animal Care. Male 8-week-old C57Bl/6 mice (Charles River Laboratories, RRID:MGI:2159769) were fed the following compounds (% w/w) mixed in with standard chow (0006955, Pico-Vac® Mouse Diet 20, LabDiet) ad libitum for 2 to 3 weeks: 0.2% CPZ (bis(cyclohexanone)oxaldihydrazone, C9012, Sigma Aldrich), 0.15% neocuproine hydrochloride hydrate (N1626, Sigma Aldrich), 0.16% to 0.25% D-(-)-Penicillamine (A11446.22, Alfa Aesar), 0.1% copper (II) sulfate pentahydrate (469130, Sigma Aldrich), or a combination of the above. To induce unambiguous Cu deficiency via chelation, the high affinity Cu chelator D-(-)-Penicillamine (D-Pen) (Walshe, 1956; Birker & Freeman, 1977) was administered in the diet of 3-week-old C57Bl/6 female mice (either 2% D-Pen for 4 weeks or 1% D-Pen without or with 0.92% copper sulfate for 12 weeks). The binding stoichiometry of D-Pen to Cu is unclear (Birker & Freeman, 1977; Gergely & Sóvágó, 1978; Ahmed & Iqbal, 2016), and so we replicated the CPZ + Cu experimental paradigm by assuming a similar binding ratio and supplementing with a 10% molar excess of Cu (2:1.1 D-Pen:Cu). Weanling females were used for the following three reasons: 1) older mice are less susceptible to the effects of D-Pen (Takeda et al., 1980; our own unpublished findings), 2) older mice are less susceptible to clinical manifestation of Cu deficiency in general (Prohaska, 1991; Prohaska & Brokate, 2002), and 3) to avoid the non-specific side effects of high-dose D-Pen observed in young male mice (Takeda et al., 1980). To compare 2% D-Pen to CPZ, 3-week-old female C57Bl6 mice were fed 0.2% CPZ for 4 weeks. Age- and sex-matched untreated control mice were fed normal chow for 2, 3, 4, or 12 weeks as appropriate.
For histological and immunohistochemical studies, mice were deeply anaesthetized with sodium pentobarbital and transcardially perfused with 12 mL 1X PBS followed by 12 mL 4% paraformaldehyde. CNS and peripheral nervous system (PNS) tissues were harvested and postfixed overnight in 4% paraformaldehyde at 4
Histology and immunohistochemistry
To examine the extent of demyelination in the corpus callosum, brain sections were stained overnight at 60
For immunohistochemistry, the M.O.M (Mouse on Mouse) Immunodetection Kit (BMK-2202, Vector Laboratories) was used for blocking and antibody dilution buffers. Sections were first delipidated in ethanol to expose the citrullinated myelin basic protein (MBP) epitope (1 min each in 50%, 70%, 95%, 100%, 100%, 95%, 70%, and 50% EtOH), then incubated with anti-MBP (RRID:AB_2920596, Citrulline R25, clone 1B8, MABT1510, Sigma-Aldrich, 1:500) and anti-Iba1 (RRID:AB_839504, Cat#:019-19741, Wako Chemicals USA, 1:500) antibodies overnight at 4
Mouse behavioural testing and clinical examination
To compare the clinical effects of D-Pen vs. CPZ treatment, mice were scored in a blinded fashion at baseline and once weekly using the hindlimb clasping test, wire hang test, and were also observed weekly for overt clinical signs such as tail flaccidity and digit paresis and paralysis (an inability to grip the metal bars of the cage top). The hindlimb clasping test was performed as previously described (Guyenet et al., 2010), wherein mice were lifted by the base of the tail, hindlimb position was observed for 10 s and scored from 0 to 3 based on limb retraction. The hindlimb clasping score for each mouse was presented as an average of three separate trials. The wire hang test was performed by placing the mouse on the wire cage top, which was then inverted. Performance was recorded as the latency from the beginning of the test to when the mouse fell. Mice were tested for a maximum of 60 s, and the score for each mouse was presented as an average of three separate trials.
Colorimetric assays of Cu chelators
Solutions of 1mM CPZ, D-Pen, and neocuproine were prepared separately in 1X PBS. To solubilize CPZ and neocuproine, 10mM was added to 30% EtOH and incubated on a shaker at 37
Image analysis and statistics
Brightfield and fluorescence images were analyzed using ImageTrak software (version 5.4.0a; https://stysneurolab.org/imagetrak) using thresholding to mask the area of LFB, citrullinated MBP, or Iba1 labeling. Statistical significance was determined using one-way ANOVA in GraphPad Prism software (version 9.3.1). P < 0.05 was considered significant. Results for each mouse are presented as an average of three tissue sections, with error bars representing standard error of the mean for each experimental group.
Data for digit paralysis and hindlimb clasping scores for mice treated with 1% D-Pen ± copper sulfate was analyzed using repeated measurements, two-way ANOVA. If the p value calculated for the main effect of the treatment was < 0.05, Fisher’s LSD post hoc tests were applied to each time point.
Results
Dietary Cu supplementation did not reduce CPZ-induced demyelination
To determine whether CPZ-induced pathology could be prevented with adequate (supra-stoichiometric) Cu supplementation, mice were fed 0.2% CPZ without or with 0.1% copper sulfate (Cu) added (i.e., a 2:1.1 stoichiometric ratio of CPZ (278g/mol) to CuSO4•5H2O (250g/mol) to ensure a slight excess of free Cu) for 3 weeks, and histopathology of the splenium of the corpus callosum was examined by LFB staining (Figure 1a). Since the extent and reproducibility of CPZ-induced pathology is highly dependent on mouse weight (Leopold et al., 2019), all mice were weighed at the start of each experiment to ensure an even distribution among groups (Figure 1b). While CPZ-fed mice exhibited reduced weight compared to controls, Cu supplementation had no effect, with CPZ ± Cu mice maintaining similar weights throughout the 3-week experiment, indicating that food intake was not altered by the addition of copper sulfate. Contrary to expectations however, Cu supplementation did not prevent myelin loss after 3 weeks of CPZ treatment (Figure 1c).
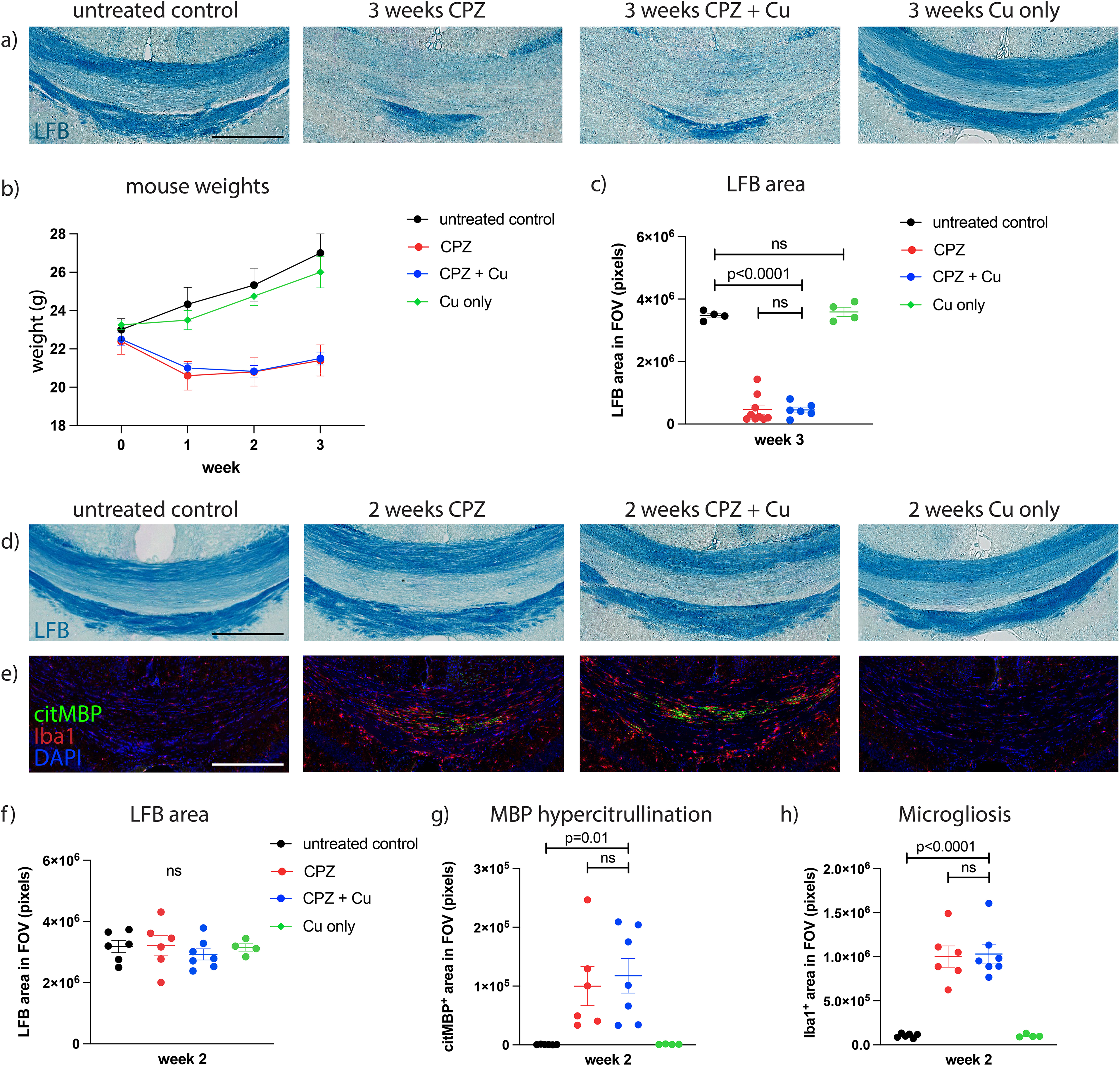
Dietary Cu supplementation did not reduce CPZ-induced myelin damage. (a) Mice were administered 0.2% CPZ ± 0.1% copper sulfate (Cu) or 0.1% Cu alone in standard rodent chow for 3 weeks and the splenium of the corpus callosum was examined via Luxol fast blue (LFB) staining. (b) All mice were weighed at baseline and once per week throughout the 3-week experiment. (c) Quantification of myelin content based on LFB staining. Cu supplementation did not prevent CPZ-induced myelin loss. (d-e) Mice were administered CPZ ± Cu for an abbreviated 2-week period and brain tissue sections stained with LFB or anti-citrullinated MBP (citMBP) and anti-microglia/macrophage (Iba1) antibodies. (f-h) 2 weeks of CPZ did not produce overt demyelination by LFB staining, but induced microgliosis and hypercitrullination of myelin in the medial corpus callosum that was not alleviated with Cu supplementation. Cu supplementation in the absence of CPZ did not cause myelin damage. Each dot on the graph in (c), (f), (g), and (h) represents one mouse. Error bars indicate SEM. Scale bars are 300 μm.
Since demyelination of the medial corpus callosum was already extensive by week 3, to see whether Cu supplementation conferred more subtle improvements to CPZ-induced pathology, we shortened the CPZ duration to 2 weeks, wherein overt demyelination is not yet visible by LFB staining but white matter is hypercitrullinated (Caprariello et al., 2018) and microglia are activated (Hiremath et al., 1998) (Figure 1d-e). Citrullinated MBP, largely absent in the naïve mouse corpus callosum, is a sensitive marker of early damage to myelin that both precedes and accompanies acute demyelination in both the CPZ model and in MS (Yang et al., 2016; Caprariello et al., 2018). This abbreviated sub-demyelinating CPZ paradigm induced citrullination of MBP and microgliosis in the medial corpus callosum as expected, but Cu supplementation again failed to rescue this subtle pathology (Figure 1f-h). Cu administration alone did not cause any white matter pathology.
Other Cu chelators failed to induce cerebral demyelination
To determine whether other well-established Cu chelating agents could create a similar pattern of demyelination as CPZ, we first identified other compounds that efficiently bind Cu. Since D-Pen, neocuproine, and CPZ each undergo color transformation in solution when complexed with Cu (to purple, yellow, and blue, respectively (Nilsson, 1950; Gahler, 1954; Birker & Freeman, 1977), Figure 2a), the relative affinity of each chelator for Cu could be examined. A solution of CPZ + Cu was prepared, and then either D-Pen or neocuproine was added. The color change in each case indicated that both D-Pen and neocuproine have a higher affinity for Cu than CPZ (Figure 2b). The final color was stable at room temperature for over 24 h, and did not change back to blue with the addition of more CPZ. From these experiments we concluded that a) compared to CPZ, both D-Pen and neocuproine will bind Cu more strongly, and b) these chelators will also extract Cu already bound by CPZ (supported by the color changes in Fig. 2). Having established that both D-Pen and neocuproine are also effective copper chelators, mice were then fed either D-Pen or neocuproine mixed in with standard rodent chow for 3 weeks. Since neocuproine also binds Cu in a 2:1 ratio (Özyürek et al., 2011), 0.15% neocuproine was used (a molar equivalent dose to 0.2% CPZ). The binding ratio of D-Pen to Cu is unclear (Birker & Freeman, 1977; Gergely & Sóvágó, 1978; Ahmed & Iqbal, 2016), and so 0.16% D-Pen was administered (a 50% higher molar amount than 0.2% CPZ). In striking contrast to CPZ, histopathology of the corpus callosum examined with LFB was unremarkable after a 3-week exposure to either of the two chelators (Figure 2c-d). Likewise, no behavioural changes were observed in any of the animals aside from weight loss in the neocuproine-treated mice (Figure 2e).
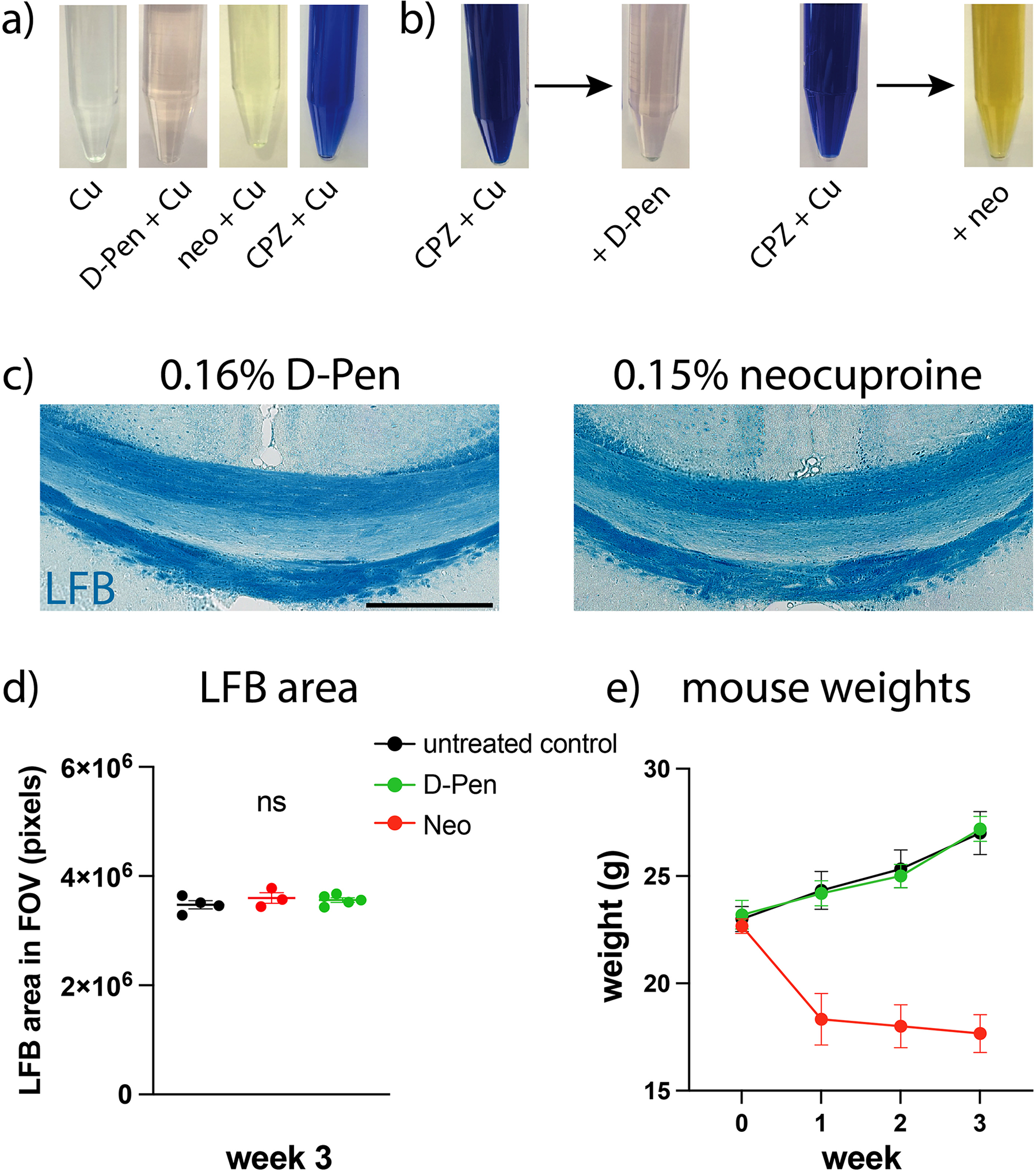
Other Cu chelators failed to induce cerebral demyelination. (a)The Cu chelators D-penicillamine (D-Pen), neocuproine (neo), and CPZ each undergo unique color change in the presence of Cu in aqueous solution. (b) When D-Pen or neocuproine was added to a deep-blue solution of 1mM CPZ + 0.5mM copper sulfate a marked color change was noted resembling the hue of each chelator without CPZ. Colors remained stable at room temperature and did not change with the addition of more CPZ (not shown). (c) 8-week-old male mice were administered 0.16% D-Pen or 0.15% neocuproine (w/w) in rodent chow for 3 weeks and the splenium examined by Luxol fast blue (LFB) staining. (d) No myelin loss was observed in the corpus callosum with either chelator, despite confirmation that both bind Cu even more tightly than CPZ. Each dot on the graph represents one mouse. (e) The only clinically notable feature was weight loss in the neocuproine-fed mice. Error bars indicate SEM. Scale bars indicate 300 μm.
D-Pen-induced pathology was very distinct from CPZ
With the aforementioned results now casting doubt on the alleged Cu depleting mechanism of CPZ, we next sought to recreate a model of unambiguous Cu depletion via chelation in C57Bl/6 mice for a direct comparison with the CPZ model. Since administration of D-Pen for the treatment of Wilson’s disease can induce hypocupremia and CNS demyelination (Narayan & Kaveer, 2006), we aimed to produce a state of Cu deficiency in 3-week-old female weanling mice by using a high dose of D-Pen (2%) in the diet. Behaviour and histopathology were compared with age- and sex-matched control mice fed standard rodent chow or 0.2% CPZ for 4 weeks.
The high D-Pen dose induced overt clinical symptoms within a few weeks. Experiments were terminated at week 4, when several mice died of sudden aortic rupture (Takeda et al., 1980). All surviving mice at week 4 presented with tail flaccidity, splayed hind toes with digit paralysis in both hindlimbs (Figure 3a), hindlimb weakness, and severely impaired hindlimb clasping and wire hang scores (Figure 3b-c). At necropsy, subcutaneous haemorrhaging was observed in all D-Pen-treated mice. Conversely, CPZ-treated mice were clinically and behaviourally indistinguishable from untreated controls at week 4, with the exception of one CPZ-treated mouse with hydrocephalus. Several CNS and PNS tissues were labeled for microglia/macrophages and citrullinated MBP; as expected, the medial corpus callosum and cerebellar white matter of CPZ-treated mice exhibited gliosis and MBP hypercitrullination (Figure 3d-e). D-Pen-treated mice, however, exhibited optic and sciatic neuropathy (Figure 3f-g). No evidence of white matter injury was found in the brain or cerebellum of D-Pen-treated mice despite using a dose tenfold higher than CPZ. No spinal cord pathology was detected in any of the treatment groups (data not shown).
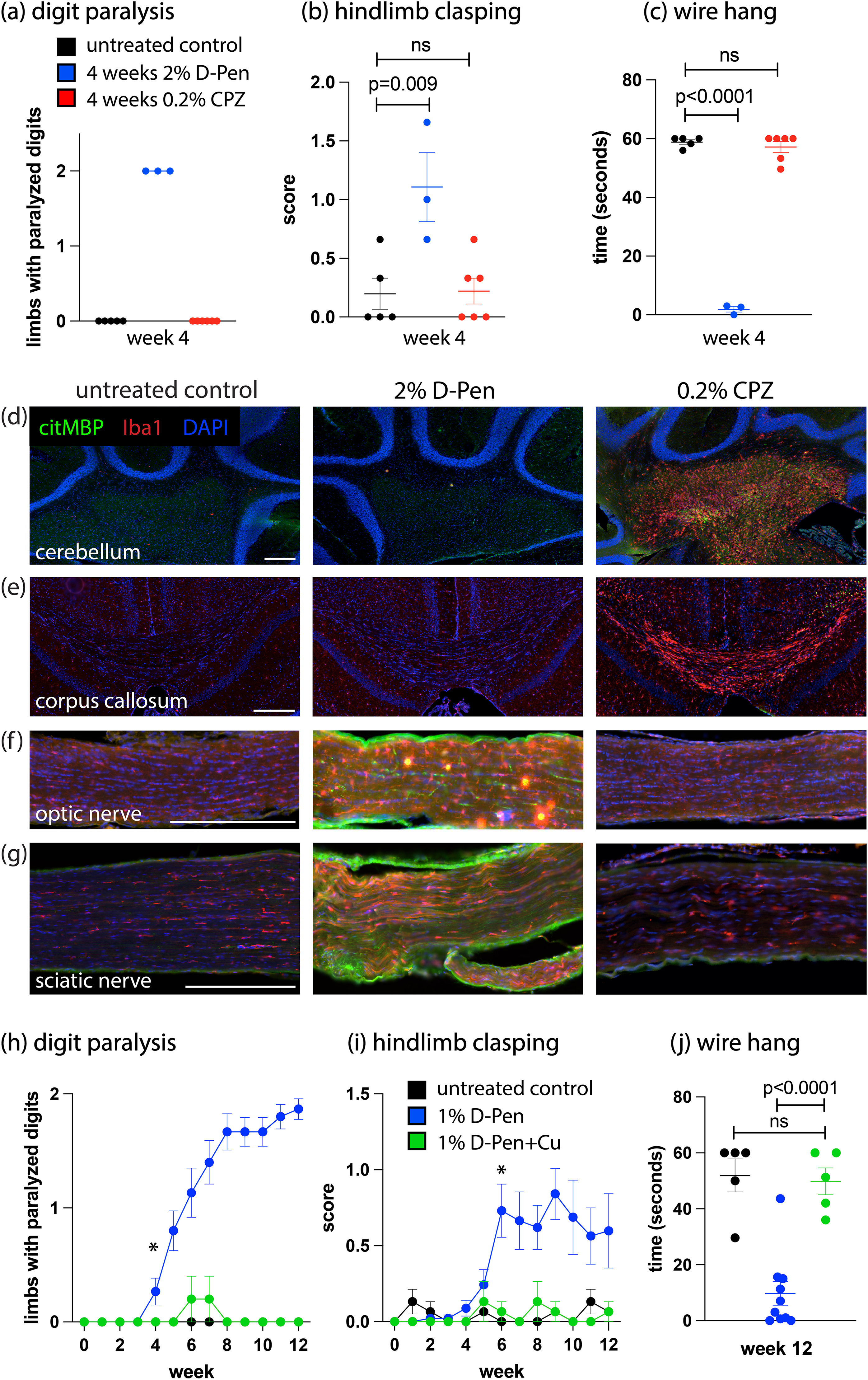
D-Pen-induced C u deficiency was unlike purported CPZ-induced Cu deficiency. (a-c) After 4 weeks of 2% D-Pen supplementation in rodent chow, mice exhibited digit paralysis in both hindlimbs (an inability to grasp the metal wires of the cage top), along with high hindlimb clasping scores and low wire hang scores. CPZ-treated mice were clinically unremarkable and behaviourally indistinguishable from untreated control mice. (d-f) mouse CNS and PNS cryosections labeled with anti-citrullinated MBP (citMBP) and anti-microglia/macrophage (Iba1) antibodies. (h-i) Mice were administered either 1% D-Pen or 1% D-Pen + 0.92% copper sulfate for 12 weeks, with digit paralysis and hindlimb clasping recorded weekly. 1%-D-Pen-treated mice began to exhibit digit paralysis at week 4, and progressed until week 12 when most mice had digit paralysis in both hindlimbs. Hindlimb clasping scores became statistically significant by week 6 of treatment. * indicates the first time point at which 1% D-Pen mice have scores that are significantly different from 1% D-Pen + Cu mice (p < 0.05, repeated measurements ANOVA with Fisher’s LSD post hoc tests). (j) At week 12, D-Pen-treated mice had significantly abnormal wire hang scores. Mice administered 1% D-Pen + Cu were clinically and behaviourally indistinguishable from untreated controls. Each dot on the graph in (a-c), and (j) represents one mouse. Error bars indicate SEM. Scale bars are 300 μm.
To confirm whether the clinical phenotype observed in D-Pen-treated mice was due to Cu deficiency, a different cohort of weanling mice was fed 1% D-Pen ± 0.92% copper sulfate for up to 12 weeks and monitored weekly for clinical signs. A lower amount of D-Pen was used in this experiment to prevent aortic rupture. Mice fed 1% D-Pen began to develop digit paralysis by week 4 (Figure 3h), displayed significantly abnormal hindlimb clasping scores by week 6 (Figure 3i), and at week 12 had severely impaired wire hang scores compared to untreated controls (Figure 3j). Cu supplementation completely prevented the appearance of clinical signs for the entire 12-week duration of the experiment.
D-Pen supplementation paradoxically prevented CPZ-induced demyelination
If the mechanism of action of CPZ-induced demyelination is depletion of Cu, one would expect that addition of a second (higher-affinity) chelator to CPZ in the chow would exacerbate callosal pathology or even induce more widespread CNS injury as is known to occur at higher doses of CPZ (Carlton, 1966; Kesterson & Carlton, 1971; Blakemore, 1972). When D-Pen was added to the 3-week 0.2% CPZ diet, it unexpectedly prevented myelin loss in a dose-dependent manner despite ostensibly causing even more Cu chelation (Figure 4a). Intriguingly, the higher the dose of D-Pen, the more protection was observed so that at 0.25% D-Pen completely protected against demyelination in all mice, resulting in the corpus callosum being histologically indistinguishable from controls (Figure 4b). All mice remained clinically unremarkable.
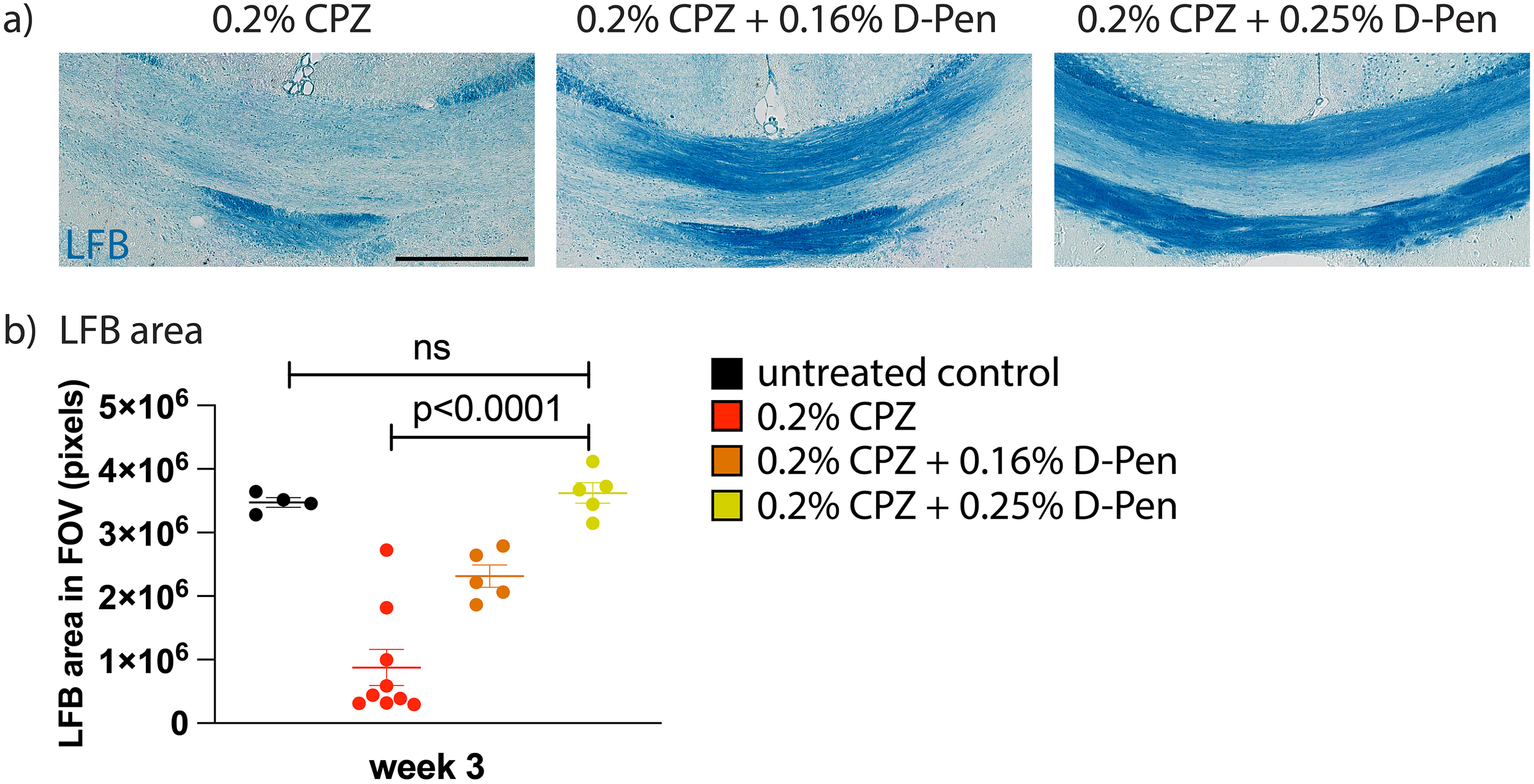
D-Pen supplementation paradoxically prevented CPZ-induced demyelination. (a) Mice were administered 0.2% CPZ ± D-Pen in rodent chow for 3 weeks and the splenium examined via Luxol fast blue (LFB) staining. (b) The addition of D-Pen to the CPZ diet prevented demyelination in a dose-dependent manner. At 0.25% D-Pen prevented all myelin loss and was statistically indistinguishable from untreated controls. Each dot on the graph represents one mouse. Error bars indicate SEM. Scale bar is 300 μm.
Discussion
The mechanism(s) of CPZ-mediated CNS demyelination in the rodent have never been firmly established. Given the selectivity and high affinity of CPZ for Cu (Peterson & Bollier, 1955; Rohde, 1966), together with reported demyelinating syndromes in hypocupremic patients (Prodan et al., 2002; Kumar, 2006), depletion of Cu as a main mechanism of toxicity of oligodendrocytes was a reasonable conclusion. If so, Cu supplementation should prevent the pathological effects of CPZ. We confirmed the well-known observation of demyelination of the medial corpus callosum by dietary CPZ (Hiremath et al., 1998; Matsushima & Morell, 2001). What was unexpected in our experiments was a complete inability of Cu (deliberately administered in stoichiometric excess to CPZ to ensure that all chelator was bound to exogenously supplied Cu) to rescue white matter pathology (Fig. 1). These results shed serious doubt on the notion that CPZ exerts toxicity by chelating and removing Cu. In other words, it appears that it is not a deficit of Cu that induces demyelination, but the presence of CPZ itself, either alone or as a CPZ:Cu complex. The fact that CPZ with Cu added in suprastoichiometric amounts had similar toxic effects implies that free CPZ was not the culprit, as all CPZ would be Cu-bound under such conditions. Nor was copper sulfate alone toxic, together indicating that a CPZ:Cu complex may be responsible. Strong evidence to support this conclusion was provided by exposing mice to CPZ together with another Cu chelator with a higher affinity for the metal (Fig. 2). Rather than exacerbating pathology as would be expected if Cu depletion were the mechanism, addition of D-Pen completely abolished the toxic effects of CPZ (Fig. 4). Taken together, we interpret the findings as being consistent with a Cu(CPZ)2 complex as the toxic species (Messori et al., 2007; Yamammoto & Kuwata, 2009). Having a higher affinity for Cu than CPZ, D-Pen would remove this metal from the Cu(CPZ)2 complex rendering it inert. How a Cu(CPZ)2 complex would exert toxicity is unclear, but one possibility may involve the complex coordination chemistry between CPZ and the metal. Notably, after binding Cu2+, studies have suggested that this process gives rise to an unusual high-valent Cu3+ state yielding a Cu3+(CPZ)2− complex (Messori et al., 2007; Yamammoto & Kuwata, 2009; Kundu et al., 2016). Such high oxidation states of Cu could disrupt vital metalloproteins and can participate in Fenton-type redox reactions that could be particularly damaging to lipid-rich structures such as myelin via peroxidation. Indeed, mature oligodendrocytes are highly vulnerable to oxidative stress, due to their high levels of intracellular iron and low endogenous levels of manganese superoxide dismutase, glutathione, and metallothioneins (Griot et al., 1990; Connor & Menzies, 1995; Nakajima & Suzuki, 1995; Thorburne & Juurlink, 1995; Back et al., 1998; Juurlink et al., 1998; Bernardo et al., 2003). In support, CPZ has been shown to cause lipid peroxidation and subsequent ferroptosis of oligodendrocytes after only a few days of treatment (Jhelum et al., 2020). Finally, experiments using protein-bound Cu or small-molecule mimics of protein-Cu binding sites suggest that rather than actively chelating away Cu, CPZ complexes with these molecules (Lindström and Pettersson, 1974; Taraboletti et al., 2017), indicating that its demyelinating action could be caused by toxic gain-of-function and/or the formation of unstable complexes with cuproproteins (Messori et al., 2007).
The above apparent toxic gain-of-function due to a presumed Cu3+(CPZ)2− complex resulted in distinctly different pathology compared with Cu depletion by traditional chelators. D-Pen-treated mice displayed symptoms more akin to mottled mutants (a murine genetic analogue of Menkes disease (Tümer & Møller, 2010), which also display hindlimb weakness and susceptibility to aortic rupture (Rowe et al., 1974; Lenartowicz et al., 2012). D-Pen mice likewise displayed symptoms similar to mice treated with Cu-deficient diets from birth, which also present with splaying of the hind toes and hindlimb weakness (Zucconi et al., 2007). Parallel experiments performed on DDD mice with low doses of D-Pen administered chronically (Takeda et al., 1980) yielded similar sporadic clinical symptoms (e.g., digit paralysis and aortic aneurysm). None of the above clinical features were present in CPZ-treated mice. Also, dietary D-Pen induced a very different histological pattern, showing myelin alterations in the optic nerve and PNS, with no evidence of callosal or cerebellar injury that is typical of CPZ. Importantly, Cu supplementation completely prevented D-Pen-induced abnormalities (Fig. 3) suggesting this chelator induced toxicity by actual depletion of Cu; this was in striking contrast to CPZ whose effects could not be rescued by Cu supplementation. Finally, pathological manifestation in the D-Pen model required several weeks of administration at high doses (2% w/w), presumably reflecting a slow depletion of total body Cu, whereas 0.2% CPZ induces significant loss of oligodendrocytes within just 48 h (Jhelum et al., 2020). To date, it remains unclear whether either of these Cu chelators enters the CNS. It is possible that CPZ more efficiently enters the corpus callosum to disrupt Cu homeostasis, resulting in differing histopathology.
Cu-bound CPZ may exert toxicity directly on oligodendrocytes, however, it may also act on CNS immune cells which then secondarily trigger cytotoxicity. Microglia and astrocyte activation are early events that precede demyelination in the CPZ model (Tezuka et al., 2013), and ablation of astrocytes reduces CPZ-induced demyelination (Madadi et al., 2019). Astrocytes are key regulators of copper homeostasis in the brain by controlling the intake, storage, and export of this metal within the cell (Dringen et al., 2013). Our data are consistent with in vivo CPZ experiments showing upregulation of astrocytic copper transporter markers in the white matter, suggesting increased (rather than decreased) Cu availability in CNS tissues (Colombo et al., 2021). The protective effect of D-Pen supplementation in the CPZ diet could therefore also be interpreted as high-affinity chelation of astrocyte-derived copper. For an extensive review on the numerous potential downstream effects of CPZ:Cu toxicity, whether via direct action on oligodendrocytes or via toxification of CNS innate immune cells, we refer to Zirngibl et al., 2022.
Although the mechanisms by which CPZ:Cu complexes damage intact white matter are unknown, the reported effects of Cu on glutamate receptors, particularly Ca-permeable N-Methyl-D-Aspartate (NMDA) receptors (You et al., 2012; Huang et al., 2018), may play an important role. NMDA receptors are dysregulated in both CPZ-treated mice and in MS NAWM, particularly in the corpus callosum (Tameh et al., 2013; Luchicchi et al., 2021), implicating the potential role of glutamate excitotoxicity, to which oligodendrocytes are exquisitely sensitive (Matute et al., 2001). We have reported on a unique signaling relationship between axons and their overlying myelin which we termed the axo-myelinic synapse, wherein glutamate is released from electrically active axons to activate NMDA and AMPA receptors on the adjacent adaxonal myelin (Micu et al., 2016; Saab et al., 2016; Micu et al., 2018). Given the potent effects of Cu ions on these receptors (You et al., 2012; Huang et al., 2018), including modulation of Ca levels in myelin itself (Tsutsui et al., 2022), it is plausible that toxic CPZ:Cu complexes perturb this signaling leading to excitotoxic damage. Recent data additionally suggest that the in vivo situation may be very complex involving indirect actions of astrocytes as important players in Cu dysregulation in the intact CNS (Colombo et al., 2021).
In conclusion, the well-known effects of the demyelinating agent CPZ are not due to depletion of Cu as might be expected, but instead are consistent with generation of toxic CPZ:Cu complexes. This may have implications for our understanding of injury mechanisms in disorders of white matter such as MS where pathological changes are similar to those induced by CPZ:Cu.
Footnotes
Abbreviations
Acknowledgements
The authors acknowledge the University of Calgary Hotchkiss Brain Institute Advanced Microscopy Platform and the Live Cell Imaging Resource Laboratory for slide scanning equipment used in sample imaging.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Multiple Sclerosis Society of Canada, Canada Research Chairs, Canadian Institutes for Health Research
