Abstract
Endometrial cancer (EC) is a gynecologic neoplasm with a constantly increasing incidence, especially in high-income countries. Obesity, diabetes, old age, and genetic predisposition account for the main risk factors. Genetic knowledge has steadily increased from the historical clinicopathological classification into two pathogenic types to the molecular subdivision into four groups, highlighting that EC is not a single entity, but rather the sum of different molecular diseases with different prognoses. About 5%–10% of ECs show a hereditary basis attributable to germline pathogenic variants (PVs) in different susceptibility genes, including MMR (MLH1, MSH2, MSH6, and PMS2), POLE/POLD1, PTEN, BRCA1/2, MUTYH, NTHL1, STK11, which confer an increased risk of developing an early onset EC. Lynch syndrome is the main inherited disorder predisposing to EC, followed by other hereditary conditions, including Cowden syndrome, polymerase proof-reading associated polyposis, NTHL1-associated syndrome, hereditary breast and ovarian cancer syndrome, MUTYH-associated polyposis, and Peutz-Jeghers syndrome. Genetics has been shown to affect several aspects of disease, including carcinogenesis, onset age, clinicopathological features, prognosis, and therapy response. In this review, we will investigate the impact of germline PVs in different genes on genetic susceptibility to the development of inherited EC, discussing the potential cancer risk in mutation carriers as well as prognostic implications and current therapeutic approaches, also evaluating the possibility of carrying out a more extensive routine genetic analysis for EC women, in order to increase the diagnostic power, improve prevention and surveillance strategies in genetically predisposed subjects, and implement tailored therapies.
Keywords
Introduction
Endometrial cancer (EC) is a common gynecological cancer worldwide, especially in high-income countries, with a constantly increasing incidence. About 90% of ECs are diagnosed at an early stage International Federation of Gynaecology and Obstetrics (FIGO I–II), with a 5-year survival rate of 95% in localized disease, which drops to 18% in advanced disease (FIGO III–IV). 1 EC was initially classified into two major pathogenetic forms. Type I ECs are more common, obesity- and estrogen-related, generally low-grade (FIGO G1–2) with superficial myometrium invasion, and show a good prognosis. Type II ECs are non estrogen-related, generally high-grade (FIGO G3) with deep myometrium invasion or metastatic spread, and show a worse prognosis. 2 Histopathological subtypes partially overlap with the above two pathogenic types of EC: endometrioid ECs (EECs) are considered prototypical type I tumors, whereas non-EECs (NEECs), mainly serous carcinomas, are considered prototypical type II tumors. EECs account for about 85% of newly diagnosed cases, serous carcinomas for about 3%–10%, clear cell carcinomas for <5%, and uterine carcinosarcomas are rare. 3
In 2013, a new molecular classification integrated previous histopathological findings with data from the combination of somatic mutational burden and somatic copy number alterations, allowing patients to be stratified with important implications for clinical decisions. Therefore, ECs have been classified into four groups: (1) DNA Polymerase Epsilon (POLE)-ultramutated; (2) microsatellite instability hypermutated (MSI-H); (3) tumors at low somatic copy number alteration (SCNA); (4) tumors at high SCNA. The first group was associated with EEC histology and good prognosis; the second and third groups were associated with EEC subtype at intermediate prognosis, whereas the fourth group was mainly composed of high-grade EECs and NEECs and was associated with the worst prognosis. 4
Obesity and prolonged exposure to estrogens have been shown to be epidemiologically important risk factors in the onset of EC. In the gynecological cancer spectrum, and more specifically in the context of uterine cancer, approximately 90%–95% of tumors are considered sporadic, whereas 5%–10% of ECs have a hereditary nature 5 (Figure 1). Inherited conditions predisposing to EC predominantly include not only the Lynch and Cowden syndromes but also the polymerase proof-reading associated polyposis (PPAP), NTHL1-associated syndrome, Hereditary Breast and Ovarian Cancer (HBOC) syndrome, MUTYH-associated polyposis (MAP), and Peutz-Jeghers syndrome (PJS)6–8 (Figure 2). Therefore, multiple genes can contribute to the increased risk of developing an inherited EC, determining a genetic overlap for the same tumor phenotype. However, assessing the contribution of single genes to the development of the disease sometimes can be difficult, creating issues in differential genetic diagnosis of EC-associated hereditary syndromes. 9

Sporadic and hereditary endometrial cancers.

Hereditary cancer syndromes and related genes predisposing to endometrial cancer.
In this review, we will investigate the impact of germline pathogenic variants (PVs) present in different genes on genetic susceptibility to the development of inherited EC, discussing the potential cancer risk in mutation carriers as well as prognostic implications and current therapeutic approaches, also evaluating the possibility of performing a more extensive routine genetic analysis for EC patients, in order to increase the diagnostic power and improve prevention and surveillance strategies in genetically predisposed subjects.
Methods: Literature search
The literature search for writing this manuscript was performed using PubMed as the major search engine and database, but also Embase, and the current guidelines, without applying no formal risk-of-bias tool. The search on PubMed was carried out by selecting and including only original, meta-analyses, and review articles regarding humans, published in English from February 01, 1983, to January 31, 2025, using the following keywords: “endometrial cancer” [MeSH Terms] OR “endometrial cancer” [All Fields] OR “hereditary” [All Fields]) AND “endometrial cancer” [MeSH Terms] OR “hereditary endometrial cancer” [All fields] OR “Lynch syndrome” [All fields] OR “Cowden syndrome” [All fields] OR “Peutz-Jeghers syndrome” [All fields] OR “POLE” [MeSH Terms] AND “POLD1” [MeSH Terms] AND “endometrial cancer” [All fields] OR “BRCA” [MeSH Terms] AND “endometrial cancer” [All fields] OR “MUTYH” [MeSH Terms] AND “endometrial cancer” [All fields] OR “NTHL1” [MeSH Terms] AND “endometrial cancer” [All fields]. Further articles not found by this search were searched manually among the references of relevant articles and reviews. Among all the articles found by this search, we preferentially selected those most updated regarding the genetic susceptibility to inherited EC, potential cancer risk in mutation carriers, prognostic and therapeutic implications.
Classification of endometrial cancer
All ECs should be tested for the expression of mismatch repair (MMR) proteins (MLH1, PMS2, MSH2, MSH6) and p53 protein by immunohistochemistry (IHC), for tumour MLH1 promoter hypermethylation status (in case of loss of MLH1 expression), and for POLE PVs, as a surrogate classification of the four The Cancer Genome Atlas (TCGA) groups. 10
Thus, based on the TransPORTEC international consortium and Proactive Molecular risk Classifier for Endometrial Cancer (ProMisE) classification system, four molecular groups of EC were redefined: (1) POLEmut group, which corresponds to the POLE-ultramutated group of TCGA, accounting for 6%–9% of cases and showing excellent prognosis; (2) non-specific molecular profile (NSMP), which corresponds broadly to the low SCNA group of TCGA, representing 45%–59% of cases and showing intermediate prognosis; (3) MMR deficient (MMRd) group, which corresponds to the MSI-H group of TCGA, accounting for 13%–30% of cases and showing intermediate-severe prognosis; (4) p53abn group, which corresponds to the high-SCNA group of TCGA, representing 9%–18% of cases and showing the worst prognosis. 11
The molecular classification of EC shows concordance between pre- and post-operative biopsies, thus facilitating pre-operative therapeutic planning and helping in assessing prognosis, as the four groups are found across all histologic types, grades, and stages of tumor.4,12–14
Only 2%–3% of ECs harbour more than one molecular signature,11,15 although MMRd and p53abn and POLEmut/p53abn mixed groups can be simply classified as MMRd and POLEmutant, respectively, because of their similar better prognosis, when compared to the single p53abn group. 15 In the recent guidelines regarding EC, ESGO/ESTRO/ESP joint scientific societies defined five risk categories by integrating the molecular classification with the tumour stage and grade, histological type, myometrial invasion, and lymphovascular space invasion (LVSI). 10
However, criticisms of the molecular classification exist. A report showed that histotype might still play a prognostic role independently of the TCGA-defined molecular groups, as NEECs are associated with worse prognosis. 16 In another work, uterine carcinosarcoma (UCS), consisting of an epithelial and a stromal component, showed an excellent prognosis in the POLEmut group, but a worse clinical outcome in the p53abn and NSMP groups compared to p53abn endometrioid or serous EC groups. 17
Endometrial carcinogenesis
The current concept of endometrial carcinogenesis integrates epidemiological, histopathological, genetic, and immunological evidences.
Epidemiological evidences
Risk factors associated with increased likelihood of developing EC include obesity, diabetes with hyperinsulinaemia, prolonged exposure to estrogens often related to nulliparity and infertility associated with metabolic syndrome or tamoxifen use, prior breast or ovarian cancer, and old age. 18
Fat-secreted cytokines in obesity, known as adipokines, are involved in carcinogenesis and include adiponectin, leptin, visfatin, resistin, apelin, chemerin, omentin, and vaspin. Among these, adiponectin acts as an anti-inflammatory adipokine, which reduces the risk of EC, being decreased in the obese state. The AMPK/mTOR pathway mediates adiponectin activity in EC. Leptin, another adipokine, is expressed in EC tissue, at the mRNA and protein level, and is involved in tumorigenesis via the JAK/STAT and MAPK/ERK pathways. 19 Hyperinsulinemia, commonly observed prior to diabetes onset, may have a causal association with EC through a direct mitogenic effect, by increasing estrogen levels through a reduction of sex hormone-binding globulin (SHBG). 20
Age and menopause determine an increased adiposity and consequent increase in circulating levels of bioactive estrogens, which stimulate endometrial proliferation through the PI3K/AKT/mTOR pathway. 19 Gestation reduces EC risk, probably due to the greater progesterone production during pregnancy with protective effects on the endometrium. 20
Notably, genetics can influence epidemiological risk factors. In fact, some body mass index (BMI)-associated single-nucleotide polymorphisms (SNPs) may increase EC risk via mechanisms other than measurable BMI. 21 Genetically predicted Vitamin C levels and intake of macronutrients (sugar and fat) have been shown to affect EC risk. 22 Ethnicities are also associated with different mutation frequency profiles and consequent different prognosis, regardless of socioeconomic status. In fact, the PIK3CA and ARID1A mutation frequencies associated with good prognosis are higher among White patients, whereas TP53 mutation frequencies associated with poor prognosis are higher among Black/African American individuals. 23 Finally, an inverse association between the risk of “hereditary” EC and late age of menarche, pregnancy, and use of hormonal contraceptives has been detected in Lynch syndrome, as also observed in the general population for “sporadic” EC risk. 24 This observation suggests that genetics might represent a common basis for both hereditary and sporadic EC risk, more than is believed.
Histopathological and genetic evidences
The ability to stratify ECs into various subgroups based on molecular characterization led to fundamental changes in clinical practice and treatment, with important implications for the anticancer immune response. Therefore, this type of analysis can also positively influence therapeutic decisions. 25
In the type I EEC, the precursor lesion is considered a complex atypical hyperplasia. Frequent mutations in PTEN and KRAS genes, alterations of beta-catenin, and microsatellite instability are considered early events, because they occur in a subset of complex atypical hyperplasia, whereas rare TP53 mutations are considered late events. 3
In the type II NEECs, the precursor lesion occurs in a background of endometrial atrophy. Frequent TP53 mutations with the role of early driver are followed by somatic alterations in PPP2R1A, FBXW7, SPOP, CHD4, and TAF1, amplification and/or overexpression of HER2, MYC, and CCNE1 (cyclin-E), and overexpression of p16 and synuclein-y. 3
POLE-ultramutated ECs are characterized by high tumor mutational burden (TMB) (>100 mut/Mb), frequent mutations in PTEN, PIK3R1, PIK3CA, and KRAS, and microsatellite stability (MSS). MSI-H ECs show high TMB (10–100 mut/Mb), frequent mutations in the MMR genes (MLH1, MSH2, MSH6, PMS2), alterations in PTEN and KRAS genes, as well as changes in the PTEN-PIK3CA pathway. Low copy number ECs are characterized by low TMB, frequent mutations in PTEN and PIK3CA, alterations in the WNT-beta-catenin pathway, and MSS. High copy-number ECs show low TMB, frequent mutations in TP53 and PIK3CA, but also in FBXW7 and PPP2R1A, which are unique for these tumors, and ERBB2 amplification. 4
Immunological evidence
Regardless of molecular classification of EC, the number of tumor-infiltrating lymphocytes (TILs) CD8+ and CD8+/FOX3+ regulatory T cells (TREGs) ratio correlates with better survival. 26 However, some immunological features are more frequently observed in specific molecular groups. In fact, MSI-H and POLEmut EC groups harbour high TMB and accumulate immunogenic neoantigens, resulting in increased amount of CD8+ cytotoxic T cells and compensatory upregulation of immune suppressive checkpoints PD-1 and PD-L1, 4 which may predict response to therapy with immune checkpoint inhibitors (ICIs). 27
In particular, genetics clearly influences immunological characteristics in POLEmut group, as activated CD8+ T cells overexpress genes involved in their cytotoxic functions (T-bet, Eomes, interferon-gamma, perforin) and exhaustion (LAG-3, Tim-3, TIGIT). 26
Lymphocyte activation gene-3 (LAG-3) and indoleamine 2,3-dioxygenase 1 (IDO1), mostly expressed in POLEmut and MMRd/MSI-H ECs, are immunosuppressive molecules involved in immune escape of tumors. 26 IDO1 is often co-expressed with PD-L1 in EC cells, especially in MMRd ECs and Lynch syndrome, and is associated with worse prognosis.26,28
Since the combined anti-LAG-3/anti-PD1 treatment has been shown to enhance antitumor response in melanoma, where mutation frequencies in POLE and MMRare increased when neoantigen production is higher, 29 similar trials are ongoing also for EC. 26
EC in hereditary cancer syndromes
Hereditary cancer syndromes (HCSs) are inherited diseases caused by germline PVs, which are associated with a high risk of developing specific malignancies at an early age. As these PVs promote carcinogenesis, their identification underlies personalized systemic target therapy. 30
ECs account for 5%–10% of HCSs, with Lynch Syndrome (LS) being responsible for the majority of these cases18,31 (Figure 1). Increased lifetime risk of EC has also been observed in subjects affected by other HCSs, including the POLE/POLD1-associated, 32 HBOC,33,34 and Cowden syndromes 35 (Table 1). Early age of tumor onset, presence of multiple affected family members, and multiple primary tumors (especially in the same organ) have long been recognized as main features of hereditary cancer predisposition, regardless of family history. 6
Lifetime risk of endometrial cancer associated with different hereditary syndromes.

Estimated lifetime risk range up to 70–80 years.
The molecular classification of EC is important for diagnosis, as it may suggest the presence of any HCSs, with important implications for the surveillance of patients and genetic counselling of the family members.
Lynch syndrome
LS is an autosomal dominant HCS, with a medium-high degree of penetrance, caused by germline PVs in MMR genes, such as MLH1, MSH2, MSH6, and PMS2, or in EPCAM. MMR system deficiency produces microsatellite instability (MSI) and increases tumor susceptibility. EC is the most common extraintestinal cancer in LS, representing a “sentinel” tumor for this syndrome. Indeed, beyond colorectal cancer (CRC), LS confers an increased risk of developing cancers also in other extraintestinal sites. 37 Approximately 2% to 6% of all ECs may be caused by germline PVs in MMR genes. 38
In a meta-analysis, an LS prevalence of 3% was observed among unselected ECs, wich increase up to 29% among MMRd or MSI ECs with negative MLH1 methylation test. 39 A further prospective study from the United Kingdom showed that LS prevalence among ECs was 3.2% but reached 12% among MMRd ECs. Interestingly, MMR analysis by IHC and MLH1-methylation testing were more sensitive for LS screening than MSI with MLH1-methylation testing. 40 In subjects with LS, the lifetime cumulative risk of EC is much higher than that found in the general population (Table 1). Moreover, MMR genes influence the organ where cancer develops, also showing different penetrance within the same organ. A prospective study showed that LS carriers develop mostly CRC and EC. Cancers were detected starting from age 25 years onwards in MLH1 and MSH2 mutation carriers, whereas from age 40 years in MSH6 and PMS2 mutation carriers. The cumulative incidence of EC in germline heterozygotes by 70 years was higher in MLH1, MSH2 and MSH6 mutation carriers than in PMS2 PV carriers. In general, the lifetime risk of LS-associated EC in carriers of germline MMR alterations is estimated to extend from 20% to 60%. The screening strategies for EC include annually gynaecological examination, transvaginal ultrasound with eventual endometrial biopsy from age 35 years. Prophylactic hysterectomy with bilateral oophorectomy is an option that might be discussed and considered in mutation carriers who have completed child- bearing or are postmenopausal. 41 Among LS patients surviving after their first cancer, EC was the most common cancer observed after CRC. Another prospective study confirmed the high cumulative incidence of EC by age 75 years for MLH1, MSH2 and MSH6 PV carriers. 42
The interaction between PVs in MMR and non-MMR genes can also cause the onset of EC. The 3′end deletion of the EPCAM gene, which is a non-MMR gene located before MSH2, is associated with Lynch syndrome. 43 EPCAM 3′end deletion can extend to the 5′end initial sequences of MSH2, which include the promoter region, resulting in the suppression of both EPCAM and MSH2 proteins.43,44 Notably, EC was observed only in carriers harbouring EPCAM deletions close to MSH2. The cumulative risk of developing EC at age 70 years in EPCAM deletion carriers was lower than that observed in MSH2 mutation carriers or in carriers showing MSH2-EPCAM combined deletion. 43 Moreover, the typology of MMR PVs may influence the probability of developing EC. In fact, MSH2 deletion carriers showed a 2.5 times higher chance of developing EC when compared to carriers of point mutations. 44
MMR PVs seem to influence the age of EC onset, as LS-related EC (LS-EC) patients are younger than those with sporadic EC. 31 Moreover, among LS-EC patients, MLH1 and MSH2 PV carriers are younger than MSH6 mutation carriers, and carriers of truncating MLH1 mutations show a later age of EC onset than those harbouring nontruncating mutations. 45
MMR genes can influence histopathological features of EC and site of disease occurrence. Endometrioid hystotype, FIGO early stage, and low grade account for the most frequent features of LS-ECs. Moreover, LS-associated ECs show a higher incidence of clear cell tumors compared to sporadic ECs, involve the inner half of the endometrium, and are more commonly localized in the uterine fundus and the lower uterine segment (LUS). 31 This latter finding may be relevant for prognosis, as LUS involvement has been shown to be associated with a higher probability of retroperitoneal lymph node metastases at surgery, even among patients with preoperative “apparent” stage IA. 46
A single MMR gene may influence sex- and age-related cancer susceptibility. In fact, among MSH6 PV carriers, the increased risk of cancers differed by sex and continued into older age, with hazard ratios higher for EC (26-fold) than any other LS-related cancer (6-fold), when compared with the general population. In particular, a study showed that the risks of EC and CRC at age 70 and 80 years were 26% and 44%, and 10% and 20%, respectively. 47
Finally, MMR PVs influence EC behaviour in the germline setting differently from the somatic. In fact, patients with LS-associated EC showed better survival, but also a higher risk, of a second LS-associated cancer, compared to patients with MMRd-EC without LS. 48
The knowledge coming from studies about LS showed that EC can be hereditary, and the MMR and EPCAM genes have different penetrance and are associated with different onset age and clinicopathological features, suggesting that EC occurrence is gene-specific. Germline MSH6 PV carriers also showed a sex-specific cancer risk. 9
POLE/POLD1-associated syndrome
The POLE and POLD1 genes code for DNA polymerases epsilon and delta, involved in DNA replication and repair, and are recognized as hereditary cancer predisposing genes. 49 Germline alterations involving the exonuclease domain (ED) determine the POLE/POLD1-associated syndrome, an autosomal dominant HCS, which was initially associated with CRC onset, and subsequently with EC, breast cancer (BC) and, probably, ovarian cancer (OC) and brain tumors. Pathogenicity adjudication should follow the hotspot ED variants and mutational signature, when available. 7 One study showed that EC risk was increased in heterozygotes of germline POLD1 and POLE ED PVs, although no EC developed before age 45 years. The limit of these risk estimations is that they rely on small numbers of carriers (27 POLD1-mutated and 43 POLE-mutated) from different studies, but, importantly, the authors used stringent classification criteria for PVs. The screening strategies for germline POLE PV carriers should begin at the age 30–35 years, considering ultrasound and endometrial biopsy every 1–2 years. 32
HBOC syndrome
Germline PVs in BRCA1 or BRCA2 are the most frequent cause of HBOC syndrome, which is an autosomal dominant HCS conferring an increased risk of developing BC and/or OC and other malignancies, probably including EC. 50
BRCA1/2 alterations are associated with homologous recombination deficiency (HRD), which is involved in the impairment of repair of DNA double-strand breaks (DSBs) and, then, in carcinogenesis. In a study including 52,426 tumors, the highest frequency of HRD was achieved in 5540 ECs, with the most frequent mutations involving the ARID1A, ATM, ATRX, and BRCA2. 51
In a report, among 21 EC patients belonging to HBOC families, 9 (42.8%) showed germline PVs in BRCA1 or BRCA2. 50 In another study, ECs occurring in germline BRCA1/2 PV carriers with loss of heterozygosity of the wild-type allele were associated with distinct clinicopathologic and molecular features, such as high-grade and nonendometrioid histology and TP53 mutations, suggesting that EC might be part of the HBOC syndrome. 52 Similarly, among germline BRCA1/2 PV carriers with HBOC syndrome, typical molecular characteristics of EC, such as high-SCNA and frequent TP53 mutations, have been observed. 53 Another study assessed EC risk in germline BRCA1/2 PV carriers, showing an increased standardized incidence ratio (3.51) for EC in BRCA1 PV carriers, when compared with the general population, which became higher for serous-like EC (12.64). This increase in incidence ratio for EC was also confirmed in BRCA2 germline PV carriers (1.70 versus 5.11). However, evidence is mixed, because the absolute risk of EC has been shown to be low (~3% by 75 years), but relative risk is higher for BRCA1 mutation carriers, particularly in women affected by serous-like EC (HR: 10.48; 95% CI: 2.95–37.20 for BRCA1 versus HR: 4.13; 95% CI: 0.83–20.50 for BRCA2). Specifically, BRCA1 mutations appeared to be associated with a higher EC risk compared to BRCA2 alterations, regardless of previous tamoxifen use. 33 Two studies confirmed an increased risk only for serous-like EC in BRCA1 PV carriers, after risk-reducing salpingo-oophorectomy.54,55 However, other studies showed no association between germline BRCA1/2 PVs and EC in Jewish and Australian women.56,57 Different ethnic/geographical distribution of BRCA PVs, small sample size, short follow-up or previous use of tamoxifen, which increases EC risk, might explain these conflicting results.
Cowden syndrome
Cowden syndrome (CS) is an autosomal dominant HCS mainly associated with an increased risk of BC, EC, thyroid and kidney cancers, and other clinical signs. CS is caused by mutations in the PTEN tumor suppressor gene, which determines chromosomal instability and cell proliferation, resulting in hamartomas and several malignancies. 58 A prospective study showed that carriers of germline PTEN PVs have an elevated age-adjusted standardized incidence ratio (SIR) for EC (42.9), with an estimated lifetime risk of 28.2%, which starts at 30–40 years of age (Table 1). According to the 2024 NCCN guidelines, the screening strategies for EC should begin starting from age 35 years, considering an endometrial biopsy every 1–2 years and discussing the hysterectomy option upon completion of childbearing and reproductive desires. 35
Additionally, PVs in other genes may have a role in CS or CS-like. Subjects referred to as having CS-like are those who met at least the criteria of the International Cowden Consortium for CS minus one criterion. A fraction of individuals with CS or CS-like and no PTEN PVs were found to have germline mutations in succinate dehydrogenase (SDH)B/C/D gene and KLLN promoter methylation. In a prospective study involving 371 EC patients, germline PTEN alterations, SDHx PVs and KLLN methylation were found in 7%, 9.8% and 10.5% of samples, respectively. 59
MUTYH-associated tumor syndrome
MUTYH-associated tumor syndrome is an autosomal recessive HCS, caused by germline biallelic alterations in MUTYH (MutY Homolog Escherichia Coli), involved in the base excision repair (BER) mechanism of endogenous DNA. Carriers of biallelic MUTYH PVs have a high lifetime risk of developing colorectal polyposis and CRC. 60 Also, other tumor types, including duodenum, breast, ovary, pancreas, bladder, and skin cancers, may be involved in this syndrome, although cancer risk evidence becomes less clear when extraintestinal tumors and monoallelic PV carriers are considered. 8 The role of germinal MUTYH PVs in predisposition to EC is controversial. In fact, the EC risk has been shown to be increased in a study involving 266 probands with MUTYH PVs and 1903 first- and 3255 second-degree relatives, 61 although another report did not find any association between heterozygosity for MUTYH PVs and EC in individuals of European ancestry. 62
Peutz-Jeghers syndrome
Peutz-Jeghers syndrome determines the development of hamartomatous polyps in the gastrointestinal level and is also characterized by pigmentation of the perioral mucosa. The STK11/LKB1 tumor suppressor gene is considered responsible for this inherited condition. The modality of inheritance is autosomal dominant. In this syndrome, the risk of gastrointestinal cancer is high, but other organs could also be involved, causing breast, ovarian, uterine cervix, and endometrial cancers. 63 The lifetime risk of uterine cancer is estimated to be approximately 9%, with an average age at diagnosis of 43 years (Table 1). According to the 2024 NCCN guidelines, surveillance is recommended from age 18–20 years by pelvic examination annually, reserving the execution of a biopsy only in case of symptoms. 64
NTHL1-associated syndrome
The NTHL1-related autosomal recessive syndrome, caused by germline biallelic NTHL1 PVs, is mainly associated with colorectal polyposis and CRC, but may predispose to a broader spectrum of extracolonic neoplasms, including breast, endometrium, skin, brain, and bladder cancers. 7 Although there is little data about the NTHL1-related genetic susceptibility to EC, some authors suggested that screening for EC in biallelic NTHL1 PV carriers should start at the age 40 years, every 1–2 years, considering ultrasound and endometrial biopsy in case of abnormal uterine bleeding. 65
New susceptibility loci for EC
The risk of EC remains higher for women who have a first-degree relative with EC, independently of Lynch syndrome. 66 Genome-wide association studies allowed the identification of seventeen EC risk loci, two of which showed reduced expression of negative regulators of oncogenic signal transduction proteins, such as SH2B3 and NF1. 67
Prognostic and therapeutic implications in EC molecular subtypes
Patients with recurrent or advanced EC show poor 5-year survival rates (about 18%), though promising results have been obtained by targeted therapy, based on molecular classification. Although approaches based on hormonal therapy or PI3K/AKT/mTOR pathway inhibitors showed promising activity in some selected NSMP cases; however, to date, trials for developing NSMP-directed therapies are needed, since adjuvant treatment decisions in this tumor group still rely on clinicopathological features. 10
MMRd/MSI-H ECs
MMRd/MSI-H occurs in 13%–30% of all cases of EC, making this the most frequent cancer with MMRd status. 68 MMRd/MSI-H ECs show an intermediate prognosis and are commonly associated with EEC. 4 Immunotherapy is used in advanced EC patients with high TMB (MMRd/MSI-H), who benefit more from it, underlining the importance of the molecular characteristics for therapy response. MMRd/MSI-H EC was associated with better response to ICI treatment than MMRp/MSS counterpart 69 (Table 2). Therefore, anti-PD-1 monoclonal antibodies (Abs), such as pembrolizumab and dostarlimab, have been approved for the treatment of patients with recurrent or advanced MMRd/MSI-H EC. 70 Avelumab, an anti-PD-L1 agent, was also proven to be effective in advanced MMRd/MSI-H EC, but not in the MMRp/MSS counterpart 71 (Table 2). A meta-analysis including advanced ECs showed that single-agent ICI (pembrolizumab or avelumab or durvalumab or dostarlimab) was associated with better response in MMRd than MMRp patients 72 (Table 2).
Molecular characteristics and response to target treatments in advanced endometrial cancer.
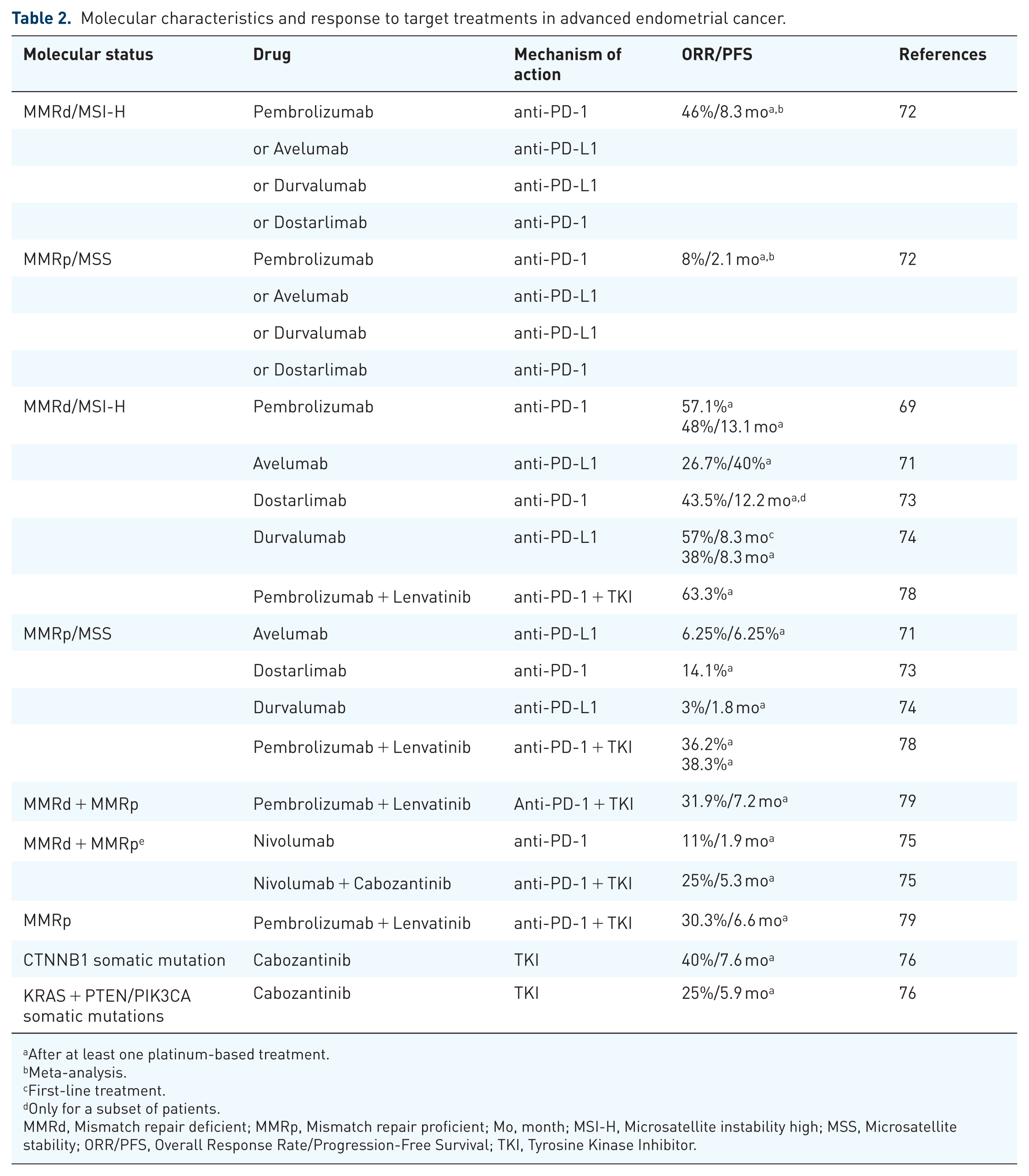
After at least one platinum-based treatment.
Meta-analysis.
First-line treatment.
Only for a subset of patients.
MMRd, Mismatch repair deficient; MMRp, Mismatch repair proficient; Mo, month; MSI-H, Microsatellite instability high; MSS, Microsatellite stability; ORR/PFS, Overall Response Rate/Progression-Free Survival; TKI, Tyrosine Kinase Inhibitor.
Also, the PD-L1 inhibitor durvalumab showed activity in advanced MMRd ECs, but not in MMRp ECs. Low PD-L1 expression showed a negative predictive value in the response to durvalumab. 77
Interestingly, the tyrosine kinase inhibitors (TKIs) cabozantinib and lenvatinib exhibited modest response as single agents in advanced ECs, but adding cabozantinib to nivolumab or lenvatinib to pembrolizumab improved response rates 78 (Table 2). The combination of lenvatinib and pembrolizumab has been approved for the treatment of advanced ECs both in MMRd and MMRp patients, although the benefit is greater in the MMRd subgroup. 79 Interestingly, these latter findings were confirmed in a subset of Japanese patients, suggesting that the combination of pembrolizumab and lenvatinib could become a new standard of care for patients of different ethnicities. 80 The role of ICI therapy is expanding to first-line treatment in EC. 81
Finally, a different mechanism of response to anti-PD-1 treatment may be involved in EC women carrying germline or somatic MMR mutations. In fact, LS/Lynch-like-associated ECs showed better response to pembrolizumab compared to sporadic MMRd/MSI-H ECs. The higher TMB observed in LS/Lynch-like-related ECs might partly explain the better response to ICIs. 82 This preliminary finding might also have therapeutic implications, as germline MMRd ECs might only need single-agent ICI therapy, avoiding the toxicities of combination treatments.
POLEmut ECs
The POLEmut group is the least common, but the most peculiar among the four molecular groups. It generally includes young women with low BMI and a high prevalence of early FIGO stages. A recent meta-analysis showed that the POLEmut prevalence was 7.95% in EEC and 4.45% in NEEC. 83 Interestingly, POLEmut ECs showed the coexistence of unfavourable histological features, such as high prevalence of grade 3, and favourable clinical charactheristics, such as FIGO stage I-II and myometrial invasion < 50%.4,83 Retrospective studies showed that POLEmut EC patients have a favourable prognosis, regardless of adjuvant therapy. 4 Two ongoing prospective trials might confirm the above findings.84,85
The excellent prognosis and clinical and molecular characteristics of POLEmut ECs do not seem to be influenced by ethnicity. Indeed, a Chinese study including 43 high-grade POLEmut ECs confirmed a good prognosis, which was not influenced by adjuvant treatment or risk groups.10,86 Among ECs with synchronous POLE and TP53 mutations, prognosis was similar to that found in ECs with only POLE PVs, underlining the clinical relevance of “true” POLE PVs in EC prognosis. Interestingly, POLE PVs might be more relevant in EC tumorigenesis when compared to TP53 alterations, but not when compared to MMR mutations. 86
There is anecdotal evidence of clinical response to pembrolizumab and nivolumab in advanced POLEmut ECs. A retrospective study, which analyzed 47,721 patients with different cancer types, showed that 189 patients with POLE/POLD1-mutated EC exhibited better overall survival (OS) and response to ICI treatment. 87
Notably, a stringent definition of POLE/POLD1 alterations in EC probably is not so important for prognosis and response to ICI treatment. Indeed, although ECs without POLE PVs are associated with worse clinical outcomes, in the rare cases of recurrence, survival rates were high. 84 Moreover, POLEmut EC patients showed the best clinical outcome across different studies, where the definition of POLE variants varies.4,88 Finally, also nonsynonymous mutations outside POLE/POLD1 ED have been shown to be associated with good OS in patients receiving ICIs. 87
Conclusions and future perspectives
EC is an extremely heterogeneous disease, and the recent molecular classification in four groups has only partially improved our understanding. Further biomarkers, tested in prospective studies, are needed. Target therapy will probably overcome conventional chemoradiotherapy in the near future. A combination of drugs targeting different cancer pathways will improve personalized treatment against EC, hopefully lowering costs and adverse events.
Germline PVs in different genes, such as MMR, PTEN, POLE/POLD1, BRCA1/2, MUTYH, NTHL1, STK11, may predispose to inherited EC, which may be part of different phenotypic profiles underlying several HCSs, which include a broad tumor spectrum (Figures 1 and 2). Therefore, multiple genes can contribute to the increased risk of developing an inherited EC, determining a genetic overlap for the same tumor phenotype. However, the lack of a standardized approach for genetic testing often makes it difficult to diagnose these HCSs, resulting in an underestimation of cases of hereditary EC.
Thanks to advances in next-generation sequencing (NGS) technology, which has revolutionized the clinical approach to genetic testing, the use of multigene panels containing multiple cancer susceptibility genes is becoming increasingly common, providing clinicians with more information about multiple inherited cancer syndromes through a single test. In the future, subjecting EC women to a genetic test by mutigenic panel could be useful to bypass the genetic overlap associated with the development of inherited ECs, in order to reduce underdiagnosis and enhance prevention and surveillance strategies in genetically predisposed probands and their family members.
Universal MMR screening is recommended for all women with EC to identify those with LS and drive treatment decisions. This testing, which is performed via somatic analysis by immunohistochemistry (IHC) or microsatellite instability (MSI), helps identify patients with a hereditary EC predisposition, also allowing for the screening of at-risk family members. In the case of immunohistochemical expression loss of MLH1 protein, a somatic analysis of MLH1 promoter hypermethylation can be used as a reflex testing strategy for identifying LS patients through a subsequent germline MMR testing, which is performed only if MLH1 promoter results are unmethylated. Furthermore, in case of stage I–II disease and in the presence of at least one factor among p53abn, FIGO IB-II, G3, or substantial LVSI, molecular evaluation of somatic POLE is indicated, as patients with the above-mentioned clinico-pathological characteristics and POLE mutation may benefit from a de-escalation of treatments (no adjuvant treatment).
Advances in molecular and genetic analysis techniques have enabled significant milestones to be achieved thanks to ongoing research in oncology. Therefore, through the use of increasingly precise and detailed classifications and methods, it is possible to integrate the purely molecular aspect with other equally important implications about the correct diagnosis, prognosis, and therapeutic decision. On the other hand, there is certainly much still to be discovered in this field, given the heterogeneity and varying scope of the analyzed studies. Furthermore, not all mutations have an established clinical significance, because some variants of unknown significance (VUS) may make it difficult to make treatment decisions. Lack of standardized protocols for testing and interpreting genetic variants could lead to inconsistent clinical application. Thus, it will be necessary to continue integrating the anatomopathological and molecular data from the literature with clinical outcomes, in order to better stratify the treatment and management of EC patients.
Footnotes
Acknowledgements
Project Title: SiciliAn MicronanOTecH Research And Innovation CEnter “SAMOTHRACE” (MUR, PNRR-M4C2, ECS_00000022), spoke 3: Università degli Studi di Palermo, “S2-COMMs - Micro and Nanotechnologies for Smart & Sustainable Communities.”
