Abstract
A key aim of therapy for multiple sclerosis (MS) is to promote the regeneration of oligodendrocytes and remyelination in the central nervous system (CNS). The present study provides evidence that the vitamin K-dependent protein growth arrest specific 6 (Gas6) promotes such repair in in vitro cultures of mouse optic nerve and cerebellum. We first determined expression of Gas6 and TAM (
Introduction
Multiple sclerosis (MS) is an autoimmune disease of the central nervous system (CNS) that involves destruction of oligodendrocytes and of the myelin sheath they produce around axons. MS features an inflammatory response that causes gradual demyelination through formation of plaques or lesions in the CNS white matter, leading to disruption in nerve impulse transmission (Saxena et al., 2011). Remyelination occurs in MS by the recruitment of oligodendrocyte progenitor cells (OPCs) to the lesion site and their subsequent differentiation into myelinating oligodendrocytes, under the control of multiple extracellular and axon-derived factors (Wolswijk, 2002). However, remyelination by OPCs ultimately fails, due in part to their differentiation being suppressed by inhibitory factors and the lack of a supportive cellular environment (Franklin, 2002; Kuhlmann et al., 2008). Hence, there is a need for a greater understanding of the factors regulating OPC differentiation and the development of novel targets for promoting remyelination.
Growth arrest specific 6 (Gas6) is a vitamin K-dependent protein that is a ligand for the TAM (
Materials and Methods
Animals
C57/BL6 mice aged postnatal day 7 (P7), P14, and adults were used for developmental studies, and 2-month-old adults were used for ex vivo experiments utilizing optic nerves and organotypic cerebellar slices cultures. Also, a transgenic mouse line was used in which green fluorescent protein (GFP) was expressed under control of the SOX10 gene promoter (Stolt et al., 2006; Azim and Butt, 2011). All experiments involving animals were approved by University of Portsmouth Ethics Committee and also by the Home Office Animals Scientific Procedures Act (1986). Animals were killed humanely by cervical dislocation, and brains were removed rapidly and placed in ice-cold saline or fixative prior to experiments.
Real-Time Quantitative PCR
Total RNA was isolated from human oligodendrocyte precursor cells (HOPC; 1600; ScienCell, CA), human astrocyte-cerebellar (HA-c; 1810; ScienCell), cultured optic nerves, and cortex and cerebellum of mice at different ages (P7, P14, and adult) using GeneJET RNA purification kit (Thermo Scientific, Loughborough, UK). The RNA was reverse transcribed into first-strand cDNA (NanoScript RT kit, Primerdesign, Southampton, UK) prior to use in real-time quantitative polymerase chain reaction (qPCR) analysis. Samples from human cell lines and from cortex and cerebellum of different ages were used to quantify gene expression, using specific primers and probes (Integrated DNA Technologies, IDT; Leuven, Belgium; Supplementary Table 1) and a qPCR master mix (FastStart Essential DNA Probes Master; Roche, Burgess Hill, UK). Also, cDNA from cultured optic nerves was analyzed for expression of 84 MS-related genes in a qPCR mini-array format (RT2 Profiler™ PCR Array Mouse Multiple Sclerosis; PAMM-125 Z; Qiagen, Hilden, Germany). The human and mouse samples were normalized to GAPDH and Cdc40 as respective housekeeping genes for each species. The Cdc40 gene was selected based on our own preliminary study of 12 reference genes (geNorm; Primerdesign Ltd), which revealed Cdc40 to be among the most stable of mouse reference genes (not shown). The qPCR data from various mouse CNS tissues were analyzed based on the relative standard curve method as described earlier (Vouri et al., 2015). The qPCR amplification data from human samples and from optic nerves used in qPCR mini-array were analyzed based on the 2−ΔΔCt method, where ΔCt is CtTarget gene – Cthousekeeping gene and ΔΔCt is ΔCtGas6 – ΔCtMock, and 2−ΔΔCt shows the fold up- or downregulation, where values >1 are upregulated and <1 are downregulated (Livak and Schmittgen, 2001). The qPCR mouse mini-array contained 84 different genes involved in different aspects of MS, including myelination, T-cell activation and signaling, adaptive immunity, inflammation, and apoptosis (listed in Supplementary Table 2).
Western Blot
Total protein extracts were obtained from fresh brain regions from mice at P7, P14, and adult ages, as well as from cultured optic nerves, using lysis buffer composed of 50 mM Tris-HCl, 150 mM NaCl, 1% Triton X-100, 0.5% NP-40, 1 mM EDTA, 10 mM Na4P2O7, pH 8.0. The extracts were loaded in equal total protein amounts on a 10% SDS-polyacrylamide gel, and proteins were separated by electrophoresis as described earlier (Goudarzi et al., 2013). Separated proteins were then transferred to a polyvinylidene fluoride membrane (Immobilon-P; Millipore, Watford, UK). Membranes were first blocked in 3% nonfat dry milk (for normal antibodies) or 3% bovine serum albumin (for phospho-specific antibodies) in 25 mM Tris, 150 mM NaCl, 0.05% Tween-20, pH 8.0, for 1 h at room temperature (RT), after which they were incubated at 4℃ overnight with primary antibodies. The primary antibodies and their dilutions were as follows: anti-Tyro3 (1:500, C-20), anti-Axl (1:500, C-20), anti-GAPDH (1:500, V-18; Santa Cruz Biotechnology, Santa Cruz, CA), anti-actin (1:10000, A2066; Sigma-Aldrich, Poole, UK), anti-phospho-STAT3 (1:500; Cell Signaling Technology, Leiden, The Netherlands), anti-phospho-Tyro3 (“anti-phospho-MER/SKY”; 1:1000, Phospho-Tyr749/681; Abbexa, Cambridge, UK), and anti-myelin basic protein (MBP, 1:500, MAB386; Millipore, Darmstadt, Germany). Postincubation washing of the membrane (25 mM Tris-HCl, 150 mM NaCl, 0.05% Tween-20, pH 8.0) was followed by 1 h incubation with a horseradish peroxidase-conjugated secondary antibody (1:5000; Promega, Madison, USA; Dako, Denmark) at RT. Chemiluminescence detection reagent (Luminata Forte Western HRP Substrate, Millipore) was used to generate the signal, and bands were visualized using a CCD-based digital gel imaging system (Bio-Rad ChemiDoc™ MP Imaging System, Hemel Hempstead, UK). The intensity of each band was quantified by densitometry using ImageJ software (Schneider et al., 2012), and query protein band intensities were normalized against those of GAPDH or actin protein in each sample.
Optic Nerve Culture
Mouse optic nerves were isolated and established in culture for several days as described earlier (Azim and Butt, 2011). Briefly, 2-month-old transgenic Sox10-GFP or wild-type mice were killed by cervical dislocation, and optic nerves were removed while still attached to the eyeball and immediately placed in ice-cold artificial cerebrospinal fluid. The tissue was placed onto semiporous membrane inserts (0.4 µm; Millipore) and covered with 2 ml culture medium containing 50% Opti-MEM™, 25% horse serum, 25% Hanks Balanced Salt Solution (Gibco Invitrogen, Paisley, UK), supplemented with 25 mM D-glucose (Sigma), and antibiotics (Penicillin G sodium 10,000 U/ml, streptomycin sulfate 1,000 µg/ml; Gibco Invitrogen) diluted to 1:500, in a six-well culture plate. For experiments, recombinant Gas6 protein (diluted in cell culture medium containing Dulbecco’s Modified Eagle Medium, supplemented with 10% fetal calf serum, 100 U/ml penicillin, 100 µg/ml streptomycin, and 2 mM L-glutamine; Lonza, Slough, UK) was added directly to the culture medium at 400 ng/ml final concentration. In some experiments, Gas6 or Axl inhibitors were added, including the Gas6 antagonists soluble Axl/Fc as ligand quencher (R&D Systems, Minneapolis, MN) and warfarin-Gas6 (produced in-house) (both previously used by us and not toxic; Stenhoff et al., 2004), and the Axl-specific small molecule inhibitor BGB324 (Stratech Scientific, Newmarket, UK), which was used at a concentration of 1 µM as previously reported by us as effective in culture (Vouri et al., 2015). The cultures were incubated for 3 days, or for 3 h in signaling activation experiments. Cell culture medium alone was used as mock treatment, the same as the medium containing recombinant Gas6, as described earlier (Vouri et al., 2015). The culture medium and treatments were replenished on the second day of incubation. After 3 days, optic nerves were detached from the eye and the tissue processed for either Western blotting or qPCR, and samples from transgenic Sox10-GFP mice were fixed (4% paraformaldehyde, 15% picric acid) for 1 h at RT, prior to whole mounting on slides in mounting medium (Vectashield; Vector Labs, Peterborough, UK) and confocal microscopic examination of Sox10+ cells. All experiments were repeated with cultures prepared from different animals at different times.
Organotypic Cerebellar Slice Culture
Sagittal sections of 300 µm thickness were made from brains of C56/BL6 mice at ages P8 to P12, using a tissue chopper (Campden Instruments, Loughborough, UK). The sections were placed onto semiporous membrane inserts (0.4 µm; Millipore) and covered with 1 ml culture medium composed of 50% Minimum Essential Medium with Glutamax-1, 18% Earle’s Balanced Salt Solution (EBSS), 5% EBSS + D-glucose, 1% penicillin-streptomycin, and 10% horse serum (Gibco Invitrogen; De Simoni and Yu, 2006). The slices were maintained on 0.4 µm semiporous membrane inserts in a six-well plate at 37℃ in a humidified atmosphere with 5% CO2, for a total of 7 days. After 3 days in culture, the slices were either treated with Gas6 for 3 h to investigate signaling mechanisms or in separate experiments to induce demyelination; on Day 3 of incubation, the medium was replaced with fresh media containing 1 mg/ml lysophosphatidylcholine (LPC, lysolecithin; Sigma) or methanol as vehicle. To investigate the effect of Gas6 on demyelination, 400 ng/ml of recombinant Gas6 in conditioned culture medium and mock medium as control were added to the same wells as the LPC treatments. All treatments with LPC were incubated further for 24 h, following which the medium was removed and the slices were incubated for another 3 days in fresh medium in the presence of freshly added Gas6 or mock treatments. Symmetrical cerebellar sections were used for direct comparisons between mock and Gas6 treatments. MBP immunofluorescent staining was then performed on the sections and analyzed by confocal microscopy. For quantification of myelination, the numbers of complete myelinated axons of length greater than 50 µm were counted for each treatment using ImageJ software. The values were converted to percentages and normalized against the values for vehicle-treated sections.
Immunofluorescence Confocal Microscopy
Cell counts of Sox10-GFP-positive oligodendrocytes or OPCs in optic nerves were performed on images taken with a laser scanning confocal microscope (Zeiss LSM 710, Cambridge, UK). z-stack images were taken from each optic nerve with dimensions of 212.34 × 212.34 µm in the x- and y-plane and 70 µm in the z-plane at 10 µm intervals. The values of cell counts represent the mean (±SEM) percentage (%) of cells per field of view.
Cerebellar sections were fixed in 4% paraformaldehyde overnight, then blocked for 1 h in 20% normal horse serum diluted in TBS-TX (41.9 mM, Tris HCl; 8 mM Tris base; 154 mM NaCl; and 4.6 mM Triton-X). The sections were then incubated overnight with primary antibodies diluted in TBS-TX at the following dilutions: anti-MBP (1:300, MAB386; Millipore), anti-APC (5ug/ml, Ab-7; Millipore), anti-NeuN (1:250, MAB377; Millipore), and anti-glial fibrillary acidic protein (GFAP, 1:400, AB5541; Millipore). The next day, after washing with TBS-TX, the slices were incubated with the secondary antibodies for 2 h.
Statistical Analysis
All statistical analyses were performed using the software IBM SPSS Statistics 20 or Prism 6 (GraphPad Inc, La Jolla, CA). Animals were randomly assigned to treatment groups. All results are expressed as mean ± SEM, with each experiment performed a minimum 3 times unless otherwise stated in the figure legend, using multiple replicates per treatments. Differences between multiple treatments in optic nerve cultures and organotypic cerebellar slice culture were compared using one-way analysis of variance followed by Bonferroni post hoc test, and comparison between two groups was carried out using unpaired Student t test. A p value of less than .05 was considered to be statistically significant.
Results
Expression of Gas6 and TAM Receptors in the Mouse CNS and in Human Glial Cells
TAM receptors are expressed in a wide variety of tissues and organs throughout the body and, while it is indicated that Tyro3 might be the main receptor in the CNS (Pierce and Keating, 2014), the expression profile of TAMs and Gas6 in the CNS remains unclear. To obtain a comprehensive picture of the whole TAM RTK subfamily and Gas6 in the mouse CNS, qPCR was performed on mRNA from different regions of the CNS and at different ages of postnatal development. TAM and Gas6 mRNA expression were detected in cortex and cerebellum of P7, P14, and adult mice and in adult optic nerve (Figure 1(a)). Furthermore, mRNA expression of all three TAMs increased significantly with age in the cortex but not in the cerebellum. This qPCR expression data are valid only for comparisons between samples per gene and not between samples for different genes. This is because the primer or probe sets for different genes will not have the same amplification dynamics and so the arbitrary relative expression values are valid only for a sample set for each gene.
Expression of TAM receptors and Gas6 in different mouse brain regions and at different postnatal ages. (a) qPCR expression analysis of mRNA for Gas6 and TAM receptors. Values represent mean ± SEM of relative gene expression; n = 4 for all samples except for optic nerve (n = 3) and P14 cerebellum (n = 2). All samples were normalized against Cdc40 as internal control; *p < .05, **p < .01, ***p < .001, ****p < .0001 for comparisons as indicated. Columns containing letter 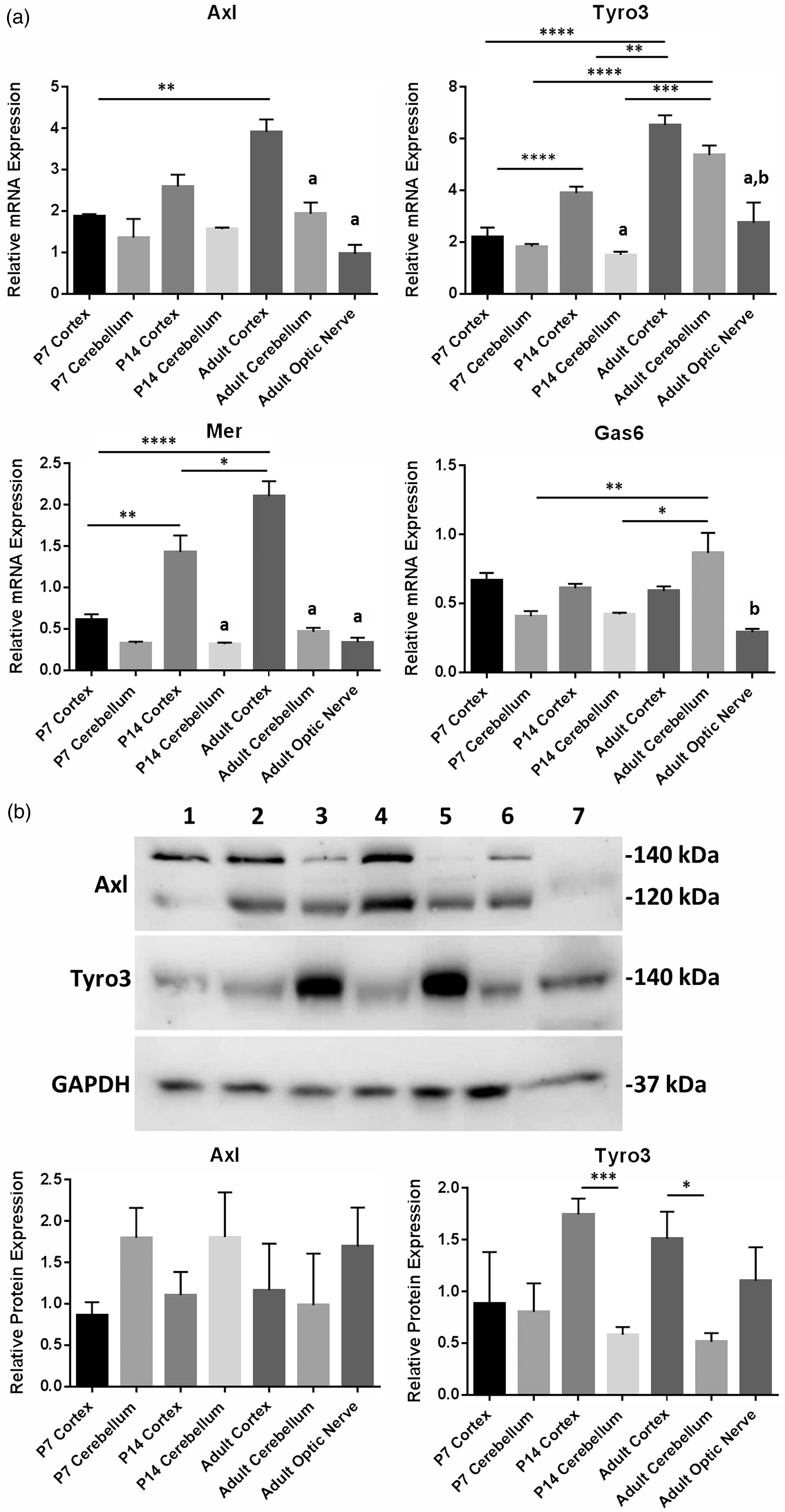
In addition, Western blots showed clear expression of both Axl and Tyro3 proteins in all mouse brain regions and at all ages (Figure 1(b)), as well as in the adult optic nerve. However, Tyro3 protein expression was significantly higher in cortex versus cerebellum in P14 and adult, while the opposite was true of Axl expression (Figure 1(b)). The two bands seen in Axl Western blots are ∼120 kDa and ∼140 kDa forms of Axl due to different levels of glycosylation, reflecting different levels of maturation of the RTK.
The mRNA expression of Gas6, Tyro3, and Axl was also analyzed by qPCR in cultured HOPCs and HA-c, using the 2−ΔCt method where ΔCt is Cttarget gene − Ctinternal control and 2−ΔCt is the relative gene expression (Schmittgen and Livak, 2008). Tyro3 was expressed clearly in HOPC, whereas Axl was absent in this type of glial cells. In stark contrast, Gas6 was highly expressed in HA-c, while it was absent in HOPC (Figure 2).
Expression of Tyro3, Axl, and Gas6 mRNA in human glial cell cultures. Extracted mRNA from human astrocytes and human oligodendrocyte precursors were analyzed by qPCR using specific primers for human Tyro3, Axl, and Gas6 genes. Results were analyzed based on 2−ΔCt method.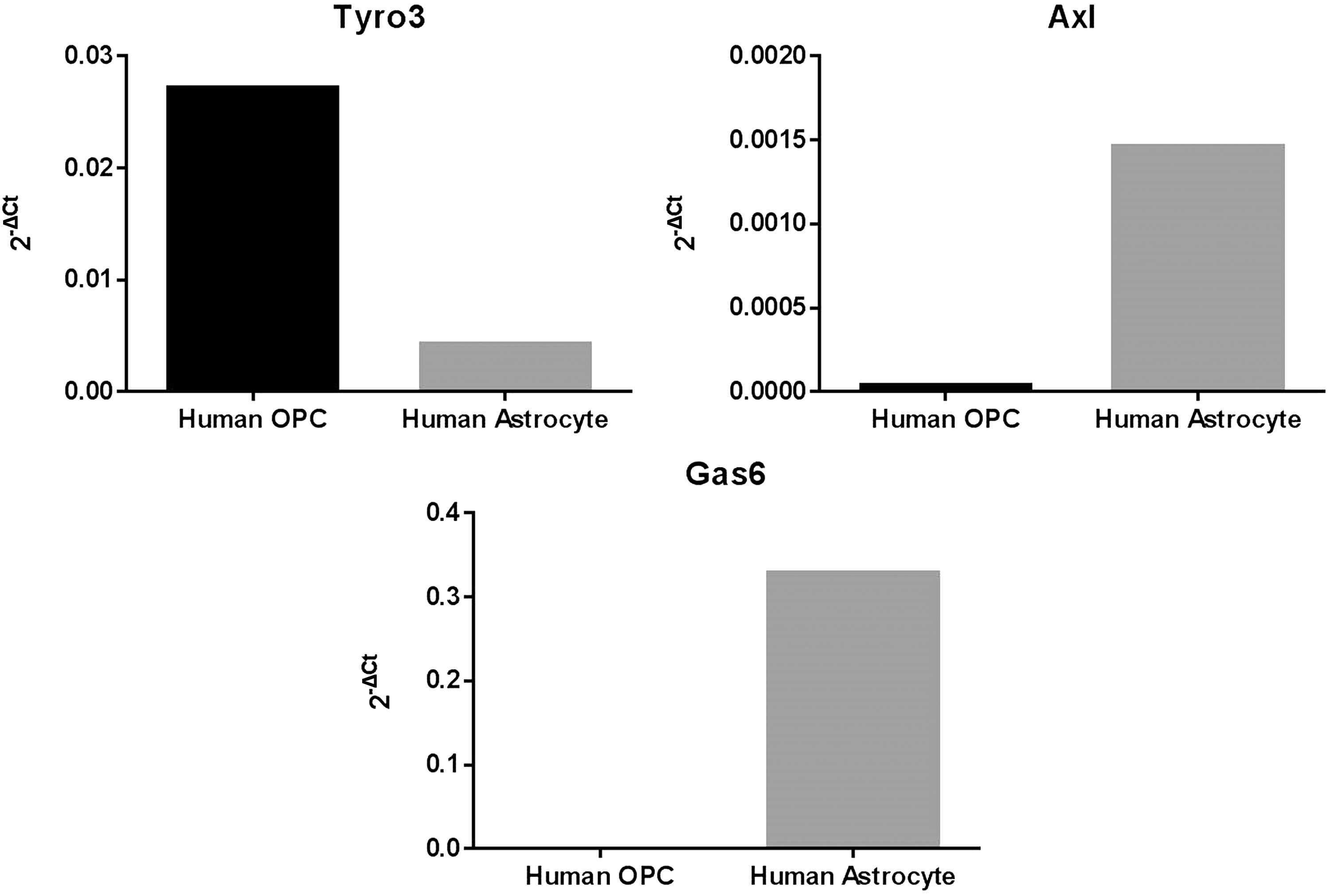
Gas6 Promotes Oligodendrogenesis in the Mouse Optic Nerve
To assess the effect of Gas6 on oligodendrogenesis, we used the optic nerve from adult Sox10-EGFP reporter mice, in which the reporter is expressed by myelinating oligodendrocytes, which make up the bulk of glia in the optic nerve, together with adult OPCs, which make up 3% to 5% of cells in the adult optic nerve (Butt et al., 2004). Optic nerves were isolated with retina intact and maintained in organotypic culture for 3 days, in control medium or medium containing Gas6. Samples also included Gas6 together with warfarin, which blocks the vitamin K-dependent γ-carboxylation of the protein posttranslation (Korshunov, 2012) and therefore produces Gas6 that is uncarboxylated and inactive, or the recombinant extracellular domain of Axl (Axl/Fc), which can act as a ligand antagonist by quenching the ligand before it binds to the receptor. After 3 days, optic nerves were fixed and whole mounted for confocal analysis and cell counts. The results show that Gas6 significantly increased the number of Sox10-GFP positive cells, and this effect was blocked by both Axl/Fc and warfarin (Figure 3; p < .001). These results were obtained from a number of experiments performed on different days, using separate cultures from different animals.
Effect of Gas6 on number of OPCs or oligodendrocytes in cultured mouse optic nerves. (a) Confocal images of optic nerves treated with mock medium (−Gas6) and Gas6, showing Sox10+ cells with green fluorescence. (b) Cell counts from images taken from optic nerves treated with Gas6 in the absence or presence of two Gas6 antagonists: soluble Axl receptor (Axl/Fc) and Warfarin-Gas6. Values represent mean ± SEM (control and Gas6, n = 8; Axl/Fc + Gas6, n = 2; Warfarin-Gas6, n = 4; n represents number of separate experiments); ***p < .001 versus control (SEM values are as follows: Mock = 5.0648, Gas6 = 3.5669, Axl/Fc + Gas6 = 4.0488, Warfarin-Gas6 = 3.4071, analysis of variance with Bonferroni correction).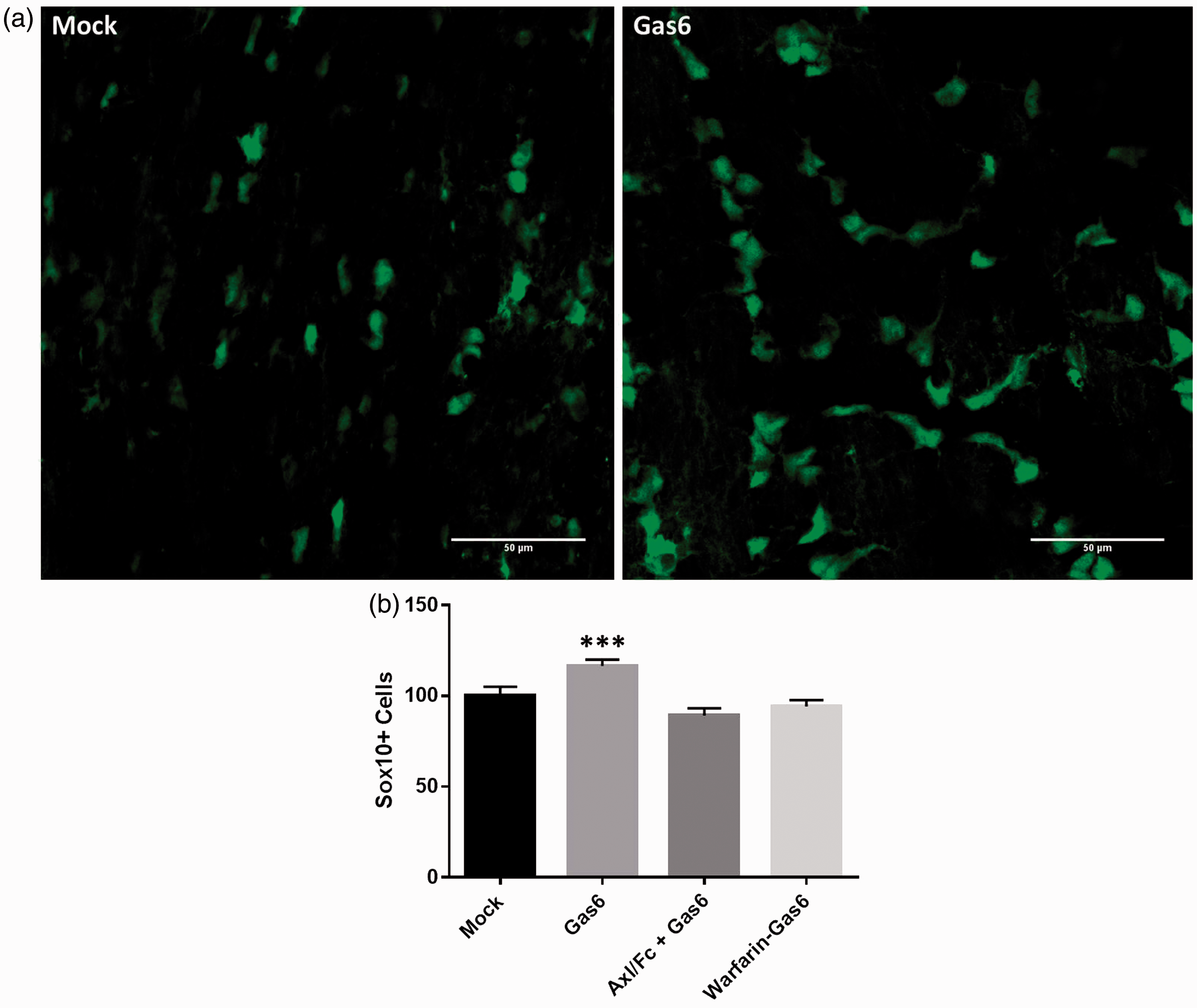
Gas6 Enhances Myelination Following Lycolecithin-Induced Demyelination in Organotypic Cerebellar Slice Cultures
Having observed that Gas6 promotes oligodendrogenesis and myelination in the adult optic nerve, we next examined whether Gas6 could promotes remyelination after toxic injury, using lysolecithin (LPC) to cause destruction to the myelin in a cerebellar slice culture model, which also features spontaneous repair (Birgbauer et al., 2004; Zhang et al., 2011). Cerebellar slices were obtained from mice aged between P8 and P12 and cultured for 3 days to allow myelin formation to occur prior to adding LPC for 24 h, in normal medium or medium containing Gas6, followed by a further 3 days culture following LPC removal. Myelin loss was observed in slices treated with LPC, whereas demyelination was significantly decreased by approximately twofold in slice cultures coincubated with Gas6 (Figure 4; p < .05).
Gas6 significantly attenuates demyelination in a toxin-induced demyelination model. (a) Cerebellar slice cultures were treated with vehicle (methanol), LPC + Mock medium (−Gas6), and LPC + Gas6 and stained with MBP antibody. For each treatment within an experiment, one cerebellar slice was used, and one field of view per slice was analyzed. Images were taken from the top end of the lobules were the axons spread out. Scale bar = 50 µm. (b) Quantification of myelination through the number of MBP + axons with lengths greater than 50 µm. Values represent mean ± SEM (n = 6 experiments); *p < .05 for comparison indicated, analysis of variance with Bonferroni correction.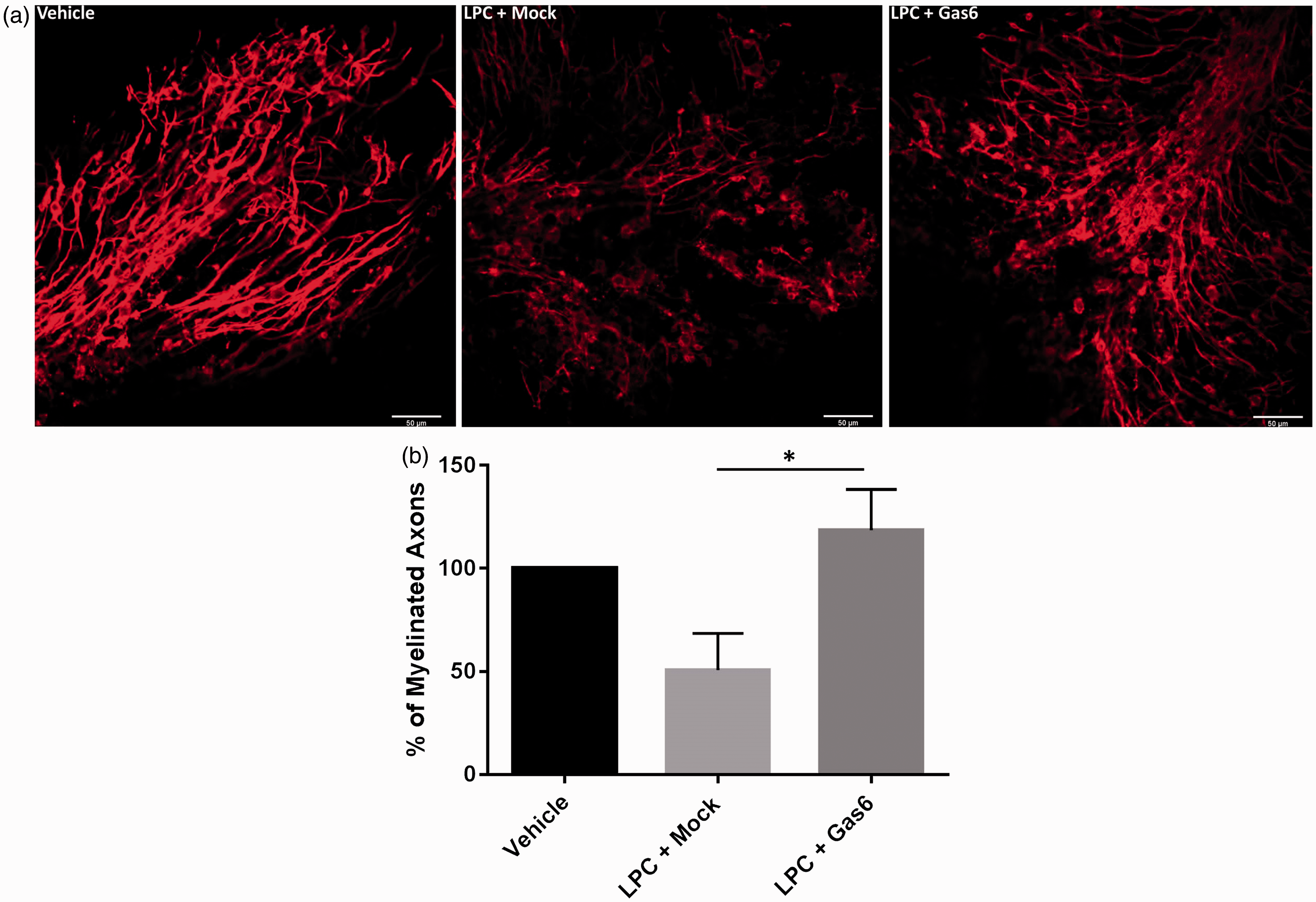
Gas6 Activates STAT3 Signaling and Myelination in Optic Nerves
The JAK/STAT signaling pathway is a primary target of Gas6 signaling (Yanagita et al., 2001) and has been shown to promote oligodendrocyte regeneration and remyelination (Hesp et al., 2015), as well as being a promising therapeutic target in multiple animal models of MS (Liu et al., 2014). Therefore, we used Western blot to determine STAT3 activation through detecting levels of phosphorylated STAT3 (p-STAT3; Figure 5(a)) as well as levels of MBP as a measure of differentiation or myelination (Figure 5(b)). The protein extracts from cultured adult optic nerves were analyzed, and Western blot band intensities were quantified by densitometric analysis, normalizing against GAPDH protein (Figure 5). The results demonstrate that compared with controls, Gas6 stimulation (400 ng/ml) resulted in a significant increases in p-STAT3 protein (Figure 5(a); p < .01) as well as MBP, which was increased approximately twofold (Figure 5(b); p < .05). The results demonstrate that Gas6 activates STAT3 signaling and stimulates the molecular process of differentiation or myelination in the adult optic nerve.
Effect of Gas6 on activation of intracellular STAT3 and myelination through MBP expression in cultured optic nerves. Representative Western blots are shown of proteins (a) pSTAT3 and (b) MBP in optic nerve lysates. The graphs accompanying each blot show the densitometric quantification of protein levels relative to GAPDH protein in those samples. Values represent mean ± SEM (n = 6 blots); *p < .05, **p < .01 versus control, Student t test. Gas6 significantly increased the phosphorylation of STAT3 as well as MBP in optic nerve cultures.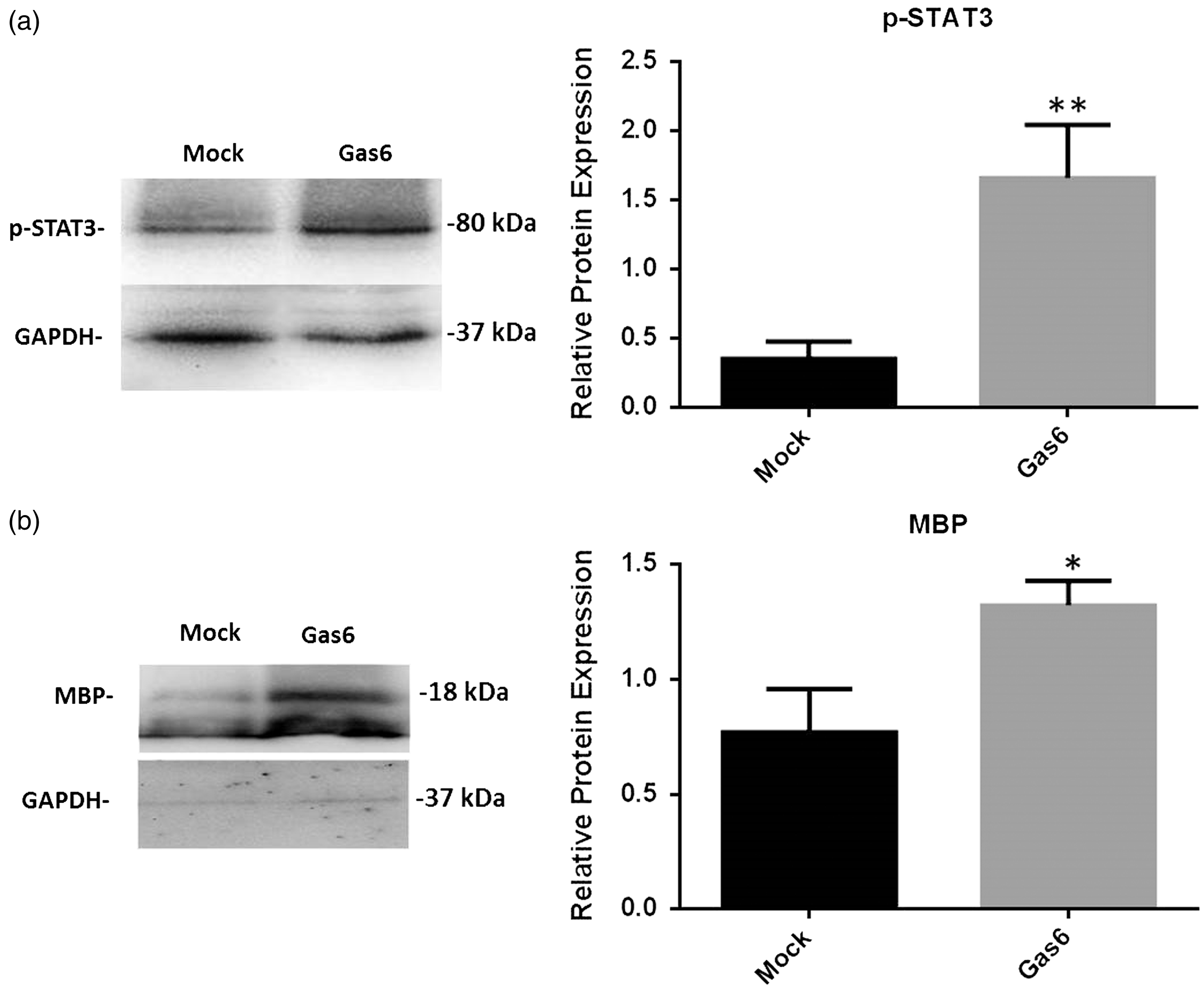
Gas6 Stimulates STAT3 Phosphorylation in Mature Oligodendrocytes and Astrocytes
Having observed by Western blot that Gas6 stimulates STAT3 phosphorylation in cultured optic nerves, we then investigated the specific cell types in which this STAT3 activation occurred, through confocal microscopic analysis of cerebellar slice cultures. After 3 h culture incubation in the presence of Gas6, fixed cerebellar sections were double stained with p-STAT3 antibody together with antibodies for mature oligodendrocyte, astrocyte, and neuronal markers. The double immunostaining of Gas6-treated tissues showed p-STAT3 to be present predominantly in mature oligodendrocytes and also in some astrocytes but not in neurons (Figure 6(a) and Supplementary Figure S3). Our data indicate that treatment of cerebellar slices with Gas6 for only 3 h is enough for activating STAT3, and thus the effect of Gas6 on increased level of STAT3 phosphorylation is not due to increased level of STAT3 protein, which is a longer term process.
Gas6 activates STAT3 in mature oligodendrocytes and astrocytes and activates Tyro3 receptor after 3 h stimulation in culture. (a) Cerebellar slices were treated with mock or Gas6 medium for 3 h, and fixed sections were double stained for p-STAT3 and APC (mature oligodendrocyte marker), p-STAT3 and GFAP (astrocyte marker), p-STAT3 and NeuN (neuronal marker; scale bar = 20 µm). The stainings reveal that p-STAT3 is present in oligodendrocytes and astrocytes but not in neurons. The arrows point to cells that are both GFAP+ and p-STAT3+. (b) Representative Western blot of p-Tyro3 protein in optic nerve lysate. The graph shows the densitometric quantification of protein level relative to actin in the same sample. Values represent mean ± SEM (n = 5 blots); **p < .01 versus control, Student t test. Gas6 significantly increased the activation of Tyro3.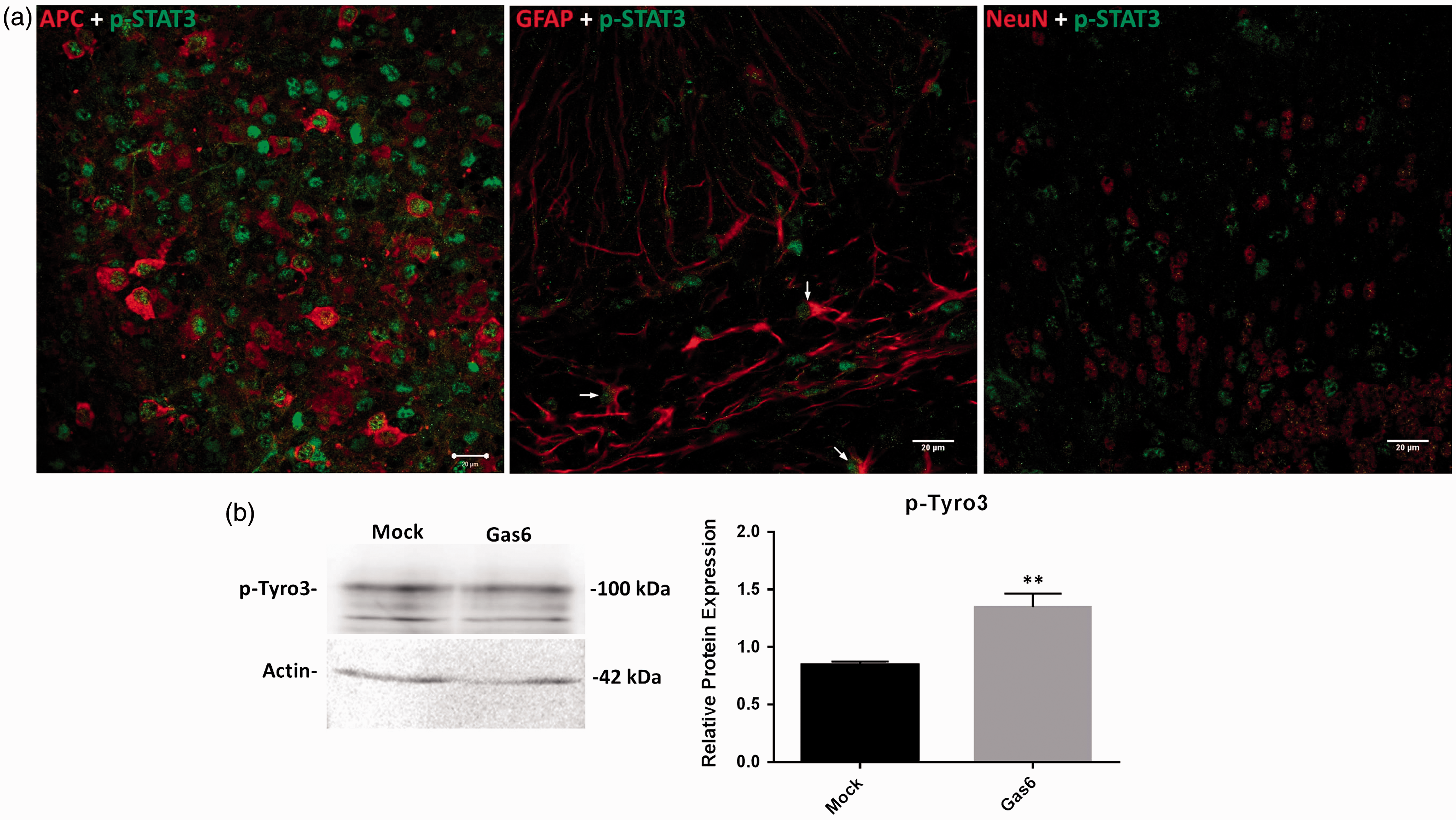
Having observed that oligodendrocytes express Tyro3, we attempted to determine whether Tyro3 was the TAM receptor that mediated the observed effects of Gas6. For this, we performed Western blots to investigate levels of phosphorylated (activated) Tyro3 (p-Tyro3) in cultured mouse optic nerves treated with Gas6 for 3 h. The Western blotting shows that compared with control, Gas6 stimulated a significant increase in p-Tyro3 protein levels in optic nerves, showing that Gas6 directly activates Tyro3 in the white matter (Figure 6(b)). The apparent discrepancy between the molecular weight of total Tyro3 (∼140 kDa) and p-Tyro3 (∼100 kDa) is due to the respective antibodies binding to different forms of Tyro3. The anti-Tyro3 antibody is highly selective for the fully mature Tyro3, whereas the anti-p-Tyro3 recognizes the smaller band species of Tyro3. We also performed these experiments in the presence of a specific small molecule inhibitor of Axl and BGB324 (Barcena et al., 2015; Vouri et al., 2015) and observed that blocking Axl did not affect the Gas6 effect on myelination, further indicating that the myelination effect occurs specifically via Tyro3 receptor (Supplementary Figure S1).
Gas6 Alters MS-Related Gene Expression in Cultured Optic Nerves in a Promyelinating Direction
Summary of Genes in Cultured Mouse Optic Nerves Altered by Gas6 Stimulation by ≥2-Fold.
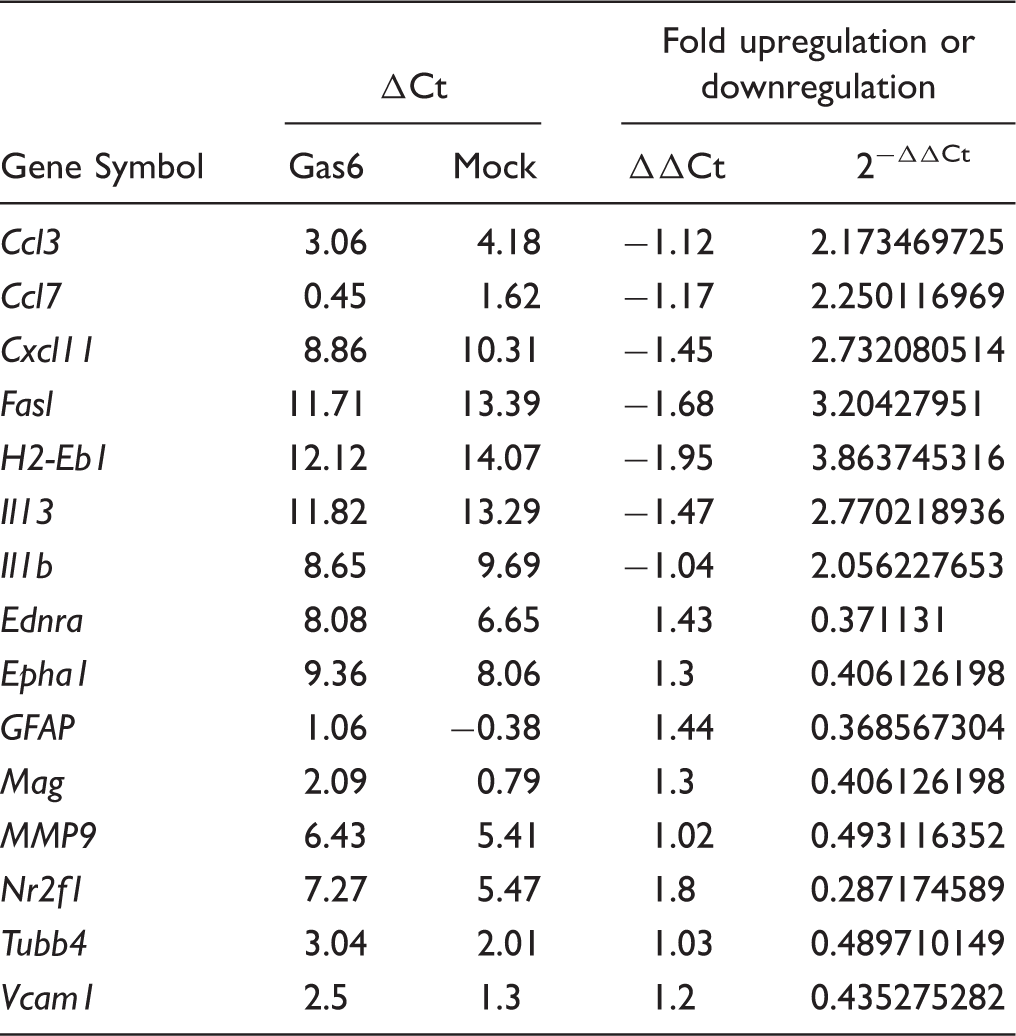
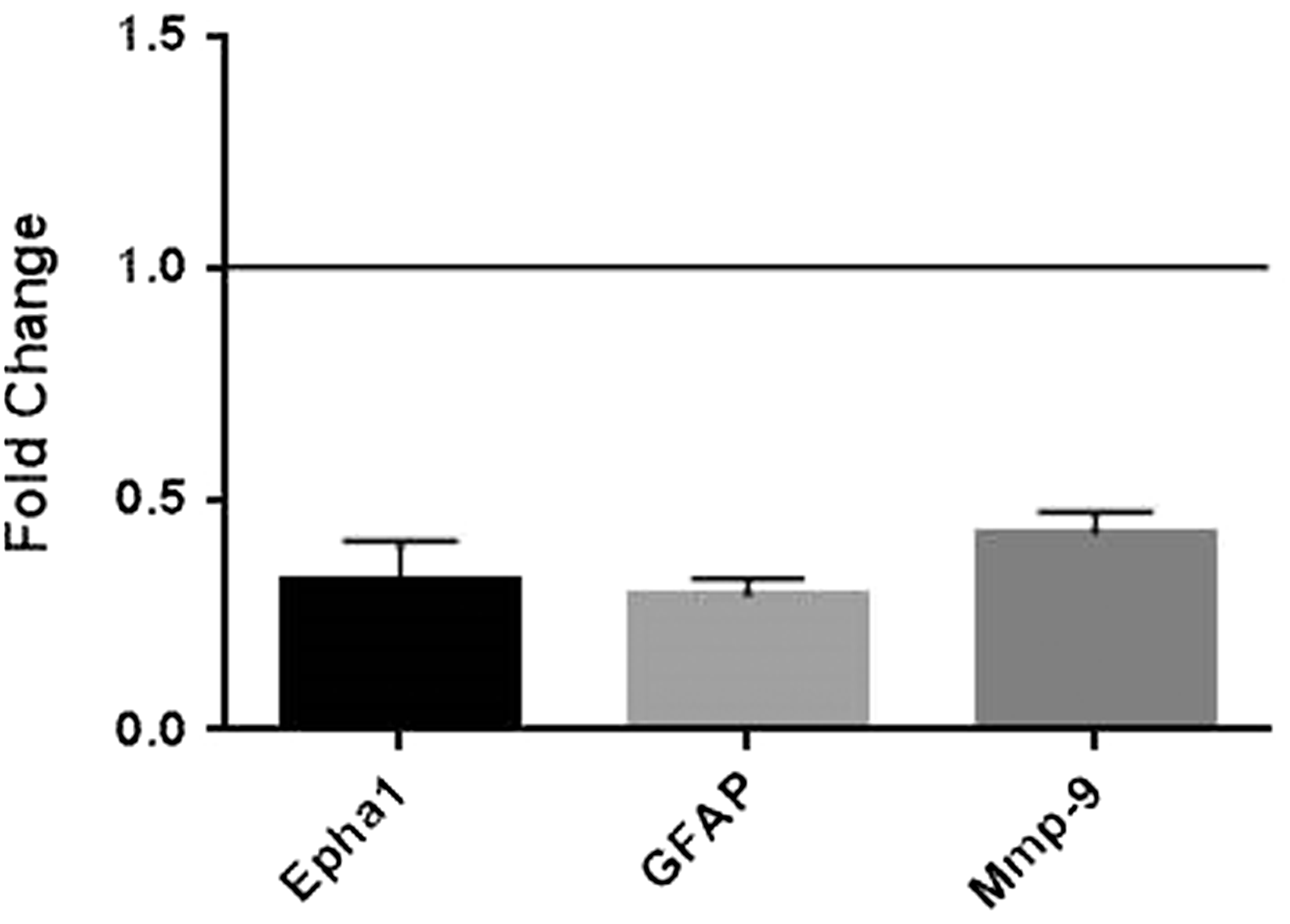
Effect of Gas6 on expression of selected MS-related genes in cultured mouse optic nerves. qRT-PCR was performed on extracts from mock and Gas6-treated optic nerve cultures, using specific primer or probe sets, and normalizing expression against the GAPDH gene. Treatment of optic nerves with Gas6 resulted in downregulation of Epha1, GFAP, and MMP9 genes. Values represent mean ± SEM (n = 4 for qPCR experiments for all genes except for Epha1 (n = 2)).
Discussion
The three homologous TAM receptors and their common ligand Gas6 are expressed widely throughout the body (Hafizi and Dahlbäck, 2006a, 2006b). However, their expression profiles in the CNS, and their functional roles therein, remain to be comprehensively characterized. In the present study, we show that Gas6 and all three TAM receptors are expressed in the mouse brain at mRNA level, as well as Tyro3 and Axl protein, and that their expression increases during myelination and is highest in the adult cortex. In addition, we show that Gas6 promotes the generation of oligodendrocytes and myelin in the white matter of the adult mouse optic nerve and also attenuates myelin loss in the lysolecithin model of demyelination in cerebellar slice cultures. Furthermore, analysis of the molecular mechanisms of action of Gas6 demonstrates that it activates the Tyro3 receptor, intracellular STAT3 signaling, and the programming of a host of MS-related genes in a prorepair and myelinating direction. These results therefore indicate that Gas6 or TAM signaling is a potential target for stimulating repair mechanisms that are relevant to demyelination and MS.
All three TAM receptors were observed to be expressed throughout the brain tissue, although Tyro3 was expressed in more regions and at higher levels compared with Axl and Mer. This supports previous indications that Tyro3 is the primary TAM receptor mediating the actions of Gas6 in the CNS (Prieto et al., 2000). In addition, our data show that Tyro3 expression increases during development in the CNS. These observations are in line with previous time-course expression data, showing Tyro3 protein to be very low at embryonic stages while its expression begins to increase shortly after birth, dramatically increasing between postnatal days P3 and P15, reaching its peak level at P24, and remaining at that level thereafter in the adult (Prieto et al., 2000). Although most myelination occurs early in life, myelination carries on at least into late adolescence and, in some regions of the CNS, may increase throughout much of adult life (Emery, 2010). Therefore, the concomitant temporal profiles of myelination and Tyro3 expression in the developing mouse CNS suggest that Tyro3 plays a role in oligodendrocyte development or maturation and myelination. In the Axl blot, a double band was observed at both 120 kDa and 140 kDa, which are the well-known alternately glycosylated versions of Axl (the 140 kDa protein being most mature; O’Bryan et al., 1995). Our results showed clear expression of Axl protein in all brain regions and ages analyzed, with no significant differences in levels of expression among them.
We also conducted qPCR analysis for Tyro3, Axl, and Gas6 genes in separate pure cultures of human OPCs and astrocytes. Tyro3 was highly expressed in oligodendrocytes, in which Axl expression was absent. This key finding therefore suggests that Gas6 acts directly on OPCs or oligodendrocytes via the Tyro3 receptor to affect their fate and function. In contrast, Tyro3 expression was lower in astrocytes compared with oligodendrocytes but instead astrocytes showed a high level of Gas6 expression. This therefore suggests an indirect and supportive role for astrocytes in regulating the fate and function of oligodendrocytes, by providing the ligand through paracrine signaling. However, this interpretation of relative TAM expression comes with a certain degree of caution as the human isolated glial cell cultures were not only from different brain regions but were also derived from different brains and at different ages. Nevertheless, within a specific cell type, our data clearly show that Tyro3 is present in oligodendrocytes, while Axl is virtually absent in these cells. Furthermore, according to Figure 1(a), Mer mRNA was also observed in the CNS, although we did not characterize its expression pattern in glial cells, but according to previous reports, it is mainly expressed in microglia within the CNS and is involved in phagocytic activity of these cells (Fourgeaud et al., 2016). Axl has also been reported to be present in microglia, and no Tyro3 was observed in these cells (Fourgeaud et al., 2016). Also, it was shown that, in addition to Mer, Axl is also essential for the phagocytic activity and noninflammatory properties of microglia cells, including removal of cell debris (Fourgeaud et al., 2016).
Moreover, as we have only analyzed glial cells in this study, it is important to note that expression of Gas6 expression has been reported in different neuronal cells including Purkinje neurons, motor neurons of spinal cord, as well as large dorsal root ganglia neurons (Li et al., 1996; Prieto et al., 1999). Therefore, it is also possible that neurons may play a similar supportive role in regulating oligodendrocyte development and function, as astrocytes do.
As well as in the brain, qPCR analysis of adult mouse optic nerve demonstrated that Gas6 or TAM signaling components are also present in this tissue, with relative levels of expression indicated in the following order Tyro3 > Axl > Mer. Also, Gas6 stimulation of optic nerve from Sox10-EGFP mice further confirmed the presence and activity of TAM receptors in CNS white matter, through which they promote the generation of oligodendrocyte lineage cells. The Sox10-EGFP reporter identifies both oligodendrocytes and OPCs, since Sox10 marks all stages of oligodendrocyte development (Stolt et al., 2006); but in the adult, OPCs are continuously generating new oligodendrocytes and are the likely targets of Gas6 in our study (Gonzalez-Pereza and Alvarez-Buyllab, 2011). In addition to oligodendrogenesis, Gas6 stimulated increases the levels of MBP protein in the adult optic nerve, which indicates that Gas6 also promotes myelination in mature oligodendrocytes (Mi et al., 2011). Therefore, these observations together demonstrate that Gas6 boosts both the number of OPCs as well as mature myelinating oligodendrocytes. In this regard, it is worthwhile considering protein S, a protein homologous to Gas6 that is also a TAM receptor ligand. However, its expression appears restricted in the CNS, with low levels of protein S mRNA observed in locus coeruleus and high levels of mRNA in the choroid plexus (Prieto et al., 1999). Furthermore, the expression of protein S appears to be primarily neuronal, having been detected in the cortex, hippocampal pyramidal neurons, and granule neurons of the dentate gyrus of the rabbit brain (He et al., 1995; Prieto et al., 2007).
Gas6 stimulation also led to a decreased demyelination in a cerebellar slice culture model of demyelination by lysolecithin (Birgbauer et al., 2004), which is one of the most commonly used toxins to induce focal demyelination in different parts of the brain (van der Star et al., 2012). Our results show that treatment with Gas6 significantly attenuated the extent of demyelination that continues for days after exposure to lysolecithin (Birgbauer et al., 2004). This is consistent with studies in the cuprizone model of demyelination, which showed systemic administration of Gas6 to enhance remyelination (Tsiperson et al., 2010), while Gas6 ablation decreased the number of myelinating oligodendrocytes and delayed recovery (Binder et al., 2011). Together, these studies demonstrate that Gas6 or TAM signaling plays a major role in oligodendrocyte generation and myelination in the adult CNS. However, our experiment was focused on the protective effect of Gas6 during demyelinating injury, whereas it is also important to investigate the acceleratory effect of Gas6 on remyelination after damage. Thus far, we did not observe an enhanced remyelination when Gas6 was added 3 days after LPC damage (Supplementary Figure S2). However, further detailed time-course studies are warranted to explore the time window during which Gas6 can be effective as an enhancer of remyelination post damage. Furthermore, although Gas6 increases MBP and blocks demyelination in culture, it is also worthwhile to measure the axon diameter or g-ratio by electron microscopy to determine specifically the location of myelination as being around axons, and hence contributing to increased axon thickness. Also, although this study was focused on the nonmicroglial response to Gas6, it is nevertheless possible, considering the phagocytosis-mediating role of Mer (Chen et al., 1997), that Gas6 could also activate Mer in microglia in our culture model; this requires further investigation.
In addition to the ligand Gas6, the role of Axl receptor has also investigated by other research groups in demyelination models. In a study using the cuprizone model of demyelination, Axl knockout mice showed no significant differences to wild-type mice in axon diameter, myelin thickness, and g-ratio of myelin-containing axons (Hoehn et al., 2008). This therefore suggests that Axl is not the main receptor involved in remyelination or that loss of signaling through all three TAM receptors is more harmful than loss of signaling through a single TAM receptor. This is consistent with our observations, which point toward the prominence of Tyro3 in oligodendrocyte biology and myelination, reinforced by its unique mRNA expression in oligodendrocytes.
As our expression analysis indicated Tyro3 as the most prominent TAM receptor in the CNS, at least as concerns oligodendrocytes, we therefore hypothesized that the Gas6 effects on OPC proliferation, oligodendrocyte maturation, and myelination are likely occurring via a direct activation of Tyro3 in these cell populations. We therefore utilized our optic nerve culture model to also investigate the relatively acute activation of Tyro3 in response to Gas6 stimulation. Thus, Gas6 stimulated a higher activation state of Tyro3 after 3 h in the optic nerve, as observed by increased p-Tyro3 protein levels. This observation is supported by a recent study in the peripheral nervous system that showed Tyro3 to be an important modulator of myelination by Schwann cells (Miyamoto et al., 2015).
We also observed that the transcriptional regulator protein STAT3 was mobilized by Gas6 stimulation using both of our in vitro culture models. Gas6 increased levels of phosphorylated STAT3 protein in the optic nerve, and using the slices for microscopy, we were able to localize the pSTAT3 to oligodendrocytes as well as to a lesser extent astrocytes. Gas6 has previously been reported to act via STAT3 to promote proliferation of mesangial cells (Yanagita et al., 2001). This is consistent with our current observations of Gas6-induced activation of STAT3 and proliferation of OPCs or oligodendrocyte in the optic nerve. STAT3 controls a large number of genes that are involved in apoptosis, cell migration, cell cycle regulation, and angiogenesis as well as modulation of immune suppressive factors (Ferguson et al., 2015). Once STAT3 becomes activated via phosphorylation, it translocates to the nucleus and binds to specific DNA sequences to activate the expression of target genes that regulate proliferation and differentiation (Qi and Yang, 2014). There is also evidence that shows STAT3 activation is potentially important during neuroinflammation by protecting myelin development (Nobuta et al., 2012). Although neurons also express Tyro3 (Schulz et al., 1995), we found Gas6 to induce STAT3 activation predominantly in OPCs or oligodendrocytes, as would be expected for differentiating or proliferating cells (Fukada et al., 1996). Therefore, STAT3 appears to be a key intracellular mediator of Gas6 signaling in oligodendrocytes, most likely via Tyro3, to affect their development and the subsequent myelination of axons.
From a mini-array screen, we also observed Gas6 to induce significant changes in expression of multiple genes relevant to glial biology and MS in the cultured optic nerve, including downregulation of MMP9, Epha1, and GFAP genes. GFAP is an astrocyte-specific protein and is used as a marker of reactive astrogliosis (Beckerman et al., 2015), suggesting that Gas6 may modulate astroglial responses in the optic nerve. MMP9 is a proteolytic enzyme that degrades type IV collagen, laminin, and fibronectin, all of which are major components of the basement membrane of the BBB (Zheng et al., 2014). During a relapse course of MS, active B cells increase the expression and secretion of MMP9 protein, ultimately resulting in digestion of myelin as well as neurological disability (Aung et al., 2015). Furthermore, it has been shown that MMP9 can be secreted by OPCs that have proliferated at sites of demyelination in the CNS, which results in opening of the BBB and the subsequent secondary cascades of cerebrovascular injury and demyelination (Seo et al., 2013). Moreover, disruption of the BBB has been considered as one of the initial steps in the development of disease in MS patients (Alvarez et al., 2011). Also, the role of MMP9, at least produced by active B lymphocytes, in the disruption of the BBB and degradation of MBP protein is well recognized (Aung et al., 2015). Thus, our observation of Gas6 causing a twofold downregulation of the MMP9 gene suggests that Gas6 may help maintain BBB integrity and hence inhibit immune cell infiltration into the CNS during the course of MS; this is a focus of investigation in subsequent studies.
In conclusion, this study shows that components of Gas6 or TAM signaling are present throughout the adult CNS, and that, specifically, Tyro3 is present on OPCs or oligodendrocytes and mediates their development and function in response to the ligand Gas6, while astrocytes are the main source of the Gas6. Gas6 promotes the development and maturation of oligodendrocytes from OPCs and myelination in the adult CNS, as well as stimulating remyelination after toxic injury by lysolecithin. In addition, we show that the effects of Gas6 involve regulation of a set of genes that, through their coordinated upregulation or suppression, push the glial cell developmental pathway in a promyelinating direction. These results support a prominent role for Gas6 in promoting CNS repair after demyelination, and thus an attractive consideration for novel therapeutic approaches for MS.
Summary
We detected regional and age-dependent expression of TAM family receptors in the mouse CNS. Their common ligand Gas6 boosted mature oligodendrocyte numbers, stimulated myelination, and dampened toxin-induced demyelination. Gas6 acted via Tyro3 receptor to activate intracellular STAT3 signaling in oligodendrocytes.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SG was supported by a PhD studentship from the Multiple Sclerosis Society UK (grant number 953/11).
