Abstract
Background:
Thermal ablation (TA) of papillary thyroid carcinoma (PTC) has been reported to have promising results. However, performing microwave ablation (MWA) on PTCs located in the isthmus remains controversial.
Objectives:
This study aimed to compare the feasibility, effectiveness, and safety of MWA for treating PTC in the isthmus and lateral lobes.
Design:
Prospective cohort study.
Methods:
In this study, between December 2019 and December 2020, patients with clinical T1N0M0 PTC who planned to receive MWA were enrolled from 12 hospitals. All tumors were evaluated by preoperative ultrasound and divided into isthmus-located tumors and lobe-located tumors. The patients were followed until 1 February 2025. The primary endpoints included technical success and disease-free survival. The secondary endpoints included treatment parameters, complications, and tumor shrinkage. Variables were compared between the two groups, and subgroup analysis was performed.
Results:
After exclusion, 652 patients (mean age, 43 years ± 11 (SD), 481 females) were included: 109 with PTC in the isthmus and 543 with PTC in the lateral lobes. One isthmic tumor terminated MWA, 108 versus 543 (mean tumor volume of 0.1 ml ± 0.1 vs 0.2 ml ± 0.2, p = 0.14), was analyzed, with a mean follow-up period of 61 months ± 5 (range, 54–67 months). Comparable technical success rates were achieved (99% (108/109) versus 100% (543/543), p = 0.17) for isthmic tumors and lobe tumors, with 1 versus 24 complications (0.9% (1/108) vs 4.4% (24/543), p = 0.08). Although fewer isthmic tumors achieved the targeted safety margin (2 mm) than did lobe tumors (50.0% (54/108) vs 61.0% (331/543), p = 0.03), no differences in disease-free survival (97.2% (105/108) vs 97.4% (529/543), p = 0.91) or tumor shrinkage (96 ± 16% (SD) vs 95 ± 17% (SD), p = 0.70) were detected. In the tumor stage, capsular invasion, safety margin, sex, age, and thyroid disease subgroups, the outcomes remained comparable.
Conclusion:
MWA was feasible for T1N0M0 PTC in the isthmus and showed comparable efficacy in comparison with tumors in the lobes.
Trial registration:
NCT04197960.
Introduction
Papillary thyroid carcinomas (PTCs) tend to have an indolent nature, most of which are low risk and have a favorable prognosis. 1 The incidence rate of PTC arising in the thyroid isthmus is 1–16.2%.2,3 Studies have reported promising results for thermal ablation (TA) in the treatment of PTC,4–6 which can provide a relatively minimally invasive treatment option compared with surgery. 7 However, performing MWA on PTCs located in the isthmus remains controversial.
Isthmic PTC is defined as thyroid cancer in which the tumor center is located between two lines perpendicular to the surface of the skin from the most lateral points of the trachea. 8 Because of the limited space, isthmic tumors tend to be more likely to have capsule invasion, 8 which may increase the risk of lymph node metastasis.9,10 Moreover, the isthmus acts as a bridge between the two lateral lobes, and tumors in this location might have a higher risk of being seeded on both sides.11,12 Thus, total thyroidectomy is always recommended for patients with isthmic PTC, but this can lead to a higher complication incidence and necessitates life-long drug replacement therapy, which may result in a decrease in patient survival. 10 In recent years, surgeons have also experimented with the local treatment of isthmic tumors with simple isthmic removal,2,13–15 which preserves the gland, but the effectiveness of this strategy remains controversial. 16
TA is also a local treatment method for tumor lesions without bilateral gland removal or preventive lymph node dissection, 17 which is similar to isthmic resection. However, it is difficult for the ablation area to reach a safety margin of 2 mm beyond the tumor edge for isthmic tumors because of the close distance to the capsule, 18 which may increase the risk of recurrence after treatment. In addition, owing to its close proximity to the trachea and superficial skin, TA for isthmic tumors may be associated with a higher risk of complications. 19 Retrospective studies have shown that TA of isthmic PTC can achieve good therapeutic effects.20–22 However, owing to insufficient evidence, the recently published Chinese Guidelines for Ultrasound-Guided Thermal Ablation of Thyroid Nodules considered isthmic PTC only as a relative indication, 23 and TA for PTC arising in the isthmus has not been fully explored.
In this prospective multicenter study, we hypothesized that MWA for the treatment of PTC might achieve similar efficacy and safety for lesions arising in the isthmus and lateral lobes. This study aimed to compare the feasibility, effectiveness, and safety of MWA for treating clinical T1N0M0 PTC arising in the isthmus and lateral lobes.
Materials and methods
Patients
The work has been reported in line with the STROBE statement (Supplemental Material 1). 24 Some of the data were from a cohort study reported previously 25 ; the purpose of the current study was different, and the follow-up was longer. The Institutional Ethics Committees (IECs) of Chinese PLA General Hospital and all other participant centers approved this study (clinicaltrials.gov: NCT04197960). All patients or their responsible caregivers provided written informed consent for MWA and inclusion in this study before enrollment. In accordance with the 2017 Thyroid Radiofrequency Ablation Guideline: Korean Society of Thyroid Radiology, TA was recommended for those who were ineligible or refused surgical resection. 26
We enrolled T1N0M0 PTC patients who planned to receive MWA from December 2019 to December 2020 from 12 hospitals. The inclusion criteria were (1) a suspicious thyroid nodule diameter of 2 cm or less in US; (2) no US- and/or Computed Tomography (CT)-detected lymph node metastasis (LNM) or distant metastasis; and (3) fine-needle aspiration or core-needle biopsy-confirmed PTC in pathology but without histopathologically or immunohistochemically proven aggressive histological types, according to the WHO classification; and (4) no prior neck irradiation or surgery. The exclusion criteria were (1) tumors with extra thyroid extension on US (replacement of the strap muscle or obtuse margins between the tumor and the trachea, esophagus, mediastinal vessels, or carotid artery),27,28 pathologically confirmed multifocal PTC, or patients with other malignancies, (2) patients with preoperative vocal cord palsy or other conditions unsuitable for MWA treatment (Supplemental Table 1), and (3) patients lost to follow-up.
The tumor location was evaluated with US, and isthmic tumors were defined as those whose tumor volume was at least 50% of the tumor volume located between two lines perpendicular to the surface of the skin from the most lateral points of the trachea; otherwise, they were recognized as lobe tumors (Figure 1). The enrolled patients were then divided into two groups: the isthmic tumor group (i-PTC) and the lobe tumor group (L-PTC). The isthmic tumors were divided into two subgroups: pure isthmic tumors (Pi-PTCs), defined as tumors entirely confined to the isthmus, and isthmic tumors extending to the lobe, defined as tumors partially protruding into the lateral lobe (Li-PTCs).
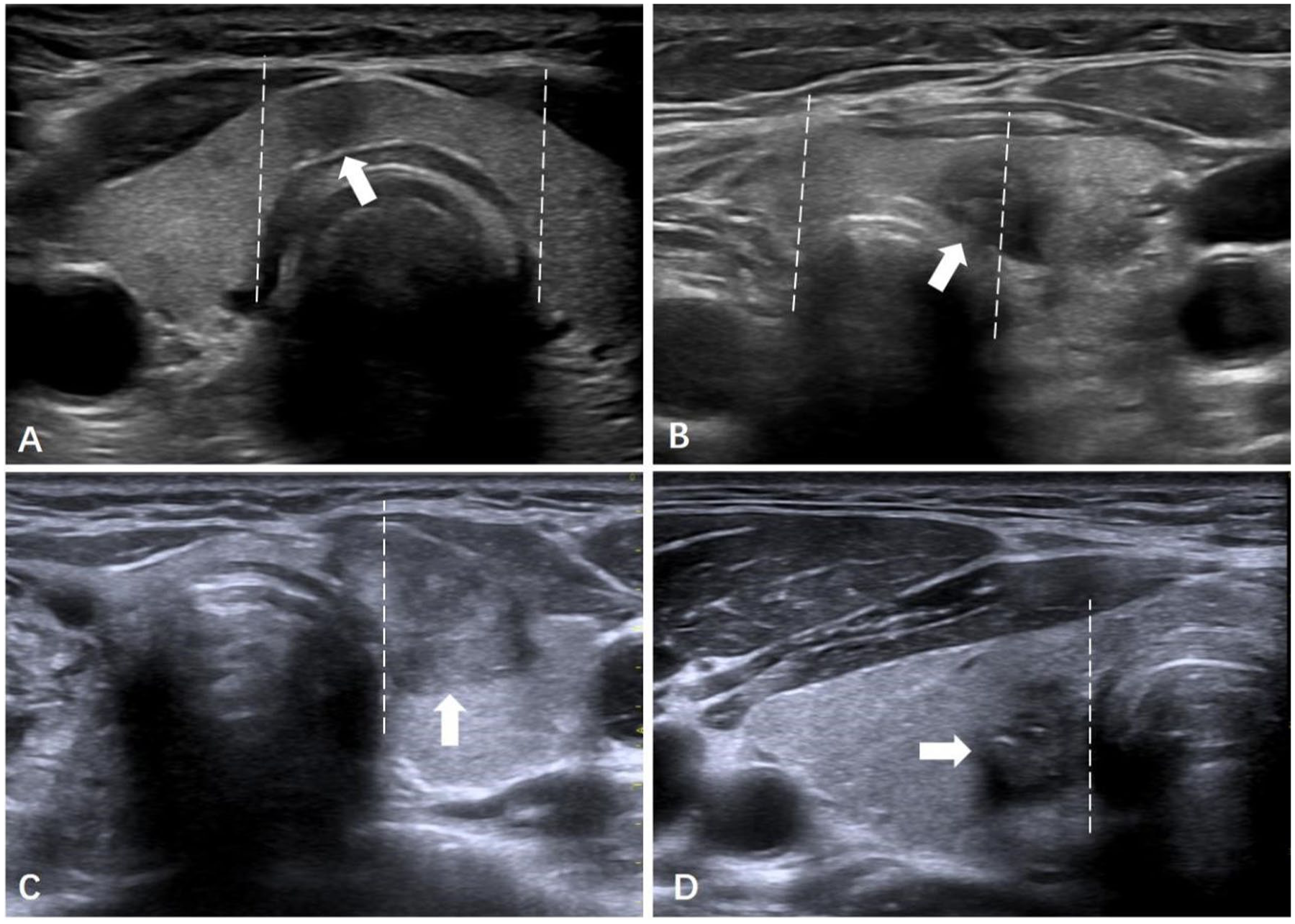
Evaluation of tumors’ location in US. (a) Pi-PTC (arrow): Pure isthmic papillary thyroid carcinoma. The tumor is entirely confined between two lines perpendicular to the surface of the skin from the most lateral points of the trachea (dotted lines). (b) Li-PTC: isthmic papillary thyroid carcinoma extending to the lobe (arrow). Less than 50% of the tumor volume protrudes into the lateral lobe. (c, d) L-lobe PTC: papillary thyroid carcinoma located in the lateral lobes (arrow). More than 50% of the tumor volume protrudes into the lateral lobe.
Preablation evaluations and determination of groups
All patients underwent laboratory tests, imaging studies, and bilateral vocal cord examinations prior to ablation (Supplemental Material 2). Anticoagulant medications were suspended for at least one week before MWA.
Preoperative US was performed to record the tumor location, size, US characteristics, and relationship with the capsule (Supplemental Material 3). 25 Capsular invasion was defined as the presence of a nodule, subtle capsular distortion or disruption, or bulging of the normal thyroid contour on the basis of capsular abutment. 25
Microwave ablation procedure
Radiologists with more than 12 years of thyroid US diagnosis experience and more than 7 years of US-guided intervention experience performed the procedure (Supplemental Table 2).
Patients and the MWA system (KY2000; Canyon Medical Instruments) were prepared as previously described. 4 Hydrodissection was performed with normal saline as isolating fluid to achieve a distance of at least 5 mm between the tumor and adjacent critical structures to prevent thermal injury. Isthmic tumors were separated from the trachea and superficial skin by hydrodissection. MWA was abandoned if the tumors could not be separated from the trachea or blood vessels. Afterward, the MWA antenna was inserted into the tumor under US guidance, and ablation was started. The safety margin was defined as the ablation zone extended at least 2 mm from the original tumor margin. The ablation zone should achieve the safety margin as much as possible unless the ablation space is limited. The patients were intermittently asked to speak to monitor the phonation during the ablation procedure.
Contrast-enhanced US (CEUS) was performed to confirm the completeness of the ablation. Additional ablation was performed immediately if residual enhancement was present within the target ablation zone. Patients remained under observation for 30 min to monitor for potential complications. All patients were advised to have their TSH suppressed in the range of 0.5–1 mU/L with levothyroxine to reduce the risk of recurrence.
Postablation evaluation and follow-up
Patients underwent a series of evaluations postablation, which included thyroid US and functional assessments (fT3, fT4, and TSH) at 3, 6, 12, 18, and 24 months, with annual follow-ups thereafter; CEUS examinations at 6 and 12 months; and annual neck and chest CT scans to surveil for potential distant metastases.
The effectiveness of ablation was determined by the absence of enhancement in all regions of the initial tumor on CEUS. Biopsies were conducted in cases where imaging suggested local tumor progression (LTP), the emergence of new thyroid cancer, or the presence of LNM.
If patients experienced complications of voice change, US was used to observe vocal cord movement and assess voice recovery at each evaluation.
Study endpoints
The primary endpoints focused on technical success and disease-free survival. Technical success was confirmed by the lack of enhancement in the targeted tumor region in CEUS after the whole ablation procedure. Disease-free survival was determined by the absence of LTP, new thyroid cancer, LNM, distant metastasis, or PTC-related death. LTP was identified by pathological confirmation of PTC at the margins of the ablation zone. New thyroid cancer was defined as pathologically confirmed malignancies in other sections of the thyroid. LNM was characterized by pathologically confirmed metastatic lymph nodes in the neck regions. Distant metastases were identified outside the neck through CT, positron emission tomography (PET), or bone scanning. The secondary endpoints included the treatment parameters, major complications, and tumor shrinkage over the monitoring duration. The ablation time was consistent with the duration of energy application. Pain levels during and immediately following ablation were assessed using the visual analog scale, which ranged from 0 to 10. Monitoring commenced after ablation until discharge. Major complications were documented according to the reporting standards for thyroid ablation, 5 and minor complications were not evaluated. The volume reduction rate was calculated as follows: (initial tumor volume – ablation zone volume) × 100/initial tumor volume (%). Complete resorption was defined as the disappearance of the original tumor and blurring of the ablation zone on US.
Statistical analysis
Quantitative data are reported as means ± standard deviations (SDs) and ranges and were compared between two groups using Student’s unpaired t test. Categorical data are presented as values and percentages, and were compared between groups with the chi-square test or Fisher’s exact test. Changes in the volume of each tumor before ablation and at each follow-up were compared with repeated-measures analysis of variance. Disease-free survival curves and cumulative complete resorption curves were generated by the Kaplan–Meier method and assessed by the log-rank test for between-group differences.
Subgroup analysis (hazard ratios and their 95% CIs) was performed using the Cox proportional hazards regression model. When p was < 0.05, a significant difference was considered. All the analyses above were performed with R Studio software (version 4.0.0; R Foundation for Statistical Computing) by L.Z. after the consultation of a statistician (Professor M.L., Statistics and Epidemiology Research Room in Chinese PLA General Hospital).
The sample size and power analysis results for disease-free survival were calculated with PASS 15.0 software (NCSS): 1 − β=0.8, R = 5.0; if disease-free survival was assumed to be 89% in the isthmic tumor group and 99% in the lobe tumor group, 107 and 532 patients, respectively, were needed.
Results
Patients and tumor characteristics
From December 31, 2019, to December 31, 2020, 658 patients with a single PTC who planned to receive MWA and 652 (mean age, 43 years ± 11 (SD), 481 females) were included after exclusion (Figure 2). Among them, 109 were isthmic tumors, and 543 were lobe tumors. The enrollment followed a fixed time window that naturally resulted in exceeding the calculated sample size. Supplemental Table 2 presents the number of enrolled patients in the 12 participating institutions. One i-PTC aborted MWA due to hydrodissection failure (recorded as a technical failure), and 651 patients were enrolled for treatment effectiveness and safety analysis. Until 1 June 2025, the mean follow-up periods for i-PTC (n = 108) and L-PTC (n = 543) were 61 months ± 4 (range, 55–67 months) and 61 months ± 4 (range, 54–67 months), respectively (p = 0.48).

Research flowchart.
The demographic and clinical characteristics of the enrolled patients are summarized in Table 1, and the features of the tumors are presented in Table 2. Compared with the L-PTC group, the i-PTC group had a greater proportion of tumors with capsular invasion (16% (91/543) vs 25% (27/108), p = 0.04). There was no evidence of differences between the two groups in terms of sex, BMI, Charlson comorbidity index, thyroid disease, laboratory studies, tumor size, US tumor features, and the grades of C-TIRADS.
General information of patients with PTC located in the isthmus or lobes.

Unless otherwise indicated, data are numbers of patients, with percentages in parentheses.
Data are means with SD in parentheses.
BMI, body mass index; CCI, Charlson comorbidity index; fT3, free triiodothyronine; fT4, free thyroxine; i-PTC, isthmic papillary thyroid carcinoma; L-PTC, papillary thyroid carcinoma located in lateral lobes; MWA, microwave ablation; PTC, papillary thyroid carcinoma; TSH, thyroid-stimulating hormone.
Characteristics of tumors located in the isthmus and lobes.

Unless otherwise indicated, data are numbers of patients, with percentages in parentheses. According to the US presentation of the internal nodule on color Doppler flow imaging, the nodular vascular scores were classified into four grades: (a) no color signal in the nodule, (b) color signals in less than 25% of the nodule (grade I), (c) color signals in 25%–50% of the nodule (grade II), and (d) color signals in more than 50% of the nodule (grade III).
Data are means with SD in parentheses.
i-PTC, isthmic papillary thyroid carcinoma; L-PTC, papillary thyroid carcinoma located in lateral lobes.
Technical success and parameters
One in the i-PTC group experienced hydrodissection failure, and the technical success rates for the i-PTC group and L-PTC group were 99% (108/109) and 100% (543/543), respectively (p = 0.17). The technique-related parameters, including the ablation energy, time, and hydrodissection, did not differ (4600 J ± 6134 J vs 4699 J ± 3134 J, 156 s ± 129 s vs 151 s ± 119 sand 28 ml ± 25 ml vs 29 ml ± 26 ml; p = 0.85, p = 0.81 and p = 0.71, respectively). All patients tolerated the MWA procedure, and the intraoperative pain score, immediate postoperative, pain score and monitoring time were similar (2.8 ± 1.2 vs 2.6 ± 1.4, 1.6 ± 1.0 vs 1.6 ± 1.1 and 1.0 d ± 1.1 vs 1.0 d ± 1.1, p = 0.30, p = 0.96 and p = 0.73, respectively; Table 3).
Technique parameters and treatment outcomes of MWA for PTC located in the isthmus or lobes.

Unless otherwise indicated, data are numbers of patients, with percentages in parentheses.
Data are means with SD in parentheses.
p was compared between the i-PTC group and the L-PTC group.
p was compared between the subgroup of Pi-PTC and the L-PTC group.
p was compared between the subgroup of Li-PTC and the L-PTC group.
fT3, free triiodothyronine; fT4, free thyroxine; i-PTC, isthmic papillary thyroid carcinoma; L-PTC, papillary thyroid carcinoma located in lateral lobes; Li-PTC, isthmic papillary thyroid carcinoma extending to lobe; Pi-PTC, pure isthmic papillary thyroid carcinoma; TSH, thyroid-stimulating hormone.
Treatment-related outcomes
Compared with the L-PTC group, a larger proportion of the i-PTC group failed to achieve a targeted 2 mm safety margin (39.0% (212/543) vs 50.0% (54/108), p = 0.03) because of space limitations (Table 3).
The incidence of major complications remained comparable between the two groups (0.9% (1/108) vs 4.4% (24/543), p = 0.08), including 1 case of cough in the i-PTC group and 19 cases of hoarseness, 12 of cough, and 1 of Horner syndrome in the L-PTC group (Table 3). All patients with complications reported in the two groups recovered within 6 months after ablation.
The disease-free survival rates in the i-PTC group and L-PTC group were 97.2% (105/108) and 97.4% (529/543), respectively, at the last follow-up (p = 0.91; Table 3). In the i-PTC group, three LNMs occurred (two in the Pi-PTC subgroup and one in the Li-PTC subgroup), and in the L-PTC group, four LTPs, three new thyroid cancers, and eight LNMs occurred. One LNM case in the i-PTC group was bilateral, and the others were unilateral in the cervical region, while all the LNMs in the lobe tumor group were ipsilateral to the original tumors. No distant metastasis was detected in either group during the follow-up period.
The ablation zone decreased gradually during follow-up, and tumor shrinkage in the i-PTC and L-PTC showed comparable results (Table 3). At the 3-year evaluation, the VRR was 96 ± 15% (SD) in the i-PTC group and 95 ± 17% (SD) in the L-PTC group (p = 0.70), and 69.6% (75/108) versus 68.8% (374/543) of the ablation zone in the two groups achieved complete resorption (p > 0.99; Figure 3).

Tumor shrinkage during the follow-up. (a) Volume reduction rates of the i-PTC group and the L-PTC group. (b) Volume reduction rates of Pi-PTC, Li-PTC, and L-PTC subgroups. (c) Complete resorption rates of i-PTC and L-PTC groups. (d) Complete resorption rates of Pi-PTC and Li-PTC groups.
During the follow-up period, 8 (1.2%) of the patients abandoned levothyroxine because of obvious side effects such as palpitations, headache, and insomnia, of whom 7 (1.3%) were in the L-PTC group, and 1 (0.9%) was in the i-PTC group. At the last follow-up, the thyroid function results revealed that the TSH level was suppressed, and there was no significant difference between the i-PTC and L-PTC groups (all p > 0.05). No treatment-related clinical hypothyroidism or hyperthyroidism was found during follow-up.
Sensitivity analysis outcomes
When Pi-PTC and Li-PTC were compared with L-PTC, the technique parameters and treatment outcomes were still similar (all p > 0.05; Table 3). Similarly, when the i-PTC and L-PTC groups were compared in terms of T1a or T1b tumor stages, tumors with or without capsule invasion and those that achieved or failed safety margins, the major complications and disease-free survival rates were still comparable (all p > 0.05; Supplemental Table 3). In the prespecified subgroups of sex, age, or thyroid disease, the treatment outcomes did not differ between the i-PTC group and the L-PTC group (all hazard ratios crossed 1, as shown in Figure 4).

Comparison of outcomes of major complications (a), disease-free survival (b), and complete resorption (c) between i-PTC and L-PTC groups under subgroups analysis.
Discussion
In our multicenter prospective cohort study, we evaluated the feasibility, effectiveness, and safety of microwave ablation (MWA) for T1N0M0 PTC located in the isthmus and lateral lobes with more than 5 years of follow-up. Our results indicated that the treatment outcomes of MWA for T1N0M0 PTC in the isthmus and lateral lobes were similar. In the i-PTC and L-PTC groups, comparable technical success rates were achieved (99% vs 100%, p = 0.17), with similar complication incidence (0.9% vs 4.4%, p = 0.08). Although fewer i-PTCs achieved the targeted safety margin (2 mm) than L-PTCs did (50.0% (54/108) vs 61.0% (331/543), p = 0.03), no differences were observed in disease-free survival (97.2% (105/108) vs 97.4% (529/543), p = 0.91) or tumor shrinkage (96 ± 16% (SD) vs 95 ± 17% (SD), p = 0.70). Multiple subgroup analyses revealed that the outcomes remained comparable between the i-PTC and L-PTC groups.
Preliminary studies with small sample sizes and short follow-up periods on TA for isthmic T1N0M0 PTC reported results similar to those of our study.20,29 Cao et al. reported no disease progression or complications approximately 17 months after TA. 20 Xiao et al. reported 2.9% LTPs and 2.9% new thyroid cancers approximately 25 months after TA, with 1% complications occurring. 29 Our study further compared the treatment outcomes of pure isthmic tumors and isthmic tumors extending to the lobe with those of lobe tumors with a larger sample size and longer follow-up. The comparable outcomes observed in our study between the groups may be partly due to the indolent nature of the tumor, 30 indicating that tumor location does not significantly impact clinical prognosis. Moreover, for patients with disease progression, including those with LTP, LNM, and new cancers, reablation was used as treatment, and good therapeutic effects (disease-free survival) were observed at subsequent follow-ups. Notably, our study design enabled us to identify substantial differences (10% or more) between tumor locations with high confidence. However, smaller discrepancies might not have been captured because of the limited number of patients, potentially masking true differences in clinical outcomes. This highlights the importance of larger studies to more accurately assess the nuanced effects of tumor location on prognosis.
Our study also revealed that i-PTCs have a higher probability of capsular invasion, and fewer could cover the target safety margin because of the limited ablation space; however, comparable results were still achieved in terms of disease-free survival between the i-PTC and L-PTC groups regardless of the negative factors. To verify the reliability of the results, we further compared the treatment outcomes of the two groups in multiple subgroups, including the degree of capsular invasion, safety margin, tumor stage, sex, age, and thyroid disease, and the outcomes remained stable. These findings indicate that for low-risk T1N0M0 PTC patients, especially those whose tumors were located in the isthmus, although safety margins were not strictly achieved, excellent therapeutic effects could still be achieved under experienced clinical management. An immediate postoperative contrast-enhanced ultrasound assessment by experienced radiologists revealed no abnormal enhancement in the tumor area, indicating that the tumor was completely eradicated. Unlike gross extra thyroid extension,31,32 capsular invasion was not proven to be a risk factor for disease progression, and previous studies have indicated that it might not moderate the treatment effect of MWA for PTC, 21 which supported our results. A number of previous articles have shown that TA therapy for stage T1b PTC can achieve efficacy similar to that of stage T1a PTC.33–35 In our study, the disease-free survival of stage T1b PTC patients was 93.5%, which is consistent with the findings of previous studies, and we further reported that for stage T1b PTC patients, good prognostic effects can also be achieved when the tumor is located in the isthmus. However, the underlying mechanism is not clear, and the number of cases in this study is limited. Further research is still necessary to verify this result.
Safety margins play crucial roles in the prognosis of aggressive tumors in MWA treatment,36,37 but MWA of PTCs that did not reach the safety margin still achieved good efficacy in our study, which may be related to the inert nature of these thyroid tumors. In addition, the proximity of the tumor to the capsule does not mean that the cancer cells have broken through the capsule, and the eighth American Joint Committee on Cancer TNM staging system has deprecated the concept of minimal extra-thyroid extension within tumor staging considering these points. 38 During the ablation procedure, the adjacent capsule was also ablated when the tumor was located near the capsule, and immediate CEUS evaluation after ablation was performed to confirm complete tumor ablation, which may further contribute to optimal therapeutic efficacy.
In the ablation of i-PTCs, hydrodissection plays a crucial role in the technical success and low complication rate because of the ample space created between the tumor and critical structures. A single instance of hydrodissection failure was encountered, and we speculate that the underlying cause was tumor invasion of the trachea, resulting in adhesions. For patients who experienced voice change, we evaluated vocal cord movement by US at each follow-up until the voice recovered, and no restriction of vocal cord movement was found. Most patients recovered their voice within 1 h after ablation, which we believed may have been transient nerve paralysis caused by anesthetics. The remaining patients with longer hoarseness might have experienced nerve heat burns, but the voice still recovered within 6 months after ablation. These findings suggest that MWA is technically feasible for treating isthmic tumors and nerve injury during the procedure, which is rare and tends to be moderate.
Our study had several limitations. First, the lack of significance might partially be due to the small number of patients, especially for subgroup analysis, and the oncological results of this study were limited. Second, the assessment of tumor location and capsular invasion depended largely on the experience of the radiologist, and occult PTCs and LNMs may have been missed. Third, the diagnosis of PTC was based only on cytological samples, and the relationship between aggressive histological subtypes and prognosis was unclear. Finally, this study followed an observational cohort design rather than a formal equivalence trial, and the comparison of subgroups was performed post hoc; thus, selection bias may have occurred.
Conclusion
Microwave ablation for T1N0M0 PTC located in the isthmus demonstrated comparable efficacy outcomes with those located in the lateral lobes, although fewer covered the safety margin. To substantiate these observations, studies with larger cohorts and extended follow-up periods are warranted.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261442628 – Supplemental material for Microwave ablation for T1N0M0 papillary thyroid carcinoma located in the isthmus: a multicenter prospective cohort study with over 5-year follow-up
Supplemental material, sj-docx-1-tam-10.1177_17588359261442628 for Microwave ablation for T1N0M0 papillary thyroid carcinoma located in the isthmus: a multicenter prospective cohort study with over 5-year follow-up by Lin Zheng, Qiao-Wei Du, Jian-Ping Dou, Fang-Yi Liu, Jie Yu, Zhi-Gang Cheng, Xiao-Ling Yu, Shui-Lian Tan, Hui Wang, Zhi-Bin Cong, Shu-Rong Wang, Ming-An Yu, Zhi-Feng Xu, Ying Che, Nan Bai, Cun Liu, Sheng-Nan Huo, Ying Hao, Xue Wang, Ying Liu, Ying Zhou, Zhi-Yu Han and Ping Liang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We acknowledged statistician Miao Liu (statistics and epidemiology research room in Chinese PLA general hospital) for the consultation and assistance with the statistical part of this research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
