Abstract
Background:
Perineural invasion (PNI) is recognized as a prognostic factor in several cancers, but its role in predicting axillary lymph node metastasis (ALNM) in early-stage breast cancer remains unclear. Understanding this association could provide insights for nodal staging in the era of axillary surgery de-escalation, guiding adjuvant treatment decisions.
Objectives:
To investigate and validate the association between PNI and both the presence and extent of ALNM in patients with early-stage operable invasive breast cancer.
Design:
A retrospective cohort study and meta-analysis.
Methods:
We conducted a retrospective study of patients with early-stage operable breast cancer who underwent surgery between June 2011 and June 2023 stratified by PNI status. Three matching methods—propensity score matching, inverse probability of treatment weighting, and overlap weighting—were used to minimize confounding. Multivariable logistic regression analysis was used to assess the association between PNI and both ALNM and lymph node ratio (LNR). In addition, a meta-analysis was conducted to verify these associations.
Results:
Among 4156 patients, 1223 (29.4%) were PNI-positive. PNI-positive patients had a significantly higher incidence of ALNM (54.0% vs 32.1%) and increased lymph node burden (LNR >0.20). These associations remained significant after adjusting for confounding variables and through sensitivity analyses using the three matching methods (all p < 0.05). In the meta-analysis, 16 cohort studies comprising 17,451 participants met the selection criteria. The analysis confirmed a significant association between PNI and increased ALNM risk (odds ratio (OR), 3.30; 95% confidence interval (CI), 2.24–4.86), poorer disease-free survival (hazard ratio (HR), 1.84; 95% CI, 1.39–2.44), and poorer overall survival (HR, 1.64; 95% CI, 1.15–2.35).
Conclusion:
PNI is significantly associated with increased ALNM and lymph node burden in early-stage operable breast cancer. These findings support the clinical utility of PNI for risk stratification and as a potential indicator for guiding adjuvant treatment decisions.
Introduction
Breast cancer is the most prevalent malignancy among women and a leading cause of cancer-related mortality worldwide. 1 Lymph node status is a key prognostic indicator, with lymphatic metastasis strongly linked to shorter overall survival (OS) and higher recurrence rates.2,3 Increasing evidence suggests that lymph node metastasis (LNM) is a critical source of tumor cells for further dissemination.4–6 Traditionally, tumor cells are believed to spread to lymphatic vessels through mechanisms such as vascular invasion and lymphangiogenesis regulation, eventually reaching lymph nodes via the lymphatic system.7,8 However, in recent decades, the role of nerves—noncellular components of the tumor microenvironment—has gained attention. 9 Crosstalk between nerves and tumor cells has been shown to regulate tumorigenesis and progression. 10 Currently, the presence of perineural invasion (PNI) is the only neuronal marker used in clinical practice; it represents the most direct relationship between cancer cells and nerves. 11
Previous studies have linked PNI to LNM in various cancers.12–14 PNI is defined as tumor cell infiltration into any of the three nerve sheath layers (epineurium, perineurium, or endoneurium) or encirclement of at least one-third of a nerve bundle by tumor cells. 15 These features facilitate the dissemination of tumor cells from primary tumors, indicating local invasion and serving as a potential route for distant spread independent of lymphatic or blood vessel invasion. 16 Clinically, PNI has been integrated into the staging systems and risk stratification frameworks of several malignancies, including penile, head and neck, esophageal, and thyroid cancers. In these contexts, PNI status provides essential guidance for determining adjuvant therapeutic strategies. 17 Current studies suggest that the occurrence and progression of PNI result from complex interactions between nerves and tumor cells, rather than from a simple one-way invasion. 18 For instance, a previous study observed that tumor cells proliferating along nerve branches, with continuity between cancer cells in the perineural space and those within certain lymph nodes, 19 indicating that neural invasion may serve as a pathway for lymphatic involvement. In addition, a recent study identified a neuropeptide as a molecular link between PNI and LNM in oral squamous cell carcinoma. 20
Although PNI has recently been recognized as a tumor hallmark, its clinical utility in breast cancer remains debated.21,22 The prevalence of PNI in breast cancer varies widely, from 1.14% to 25.7%.23–25 Some studies show that PNI is associated with axillary lymph node metastasis (ALNM), an increase in local recurrence, and a poor prognosis,21,26,27 whereas others argue that it lacks prognostic significance.25,28 Many of these studies are limited by small sample sizes, single-center designs, or a lack of focus on PNI as the primary research objective.24,26,28 Moreover, PNI is not a mandatory component of routine pathological assessments in breast cancer guidelines, which may contribute to its underreporting in clinical practice. 29 The established mechanisms of tumor dissemination, together with reliance on tumor-node-metastasis staging, continue to fuel debate over the clinical significance of PNI.21–24 The approach to axillary surgery in breast cancer has evolved from axillary lymph node dissection (ALND) to sentinel lymph node biopsy, with the potential for exemption from axillary surgery.30–32 However, this shift may result in the loss of valuable pathological information about lymph node status, potentially impacting adjuvant treatment planning. 33 Therefore, exploring alternative pathways for ALNM and identifying biomarkers associated with lymph node burden are crucial for guiding adjuvant therapies.
Given these concerns, this study aimed to conduct a large-scale cohort analysis to investigate the correlation between PNI and ALNM, including the extent of nodal involvement in early-stage operable breast cancer, and to perform a meta-analysis to synthesize existing data and validate these findings.
Materials and methods
Study design and participants
This retrospective study enrolled patients who underwent surgical treatment for breast cancer at our hospital between June 2011 and June 2023. The study was reviewed and approved by the Ethics Committee of Fujian Medical University Union Hospital (approval number: 2024KY276) and was conducted in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki). Since the data employed were extracted from electronic medical records and clinical follow-up information and were devoid of unique personal identifiers, the requirement for written informed consent was waived. The reporting of the clinical cohort study follows the STROBE statement, and the supplementary meta-analysis conforms to the PRISMA 2020 statement.34,35 The completed STROBE and PRISMA 2020 checklists are provided as Supplemental File Checklists.
Patients were included if they had a pathological diagnosis of invasive breast cancer and had undergone radical surgery. The exclusion criteria were distant metastasis on diagnosis, preoperative neoadjuvant chemotherapy, breast cancer history, or incomplete clinicopathological data (e.g., key tumor characteristics or lymph node status). A total of 4156 patients with operable invasive breast cancer were included in this study. These patients were categorized into PNI-positive and PNI-negative groups based on the occurrence of PNI, as identified in their postoperative pathology reports (Figure S1(A)).
PNI was defined as the infiltration of tumor cells into the endoneurium, epineurium, or perineurium of the nerve sheath, as identified on hematoxylin and eosin-stained histologic slides. Pathological features were independently assessed and cross-validated by two certified pathologists. Any discrepancies were resolved through joint re-evaluation until a consensus was achieved.
The study variables included basic clinical information such as age at diagnosis, body mass index, menstrual status, and underlying diseases (hypertension and diabetes), along with postoperative pathological data comprising breast cancer subtypes, Ki67, pathological type, histological grade, PNI, lymphovascular invasion (LVI), tumor size in the breast (pT), lymph node status (pN), and surgical procedures. All data were extracted from the electronic medical record system of the hospital using a standardized data collection form and cross-validated by two trained physicians.
Outcomes
The primary endpoint was ALNM. The secondary endpoint was the lymph node ratio (LNR), calculated as the number of positive lymph nodes divided by the total number of examined lymph nodes. This endpoint was evaluated in the subset of 1423 patients with breast cancer who had at least one metastatic axillary lymph node and underwent subsequent ALND. The cohort was stratified into two risk groups based on the established LNR cut-off value 36 : a low-risk group (LNR ⩽0.20) and a high-risk group (LNR >0.20).
Literature review and meta-analysis
A systematic literature search and meta-analysis were performed to further investigate the correlation between PNI and ALNM in early-stage breast cancer. Considering the strong association between ALNM and the survival outcomes of patients with breast cancer, these outcomes were also evaluated in this meta-analysis. PubMed, Embase, Web of Science, and the Cochrane Library were searched from their inception until June 1, 2024. The inclusion criteria were as follows: (1) a pathological diagnosis of breast cancer; (2) studies reporting the association between PNI, LNM, and breast cancer prognosis; (3) studies with available or calculable measures of association, such as odds ratio (OR) or hazard ratio (HR), along with their 95% confidence intervals (CIs); and (4) English publications. The following studies were excluded: (1) reviews, meeting abstracts, comments, case reports, letters, and other communications lacking original data; (2) studies with duplicate patients; (3) animal studies; and (4) studies reporting only sentinel LNM or those with a PNI incidence <5%. The PICO framework for the current study was as follows: P (population)—women with breast cancer; I (intervention, risk factor)—the presence of PNI; C (comparison)—the absence of PNI; O (outcome)—ALNM and survival outcomes (OS and disease-free survival (DFS)). The full search strategy for each database is detailed in Table S1. Keywords related to predefined outcomes and study designs were not used in the database search to maximize the number of studies meeting the inclusion criteria. In addition, literature retrieval was conducted by reviewing the reference lists of relevant studies to identify any studies not initially found. EndNote software version 21 (Thomson Reuters, New York, NY, USA) was used to organize all studies. After removing duplicates, the titles and abstracts were reviewed, followed by a preliminary screening of the relevant literature. Subsequently, the full texts of the studies were retrieved for a thorough eligibility assessment. The risk of bias and study quality were assessed using the cohort quality assessment instrument provided by the Newcastle–Ottawa Quality Assessment Scale. 37 Two investigators independently performed the literature search, study screening, and data extraction, with any discrepancies resolved through consensus or consultation with a third reviewer. The following data were extracted: (1) author names, publication year, and country; (2) study design and follow-up duration; (3) study population characteristics, including data source, sample size, and mean age; (4) outcomes and determination of outcomes; and (5) risk estimates and adjustments. We sought all available results compatible with these outcome domains in each study; if multiple multivariable models were reported, the one adjusting for the most comprehensive set of confounders was prioritized.
Statistical analysis
Statistical analysis and data visualization were performed using R version 4.2.3 (R Foundation for Statistical Computing, Vienna, Austria), Review Manager version 5.3 (The Cochrane Collaboration, London, UK), and GraphPad Prism version 9 (GraphPad Software, Inc., San Diego, CA, USA). Categorical variables, presented as frequencies and percentages, were analyzed for group differences using Pearson’s Chi-squared or Fisher’s exact test. Continuous variables that were non-normally distributed are presented as median with interquartile range and were compared using the Mann–Whitney U test. We reduced the impact of confounding factors by matching PNI-positive and PNI-negative groups using propensity score matching (PSM), robust inverse probability of treatment weighting (IPTW), and overlap weighting (OW) methods, using the following matching factors: age, breast cancer subtype, pathological type, Ki67, and LVI. The balance of these baseline characteristics was assessed using standardized mean differences (%). Multivariable logistic regression analyses were conducted to compare ALNM and involvement burden between the two groups. Regarding the meta-analysis, the clinical and methodological characteristics of the eligible studies were examined against the PICO framework to determine the appropriateness of quantitative synthesis. ORs and HRs with 95% CI from primary studies were pooled to estimate the role of PNI in determining ALNM and survival outcomes in patients with breast cancer. Heterogeneity across studies was assessed for each pooled analysis using Cochran’s Q test and the I2 statistic. The choice of effect model was pre-specified: a fixed-effects model was applied when heterogeneity was low (I2 ⩽50% and p ⩾0.10); otherwise, a random-effects model was used. When ORs for the association between PNI and ALNM were not directly reported, they were calculated from the corresponding 2 × 2 contingency tables. 38 The 2 × 2 contingency tables used to calculate ORs are provided in Table S2. Forest plots were generated to visually display the effect sizes and 95% CIs of individual studies and the pooled syntheses. A sensitivity analysis was used to assess the stability of the pooled data. The risk of bias across the studies (publication bias) was assessed using Begg’s and Egger’s tests. Statistical significance was set at the 0.05 level for two-tailed tests, and conclusions were based on 95% CIs.
Results
Patient characteristics
A total of 4156 patients with operable early-stage breast cancer were included, with a median age of 52 years. Among these, 2181 (52.5%) were postmenopausal. PNI was identified in 1223 patients (29.4%). Significant differences in clinicopathological features were observed between the PNI-positive and PNI-negative patients. Compared with PNI-negative cases, PNI-positive tumors were more frequently associated with LVI, higher pathological T stage, and an increased incidence of ALNM. In contrast, PNI-positive patients were more likely to present with the HR+/HER2− subtype, low Ki-67 expression (⩽20%), and invasive carcinoma of no special type. Of the patients with ALNM, 1423 underwent ALND. The axillary lymph node burden, quantified by LNR, was significantly higher in the PNI-positive group. Detailed baseline characteristics are in Table 1.
Clinicopathological characteristics of 4156 operable breast cancer patients.

Bold font indicates the presence of statistically significant differences.
The Chi-square test or Fisher’s exact test was used for categorical variables, while continuous variables were assessed with the Mann–Whitney U test due to their non-normal distribution.
The underlying disease was hypertension or diabetes.
Includes only the 1423 patients who had at least one axillary lymph node metastasis and underwent axillary lymph node dissection.
AJCC, American Joint Committee on Cancer; BMI, body mass index; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; LNR, lymph node ratio; LVI, lymphovascular invasion; PNI, perineural invasion; PR, progesterone receptor; SD, standard deviation.
Factors affecting ALNM
ALNM incidence was significantly higher in PNI-positive patients than in PNI-negative patients (54.0% vs 32.1%, p < 0.001). This association remained significant after adjusting for covariates in multivariable logistic regression (OR = 1.29, 95% CI: 1.10–1.52; p = 0.002). Owing to baseline imbalances between groups, sensitivity analyses were conducted using three matching methods (PSM, IPTW, and OW) to minimize confounding. Baseline characteristics before and after matching are detailed in Table S3. Multivariable logistic regression after matching consistently indicated PNI positivity as a risk factor for ALNM (Figure 1). LVI positivity and tumor size >2 cm (pT2–4), known risk factors, were also significant predictors of ALNM in multivariable analysis using the three matching methods (Table S4). Furthermore, analyses before and after matching showed that underlying diseases were associated with ALNM.
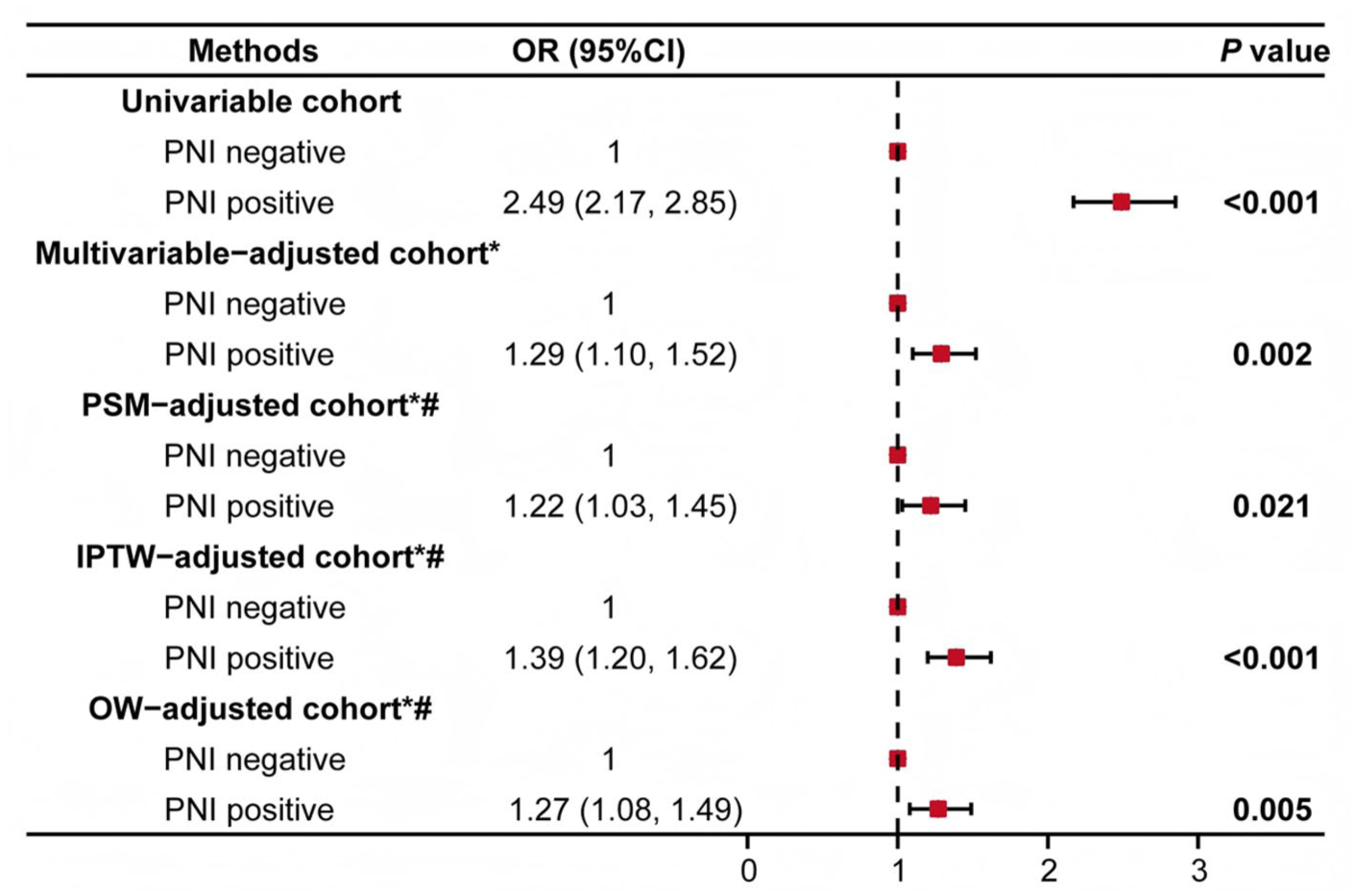
Multivariable-adjusted associations between PNI and axillary lymph node metastasis in patients with operable breast cancer.
Factors affecting LNR
To evaluate the relationship between PNI and lymph node burden, we analyzed LNR in 1423 patients with ALNM who underwent axillary dissection. Univariable and multivariable logistic regression showed that PNI positivity was significantly associated with a high LNR (>0.2; OR = 1.38, 95% CI: 1.07–1.77, p = 0.012). Sensitivity analyses using multiple matching methods confirmed this association (Figure 2). Baseline characteristics before and after matching are shown in Table S5. LVI positivity and pT2–4 were also identified as risk factors for high LNR in multivariable analysis using all three matching methods (Table S6), consistent with previous ALNM findings.

Multivariable-adjusted association between PNI and LNR in patients with node-positive breast cancer.
Meta-analysis
The literature search identified 1358 potential articles, with duplicates removed to yield 1016 studies (Figure S1(B)). After screening, 78 studies were reviewed in full. In the end, 16 cohort studies with 17,451 participants met the inclusion criteria for the meta-analysis. The study characteristics are summarized in Table S7. The Newcastle–Ottawa scale scores of these included studies ranged from 6 to 9, indicating generally high study quality. These studies, published between 2011 and 2024, had sample sizes ranging from 105 to 7371. Six studies reported risk estimates for ALNM, and the pooled results showed that PNI was associated with increased ALNM risk (OR = 3.30, 95% CI: 2.24–4.86; p < 0.001). Eight studies linked PNI to poorer DFS (HR = 1.84, 95% CI: 1.39–2.44; p < 0.001), and nine studies showed that PNI was associated with higher all-cause mortality (HR = 1.64, 95% CI: 1.15–2.35; p = 0.007). Figure 3 presents the forest plot illustrating these associations. Publication bias was observed in DFS studies according to Egger’s test (p = 0.002) but not Begg’s test (p = 0.266; Figure S2(C) and (D)). The trim-and-fill method adjusted the pooled HR slightly but results remained significant (HR = 1.78, 95% CI: 1.36–2.33; p < 0.001; Figure S3). No publication bias was detected in the ALNM or OS analyses (Figure S2(A), (B), (E), and (F); Begg’s: p = 1.000 for ALNM and p = 0.175 for OS; Egger’s: p = 0.900 for ALNM and p = 0.267 for OS). Sensitivity analyses showed consistent pooled effects after the sequential removal of any single included study, confirming result stability (Figure S4).

Meta-analysis of the effect of PNI on ALNM, DFS, and OS in patients with breast cancer. (a) Forest plot of the meta-analysis for ALNM. (b) Forest plot of the meta-analysis for DFS. (c) Forest plot of the meta-analysis for OS.
Discussion
The clinical relevance of PNI and its association with ALNM in breast cancer remains debated. In this study, based on a large cohort of patients with early-stage operable invasive breast cancer, we demonstrated that PNI was independently associated with both ALNM and a high LNR. The association remained robust across multivariable regression and three propensity score-based approaches (PSM, IPTW, and OW), which minimized confounding and simulated randomization effects. To complement these findings, we conducted a meta-analysis of existing studies, which confirmed that PNI was correlated with increased lymph node burden and poorer DFS and OS, underscoring its prognostic relevance.
From a clinical perspective, our findings highlight the importance of incorporating PNI into routine pathological reporting and risk assessment. In contemporary breast cancer management, axillary surgery is undergoing significant de-escalation. Trials such as SOUND and INSEMA suggest that sentinel lymph node biopsy may be safely omitted in selected early-stage patients.31,32 However, the omission of axillary staging inevitably leads to a loss of pathological nodal information, which is traditionally critical for guiding adjuvant systemic therapy. 33 While preoperative radiological assessment plays a pivotal role in selecting candidates for surgical de-escalation, imaging modalities may be limited in their ability to detect microscopic disease or fully capture tumor biological aggressiveness. 39 In this context, PNI may serve as a biologically informative marker to help compensate for missing nodal staging data and refine postoperative risk stratification. This post-resection confirmation of PNI offers a definitive biological risk indicator that complements preoperative imaging. 40 In this sense, PNI serves as a postoperative biological cross-check of imaging-based de-escalation strategies, helping to recalibrate adjuvant treatment intensity when nodal disease may have been underestimated. Clinically, PNI is already a recognized indicator for adjuvant radiotherapy in several other malignancies. 17 Specifically, regarding locoregional control in breast cancer, the evidence demonstrates that outcomes in patients with limited nodal involvement are heterogeneous, where biologically low-risk subsets achieve excellent locoregional control even without irradiation. 41 In this context, PNI may serve as a valuable biological indicator to help refine radiotherapy decision-making, potentially identifying patients who would benefit from escalated treatment versus those suitable for de-escalation, even within comparable anatomical stages. Although our study did not directly assess survival outcomes or treatment benefit, these observations suggest that PNI status provides essential data for refining risk stratification in the era of minimal axillary surgery.
Although previous studies have reported the adverse prognostic significance of PNI in breast cancer, many were limited by small sample sizes and inconsistent reporting practices.21,24 In addition, PNI has often been regarded as a secondary feature, or it has been combined with LVI into a composite variable, a practice that can obscure its independent contribution.42,43 Narayan et al. 21 demonstrated that PNI significantly increases the risk of local recurrence in breast cancer, with PNI-positive patients showing a higher 7-year locoregional recurrence rate than PNI-negative patients (7.1% vs 4.7%, HR = 1.57, 95% CI: 1.20–2.07, p = 0.001). Importantly, the prognostic effect of PNI was stronger than that of LVI (HR = 1.24, 95% CI: 1.10–1.54), for which statistical significance was only borderline (p = 0.054). In our study of more than 4000 patients, the association between PNI and ALNM persisted even after adjusting for LVI and other clinicopathological variables, reinforcing the view that PNI is not simply an extension of lymphatic dissemination but an independent factor in tumor progression (Tables S3 and S5). Our meta-analysis further supported these findings. PNI was associated with an increased risk of ALNM (OR = 3.30, 95% CI: 2.24–4.86), consistent with the results of our cohort. Exploratory analyses of survival outcomes suggested that PNI was linked to poorer DFS (HR = 1.84, 95% CI: 1.39–2.44) and increased overall mortality (HR = 1.64, 95% CI: 1.15–2.35; Figure 3), consolidating its status as a robust prognostic indicator.
Despite its association with poor prognosis, PNI was more frequently observed in patients with ER-positive, HER2-negative tumors and low Ki-67 expression (Table 1), consistent with previous reports.21,25 Although the underlying mechanisms remain to be fully elucidated, several lines of evidence provide biological plausibility. In endocrine-driven tumors such as prostate cancer, PNI is highly prevalent and is associated with the enrichment of estrogen response pathways and increased ESR1 expression. 44 Cross-talk between steroid receptors and neurotrophin pathways, such as NGF/tropomyosin receptor kinase A has been shown to promote tumor cell migration. 45 In addition, experimental models indicate that estradiol/ERα signaling can enhance neurite outgrowth in sensory neurons, supporting the possibility that hormonal signaling may influence the neural microenvironment. 46 These observations suggest that PNI may reflect complex tumor–nerve interactions beyond what is predicted by classical markers of tumor aggressiveness. These interactions promote tumor dissemination through unconventional pathways, including direct perineural tracking along nerve sheaths and nerve-guided local spread independent of vascular or lymphatic routes, which may not be adequately addressed by standard surgical margins.47,48 Furthermore, nerve–tumor crosstalk establishes a bidirectional signaling network, whereby nerves secrete neurotransmitters and neurotrophic factors that enhance tumor cell motility and invasiveness while cancer cells actively trigger innervation within the tumor microenvironment.22,49,50 Concurrently, the definition of HER2-negativity is being critically refined to address biological heterogeneity, marked by the formal delineation of the HER2-low and HER2-ultralow categories.51,52 In light of the biological association of PNI with the hormone receptor-positive tumors described above, and given that the majority of HER2-low tumors are characterized as hormone receptor-positive, PNI emerges as a potential risk stratifier within this newly defined heterogeneous population.53,54 Future studies should, therefore, investigate whether the prognostic weight of PNI differs across these refined expression categories to potentially guide more precise therapeutic sequencing.
Building on these findings, our study represents the largest cohort to date examining the correlation between PNI and ALNM in early-stage operable breast cancer, addressing the sample size limitations of previous studies in which PNI reporting was not mandatory. We used multivariable regression analysis and three robust matching methods to adjust for confounding factors, enhancing the rigor of this study by simulating the randomization effects of controlled trials. By examining the relationship between PNI and lymph node status and burden, our study provides new insights into the potential role of PNI in compensating for missing nodal pathological information in the era of axillary surgery de-escalation. Nevertheless, the clinical utility of PNI as a biomarker requires further validation in prospective studies.
This study has limitations. First, as PNI is not currently a mandatory component of routine pathologic reporting for breast cancer, our sample was limited to a single center. To address this, we conducted a supplementary meta-analysis using data from multiple institutions. However, all included studies were retrospective in nature, which inherently increases the risk of selection and information bias. In addition, the literature search was restricted to English-language publications and excluded gray literature, which may have introduced reporting bias. Prospective studies would provide higher-level evidence but are currently lacking in this field. Second, while the meta-analysis provided insights into long-term survival, our single-center cohort primarily focused on ALNM and lymph node burden. Lastly, although we highlighted the clinical value of PNI in breast cancer, we did not explore the molecular mechanisms underlying tumor–nerve crosstalk. Future large-scale, multicenter, longitudinal studies are needed to investigate the long-term prognostic significance of PNI and its molecular mechanisms across refined subtypes, including HER2-low populations.
Conclusion
PNI is strongly and independently associated with ALNM and lymph node burden in early-stage operable breast cancer. Together with evidence from our meta-analysis, these findings support the prognostic value of PNI and its potential clinical utility. As axillary treatment is increasingly de-escalated in selected patients, PNI may emerge as an important pathological biomarker that complements missing nodal data and informs adjuvant treatment decisions.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261424685 – Supplemental material for Perineural invasion and risk of lymph node metastasis in early-stage operable breast cancer: a large-scale propensity-matched cohort study and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359261424685 for Perineural invasion and risk of lymph node metastasis in early-stage operable breast cancer: a large-scale propensity-matched cohort study and meta-analysis by Weifeng Cai, Yali Wang, Yibin Qiu, Peng He, Qindong Cai, Shunyi Liu, Jiaqi Zhan, Minyan Chen, Lili Chen, Chunsen Xu, Shunguo Lin, Fangmeng Fu and Chuan Wang in Therapeutic Advances in Medical Oncology
