Abstract
Background:
Primary central nervous system lymphoma (PCNSL) is a rare but aggressive non-Hodgkin lymphoma. Currently, high-dose methotrexate (HD-MTX)-based chemotherapy remains the standard first-line treatment. Previous clinical trials have reported that fotemustine-containing regimens offer promising efficacy and tolerability as an alternative option.
Objectives:
To compare the efficacy, safety, and feasibility of fotemustine-containing regimens with HD-MTX-containing regimens for newly diagnosed PCNSL.
Design:
A single-center, retrospective cohort study.
Methods:
We retrospectively analyzed 114 newly diagnosed PCNSL patients treated between April 2011 and December 2021. Patients were classified into two cohorts: those receiving fotemustine-containing regimens (n = 72) and those receiving HD-MTX-containing regimens (n = 42). The primary efficacy endpoint was the objective response rate (ORR). Secondary endpoints included complete response rate, progression-free survival (PFS), and overall survival (OS). Adverse events (AEs) were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0.
Results:
No significant difference in ORR was observed between the fotemustine-containing group and HD-MTX-containing group (68% vs 67%, p = 0.879). At a median follow-up of 28.5 months, the survival outcomes were comparable, with no statistically significant differences in either PFS (hazard ratio (HR) = 0.887, 95% confidence interval (CI): 0.522–1.508; p = 0.654) or OS (HR = 0.966, 95% CI: 0.494–1.888; p = 0.918). Notably, fotemustine-based therapy was associated with significantly fewer AEs, including leukopenia, thrombocytopenia, digestive tract toxicity, and mucositis (all p < 0.05).
Conclusion:
Fotemustine-containing chemotherapeutics appear to confer a safer profile with comparable efficacy relative to HD-MTX-based regimens in newly-diagnosed PCNSL patients.
Keywords
Introduction
Primary central nervous system lymphoma (PCNSL) is a rare but aggressive subtype of non-Hodgkin lymphoma that originates in the brain, eyes, leptomeninges, or spinal cord. 1 The majority of PCNSL patients are diagnosed with diffuse large B-cell lymphoma (DLBCL) of the non-germinal center-B-cell origin. 2 Currently, high-dose methotrexate (HD-MTX)-based chemotherapy followed by whole-brain radiotherapy is the most commonly used approach for patients with newly diagnosed PCNSL, extending median disease-free and overall survival (OS) to approximately 30–40 months compared with the prior 12–18 months.3,4 However, HD-MTX is a hospital-based drug requiring adequate fluid management and may be poorly tolerated in elderly patients with an increased prevalence of comorbid illness. 5 Therefore, there is a clear need to find more effective and safer innovative therapeutic strategies for newly diagnosed PCNSL.
Fotemustine is a third-generation nitrosourea, which is easily penetrated through the blood-brain barrier (BBB) due to its high lipophilicity and low molecular weight. It is indicated for the treatment of primary brain tumors and disseminated malignant melanoma, including those involving internal brain sites. 6 Our team has innovatively conducted prospective clinical trials using fotemustine-based regimens for newly diagnosed PCNSL patients. The results of the above studies7,8 suggest that the fotemustine-containing regimens are effective in treating newly diagnosed PCNSL with minimal toxic side effects. Therefore, to further validate these results, this study increased the sample size, extended the follow-up period, and included a control group to analyze the efficacy and safety of fotemustine-containing regimens compared with HD-MTX-containing regimens in newly diagnosed PCNSL patients.
Materials and methods
Study design
This single-center, retrospective cohort study was conducted and reported in compliance with the STROBE guidelines 9 (Supplemental File). The study protocol was approved by the ethics committee of the First Affiliated Hospital of Zhengzhou University (Approval No: 2022-KY-0869-001). The study complied with the Declaration of Helsinki. All patients or authorized family members were fully informed about the treatment and provided written informed consent before receiving treatment.
Patients
We retrospectively included all patients with newly diagnosed PCNSL at the First Affiliated Hospital of Zhengzhou University between April 2011 and December 2021 who met the following criteria: (1) diagnosis confirmed by histopathology of brain tissue and/or cerebrospinal fluid (CSF) flow cytometry; (2) first-line treatment with either a HD-MTX-containing or a fotemustine-containing regimen; and (3) availability of baseline clinical data and follow-up information. Exclusion criteria included: (1) secondary central nervous system (CNS) lymphoma; (2) prior chemotherapy or radiotherapy for lymphoma; (3) other active systemic malignancies; (4) severe comorbidities; or (5) insufficient clinical or survival data for outcome assessment. Follow-up was conducted through chart reviews, outpatient visits, and telephone visits, with the final follow-up completed in July 2022.
Treatment protocol
A total of 114 eligible patients were included in the final analysis. Forty-two patients were treated with HD-MTX-containing protocols, and 72 patients were treated with fotemustine-containing protocols. In the fotemustine-containing group, 15 patients were treated with FTD protocols (fotemustine + teniposide + dexamethasone), 12 patients were treated with FVD protocol (fotemustine + temozolomide + dexamethasone), and 45 patients were treated with RFPD protocol (rituximab + fotemustine + pemetrexed + dexamethasone). Twenty-seven patients were treated with the HD-MA protocol (HD-MTX + cytarabine), and 15 patients were treated with the R-MT protocol (rituximab + HD-MTX + temozolomide). The specific details of HD-MTX-containing protocols and fotemustine-containing protocols are shown in Table 1. Figure 1 illustrates the full study profile.
The fotemustine-containing regimens and HD-MTX-containing regimens.

FTD, fotemustine + teniposide + dexamethasone; FVD, fotemustine + temozolomide + dexamethasone; HD-MA, HD-MTX + cytarabine; HD-MTX, high-dose methotrexate; RFPD, rituximab + fotemustine + pemetrexed + dexamethasone; R-MT, rituximab + HD-MTX + temozolomide.

The profile of this study.
Assessment of response and adverse effects
The primary efficacy endpoints were objective response rate (ORR). Secondary endpoints included complete response rate (CRR), progression-free survival (PFS), and OS. The assessment of response was based on changes in the tumor size of enhanced lesions on MRI and CSF exams and was performed on the 2nd and 4th cycles, following the International PCNSL Collaborative Group Response Criteria. 10 Complete response (CR) was defined as the complete disappearance of all tumor lesions, partial response (PR) as a 50% or greater reduction in tumor volume, progressive disease as the emergence of a new lesion or a 25% or greater increase in tumor volume, and stable disease (SD) as situations that did not meet these criteria. In cases with concomitant positive CSF, cytology examination was performed after the 2nd and 4th cycles of chemotherapy and after treatment completion. PR was defined as a reduction of >50% in cell number, and SD was defined as a lower reduction. Adverse events (AEs) were graded according to National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE, version 5.0). 11
Statistical analysis
Descriptive statistics were used to summarize patient baseline characteristics. Continuous variables were expressed as mean ± standard deviation or median (interquartile range) based on their distribution, while categorical variables were summarized as frequencies and percentages. We analyzed clinical data, response rate, and adverse effects between therapy groups with the χ2 test and Mann–Whitney U test. Survival curves were estimated using the Kaplan–Meier method and compared with the log-rank test. Multivariable analysis was performed using the Cox proportional hazards regression model to adjust for pre-specified potential confounders, including age, sex, Eastern Cooperative Oncology Group Performance Score (ECOG PS), lactate dehydrogenase (LDH) level, CSF protein level, deep brain parenchyma involvement, number of lesions (single vs multiple), and International Extranodal Lymphoma Study Group (IELSG) risk score. A complete-case analysis approach was used for handling missing covariate data in the multivariable models. PFS was defined as the time from the diagnosis date to the first documented disease progression, death from any cause, or the last follow-up. OS was defined as the time from the diagnosis date to death from any cause or the last follow-up. All p-values were two-sided, and statistical significance was deemed at a level of p < 0.05. All statistical analyses were carried out with SPSS version 25 (SPSS, Inc., Chicago, IL, USA) and GraphPad Prism 8 (GraphPad Software Inc., La Jolla, CA, USA).
Results
Baseline characteristics of patients
Table 2 displayed the comparative clinical presentations of the fotemustine-containing group and the HD-MTX-containing group. The pathological type of 111 (97.4%) cases was DLBCL. One (0.9%) patient was diagnosed by CSF. The median age at first diagnosis of PCNSL was 55 (47–61) years, 59 (51.8%) patients were male, 30 (26.3%) patients were older than 60 years, and 98 (86.0%) patients had ECOG PS ⩾2. A total of 21 (25.6%) patients had a high IELSG risk score, 40 (35.1%) patients had elevated LDH, and 54 (65.9%) had elevated CSF protein. The patient characteristics were well balanced and did not differ significantly between the fotemustine-containing group and the HD-MTX-containing group.
Baseline patient characteristics.

CSF, cerebrospinal fluid; ECOG PS, Eastern Cooperative Oncology Group performance status; HD-MTX, high-dose methotrexate; IELSG, International Extranodal Lymphoma Study Group; LDH, lactate dehydrogenase.
Response and survival
A total of 114 patients were evaluable for response. After 4 cycles of chemotherapy, CR was achieved in 20 (28%) patients in the fotemustine-containing group (n = 72) and 10 (24%) patients in the HD-MTX-containing group (n = 42). PR was observed in 29 (40%) and 18 (43%) patients in the respective groups. The ORR (CR plus PR) was 68% in the fotemustine-containing group and 67% in the HD-MTX-containing group, with no statistically significant difference between the two groups (p = 0.879; Table 3). The median follow-up time was 28.5 months (range 2–122 months). Survival analysis revealed no significant difference in PFS between the two groups (hazard ratio (HR) = 0.887, 95% CI: 0.522–1.508, p = 0.654; Figure 2). The 3-year PFS rate was 40.9% for the HD-MTX-containing group and 46.7% for the fotemustine-containing group (p = 0.624), while the 5-year PFS rate was 26.3% versus 26.7% (p = 0.976), respectively. The median PFS was 24 months (95% CI: 9.310–38.690) in the HD-MTX-containing group and 36 months (95% CI: 29.535–42.465) in the fotemustine-containing group. No significant difference was found in OS between the treatment groups (HR = 0.966, 95% CI: 0.494–1.888, p = 0.918; Figure 2). The 3-year OS rate was 63.2% in the HD-MTX-containing group, and 61.8% in the fotemustine-containing group (p = 0.902), and the 5-year OS rate was 52.1% versus 61.8% (p = 0.434), respectively. The median OS was not reached in either group.
Response rates among groups.
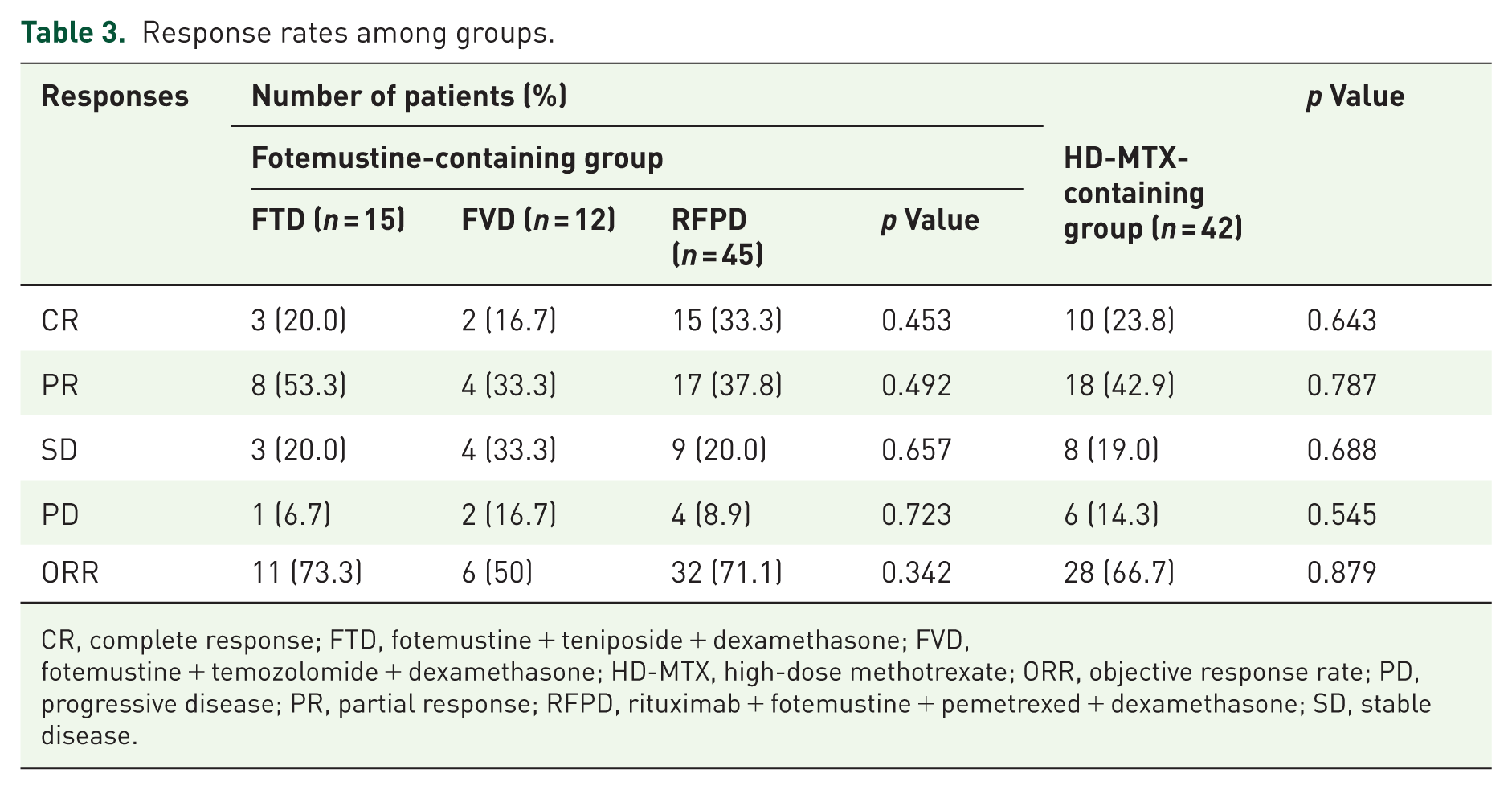
CR, complete response; FTD, fotemustine + teniposide + dexamethasone; FVD, fotemustine + temozolomide + dexamethasone; HD-MTX, high-dose methotrexate; ORR, objective response rate; PD, progressive disease; PR, partial response; RFPD, rituximab + fotemustine + pemetrexed + dexamethasone; SD, stable disease.

Progression-free survival curves (a) and overall survival curves (b) between the HD-MTX containing group and the fotemustine-containing group.
Among the patients receiving fotemustine-containing regimens, the ORR were 73% with FTD, 50% with FVD, and 71% with RFPD, with no statistically significant difference observed between the subgroups (p = 0.342; Table 3). In the subgroup survival analysis, neither PFS (p = 0.602) nor OS (p = 0.742) differed significantly across the regimens (Figure 3). Compared with the RFPD subgroup, the HRs for PFS were 0.640 (95% CI: 0.251–1.629) for FTD and 0.769 (95% CI: 0.313–1.888) for FVD; the corresponding HRs for OS were 1.272 (95% CI: 0.474–3.416) and 0.750 (95% CI: 0.210–2.681), respectively.

Progression-free survival curves (a) and overall survival curves (b) among RPFD, FTD, and FVD.
Multivariate Cox regression analysis
CSF protein levels were missing for 32 patients (28.1%). Therefore, the multivariate Cox regression, which included this variable as a covariate, was performed on the complete-case subset of 82 patients. After adjustment for potential confounders, no significant association was observed between the type of chemotherapy regimen (fotemustine-based vs HD-MTX-based) and patient outcomes, either in PFS (HR = 1.314, 95% CI: 0.647–2.671, p = 0.450) or OS (HR = 1.250, 95% CI: 0.534–2.929, p = 0.607; Table 4).
Multivariate Cox regression analysis.

The treatment groups consisted of the fotemustine-containing group and the HD-MTX-containing group.
Adj. HR (95% CI), adjusted hazard ratio (95% confidence interval); CSF, cerebrospinal fluid; ECOG PS, Eastern Cooperative Oncology Group performance status; HD-MTX, high-dose methotrexate; HR, hazard ratio; IELSG, International Extranodal Lymphoma Study Group; LDH, lactate dehydrogenase; OS, overall survival; PFS, progression-free survival.
Efficacy of fotemustine-containing regimens in elderly patients
Based on the established prognostic value of advanced age in PCNSL and its implications for treatment tolerance, we performed a subgroup analysis evaluating the efficacy and safety of fotemustine-containing regimens in patients aged >60 years. The ORR was 59.1% in patients >60 years, compared with 72.0% in those ⩽60 years (p = 0.279). In survival analysis, no significant difference was observed between the two age subgroups in terms of PFS (p = 0.296) or OS (p = 0.616). Furthermore, the incidence of AEs did not differ significantly between older and younger patients (all p > 0.05; Table 5).
Incidence of AEs stratified by age.

AE, adverse events.
Toxicity
The AEs observed in all eligible patients are shown in Table 6. The primary AEs in both groups were hematologic toxicities, including leukopenia, anemia, and thrombocytopenia. The most common non-hematologic toxicities were hepatic dysfunction, infection, and electrolyte imbalance, with most cases being relieved after active symptomatic treatment. Compared with the fotemustine-containing group, the HD-MTX-containing group showed more serious leukopenia (p = 0.009), thrombocytopenia (p = 0.022), digestive tract toxicity (p = 0.004), and mucositis (p = 0.048).
AEs between fotemustine-containing group and the HD-MTX-containing group.

AE, adverse events; HD-MTX, high-dose methotrexate.
Discussion
The treatment of PCNSL has been a significant challenge for decades, with HD-MTX-based polychemotherapy playing a critical role in induction therapy for PCNSL. 12 This study is the largest retrospective study to date comparing the efficacy and safety of HD-MTX-containing regimens versus non-HD-MTX-containing regimens in newly diagnosed PCNSL patients. The results suggest that fotemustine-containing regimens offer comparable efficacy with lower toxicity compared to HD-MTX-containing regimens.
HD-MTX has several limitations in its application. First, treatment-related mortality in HD-MTX-containing groups has ranged from 5% to 11% in previous trials.13,14 Severe hematological toxicity, mucositis, hypersensitivity, and decreased renal function are common side effects in patients receiving HD-MTX. 13 Additionally, blood concentration monitoring is essential after administration of MTX; however, such monitoring is not feasible during partial hospital stays, which limits the drug’s usage. Importantly, HD-MTX requires adequate hydration, urinary alkalinization, and folinic acid rescue both before and after administration. However, there is no consensus on the appropriate dose of folinic acid or the timing of rescue, which affects the consistency of evaluations of HD-MTX-containing regimens.
Fotemustine is characterized by elevated lipophilicity and low molecular weight, which facilitate its efficient penetration through the BBB. 15 Additionally, it exhibits significant antitumor activity and has been used to treat CNS tumors. 16 Clinical trials have shown that fotemustine is an effective and well-tolerated treatment for newly diagnosed PCNSL patients.7,8 Compared to methotrexate, pemetrexed shares a similar ability to penetrate the CNS but offers the advantage of targeting multiple points in folate metabolism. 17 Moreover, pemetrexed is easier to produce and has a lower toxicity profile. A study demonstrated that single-agent pemetrexed is feasible, effective, and well tolerated in elderly patients with PCNSL. 18 Teniposide, a topoisomerase inhibitor, has been shown to penetrate both normal brain tissue and brain tumors. Our previous studies have shown the efficacy and safety of teniposide in the treatment of PCNSL.7,19 Notably, the HD-MA regimen does not contain dexamethasone. In addition to its direct antitumor effects, which are mediated through anti-inflammation, anti-angiogenesis, and regulation of androgens, dexamethasone plays a crucial supportive role in PCNSL therapy. When combined with chemotherapy, it not only mitigates treatment-related toxicities but may also act as a sensitizer, enhancing chemotherapeutic efficacy. 20 Existing studies have shown that glucocorticoids, by activating their receptors, can induce cell cycle arrest and apoptosis in lymphoid malignancy cells, providing theoretical support for their potential as chemosensitizers. 21
Currently, there are limited studies comparing the clinical efficacy and safety of fotemustine-containing regimens versus HD-MTX-containing regimens in patients with newly diagnosed PCNSL. In our previously published prospective, randomized clinical trial, we compared the efficacy and safety of FTD and HD-MA in 49 patients with newly diagnosed PCNSL. 7 The ORR (88% vs 84%, p = 0.628), 2-year PFS (37% vs 39%, p = 0.984), and 3-year OS (51% vs 46%, p = 0.509) did not differ significantly between the FTD and HD-MA groups. However, the HD-MA group exhibited more severe neutropenia (p = 0.009) than the FTD group. Our current study further expanded the sample size to include 114 patients with untreated PCNSL and extended the follow-up period. Consistent with prior findings, fotemustine-based regimens were associated with a more favorable safety profile, including significantly lower rates of hematologic (leukopenia, thrombocytopenia) and non-hematologic toxicities (gastrointestinal toxicity, mucositis) relative to HD-MTX-based regimens. Fotemustine demonstrates not only satisfactory BBB penetration but also exhibits stable pharmacokinetic properties, maintaining a favorable clearance rate. 22 Dexamethasone rapidly alleviates PCNSL-related brain edema and neurological symptoms, and reduces chemotherapy-induced mucosal toxicity and gastrointestinal reactions, thereby improving patient treatment adherence.23,24 As expected, no significant differences were observed between the two groups in terms of ORR, median PFS, or median OS. Additionally, the 68% ORR in the fotemustine-containing group was generally consistent with previous studies.7,8 In clinical practice, patients with poorer performance status were more likely to be administered the fotemustine-containing regimen. Nevertheless, the fotemustine cohort still exhibited survival outcomes comparable to those of the HD-MTX-based group.
In the fotemustine-containing group, we compared the efficacy of three combination regimens (FTD, FVD, and RFPD). Although the differences between the groups did not reach statistical significance, the RFPD regimen, which included rituximab, showed the highest CRR. Rituximab targets and eliminates B-cell tumors through mechanisms such as antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity. 25 While the therapeutic value of rituximab in PCNSL remains a subject of debate, 26 the existing overall evidence continues to support its role as an important component of combination therapy strategies. It is worth noting that in this study, the ORR in the RFPD group was lower than in the FTD group. This discrepancy may be attributed to the sample size imbalance between the subgroups, as the smaller sample size in the FTD group could have led to greater variability in the response rate.
According to a study of 579 patients diagnosed with PCNSL, age was inversely associated with the chance of receiving treatment with MTX.16,27 Elderly patients clinically exhibit lower response rates and a higher incidence of toxicities than in younger patients. 28 Age >60 years is a well-established adverse prognostic factor for PCNSL. 29 The age-stratified analysis showed no statistically significant differences in efficacy or the incidence of AEs between different age groups in the fotemustine group, with 60 years used as the cutoff. This suggests that fotemustine’s favorable safety profile is maintained in the elderly population. By reducing treatment-related toxicity associated with aging, fotemustine may support higher treatment completion rates and better maintenance of quality of life, positioning it as a promising and better-tolerated therapeutic alternative for elderly or frail PCNSL patients.
Our findings suggest that fotemustine may be a potential alternative to HD-MTX. It should be acknowledged that current HD-MTX-based first-line regimens, such as R-MPV (rituximab, methotrexate, procarbazine, and vincristine) and MATRix (methotrexate, cytarabine, thiotepa, and rituximab), have demonstrated superior efficacy in multiple high-level clinical studies.13,30 However, the toxicity associated with intensive chemotherapy may limit its use in vulnerable populations. For example, treatment‑related mortality with R‑MPV has been reported at up to 6%, alongside a high incidence of treatment discontinuation due to severe AEs. 30 Consequently, future research should focus on prospective trials comparing fotemustine-based regimens with contemporary standard therapies, particularly in elderly patients, those with complex comorbidities, or those who are intolerant to intensive chemotherapy. Such studies are essential to inform precise, stratified treatment strategies for PCNSL.
Several limitations of this study merit discussion. First, its retrospective, single-center design may introduce selection bias and could limit the generalizability of the findings. Second, due to the retrospective nature of the study, the lack of systematically collected patient‑reported quality of life data and standardized neurological assessments restricts a more comprehensive evaluation of patient‑centered outcomes.
Conclusion
Our study suggests that fotemustine-containing chemotherapy is a promising alternative for newly diagnosed PCNSL, particularly for the elderly or those intolerant to HD-MTX-based therapy. Future prospective studies comparing it with contemporary standard regimens are warranted to confirm these findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261426830 – Supplemental material for Fotemustine-containing chemotherapy suggests lower toxicity with comparable efficacy versus high-dose methotrexate-based regimens: a retrospective cohort analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359261426830 for Fotemustine-containing chemotherapy suggests lower toxicity with comparable efficacy versus high-dose methotrexate-based regimens: a retrospective cohort analysis by Xudong Zhang, Wanyue Zhao, Wenhua Wang, Jingjing Wu, Mengke Fan, Shanshan Ma, Meng Dong, Linan Zhu, Xiaolong Wu, Xiaoyan Feng, Xin Li, Ling Li, Zhenchang Sun, Xinhua Wang, Xiaorui Fu, Zhaoming Li, Jiaqin Yan, Yu Chang, Hui Yu, Feifei Nan, Zhiyuan Zhou, Mengjie Ding, Jieming Zhang, Lei Zhang and Mingzhi Zhang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to express our sincerest appreciation to all study participants. Preliminary findings of this study were previously presented in abstract form at the 2024 American Society of Hematology (ASH) Annual Meeting.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
