Abstract
Background:
Low-level hepatitis B viremia is recognized as a potential driver of hepatocarcinogenesis; however, its prognostic impact on patients with advanced hepatocellular carcinoma (HCC) receiving systemic therapy remains poorly defined.
Objective:
To determine the independent prognostic impact of low-level viremia (LLV; HBV DNA 20–2000 IU/mL) compared with maintained virologic response (MVR; HBV DNA < 20 IU/mL) in patients with intermediate-to-advanced HBV-related HCC undergoing systemic therapy, and to evaluate whether on-treatment viral status serves as a superior predictor to baseline HBV DNA levels.
Design:
This was a multicenter retrospective cohort study.
Methods:
A total of 1814 patients treated at three centers between 2020 and 2024 were retrospectively enrolled. After ⩾4 months of follow-up, patients were classified into LLV (n = 860) or maintained virologic response (MVR; HBV DNA < 20 IU/mL, n = 954) groups. Overall survival (OS) and progression-free survival (PFS) were the primary endpoints. Logistic regression identified risk factors for LLV; Cox proportional hazards models assessed independent prognostic factors. Subgroup analyses were performed by treatment regimen and baseline HBV DNA load.
Results:
Multifactorial analysis identified HBeAg positivity (OR 1.971, 95% CI 1.53–2.541, p < 0.001), AST > 40 U/L (OR 1.437, 95% CI 1.126–1.832, p = 0.004), extrahepatic metastasis (OR 1.640, 95% CI 1.187–2.266, p = 0.003), and detectable baseline HBV DNA (OR 2.482, 95% CI 2.002–3.077, p < 0.001) as independent risk factors for LLV. Compared with the MVR group, patients in the LLV group had significantly reduced PFS (8.3 vs 14.3 months, p < 0.001) and OS (25.5 vs 37.8 months, p < 0.001). Multivariate analysis identified LLV as an independent predictor of worse OS (HR 1.506, 95% CI 1.299–1.746). Baseline HBV DNA load was not associated with survival.
Conclusion:
LLV is a robust, independent indicator of poor prognosis in systemic-treated intermediate-to-advanced HBV-HCC and should be preferred to baseline HBV DNA load for risk stratification and to guide intensified antitumor therapy.
Plain language summary
Introduction: Low-level hepatitis B viremia viremia is recognized as a potential driver of hepatocarcinogenesis; however, its prognostic impact on patients with advanced hepatocellular carcinoma (HCC) receiving systemic therapy remains poorly defined. Aim: We wanted to see if LLV (20–2,000 virus copies per mL) detected during treatment predicts shorter survival in patients with intermediate or advanced hepatitis B-related liver cancer who are receiving medicines such as lenvatinib or immunotherapy. Methods: A total of 1,814 patients treated at three centers between 2020 and 2024 were retrospectively enrolled. After ⩾4 months of follow-up, patients were classified into LLV (n = 860) or maintained virologic response (MVR; HBV DNA < 20 IU/mL, n = 954) groups. Overall survival (OS) and progression-free survival (PFS) were the primary endpoints. Logistic regression identified risk factors for LLV; Cox proportional hazards models assessed independent prognostic factors. Subgroup analyses were performed by treatment regimen and baseline HBV DNA load. Results: Multifactorial analysis identified HBeAg positivity (OR 1.971, 95% CI 1.53–2.541, p < 0.001), AST > 40 U/L (OR 1.437, 95% CI 1.126–1.832, p = 0.004), extrahepatic metastasis (OR 1.640, 95% CI 1.187–2.266, p = 0.003), and detectable baseline HBV DNA (OR 2.482, 95% CI 2.002–3.077, p < 0.001) as independent risk factors for LLV. Compared to the MVR group, patients in the LLV group had significantly reduced PFS (8.3 vs. 14.3 months, p < 0.001) and OS (25.5 vs. 37.8 months, p < 0.001). Multivariate analysis identified LLV as an independent predictor of worse OS (HR 1.506, 95% CI 1.299–1.746). Baseline HBV DNA load was not associated with survival.
Keywords
Introduction
Globally, primary hepatic malignancies are the sixth most prevalent cancer type, with HBV infection implicated in over half of all cases of HCC.1,2 The contribution of HBV has increased substantially in China and is now associated with 69.9% of all HCC cases. 3 Owing to the insidious onset and highly aggressive nature of HCC, most patients are diagnosed when the disease is at an intermediate or advanced stage. Current first-line therapeutic strategies for intermediate or advanced HCC predominantly involve tyrosine kinase inhibitor (TKI) monotherapy, for example, lenvatinib or sorafenib, or immune checkpoint inhibitor-based combination regimens such as atezolizumab plus bevacizumab. 4 The median overall survival (OS) of patients with intermediate or advanced HCC has improved from 10.7 months in 2018 to 19.2 months in 2022.5,6 The optimization of the management of etiological factors, including antiviral therapy for HBV/HCV and metabolic dysfunction-associated steatohepatitis control, over the same period has contributed to this improvement in patient prognosis. 4 Current clinical guidelines recommend that patients with HBV-associated HCC undergoing systemic antitumor therapy receive concomitant prophylactic antiviral therapy to suppress HBV replication. 7 However, existing nucleos(t)ide analogs are unable to eradicate covalently closed circular DNA, the persistent transcriptional template for HBV in infected hepatocytes. 8
The association between HBV DNA levels and HCC risk has been well studied, with a consensus that HBV DNA levels > 2000 IU/mL are significantly associated with hepatic inflammatory activity, fibrosis progression, and HCC risk. Therefore, comprehensive virologic surveillance, especially quantitative HBV DNA monitoring, is essential for patient risk stratification. Standardized management protocols, including viral surveillance and antiviral therapy, have been developed for this high-risk group. 9 The pathological significance of low-level viremia (LLV 9 ; defined as serum HBV DNA levels of 20–2000 IU/mL) in driving hepatic fibrosis and hepatocarcinogenesis remains controversial. A study by Sinn et al. involving patients with compensated cirrhosis demonstrated that patients with LLV were at a significantly higher risk of HCC development than those with undetectable HBV DNA. 10 By contrast, Lee et al. reported no statistically significant association between LLV and HCC incidence in this population. 11
A limited number of studies have evaluated the prognostic implications of LLV in patients with HCC. Although a study by Kim et al. demonstrated no significant association between LLV and clinical outcomes, these findings may have been affected by substantial selection bias, specifically the underrepresentation of intermediate or advanced -stage patients, with only 31.3% of patients categorized as Barcelona Clinic Liver Cancer (BCLC) stage B/C/D, and the minimal use of systemic therapies, with only 2.5% of patients receiving sorafenib treatment. 12 As systemic treatment is now a mainstay of care, the present study investigated the prognostic impact of LLV and its contributing factors in patients with intermediate or advanced stage HBV-related HCC undergoing systemic therapy. Furthermore, we comprehensively evaluated the association between baseline HBV DNA levels and patient prognosis across the virological strata.
Methods
Study design and participants
This multicenter retrospective study was conducted at the Fifth Medical Center of the General Hospital of the Chinese People’s Liberation Army, Youan Hospital of Capital Medical University, and the Affiliated Hospital of Guizhou Medical University. All patients with HCC who attended the three study centers between January 2020 and January 2024 were screened for inclusion in the study. The inclusion criteria were as follows: (1) diagnosis of intermediate-to-advanced HCC, (2) acceptance of first-line systemic therapy, (3) seropositive for HBsAg, and (4) patients with BCLC stage C or BCLC stage B. The exclusion criteria were as follows: (1) co-infection with other viruses, (2) incomplete follow-up data, (3) follow-up interval of more than 2 months, (4) regular use of antiviral drugs prescribed during antitumor therapy, (5) HBV DNA ⩾2000 IU/mL after 4 months of follow-up, (6) OS of less than 4 months, and (7) received regional antitumor therapy at baseline. The patients were categorized into two groups according to their serum HBV DNA levels after at least 4 months of follow-up: LLV group, with serum HBV DNA levels of 20–2000 IU/mL on at least one occasion; and maintained virologic response (MVR) group, with serum HBV DNA persistently undetectable (<20 IU/mL). The reporting of this study conforms to Strengthening the reporting of observational studies in epidemiology (STROBE) (Supplemental Table 1). 13
Related definitions and study endpoints
OS was measured from protocol-specified treatment commencement until all-cause mortality or censoring at the final evaluable time point (cutoff, January 2025). Progression-free survival (PFS) was defined as the time interval from baseline imaging to the earliest occurrence of radiologically confirmed disease progression (centrally blinded) or death from any cause according to RECIST 1.1. Antiviral history was defined as the receipt of regular antiviral therapy for at least 6 months prior to inclusion in the study. Baseline HBV DNA was defined as the level measured at enrollment, that is, immediately before the initiation of systemic antitumor therapy.
Data collection
Baseline data collected included information on sex, age, other population characteristics, serologic markers such as alpha-fetoprotein (AFP) and bilirubin, HBV DNA levels, radiography, antiviral regimens, and antitumor regimens. Thereafter, HBV DNA levels were measured, and abdominal imaging was performed every 1–2 months during follow-up.
Statistical analysis
Categorical and continuous variables are expressed as frequencies and mean ± SD or median and IQR (as applicable), respectively. Categorical variables were analyzed using the chi-squared test. Continuous variables were analyzed using the Student’s t-test or the Wilcoxon test, as appropriate. Prognosis was assessed using the Kaplan–Meier method. Logistic regression modeling was used to identify risk factors for LLV. In addition, Cox proportional hazards regression analyses were used to estimate adjusted HRs. A two-tailed p value ⩽0.05 was considered to indicate statistical significance. All statistical analyses were performed using SPSS software version 26.0 (IBM Corp., Armonk, NY, USA).
Results
Patient characteristics
Among all screened patients, 1814 met the inclusion criteria, 954 (52.6%) in the MVR group and 860 (47.4%) in the LLV group (Figure 1). Follow-up was discontinued in January 2025, with a median follow-up duration of 28 months (95% CI 26–30 months). The median age of the patients was 57 (IQR, 50–64) years; the majority were male (n = 1569; 86.5%). Of the 1814 patients, 965 (53.2%) had undetectable HBV DNA at baseline, and 849 (46.8%) had detectable HBV DNA at baseline. All patients received nucleos(t)ide analog antiviral therapy, primarily with entecavir (n = 1273; 70.2%).

Patients flow chart.
Antitumor regimens typically involved either TKI monotherapy (n = 797; 43.9%) or TKI combined with programmed cell death protein 1 (PD-1) inhibitors (n = 1017; 56.1%). Six TKI monotherapies were used: lenvatinib (n = 443; 55.6%), sorafenib (n = 283; 35.5%), donafenib (n = 45; 5.6%), apatinib (n = 11; 1.4%), regorafenib (n = 10; 1.3%), and anlotinib (n = 5; 0.6%). The TKI combined with PD-1 inhibitor regimens included lenvatinib plus sintilimab (n = 409; 40.2%), lenvatinib plus camrelizumab (n = 151; 14.9%), and sorafenib plus sintilimab (n = 98; 9.6%). The baseline characteristics of the two patient groups are presented in Table 1 and Supplemental Figure 1.
Baseline characteristics of the study population.

AFP, alpha-fetoprotein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCLC, Barcelona Clinic Liver Cancer; HBeAg, hepatitis B e antigen; LLV, low-level viremia; MVR, maintained virologic response; TKI, tyrosine kinase inhibitor; TKI plus PD-1 inhibitors, tyrosine kinase inhibitor plus programmed cell death protein-1 inhibitors; Tumor size, maximum tumor diameter.
Factors associated with LLV
As shown in Table 1, LLV was positively correlated with AFP > 400 ng/mL, HBeAg seropositivity, ALT > 40 U/L, AST > 40 U/L, tumor size > 5 cm, extrahepatic organ metastasis, detectable baseline HBV DNA, and BCLC stage C. These factors were therefore included in a multifactorial analysis (Supplemental Table 2), which identified HBeAg seropositivity (OR 1.971, 95% CI 1.53–2.541, p < 0.001), AST > 40 U/L (OR 1.437, 95% CI 1.126–1.832, p = 0.004), extrahepatic organ metastasis (OR 1.640, 95% CI 1.187–2.266, p = 0.003), and detectable baseline HBV DNA (OR 2.482, 95% CI 2.002–3.077, p < 0.001) as independent risk factors for LLV.
LLV and prognosis
Univariate and multivariate analyses of PFS and OS were performed using Cox regression analysis (Table 2). Univariate analyses showed that vascular invasion, AFP > 400 ng/mL, AST > 40 U/L, detectable baseline HBV DNA, Child-Pugh Class B, BCLC stage C, and LLV were associated with reduced PFS. Reduced OS was correlated with the same factors, and the additional factor was tumor size >5 cm. Multifactorial analysis revealed that PFS was reduced in patients with AFP > 400 ng/mL (HR 1.173, 95% CI 1.032–1.332, p = 0.015), AST > 40 U/L (HR 1.191, 95% CI 1.049–1.352, p = 0.007), LLV (HR 1.656, 95% CI 1.468–1.868, p < 0.001) and that OS was reduced in those with AFP > 400 ng/mL (HR 1.237, 95% CI 1.062–1.441, p = 0.006), tumor size > 5 cm (HR 1.240, 95% CI 1.060–1.450, p = 0.007), Child-Pugh Class B (HR 1.380, 95% CI 1.186–1.607, p < 0.001), BCLC stage C (HR 1.421, 95% CI 1.058–1.907, p < 0.019), and LLV (HR 1.506, 95% CI 1.299–1.746, p < 0.001). In addition, a comparison of the prognosis of patients in the LLV and MVR groups using Kaplan–Meier curves revealed that the median OS (25.5 vs 37.8 months, p < 0.001) and PFS (8.3 vs 14.3 months, p < 0.001) were significantly shorter in the LLV group than in the MVR group (Figure 2).
Analysis of prognostic risk factors for patients receiving systemic therapy.

AFP, alpha-fetoprotein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; HBeAg, hepatitis B e antigen; HR, hazard ratio; LLV, low-level viremia; TKI, tyrosine kinase inhibitor; Tumor size, maximum tumor diameter.
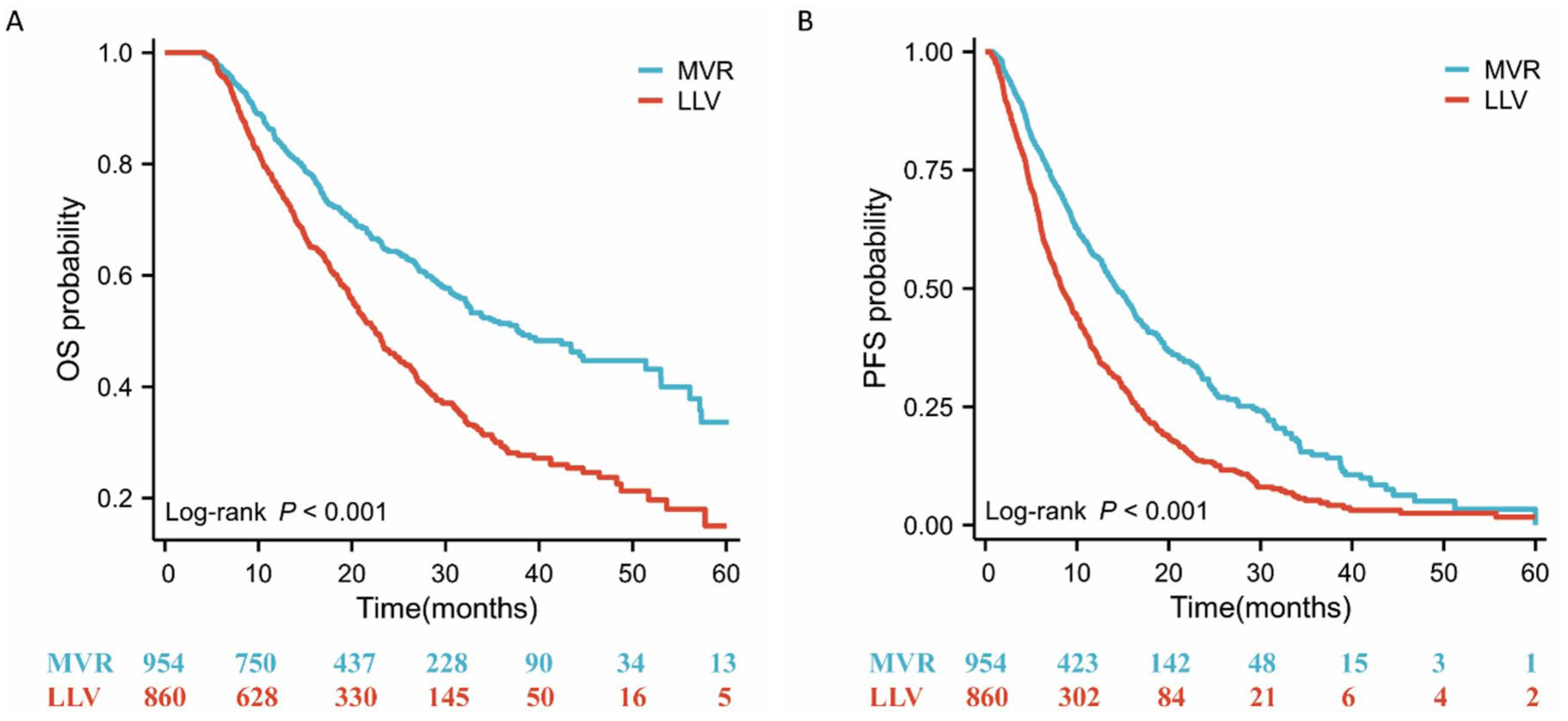
K-M survival curves were used to analyze OS and PFS in patients in the LLV and MVR groups.
Subgroup analysis of LLV and prognosis
These results indicate that patients with LLV have a poorer prognosis than those with MVR. To exclude the influence of the antitumor regimen on the results, we divided the patients into subgroups according to treatment regimen. The median OS of the LLV group remained significantly shorter than that of the MVR group in patients treated with TKI monotherapy (20.9 vs 37.8 months, p < 0.001), lenvatinib monotherapy (20.0 vs 32.1 months, p = 0.001), a TKI plus a PD-1 inhibitor (23.1 vs 39.6 months, p < 0.001), and lenvatinib plus sintilimab (23.1 vs 44.7 months, p < 0.001). Similarly, the median PFS of the LLV group was significantly shorter than that of the MVR group in all four of these subgroups (Supplemental Figure 2). We further stratified patients by BCLC stage into BCLC-B and BCLC-C cohorts and compared outcomes between the LLV and MVR groups within each subset. In both BCLC-B and BCLC-C, patients with LLV exhibited significantly shorter progression-free survival and overall survival than those with MVR (Supplemental Figure 3).
Patients were also divided into subgroups according to baseline HBV DNA load. Patients in the LLV group had significantly reduced OS compared to those in the MVR group in patients with undetectable baseline HBV DNA (26.3 vs 43.3 months, p < 0.001) and in all patients with detectable baseline HBV DNA (20.1 vs 28.7 months, p < 0.001). Patients with detectable baseline HBV DNA were further divided into four subgroups based on the baseline HBV DNA level (20–500, >500, 20–2000, and ⩾2000 IU/mL). In all four subgroups, the OS of patients in the LLV group was significantly shorter than that of patients in the MVR group (Figure 3). Similarly, the LLV group had a significantly shorter median PFS than the MVR group in all six of these subgroups (Supplemental Figure 4).

K-M survival curves were used to analyze the effect of LLV and MVR on OS in different subgroups of patients: baseline HBV DNA undetectable (a), baseline HBV DNA detectable (b), baseline HBV DNA = 20–500 IU/mL (c), baseline HBV DNA=20–2000 IU/mL (d), baseline HBV DNA > 500 IU/mL (e), baseline HBV DNA > 2000 IU/mL (f).
Baseline HBV DNA and prognosis
As shown in Table 2, univariate analyses revealed that detectable baseline HBV DNA was associated with shorter OS and PFS; however, in multivariate analyses, this association was offset by other factors. A further multifactorial analysis that did not consider LLV as an influencing factor (Supplemental Table 3) showed that detectable baseline HBV DNA significantly reduced the OS of patients compared with undetectable baseline HBV DNA (HR 1.239, 95% CI 1.061–1.448, p = 0.007). Baseline HBV DNA load subgroup analysis demonstrated that although patients with detectable baseline HBV DNA had a shorter OS and PFS than those of patients with undetectable baseline HBV DNA, among patients with detectable baseline HBV DNA, associations of loads of ⩾500 versus <500 IU/mL and ⩾2000 versus <2000 IU/mL showed no significant association between baseline HBV DNA load and patient prognosis (Figure 4).

K-M survival curves were used to analyze the effect of baseline HBV DNA levels on patient OS and PFS. Patients were stratified into two cohorts based on HBV DNA detection status (detectable vs undetectable), followed by comparative analysis of OS and PFS between the groups (a, d). In patients with detectable HBV DNA, the effects of high versus low HBV DNA load on OS and PFS were compared in subgroup analyses with different HBV DNA levels (b, e, c, f).
Dynamic changes in HBV DNA load affect the ability of HBV DNA load to predict prognosis. As shown in Supplemental Figure 5, of patients with detectable baseline HBV DNA, HBV DNA loads became undetectable in 377 (44.4%) and 455 (53.6%) at the first and second follow-up visits, respectively. HBV DNA levels were significantly lower during follow-up than at baseline. In addition, as shown in Supplemental Figure 6, the median baseline HBV DNA load was significantly higher in patients without antiviral history than in patients with antiviral history (5030 vs 200 IU/mL). After two follow-up visits, the median HBV DNA level was zero in both groups, and the proportion of patients with undetectable HBV DNA was almost the same in both groups (53.2% vs 54.1%).
Discussion
This study comprehensively evaluated the prognostic impact of LLV in patients with intermediate or advanced HBV-related HCC undergoing systemic therapy. Our findings demonstrated significantly shorter median PFS and OS in patients with LLV compared to those with MVR. These associations remained robust through Cox regression analyses and subgroup analyses stratified by antitumor regimens and baseline HBV DNA levels. Multivariate analyses revealed no association between baseline HBV DNA load and patient prognosis, which was further supported by subgroup analyses according to detectable baseline HBV DNA levels.
LLV is defined only in the guidelines of the American Association for the Study of Liver Diseases (2018), as detectable HBV DNA after 48 weeks (12 months) of antiviral therapy and HBV DNA < 2000 IU/mL. 9 However, in the present study, we modified this criterion to a minimum follow-up duration of 4 months. This adjustment was necessitated by the unique clinical characteristics of intermediate-to-advanced HCC patients. First, previous prospective studies have reported median PFS of 4.3–8.2 months in this population.6,14 A longer minimum follow-up period would result in therapeutic modifications in approximately 50% of patients during observation, thereby confounding LLV stratification. Second, the median OS for intermediate-to-advanced HCC ranges from 10.7 to 21.2 months.5,14 Implementing a 12-month follow-up requirement would exclude nearly half of potential enrollees, substantially limiting the prognostic utility of LLV assessment. Notably, even with this shortened 4-month criterion, 469 patients (25.9%) died or were lost to follow-up within the initial 4-month period according to our data. These considerations collectively justify our adoption of a 4-month rather than 48-week follow-up threshold.
The incidence of LLV in the present study was 47.4%, within the wide range of 32.0%–69.0% reported in previous studies.11,12,15,16 The limited reference guidelines available have led to inconsistencies in the definition of LLV and, therefore, large differences in reported LLV incidences. Reviewing the literature revealed the main reasons for the variation in these reported incidences of LLV as differences in baseline HBV DNA levels, primary disease status, patient enrollment rules, and antiviral status. First, baseline HBV DNA levels in previous studies were categorized as ⩾2000 and < 2000 IU/mL. In our study, additional groups were used, with varying incidences of LLV: undetectable, 34.5%; detectable, 62.1%; <2000 IU/mL, 42.1%; 20–2000 IU/mL, 56.1%; and ⩾2000 IU/mL, 70.4%. Patients with higher baseline HBV DNA levels had a higher incidence of LLV than those with lower baseline HBV DNA levels (Supplemental Table 4). Second, with the exception of the study by Tae-Se et al., previous studies on LLV have focused on patients with compensated cirrhosis without HCC. 12 The patients included in the study by Tae-Se et al. were predominantly BCLC stage O/A (73.1%), whereas the patients with HCC included in our study were BCLC stage B/C. Third, in terms of patient enrollment rules, the study by Jung et al. had the same enrollment rules as our study: regardless of the detection of baseline HBV DNA, patients could be classified as LLV or MVR during follow-up. 15 However, in most studies, only patients with detectable baseline HBV DNA could be classified as LLV. Fourth, previous studies often varied in the proportion of patients receiving antiviral therapy. Moreover, it is difficult to achieve uniformity in antiviral treatment regimens among patients undergoing antiviral therapy.
In the present study, PFS and OS were significantly shorter in the LLV group than in the MVR group, even after subgroup and Cox regression analyses. However, in the study by Tae-Se et al., although the 5-year survival rate of patients in the LLV group was lower than that of patients in the MVR group (67.3% vs 74.3%), the difference was not statistically significant (p = 0.076). 12 The association between LLV and the risk of HCC in patients with cirrhosis is controversial; Jiwon et al. found that patients in the LLV group had a higher risk of HCC than those in the MVR group (adjusted HR 2.36), 17 whereas Daniel et al. reported a comparable HCC risk between the two groups. 16 Despite this controversy, the conclusion that LLV affects the prognosis of patients with HCC is generally reliable.
The association between the baseline HBV DNA load and oncological prognosis also remains controversial. Some studies have reported that in patients treated with various approaches, such as sorafenib and hepatic resection, those with higher baseline HBV levels have a poorer prognosis than those with lower baseline HBV levels.18,19 By contrast, some studies have shown that patients with high and low baseline HBV DNA levels have a similar prognosis following systemic antitumor therapy, regardless of whether the threshold was set at 2000 or 500 IU/mL.20,21 This instability in the association between baseline HBV DNA levels and patient prognosis is mainly due to two reasons. First, HBV DNA levels are highly variable. Baseline levels were higher among patients with detectable baseline HBV DNA and no history of antiviral treatment than those with a history of antiviral therapy (median, 5030 vs 200 IU/mL). However, at the second follow-up visit after concurrent antitumor and antiviral therapy, median HBV DNA levels were similar in both groups, with similar rates of conversion to undetectable HBV DNA status. Second, transiently elevated HBV DNA levels have limited effects on disease progression. The prognosis of patients with high baseline HBV DNA levels can improve after viral reduction, whereas those with low baseline levels may experience disease exacerbation upon viral rebound.22,23 Our data suggested that patients in the LLV group had worse PFS and OS than those in the MVR group, regardless of baseline HBV DNA levels, and we found no significant differences in the prognosis of patients with high and low baseline HBV DNA levels (Supplemental Figure 6). Therefore, in clinical practice, LLV/MVR should be preferred to baseline HBV DNA levels as a predictive indicator of prognosis in patients with HCC.
This study has several limitations. First, as this is a retrospective study, there may be bias. However, we attempted to overcome this by including a large patient cohort from multiple centers and by performing subgroup analyses. Second, we included only patients with BCLC stage B/C. Although this reduces the prognostic impact of differences in disease severity, data on the prognostic impact of LLV in patients with early-stage HCC are lacking. Third, the imaging follow-up time window was non-uniform, which may have introduced bias into the PFS evaluation results. Fourth, we included patients with a minimum follow-up period of only 4 months, shorter than the period specified in previous similar studies. Although this decision was made based on factors related to PFS and OS of patients with intermediate or advanced HCC, there may be issues associated with the short follow-up period. Given these limitations, further prospective, multidisease, multistratified studies are required to optimize the surveillance, prevention, and management of patients with LLV.
Conclusion
The incidence of LLV is high in patients with intermediate or advanced HBV-related HCC. The main independent risk factors for LLV are HBeAg seropositivity, AST levels > 40 U/L, extrahepatic organ metastasis, and detectable baseline HBV DNA levels. Compared with patients with MVR, those with LLV experienced reduced PFS and OS. Predicting patient prognosis based on viral persistence status is more valuable than relying solely on baseline HBV DNA levels.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261435330 – Supplemental material for Low-level HBV viremia independently predicts poor outcomes in patients with intermediate-to-advanced HBV-related hepatocellular carcinoma receiving systemic therapy: a multicenter retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359261435330 for Low-level HBV viremia independently predicts poor outcomes in patients with intermediate-to-advanced HBV-related hepatocellular carcinoma receiving systemic therapy: a multicenter retrospective study by Jin Lei, Huaxing Ma, Xiong Chen, Ling Lin, Zhipeng Liang, Guangling Ou, Jiliang Jin, Hongyuan Dai, Ya Zhang, Mei Liu, Haiyang Li and Yinying Lu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261435330 – Supplemental material for Low-level HBV viremia independently predicts poor outcomes in patients with intermediate-to-advanced HBV-related hepatocellular carcinoma receiving systemic therapy: a multicenter retrospective study
Supplemental material, sj-docx-2-tam-10.1177_17588359261435330 for Low-level HBV viremia independently predicts poor outcomes in patients with intermediate-to-advanced HBV-related hepatocellular carcinoma receiving systemic therapy: a multicenter retrospective study by Jin Lei, Huaxing Ma, Xiong Chen, Ling Lin, Zhipeng Liang, Guangling Ou, Jiliang Jin, Hongyuan Dai, Ya Zhang, Mei Liu, Haiyang Li and Yinying Lu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359261435330 – Supplemental material for Low-level HBV viremia independently predicts poor outcomes in patients with intermediate-to-advanced HBV-related hepatocellular carcinoma receiving systemic therapy: a multicenter retrospective study
Supplemental material, sj-docx-3-tam-10.1177_17588359261435330 for Low-level HBV viremia independently predicts poor outcomes in patients with intermediate-to-advanced HBV-related hepatocellular carcinoma receiving systemic therapy: a multicenter retrospective study by Jin Lei, Huaxing Ma, Xiong Chen, Ling Lin, Zhipeng Liang, Guangling Ou, Jiliang Jin, Hongyuan Dai, Ya Zhang, Mei Liu, Haiyang Li and Yinying Lu in Therapeutic Advances in Medical Oncology
