Abstract
Background:
Pancreatic adenocarcinoma has a high mortality rate. Nanoliposomal irinotecan plus 5-fluorouracil and leucovorin (nal-IRI/FL) is the standard second-line chemotherapy after gemcitabine-based therapy. Although nanoliposomal irinotecan plus oxaliplatin, fluorouracil, and leucovorin (NALIRIFOX) has shown superior efficacy as first-line therapy over gemcitabine and nab-paclitaxel, its roles as second-line therapy remains undefined.
Objectives:
We aimed to compare the clinical outcomes and safety profiles of NALIRIFOX and nal-IRI/FL when used as a second-line treatment for patients with pancreatic adenocarcinoma who have progressed on a gemcitabine-based therapy.
Designs:
This was a single-center retrospective cohort study.
Methods:
We included patients with locally advanced or metastatic pancreatic adenocarcinoma who received NALIRIFOX or nal-IRI/FL following progression on first-line gemcitabine-based therapy from September 2020 to October 2024.
Results:
A total of 62 patients in the NALIRIFOX group and 131 in the nal-IRI/FL group were analyzed. Cumulative dose intensity ⩾80% over four cycles was achieved in 81.4% of the NALIRIFOX group and 73.1% of the nal-IRI/FL group (p = 0.32). The median progression-free survival (PFS) was 4.0 months (95% confidence interval (CI): 3.6–4.5) in the NALIRIFOX group versus 2.5 months (95% CI: 2.1–3.0) in the nal-IRI/FL group (hazard ratio (HR): 0.686; 95% CI: 0.497–0.947; p = 0.021), and the median overall survival was 7.0 months (95% CI: 5.0–9.1) versus 6.3 months (95% CI: 4.4–8.1), respectively (p = 0.827). Of the patients with disease progression after nal-IRI/FL, 69.1% received oxaliplatin-containing chemotherapy. Grade 3–4 adverse events were more frequent with NALIRIFOX, including febrile neutropenia, neutropenia, thrombocytopenia, and peripheral neuropathy; however, most were transient and manageable.
Conclusion:
NALIRIFOX provides superior PFS compared to nal-IRI/FL as second-line therapy for advanced or metastatic pancreatic cancer after gemcitabine failure. Although NALIRIFOX was associated with higher rates of hematologic and neurologic toxicities, they were manageable with careful monitoring.
Plain language summary
NALIRIFOX provides longer disease control than nal-IRI/FL in patients with advanced pancreatic cancer who has worsened after initial gemcitabine-based treatment. This real-world study from Taipei Veterans General Hospital compared two chemotherapy combinations to evaluate the efficacy and safety as a second-line therapy.
Key findings
1. Disease Control: Patients receiving NALIRIFOX went longer without their cancer growing (median of 4.0 months) compared to those receiving nal-IRI/FL (median of 2.5 months). 2. Overall Survival: While NALIRIFOX controlled the disease longer, the total survival time was similar between both groups (7.0 months for NALIRIFOX vs. 6.3 months for nal-IRI/FL).
The NALIRIFOX regimen is more intense and was associated with a higher risk of severe side effects compared to nal-IRI/FL:
1. Blood Counts: Significant drops in white blood cells and platelets were more common with NALIRIFOX 2. Nerve Damage: Patients on NALIRIFOX experienced more frequent numbness or tingling in their hands and feet. 3. Management: Most side effects were temporary and could be managed by doctors through careful monitoring and dose adjustments.
Summary for Patients
For patients whose pancreatic cancer has progressed, NALIRIFOX offers a more powerful option that can keep the disease stable for a longer period than the standard nal-IRI/FL. However, because it is more likely to cause side effects like low blood counts and nerve numbness, the choice of treatment should be tailored to each individual’s health status and personal goals.
Background
Pancreatic adenocarcinoma is one of the most lethal malignancies worldwide, with persistently poor survival outcomes despite therapeutic advances. Patients with metastatic disease have the worst prognosis, with a 5-year survival rate as low as 2.2% for inoperable patients. 1 For patients with metastatic disease, first-line chemotherapy options have evolved remarkably over the past decade.2–4 Unfortunately, the choice of second-line chemotherapy remains quite limited, underscoring the urgent need for more effective regimens.
Nanoliposomal irinotecan in combination with fluorouracil/leucovorin (nal-IRI/FL) has become the standard second-line treatment, since publication of the NAPOLI-1 trial.5,6 The liposomal formulation of irinotecan may provide preferential tumor accumulation and prolonged circulation, potentially overcoming stromal barriers in pancreatic cancer while reducing systemic toxicity.7–9 Pooled data from real-world experience with nal-IRI/FL in a recent meta-analysis revealed modest outcomes, with progression-free survival (PFS) of 2.87 months and overall survival (OS) of 6.06 months. 10 Considerable efforts have been made to identify superior chemotherapy regimens; however, treatment outcomes remain unsatisfactory. 11
The NAPOLI-3 trial evaluated the use of nanoliposomal irinotecan plus oxaliplatin, fluorouracil and leucovorin (NALIRIFOX; nal-IRI in combination with oxaliplatin, fluorouracil, and leucovorin) in the first-line setting and demonstrated superior efficacy, with improved survival outcomes compared to gemcitabine plus nab-paclitaxel. 3 The oxaliplatin component in NALIRIFOX may provide additional benefits in patients harboring homologous recombination repair (HRR) deficiency. 12 The use of NALIRIFOX as second-line chemotherapy may, therefore, offer additional benefits compared to nal-IRI/FL. Importantly, patients who progress on first-line chemotherapy often have poor performance status, 13 raising concerns regarding tolerability. Therefore, real-world studies comparing the efficacy and safety of nal-IRI/FL and NALIRIFOX are urgently needed.
In this retrospective study, we aimed to compare the efficacy and safety of nal-IRI/FL versus NALIRIFOX as second-line treatment options for patients with pancreatic adenocarcinoma who progressed on gemcitabine-based chemotherapy. By analyzing real-world outcomes, we sought to provide evidence to guide treatment decisions in this challenging clinical context.
Materials and methods
Patients
We conducted this retrospective single-center cohort study at Taipei Veterans General Hospital. The study population comprised patients with histologically confirmed locally advanced or metastatic pancreatic adenocarcinoma who received either NALIRIFOX or nal-IRI/FL as second-line chemotherapy following disease progression on gemcitabine-based chemotherapy. The choice of regimen was determined by the treating physician based on individual patient characteristics and clinical practice.
Patients who received an average dosage of less than 60% of the standard regimen during the first four treatment cycles were excluded, as previous studies have suggested that maintaining an adequate early cumulative dose is associated with better outcomes. 14 Patient records from September 1, 2020 to October 1, 2024 were reviewed, with follow-up extending to April 1, 2025. The study protocol was approved by the Institutional Review Board of Taipei Veterans General Hospital (IRB No. 2025-08-013BC), and the need for informed consent was waived due to the retrospective study design.
Treatment and assessments
The included patients received one of the following regimens. In the NALIRIFOX group, nal-IRI 50 mg/m2 was administered intravenously over 90 min, oxaliplatin 60 mg/m2 over 2 h, leucovorin 400 mg/m2 over 30 min, followed by fluorouracil 2400 mg/m2 as a 46-h continuous infusion, repeated every 2 weeks as described in the NAPOLI-3 trial. 3 In the nal-IRI/FL group, the patients received nal-IRI 70 mg/m2 intravenously over 90 min, followed by leucovorin 400 mg/m2 over 30 min, and then fluorouracil 2400 mg/m2 as a 46-h continuous infusion, repeated every 2 weeks as described in the NAPOLI-1 trial. 5 Treatment modifications were conducted based on physician discretion, toxicity, and recorded patient characteristics. Therapy duration, as documented in medical records, was determined by radiologically confirmed disease progression, development of unacceptable toxicity, patient refusal, or the physician decided to discontinue.
In accordance with standard institutional protocols, baseline evaluations included comprehensive medical history, physical examination, Eastern Cooperative Oncology Group (ECOG) performance status assessment, complete blood count, serum chemistry panel, carbohydrate antigen 19-9 (CA19-9) level, and radiological tumor assessment were performed before each regimen initiation. Radiological assessments and toxicity monitoring were performed every 3 months and prior to each cycle, respectively; these records were then retrospectively reviewed. We collected demographic data, disease characteristics, treatment details, laboratory values, response outcomes, and survival information from electronic medical records. Overall tumor response was evaluated according to Response Evaluation Criteria in Solid Tumors v1.1, and adverse events were graded using Common Terminology Criteria for Adverse Events v5.0. Outcomes analyzed included PFS, OS, objective response rate, best overall response, disease control rate, serum CA19-9 response (⩾50% reduction from baseline at least once during treatment), and safety profile.
Statistical analysis
Patient characteristics were summarized using descriptive statistics. Categorical variables were compared using Pearson’s Chi-square test or Fisher’s exact test, while continuous variables were analyzed using the Mann–Whitney U test. Survival analyses were performed using the Kaplan–Meier method with log-rank tests for group comparisons. PFS was defined as the duration from starting second-line chemotherapy to disease progression or death from any cause. OS was defined as the interval from the initiation of second-line chemotherapy to death from any cause. Potential prognostic factors were identified using univariate analysis, and those with p < 0.1 were included in a multivariate Cox proportional hazards regression model. Statistical significance was defined as p < 0.05 (two-sided). All statistical analyses were performed using SPSS version 28.0 (IBM Corp., Armonk, NY, USA).
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement guidelines for observational studie. 15 The completed STROBE checklist is provided as a Supplemental File.
Results
Patient characteristics
From September 1, 2020 to October 1, 2024, a total of 218 patients with pancreatic adenocarcinoma received NALIRIFOX or nal-IRI/FL. After excluding those with insufficient dose intensity, 193 patients—comprising 62 in the NALIRIFOX group and 131 in the nal-IRI/FL group—were included in the effectiveness analysis (Figure 1).

Consort diagram of patient selection and analysis populations.
There were no significant differences in most of the baseline characteristics between the two groups (Table 1). The median age was similar (65.3 vs 66.4 years, p = 0.875), and the proportion of older patients (⩾65 years) was comparable (51.6% vs 55.0%).
Baseline patient characteristics.
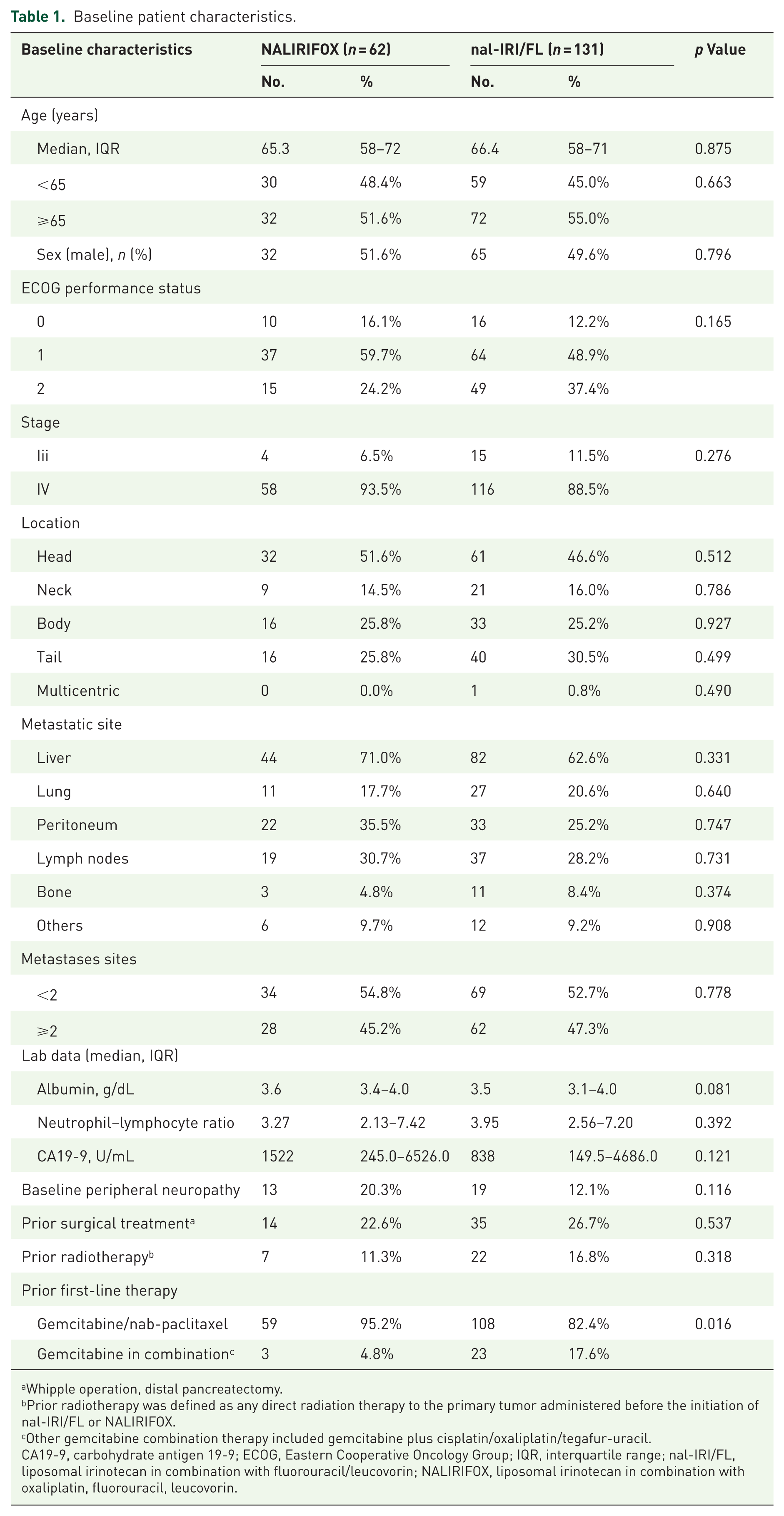
Whipple operation, distal pancreatectomy.
Prior radiotherapy was defined as any direct radiation therapy to the primary tumor administered before the initiation of nal-IRI/FL or NALIRIFOX.
Other gemcitabine combination therapy included gemcitabine plus cisplatin/oxaliplatin/tegafur-uracil.
CA19-9, carbohydrate antigen 19-9; ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range; nal-IRI/FL, liposomal irinotecan in combination with fluorouracil/leucovorin; NALIRIFOX, liposomal irinotecan in combination with oxaliplatin, fluorouracil, leucovorin.
The distribution of tumor location was similar, and there was no significant difference in prior surgical history (22.6% vs 26.7%, p = 0.537). Regarding the sites of metastasis, the liver was the most common site in both groups (71.0% vs 62.6%, p = 0.331). The proportion of patients having ⩾2 metastatic sites was comparable (45.2% vs 47.3%, p = 0.778). The baseline peripheral neuropathy after first-line chemotherapy was similar (20.3% vs 12.1%, p = 0.116). However, although gemcitabine plus nab-paclitaxel (G + A) was the predominant first-line chemotherapy in both groups, it was used significantly more frequently in the NALIRIFOX group (95.2% vs 82.4%, p = 0.016). Other gemcitabine-based combinations were less common.
Effectiveness outcomes
The median follow-up was 5.2 months (range, 0.2–28.0) for all patients, 5.5 months (range, 0.5–24.5) for those treated with NALIRIFOX, and 4.7 months (range, 0.2–28.0) for those treated with nal-IRI/FL. At the time of median follow-up (5.2 months), the NALIRIFOX group had 40 PFS events (64.5%) and 28 deaths (45.2%), while the nal-IRI/FL group had 97 PFS events (74.0%) and 67 deaths (51.1%). The 6-week cumulative dose of liposomal irinotecan was analyzed in 110 eligible patients (43 in the NALIRIFOX group and 67 in the nal-IRI/FL group). All patients in both groups maintained a relative dose intensity (RDI) of at least 60%. A similar proportion of patients achieved an RDI ⩾80% (81.4% vs 73.1% in the NALIRIFOX and nal-IRI/FL groups, respectively, p = 0.320). The mean RDI of liposomal irinotecan was 89.6% ± 11.7% in the NALIRIFOX group and 87.3% ± 10.6% in the nal-IRI/FL group. For fluorouracil, the mean RDI was 65.6% ± 10.3% and 77.6% ± 14.2%, respectively. The mean RDI of oxaliplatin in the NALIRIFOX group was 84.6% ± 13.5%.
The effectiveness outcomes are summarized in Table 2. NALIRIFOX demonstrated a significantly longer PFS compared to nal-IRI/FL, with a median PFS of 4.0 months (95% confidence interval (CI): 3.6–4.5) versus 2.5 months (95% CI: 2.1–3.0), respectively (p = 0.021, Figure 2(a)). The 6-month PFS rates were 30.0% and 19.7% in the NALIRIFOX and nal-IRI/FL groups, respectively. The median OS was comparable between the two groups (7.0 months (95% CI: 5.0–9.1) vs 6.3 months (95% CI: 4.4–8.1), respectively (p = 0.827, Figure 2(b))), and the 6-month OS rates were 54.2% and 53.1%, respectively. The objective response rate was significantly higher in the NALIRIFOX group compared to the nal-IRI/FL group (9.7% vs 2.3%, p = 0.019), and the disease control rate was also significantly better in the NALIRIFOX group (58.1% vs 36.6%, p = 0.005). The CA19-9 response rate was significantly better in the NALIRIFOX group, with a decrease >50% observed in 30.6% of the NALIRIFOX patients versus 16.8% of the nal-IRI/FL patients (p = 0.028).
Effectiveness outcomes.

CA19-9, carbohydrate antigen 19-9; CI, confidence interval; nal-IRI/FL, liposomal irinotecan in combination with fluorouracil/leucovorin; NALIRIFOX, liposomal irinotecan in combination with oxaliplatin, fluorouracil, leucovorin; No, number; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.

Kaplan–Meier curves of the two chemotherapy regimens. (a) Progression-free survival. (b) Overall survival.
Subgroup analysis of PFS (Figure 3) showed consistently better results with NALIRIFOX treatment. Significant benefits were observed in male patients (hazard ratio (HR) = 0.531, 95% CI: 0.335–0.844, p = 0.007), those aged >65 years (HR = 0.541, 95% CI: 0.346–0.846, p = 0.007), those with liver metastasis (HR = 0.620, 95% CI: 0.421–0.914, p = 0.016), lung metastasis (HR = 0.638, 95% CI: 0.446–0.914, p = 0.014), serum albumin <3.5 g/dL (HR = 0.563, 95% CI: 0.324–0.979, p = 0.042), prior chemotherapy (G + A; HR = 0.602, 95% CI: 0.428–0.845, p = 0.003), and no prior radiotherapy (HR = 0.648, 95% CI: 0.458–0.917, p = 0.014). In overall analysis, the patients who received NALIRIFOX had a better PFS than those who received nal-IRI/FL (HR = 0.686, 95% CI: 0.497–0.947, p = 0.021). In contrast, in subgroup analysis of OS (Figure S1), no statistically significant differences were observed between the two groups.

Forest plot with subgroup analysis of progression-free survival.
Information on subsequent therapy after disease progression is summarized in Table S1. In total, 83 patients (43.0%) received subsequent chemotherapy following progression on second-line NALIRIFOX or nal-IRI/FL, with similar rates between groups (48.3% vs 43.7%, p = 0.576). Notably, the subsequent use of oxaliplatin-containing regimens was significantly more frequent in the nal-IRI/FL group than in the NALIRIFOX group (69.1% vs 39.3%, p = 0.009).
Univariate and multivariate analyses were performed to identify prognostic factors associated with PFS (Table S2). Significant factors in univariate analysis included treatment with NALIRIFOX, ECOG performance status, disease stage, pancreatic head location, multicentric location, metastatic sites (liver and peritoneum), and baseline laboratory values (albumin, total bilirubin, creatinine, and neutrophil-to-lymphocyte ratio). In the multivariate analysis, NALIRIFOX treatment remained an independent favorable prognostic factor with an adjusted HR of 0.601 (p = 0.008). Other significant independent prognostic factors included ECOG performance status (HR = 2.064, p = 0.002), multicentric tumor location (HR = 19.853, p = 0.005), peritoneal metastasis (HR = 2.276, p < 0.001), and baseline neutrophil-to-lymphocyte ratio (HR = 1.752, p = 0.002).
Safety profiles
All patients receiving nal-IRI/FL (n = 154) or NALIRIFOX (n = 64) were included in the adverse event analysis, regardless of whether the average dose was below 60% of the standard dose.
Grade 3 or higher adverse events are summarized in Table 3. For hematologic toxicities, significantly higher rates of febrile neutropenia (14.1% vs 3.2%, p = 0.003), neutropenia (42.2% vs 14.6%, p = 0.001), and thrombocytopenia (29.7% vs 10.2%, p = 0.001) were observed in the NALIRIFOX group. Febrile neutropenia after chemotherapy was mostly brief and self-limited. However, during the study period, one patient in the NALIRIFOX group with pre-existing spontaneous bacterial peritonitis and liver dysfunction developed febrile neutropenia and subsequently died due to infection progression, while one patient in the nal-IRI/FL group died due to concurrent SARS-CoV-2 infection and febrile neutropenia. For non-hematologic toxicities, grade 3–4 peripheral neuropathy (10.9% vs 0.6%, p < 0.001) and hypokalemia (40.6% vs 26.1%, p = 0.033) were significantly more common in the NALIRIFOX group. Rates of diarrhea and vomiting were numerically higher in the NALIRIFOX group but did not reach statistical significance (4.7% vs 1.3%, p = 0.122 and 7.8% vs 3.2%, p = 0.133, respectively).
Treatment-emergent adverse events of Grade 3 or higher.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; nal-IRI/FL, liposomal irinotecan in combination with fluorouracil/leucovorin; NALIRIFOX, liposomal irinotecan in combination with oxaliplatin, fluorouracil, leucovorin.
Discussion
This retrospective study provides important comparative data on the efficacy and safety of NALIRIFOX versus nal-IRI/FL as second-line treatment options for patients with pancreatic cancer who progress on first-line gemcitabine-based chemotherapy. To our knowledge, this is one of the first direct comparisons between these two regimens in the second-line setting. Our findings demonstrated that NALIRIFOX was associated with significantly improved PFS compared to nal-IRI/FL, and that this improvement was largely consistent across the evaluated subgroups. The significantly increased hematologic toxicities and neurotoxicity with NALIRIFOX may require careful monitoring; however, they were manageable. The results suggest that NALIRIFOX represents an effective second-line therapeutic option for pancreatic cancer patients.
The superior PFS observed with NALIRIFOX likely reflects the synergistic benefit of incorporating oxaliplatin into this combination regimen. Oxaliplatin is a platinum compound that forms DNA adducts and inhibits DNA replication, and it may provide additional therapeutic advantages, particularly in specific patient subgroups. Recent evidence suggests that platinum agents significantly improve survival outcomes in patients with HRR-deficient pancreatic cancer. 12 The NAPOLI-3 trial demonstrated the superior efficacy of NALIRIFOX in the first-line setting for metastatic pancreatic ductal adenocarcinoma. 3 Our study extends these findings to the second-line setting, suggesting that the triplet combination may offer enhanced efficacy compared to the doublet regimen after progression on gemcitabine-based therapy.
Of note, although NALIRIFOX significantly extended PFS, this advantage did not translate to improved OS. This apparent paradox may be explained by the significant difference in subsequent treatment patterns. In this study, more than 40% of the patients received third-line chemotherapy after nal-IRI/FL or NALIRIFOX failure. Notably, patients in the nal-IRI/FL group were more likely to receive oxaliplatin-containing regimens as their next chemotherapy compared to those in the NALIRIFOX group. This comparable transition rate to third-line treatment, coupled with more subsequent oxaliplatin use in the nal-IRI/FL group, may partly explain why the PFS advantage with NALIRIFOX did not ultimately result in a significant difference in OS.
This retrospective study included patients with stage III and stage IV pancreatic adenocarcinoma. Although the nal-IRI/FL group numerically had more Stage III patients (11.5% vs 6.5%), this difference was not statistically significant (p = 0.276). While Stage III disease is generally associated with a better prognosis than Stage IV, the nal-IRI/FL group still exhibited shorter median PFS compared to the NALIRIFOX group. This suggests that the superior efficacy of NALIRIFOX was not masked by the slightly higher proportion of advanced-stage patients in that arm. Furthermore, our subgroup analysis of Stage IV patients—who represent most of our cohort—confirmed a significant PFS benefit for NALIRIFOX (HR 0.659, p = 0.015). In the multivariate analysis, the NALIRIFOX regimen remained an independent favorable prognostic factor for PFS after adjusting for stage. Due to the small sample size of Stage III patients (n = 19), specific conclusions regarding the efficacy of NALIRIFOX for Stage III disease could not be drawn, and further prospective studies are needed to validate these findings.
In this cohort, the proportion of patients previously treated with gemcitabine plus nab-paclitaxel differs between groups. This distribution was primarily driven by individual physician discretion and patient selection. Gemcitabine/nab-paclitaxel was the predominant first-line chemotherapy throughout the entire study period in both cohorts. Furthermore, as this was a single-center study, all patients had uniform access to the study medications. Crucially, multivariate analysis confirmed NALIRIFOX rather than the specific type of prior first-line treatment as significant prognostic factor. These findings support the validity of our survival analysis and suggest that the observed efficacy of NALIRIFOX remains robust despite the baseline imbalance in first-line treatment patterns.
While improved PFS did not translate to better OS in this study, the importance of early tumor control should not be underestimated. The extended time to progression observed with NALIRIFOX may offer a valuable window of disease stability, though this potential benefit must be carefully balanced against treatment-related toxicities. Another relevant consideration is that third-line chemotherapy may not be routinely offered following failure of second-line regimens in different healthcare settings due to diminishing returns, cumulative toxicity, and declining performance status. In these scenarios, optimizing the efficacy of second-line therapy becomes particularly crucial, as improved PFS with second-line chemotherapy may directly translate to enhanced clinical outcomes when subsequent therapeutic options are limited or unavailable.
Patients with pancreatic cancer typically experience declining performance status with each episode of disease progression, making the toxicity of second-line chemotherapy an important consideration when choosing treatment. The safety profile results in this study revealed that febrile neutropenia, neutropenia, and thrombocytopenia were significantly more common in the NALIRIFOX group. Peripheral neuropathy also occurred more frequently with NALIRIFOX, consistent with the known neurotoxicity profile of oxaliplatin. Given the potential for cumulative neurotoxicity in patients previously treated with gemcitabine plus nab-paclitaxel, the choice between NALIRIFOX and nal-IRI/FL should carefully account for the patient’s baseline neurological status. Management of emergent Grade 3–4 sensory neuropathy involves the addition of neuropathic agents, oxaliplatin dose reduction, or discontinuation to mitigate toxicity while preserving efficacy. In addition, recent randomized controlled evidence supports the use of cooling and compression as an effective strategy to mitigate the risk of severe chemotherapy-induced peripheral neuropathy. 16 Despite the increased risk of hematologic toxicities, most of the febrile neutropenia episodes were brief and well tolerated, suggesting that both regimens remain viable second-line options, although NALIRIFOX requires more vigilant monitoring for toxicities. This retrospective study provides important real-world data demonstrating that NALIRIFOX is well tolerated as second-line chemotherapy in patients with pancreatic cancer.
Conclusion
The results of this study demonstrated that NALIRIFOX provided superior PFS compared to nal-IRI/FL as second-line therapy in pancreatic adenocarcinoma. However, given the lack of OS benefit and the increased risk of adverse events such as neutropenia and peripheral neuropathy, NALIRIFOX should be considered an individualized option. The benefits of NALIRIFOX as second-line chemotherapy compared to nal-IRI/FL deserve further investigation in prospective studies.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261431386 – Supplemental material for NALIRIFOX versus liposomal irinotecan plus fluorouracil/leucovorin as the second-line chemotherapy in gemcitabine refractory pancreatic adenocarcinoma: a real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359261431386 for NALIRIFOX versus liposomal irinotecan plus fluorouracil/leucovorin as the second-line chemotherapy in gemcitabine refractory pancreatic adenocarcinoma: a real-world study by Wei-Ze Wong, Nai-Jung Chiang, Kuei-Chuan Lee, Hung-Yuan Yu, Chia-Jui Tsai, Shao-Jung Hsu, Pei-Chang Lee, Ming-Huang Chen, Hui-Chun Huang, Chung-Pin Li, Jiing-Chyuan Luo and Fah-Yauh Lee in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261431386 – Supplemental material for NALIRIFOX versus liposomal irinotecan plus fluorouracil/leucovorin as the second-line chemotherapy in gemcitabine refractory pancreatic adenocarcinoma: a real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359261431386 for NALIRIFOX versus liposomal irinotecan plus fluorouracil/leucovorin as the second-line chemotherapy in gemcitabine refractory pancreatic adenocarcinoma: a real-world study by Wei-Ze Wong, Nai-Jung Chiang, Kuei-Chuan Lee, Hung-Yuan Yu, Chia-Jui Tsai, Shao-Jung Hsu, Pei-Chang Lee, Ming-Huang Chen, Hui-Chun Huang, Chung-Pin Li, Jiing-Chyuan Luo and Fah-Yauh Lee in Therapeutic Advances in Medical Oncology
