Abstract
Radioligand therapy (RLT) has reshaped the treatment landscape of advanced prostate cancer (PCa), offering a precision medicine approach that integrates molecular imaging with targeted radionuclide delivery, in which radioactive isotopes are bound to molecules that aim to selectively target cancer cells. The approval of 177Lu-prostate-specific membrane antigen (PSMA)-617 for metastatic castration-resistant prostate cancer marked a major milestone, with randomized trials (VISION, PSMAfore) demonstrating significant improvements in overall and progression-free survival compared to standard therapies. Beyond beta-emitting agents, next-generation alpha emitters such as actinium-225 and beta/Auger electron-emitting isotopes like terbium‑161 are in active development, aiming to overcome resistance and target micrometastases with greater potency. Combination approaches, pairing RLT with poly(ADP-ribose) polymerase inhibitors, immune checkpoint blockade, or androgen receptor pathway inhibitors, for example, are under intense investigation, with early phase data indicating potential efficacy. Technical advances in imaging, personalized dosimetry, and molecular diagnostics may enable more precise patient selection and adaptive treatment strategies, such as dose adjustment based on dosimetry or target expression. Emerging RLT platforms target additional tumor markers, including human kallikrein 2 and six-transmembrane epithelial antigen of the prostate-2 as well as bispecific ligands, addressing disease heterogeneity and expanding therapeutic reach. Nonetheless, challenges remain around long-term hematologic and renal safety, radionuclide supply, protocol standardization, and global accessibility. Ongoing and future multicenter trials, collaborative consortia, and innovations in theranostics will be critical to defining optimal patient selection, sequencing with existing therapies, and embedding RLT as a key pillar in the management of advanced PCa.
Introduction
Prostate cancer (PCa) remains one of the most frequently diagnosed malignancies in men worldwide, with an estimated lifetime risk of approximately 1 in 9. 1 Approximately 3 million men in the United States live with PCa, 1 and advanced forms, including metastatic hormone-sensitive prostate cancer (mHSPC) and metastatic castration-resistant prostate cancer (mCRPC), represent incurable diseases where the aim of therapy is to prolong life, with survival typically ranging from 4 to 6 years for mHSPC and 1 to 3 years for mCRPC.2,3 The standard first-line management of mHSPC typically combines androgen deprivation therapy (ADT) with novel hormonal agents (androgen-receptor pathway inhibitors, ARPIs) such as abiraterone and enzalutamide. 4 Increasingly, the treatment paradigm for mHSPC has evolved beyond doublet combinations to include triplet therapy. Recent phase III trials, including ARASENS 5 and PEACE-1, 6 have shown significant survival benefit for patients who receive ADT, docetaxel, and an ARPI (such as darolutamide or abiraterone) compared to doublet regimens, establishing triplet therapy as a potential standard-of-care for patients with high-volume disease in particular. Although many patients initially respond, resistance ultimately develops in the majority of patients with eventual progression to CRPC in around 2–5 years with intensified therapies for mHSPC.6,7 Existing systemic therapies, including ARPIs, taxane chemotherapies (docetaxel, cabazitaxel), and the α-emitter 223Ra-dichloride, have all been shown to prolong overall survival (OS) in CRPC, but the disease remains incurable, and there is still an unmet need to develop novel therapeutic strategies.2,8
Theranostic strategies, which combine diagnostic imaging and therapy targeting the same molecular target, have significantly changed the treatment paradigm for advanced PCa. 9 Prostate-specific membrane antigen (PSMA) is overexpressed by most PCa cells (especially in high-grade and metastatic disease) and is a promising target for both imaging and targeted radioligand therapy (RLT). PSMA-targeted RLT agents (either small-molecule ligands or antibodies) deliver cytotoxic radionuclides directly to PSMA-expressing tumor cells, enabling focused irradiation of PCa lesions throughout the body. 9 The first-in-class agent, 177Lu-PSMA-617 (vipivotide tetraxetan, FDA-approved as Pluvicto), is a small-molecule ligand that binds the extracellular domain of PSMA and is internalized by tumor cells, delivering medium-energy β-particles that induce DNA double-strand breaks. 10 The short-range β-emissions (~0.7 mm mean range) may produce a “crossfire” effect, whereby radiation from the radionuclide engagement to the target cell may additionally damage neighboring cells. PSMA-PET/CT imaging (e.g., 68Ga-PSMA-11 PET/CT) is integral to patient selection, ensuring that metastatic lesions have sufficient PSMA uptake before administering therapy.11,12
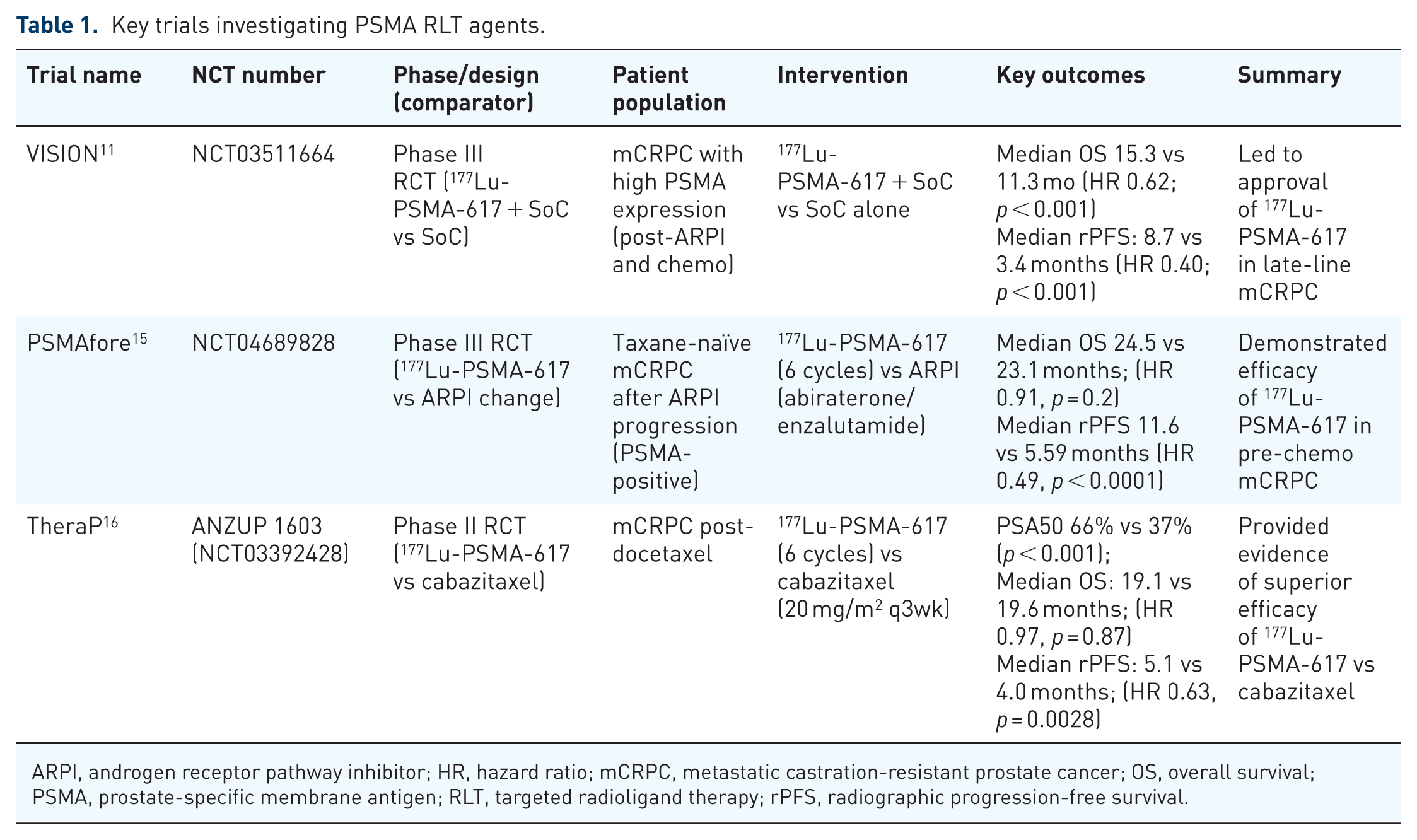
The pivotal phase III VISION trial provided high-level evidence of 177Lu-PSMA-617 efficacy in heavily pretreated mCRPC patients.11,13 In VISION (NCT03511664), the addition of 177Lu-PSMA-617 to best supportive care produced a statistically significant improvement in OS (median 15.3 vs 11.3 months; hazard ratio (HR) 0.62; p < 0.001), as well as radiographic progression-free survival (rPFS).11,13 These results, reflecting an absolute 4-month extension in median OS, led to the FDA approval of 177Lu-PSMA-617 in 2022 for PSMA-positive mCRPC. This review outlines current RLT modalities and key clinical trial data, including evidence from combination strategies with poly(ADP-ribose) polymerase (PARP) inhibitors, immune checkpoint inhibitors, and ARPIs. We also survey next-generation isotopes (including alpha-emitters such as actinium-225 (225Ac) and Lead‑212 (212Pb), and the Auger-emitting terbium‑161 (161Tb)) and antibody-based platforms (such as 225Ac-J591) that are under active investigation. In addition, we discuss emerging targets as well as technical innovations in imaging and personalized dosimetry that will shape the future of RLT in advanced PCa.
Current radioligand therapies in advanced prostate cancer
PSMA-targeted β-emitter therapy (177Lu-PSMA-617)
177Lu‑PSMA‑617 is a small‑molecule ligand that binds the extracellular domain of PSMA with high affinity and specificity. Upon binding, the complex is internalized into the tumor cell where the emitted beta particles, characterized by a mean tissue range of approximately 0.7 mm, induce lethal double‑strand DNA breaks both in targeted cells and adjacent tumor tissue through a crossfire effect. Moreover, PSMA internalization may contribute to sustained tumor retention of the radionuclide. 9 The efficacy of 177Lu‑PSMA‑617 has been demonstrated in multiple studies, with approximately 40%–50% of mCRPC patients achieving a ⩾50% decline in PSA (PSA50). The phase III VISION trial included 831 mCRPC patients who had received ⩾1 ARPI and ⩾1 taxane chemotherapy. Patients treated with 177Lu‑PSMA‑617 plus standard-of-care had a 4-month increase in median OS compared to those receiving standard-of-care alone.11,13 Notably, patient selection relies on PSMA‑PET imaging (e.g., using an SUVmax cut-off or avidity greater than a reference organ such as liver) to ensure that metastatic lesions exhibit a minimum threshold of PSMA expression. 14
Building upon these findings, the recently published PSMAfore trial demonstrated the efficacy of 177Lu‑PSMA‑617 in the pre-chemotherapy mCRPC setting. This phase III study randomized 468 taxane-naïve mCRPC patients who had progressed on one prior ARPI to receive either 177Lu‑PSMA‑617 or a change of ARPI therapy. The trial showed a significant improvement in rPFS with 177Lu‑PSMA‑617, with a median of 11.6 months compared to 5.6 months for ARPI change (HR 0.49), though OS was similar between the two groups (median OS 23.7 vs 23.9 months; HR 0.98). Notably, 177Lu-PSMA-617 demonstrated a more favorable safety profile, with common adverse effects including dry mouth (xerostomia), fatigue, and mild hematologic toxicity, a lower incidence of grade 3–5 adverse events (36% vs 48% as per PSMAfore) and few patients requirement treatment discontinuation for toxicity). 15 The PSMAfore results led to an expansion in the FDA label for 177Lu‑PSMA‑617 as a means to defer taxane-based chemotherapy in patients with mCRPC who have progressed after an ARPI.
Data from the phase II TheraP trial further reinforce findings from VISION and PSMAfore; in this trial, 177Lu‑PSMA‑617 was tested against an active comparator, cabazitaxel, in mCRPC patients who had received prior docetaxel and, in many cases, a prior ARPI. 16 Notably, patient selection for this trial was different to VISION and PSMAfore by requiring no discordance between FDG-PET and PSMA-PET as well as requiring all PSMA-positive measurable lesions to have an SUVmax of ⩾10. The trial demonstrated superior PSA50 rates and longer progression-free survival, as well as a more favorable toxicity profile with 177Lu-PSMA-617. 16 A summary of these studies in shown in Table 1.
Key trials investigating PSMA RLT agents.
ARPI, androgen receptor pathway inhibitor; HR, hazard ratio; mCRPC, metastatic castration-resistant prostate cancer; OS, overall survival; PSMA, prostate-specific membrane antigen; RLT, targeted radioligand therapy; rPFS, radiographic progression-free survival.
Other PSMA-RLT agents
In addition to 177Lu‑PSMA‑617, several other PSMA‑targeting agents have been developed. Table 2 highlights their key characteristics, study details, dosing regimens, efficacy, and notable considerations.
Summary of PSMA RLT agents other than 177Lu‑PSMA‑617.

ARPI, androgen receptor pathway inhibitor; HR, hazard ratio; I&T, Imaging and therapy; mCRPC, metastatic castration-resistant prostate cancer; PSMA, prostate-specific membrane antigen; RLT, targeted radioligand therapy; rPFS, radiographic progression-free survival; N/A, Not Applicable.
177Lu-PSMA-I&T is currently being assessed in two phase III trials. The SPLASH trial evaluated 177Lu-PSMA-I&T at a dose of 6.8 GBq every 8 weeks for up to 4 cycles versus ARPI switch in patients with PSMA-positive, prechemotherapy mCRPC who had received a prior ARPI. 17 The primary endpoint (rPFS) was met, with a median of 9.5 versus 6.0 months (HR 0.71, p < 0.001), while the PSA50 rate was 36% with 177Lu-PSMA-I&T compared to 15% with ARPI switch. At the first interim analysis, median OS was not significantly different between the arms (HR 1.11, p = 0.615). Overall, while accepting the limitations of cross-trial comparisons, the efficacy data from 177Lu-PSMA-I&T in the SPLASH trial are weaker than those seen in the PSMAfore trial, with a lower median rPFS and PSA50 rate, suggesting that dose intensity and schedule may be important in determining response to RLT. 15 The ongoing ECLIPSE trial (NCT05204927) is also evaluating 177Lu-PSMA-I&T in a similar population but utilizing a dose of 7.4 GBq every 6 weeks for up to 6 cycles compared to ARPI switch; while data from the trial are not yet available, a press release indicated that the primary endpoint (rPFS) had been met. 20
Antibody-based RLTs are also advancing. 177Lu-rosopatamab (TLX591) is under evaluation in the ProstACT GLOBAL trial, a multiregional phase III study enrolling mCRPC patients post-ARPI, with or without prior chemotherapy. The regimen involves two doses, 14 days apart, in conjunction with standard-of-care, with results expected in 2026. The aim of fractionated dosing is to improve tolerability while maintaining efficacy by reducing hematologic toxicity, particularly myelosuppression, through allowing bone marrow recovery between doses, while preserving cumulative tumoricidal radiation exposure. 21
In the alpha-emitter domain, 225Ac-J591 is an antibody-based PSMA-RLT that is currently being evaluated in the phase II CONVO1-alpha trial. In a first-in-human phase I dose-escalation study of 32 heavily pretreated mCRPC patients, 7 ascending dose levels (up to 93.3 kBq/kg) were tested: dose-limiting toxicity occurred in 1 of 22 patients at 80 kBq/kg, the maximum tolerated dose was not reached, and 93.3 kBq/kg was selected as the RP2D. 22 Treatment was generally tolerable, with grade ⩾3 adverse events predominantly hematologic, and preliminary efficacy signals included PSA50 responses in 47% (34% confirmed) of patients and a circulating tumor cell response in 59% of evaluable cases. 22
These ongoing studies reflect a rapidly expanding PSMA-RLT pipeline, with multiple scaffolds and isotopes under investigation across earlier disease settings. If successful, they may lead to regulatory approvals that enhance global access, increase therapeutic choice, and enable more individualized treatment strategies for patients with mCRPC.
Combination strategies with PSMA-RLT
Although 177Lu‑PSMA‑617 monotherapy has yielded measurable benefits in mCRPC, many patients eventually progress, with the resistance mechanisms to PSMA-RLT remaining poorly defined at present. Consequently, several studies are testing combinations of RLT with other systemic therapies as a means to potentially enhance antitumor efficacy.
PARP inhibition
The rationale for combining RLT with PARP inhibitors stems from the complementary mechanisms of action. Beta emissions from 177Lu generate single‑strand and double‑strand DNA breaks, and inhibiting PARP, an enzyme involved in repairing single‑strand DNA lesions, can potentiate radiation‑induced cell death. The phase I LuPARP trial (NCT03874884) evaluated 177Lu‑PSMA‑617 combined with the PARP inhibitor olaparib in biomarker-unselected patients with mCRPC. 23 This trial enrolled patients with mCRPC who had confirmed high PSMA expression, defined as SUVmax ⩾15 in at least one lesion and ⩾10 in additional lesions, with no discordant imaging findings between PSMA-PET and FDG-PET. Interim results demonstrated deep PSA responses, with 66% of patients achieving a 50% decline in PSA and 44% achieving a 90% decline, with five of seven patients with RECIST-evaluable disease having a partial response. 23
Immunotherapy
Despite the limited single-agent activity of immune checkpoint inhibitors in unselected mCRPC, early-phase studies suggest that combining RLT with immunotherapy may “prime” the tumor microenvironment. In the phase I PRINCE study (NCT03658447), patients received 7.4 GBq 177Lu-PSMA-617 every 6 weeks for up to six cycles alongside pembrolizumab 200 mg every 3 weeks for up to 2 years, demonstrating acceptable tolerability and preliminary signals of activity. 24 In a separate first-in-human phase I trial, a single priming dose of 7.4 GBq 177Lu-PSMA-617 was administered 28 days before initiation of pembrolizumab maintenance, yielding an objective response rate of 56% and a PSA50 response in a substantial proportion of patients. 25 Building on this, the phase II EVOLUTION trial (ANZUP2001) randomized mCRPC patients to receive either 177Lu-PSMA-617 alone or 177Lu-PSMA-617 in combination with ipilimumab and nivolumab. At 12 months, PSA-progression-free survival was 33% in the combination arm versus 17% in the 177Lu-PSMA-617-only arm, suggesting improved disease control with dual-checkpoint blockade. 26 However, the triplet approach was associated with increased immune-related toxicity, most notably checkpoint inhibitor-related myocarditis, leading to early trial discontinuation for some participants. 26 These findings highlight the potential of adding CTLA-4/PD-1 blockade to PSMA-targeted RLT and also the need for careful patient selection and vigilant toxicity monitoring. Further definitive studies will be required to clarify long-term benefits and optimal sequencing of these combination strategies.
Androgen receptor pathway inhibitors
ARPIs remain the cornerstone of systemic therapy in metastatic PCa. Preclinical studies have demonstrated that AR blockade can upregulate PSMA expression, thereby potentially enhancing the therapeutic efficacy of PSMA‑targeted RLT. 27 The phase II ENZA‑P trial (NCT04419402) has reported results comparing the efficacy of 177Lu‑PSMA‑617 plus enzalutamide versus enzalutamide alone in mCRPC, demonstrating potential evidence of significant clinical benefit with the combination of an ARPI and 177Lu-PSMA-617. In this randomized trial of 162 patients with mCRPC and risk factors for early progression on enzalutamide, the combination of 177Lu-PSMA-617 and enzalutamide significantly improved PSA progression-free survival (median 13.0 vs 7.8 months; HR = 0.43, p < 0.0001) as well as OS (median 34 vs 26 months, HR = 0.55, p = 0.0053) compared to enzalutamide alone. 28 Notably, this was in a population of patients who had generally not been treated with a prior ARPI, though ~50% had received prior docetaxel for mHSPC. The safety profile was acceptable, with grades 3–5 adverse events occurring in similar proportions between treatment groups (40% combination vs 41% enzalutamide alone). 28 Post hoc exploratory analyses from the VISION trial also suggested better outcomes in mCRPC patients randomized to 177Lu-PSMA-617 who also received a concurrent ARPI, 13 and there is an ongoing randomized phase II trial to evaluate 177Lu-PSMA-617 with or without an ARPI in first-line mCRPC (NCT05849298).
Other combinations
Dedicated chemotherapy combination trials include the phase I/II LuCAB study (NCT05340374), which evaluates cabazitaxel (12.5–20 mg/m2 on days 2 and 23) plus 7.4 GBq 177Lu-PSMA-617 every 6 weeks to establish safety, RP2D, and preliminary efficacy in mCRPC patients, 29 and the phase Ia/Ib LuCarbo trial (NCT06303713), combining carboplatin with 177Lu-PSMA-617 in a 42-day cycle to define RP2D and assess tolerability. 18 Other ongoing studies include the combination of PSMA-RLT with Ra-223 (Alphabet; NCT05383079) as well as combination of alpha- and beta- PSMA-RLT with 225Ac-J591 and 177Lu-PSMA-I&T (NCT04886986).
Novel isotopes and radioligands in development
Next‑generation innovations are focused on enhancing the potency of RLT through alternative radionuclides, improved ligand design, and antibody‑based constructs.
Alpha‑emitters (225Ac)
Alpha-emitters such as 225Ac deliver multiple high-energy alpha particles with very short range (approximately 50–100 µm), resulting in ultra-high linear energy transfer (i.e., high energy deposition density over a short distance). Early clinical series with 225Ac‑PSMA‑617 have reported PSA declines in a high proportion of patients, including those who had received prior 177Lu‑PSMA‑617, with overall rates of any PSA decline reaching up to 87% and PSA50 responses in approximately 66% of patients.30,31 The large multinational WARMTH-Act study has provided robust evidence for 225Ac-PSMA-RLT efficacy, reporting outcomes from 488 men with mCRPC treated across 7 centers worldwide. 32 This retrospective analysis demonstrated a median OS of 15.5 months and median progression-free survival of 7.9 months with 225Ac-PSMA-RLT, with 73% of patients achieving any PSA decline, and 57% experiencing PSA reductions of 50% or greater. Notably, 225Ac-PSMA RLT showed activity even in heavily pretreated patients, including in patients who had received prior 177Lu-PSMA-617 therapy. The major dose-limiting toxicity was xerostomia, reported in 68% of evaluable patients after the first cycle, with all patients receiving >7 cycles affected; grade ⩾3 hematologic adverse events included anemia (13%), thrombocytopenia (7%), leukopenia (4%), and renal toxicity (5%), while no treatment-related deaths occurred. 32
While supply constraints may limit widespread therapy, production initiatives are underway. The development of 225Ac‑PSMA‑I&T and 225Ac‑J591 (an anti‑PSMA antibody, see earlier section) are further broadening availability. For instance, a phase I dose‑escalation study of fractionated 225Ac‑J591 in mCRPC has presented promising early results showing acceptable hematologic toxicity and encouraging PSA responses. 33 There are also large phase III trials of 225Ac-PSMA-617 in both the first-line mCRPC setting (AcTFirst, NCT06855277) and post-177Lu-PSMA-617 setting (PSMAcTION, NCT06780670) currently underway.
Terbium‑161
161Tb is a β‑emitter similar to 177Lu but has the additional feature of emitting low‑energy Auger electron. Unlike β-particles, which traverse cells and tissues over distances of millimeters, Auger electrons deposit energy over an exceptionally limited spatial range of 2–500 nm in biological tissue—a distance comparable to the diameter of a single cell nucleus, making them particularly useful for destroying micrometastases. Preclinical studies demonstrate that 161Tb induces two to three times greater DNA double-strand break density compared to 177Lu in PCa models, particularly in submillimeter tumor clusters and single-cell metastases. 34 This “micro-dosimetry” effect suggests a theoretical gain in killing efficiency, particularly against micrometastases or small-volume tumor deposits where maximal precision and minimal collateral damage are necessary. 34
The VIOLET trial (NCT05521412) is a first-in-human study of 161Tb‑PSMA‑I&T in mCRPC. In this single-center phase I/II study, 30 mCRPC patients with PSMA-avid, FDG-negative disease received up to six cycles of 161Tb‑PSMA‑I&T at escalating doses (4.4, 5.5, and 7.4 GBq), with 7.4 GBq established as the recommended phase II dose. 35 Unlike 177Lu, 161Tb emits both beta and high-LET Auger/conversion electrons, yielding 2–4× higher subcellular radiation doses in micrometastatic models. Early clinical outcomes were promising, with PSA50 and PSA90 responses of 70% and 40%, respectively, a median rPFS of 11.1 months, and low rates of grade ⩾3 toxicity (7%). 35 These results highlight that further study of 161Tb in mCRPC is warranted.
Lead‑212 and Bismuth‑213
212Pb is particularly interesting for its potential in targeted alpha therapy. After decay, 212Pb gives rise to Bismuth‑213 (212Bi), yielding high‑energy alpha emissions. Early imaging and biodistribution studies with 212Pb‑PSMA ligands have shown promising results, with favorable tumor uptake and acceptable normal tissue clearance. Phase I trials (TheraPb, NCT05720130) are underway to define the safety and efficacy of 212Pb‑based PSMA inhibitors. 36 In parallel, the very short half‑life 213Bi has been explored experimentally; however, limitations in radionuclide half‑life complicate its use for systemic RLT delivery. 37
Antibody‑based and dual‑targeting approaches
Beyond small‑molecule ligands, antibody‑based RLT is emerging as a promising strategy. Radiolabeled monoclonal antibodies such as 225Ac‑J591 or 225Ac-pelgifatamab offer a different pharmacokinetic profile compared to small molecules and the potential for either single or fractionated dosing. Although full‑length antibodies may increase marrow exposure and prolong circulation, optimizing dosing schedules (e.g., fractionated, dose‑escalating regimens) can mitigate toxicity. 33 Meanwhile, dual‑targeting ligands that combine PSMA targeting with additional targets such as the gastrin‑releasing peptide receptor (GRPR) are in preclinical development. These bispecific agents may offer an advantage in patients with heterogeneous receptor expression, especially when PSMA loss is observed. 38
Preclinical data on 225Ac-pelgifatamab, an antibody conjugate using pelgifatamab with a novel chelator (Macropa), have demonstrated robust tumor targeting and antitumor efficacy in PSMA-positive cell lines and animal models. 39 This emerging platform represents a promising addition to the existing PSMA-617 and J591 therapies, reinforcing ongoing efforts to expand and improve RLT development.
Novel isotopes such as 67Cu, 90Y, and 47Sc are also being explored for RLT, though these remain largely at the preclinical stage. The evolution of optimized chelators and improved ligand modifications (e.g., albumin‑binding derivatives like 177Lu‑PSMA‑ALB‑56 and 177Lu‑EB‑PSMA‑617) promises enhanced tumor uptake and better dosimetry, albeit with a trade‑off in renal or marrow exposure that must be carefully balanced. 40 Three of the key isotope mechanisms are highlighted in Figure 1.

Summary of isotope mechanisms.
Future perspectives
Next‑generation RLT for advanced PCa is expected to evolve along several complementary lines (Figure 2). First, the integration of novel isotopes such as 225Ac, 212Pb, and 161Tb into clinical practice promises increased potency and the ability to overcome resistance observed with beta‑emitting agents like 177Lu‑PSMA‑617. It is anticipated that improvements in radionuclide production, such as reactor‑based or accelerator‑based methods, will mitigate current supply constraints. 41

Summary of future perspectives in RLT.
Second, the field is moving toward new molecular targets. Although PSMA remains the best validated target, other ones such as fibroblast activation protein (FAP) have emerged in early studies. FAP is expressed on cancer‑associated fibroblasts and may be particularly valuable in PSMA‑negative or mixed tumors. Early imaging studies have demonstrated substantial FAP uptake in advanced PCa, raising the prospect of dual‑targeting strategies or sequential use of PSMA‑ and FAP‑targeted RLT to overcome tumor heterogeneity. 42
Combination regimens represent another important future direction. Ongoing and planned clinical trials are exploring the optimal sequencing and combination of RLT with PARP inhibitors, immunotherapies, and ARPIs. Personalization of RLT through genomic profiling and development of rational therapeutic combinations (e.g., combination with PARP inhibitors in patients with BRCA-mutated tumors) and advanced imaging (e.g., leveraging artificial intelligence or machine learning approaches for more precise quantification of disease burden and metrics on PSMA-PET/CT scans) will likely guide adaptive dosing and fractionation schedules, aiming to move from fixed‑dose protocols toward personalized, dosimetry‑based treatments. 43
Expanding indications is another key area of research. There is active interest in moving RLT from the late-line mCRPC setting into earlier stages of disease. For instance, trials are enrolling patients with mHSPC to assess whether the addition of 177Lu-PSMA-617 to ADT and ARPIs can improve outcomes beyond ADT and ARPIs alone. Recently, the PSMAddition trial demonstrated a significant benefit in rPFS with the addition of 177Lu-PSMA-617 to standard-of-care (ADT + ARPI) in men initiating therapy for mHSPC, and we await further mature data on OS as well as longer-term toxicity from this trial. 44 Additionally, the phase II CONSOLIDATE trial, showed preliminary evidence of efficacy with the addition of 177Lu-PSMA-617 in patients with a suboptimal PSA response after 6 months of ADT plus docetaxel for mHSPC. 45
Other important ongoing studies further highlight this trend. The PSMA-DC trial (NCT 05939414) is a phase III, international, randomized study evaluating 177Lu-PSMA-617 versus observation after stereotactic body radiotherapy (SBRT) in men with biochemical recurrence of PCa. The trial is currently accruing and is designed to assess whether the addition of RLT can extend metastasis-free survival compared to standard management with SBRT in this early recurrence setting. 46 Meanwhile, the STAMPEDE-2 trial (NCT06320067) is investigating an accelerated dosing schedule of 177Lu-PSMA-617 in mHSPC, with more cycles delivered at shorter intervals (every 4 weeks) alongside ADT and ARPI. Early safety data from similar protocols show manageable toxicity, but definitive efficacy results—such as rPFS and OS—are still awaited. 47 These trials represent important steps in moving PSMA-RLT into earlier stages of PCa, with the potential to improve outcomes for broader patient populations. Similarly, neoadjuvant approaches such as the LuTectomy trial (NCT04430192) seek to downstage high‑risk localized tumors prior to definitive surgery, which could translate into better long‑term outcomes. 48
Advances in imaging and theranostics will underpin these improvements. Newer PSMA PET tracers such as 18F‑PSMA‑1007 and the use of total‑body PET scanners promise to enhance lesion detection and allow for more accurate patient‑specific dosimetry. 49 Furthermore, novel PET tracers targeting alternative pathways (e.g., GRPR‑targeted tracers) may complement PSMA imaging and provide improved characterization of tumor heterogeneity. 50 Integration of liquid biopsies and circulating tumor cell analyses may further refine patient selection and early detection of resistance mechanisms. 51
Potential challenges
RLT has demonstrated clinical effectiveness in advanced PCa, yet its widespread adoption faces significant global barriers. Production of essential radioisotopes such as 177Lu depends on a limited number of aging nuclear reactors located predominantly in high-income countries, creating supply vulnerabilities. Infrastructure gaps, especially in low- and middle-income regions, including access to cyclotrons, PET/SPECT scanners, and radiation-safe facilities hinder safe and effective RLT delivery. 52 Substantial financial requirements for establishing and maintaining nuclear medicine centers, alongside high radiopharmaceutical costs and inconsistent reimbursement, further restrict accessibility, as outlined by the Lancet Oncology Commission.53,54 Additionally, complex logistics, political factors, and supply chain disruptions exacerbate inequities in care. Practical hurdles such as the need for standardized protocols (e.g., dosing, imaging criteria), multidisciplinary collaboration, and reliable radionuclide production infrastructure must be addressed to expand global access.53,54
However, long-term hematologic safety remains a concern. While acute cytopenias (e.g., grade ⩾3 anemia in ~10%–20%, thrombocytopenia in ~4%–11%) are well documented in trials such as VISION, 13 PSMAfore, 15 and TheraP, 16 therapy-related myeloid neoplasms (tMNs; i.e., myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML)) have not thus far been reported, accepting that follow-up in these trials is relatively short. Nevertheless, MDS was seen in 2% of patients with neuroendocrine tumor treated with 177Lu-DOTATATE in the NETTER-1 trial, highlighting the importance of long-term follow-up to monitor for these late adverse effects.55,56 In a single-center case series of 381 mCRPC patients treated with 177Lu-PSMA, 5 (1.3%) developed tMN after a median of 33.6 months (range, 6–58.8 months) following initiation of therapy. 57 A case report described low-risk MDS with SF3B1 mutation diagnosed just 1 month after the sixth cycle of 177Lu-PSMA-617, while studies in analogous peptide receptor radionuclide therapy (PRRT) milieus noted MDS/AML rates of approximately 2%–3%, with latency windows spanning 15 months to several years. 58 Predisposing factors include prior alkylator therapy and high marrow radiation exposure, particularly in patients with diffuse bone metastases.
Another emerging issue is clonal hematopoiesis (CH), which refers to somatic mutations in hematopoietic stem cells that may increase the risk of tMNs. Studies in PRRT-naïve patients treated with 177Lu-DOTATATE have reported a CH prevalence rates of 35%–46%, with frequent mutations in genes such as DNMT3A and TET2. Importantly, the presence of CH has been significantly associated with persistent thrombocytopenia and other cytopenias following therapy. 59 Data in PCa cohorts treated with PSMA-RLT is emerging, with a clear trend that RLT may lead to development of new CH clones or expansion of existing CH clones, particularly in DNA-damage response genes such as PPM1D and TP53; the long-term implications of such clonal emergence or expansion is unclear, but will likely have more impact as RLT is moved earlier in the PCa disease spectrum where survival may be many years.60,61
Renal toxicity appears less frequent—short-term grades 3–4 nephrotoxicity is rare (<5%), though mild renal function decline is observed in a minority of patients—yet long-term data remain limited. 55 In a retrospective cohort of 106 mCRPC patients treated with 177Lu-PSMA-617, a moderate or severe decrease in estimated glomerular filtration rate (eGFR) at 1 year post-therapy was seen in 45% of patients, with nearly half of those (21.7% overall) experiencing severe or very severe eGFR loss; importantly, each additional baseline nephrotoxic risk factor was associated with a 4.51 mL/min/1.73 m2 greater eGFR decline. 62
In summary, although RLT is generally well tolerated in the short term, the potential for long-term hematologic and renal toxicities, including MDS and CH, highlights the importance of ongoing surveillance. This should include regular hematologic and renal monitoring and, where feasible, genomic profiling to identify patients at higher risk of late adverse effects.
Future research directions will focus on key questions such as which patients derive the most benefit from alpha‑ versus beta‑emitting therapies, if there is a role for sequencing these agents after each other, how best can RLT be integrated with newer hormonal agents or chemotherapeutic regimens, and whether biomarkers reliably predict response to RLT. Large, multicenter trials and collaborative research consortia are needed to answer these questions.
Conclusion
RLT has rapidly evolved from a niche experimental approach into a validated, clinically impactful modality in advanced PCa. The approval of 177Lu‑PSMA‑617 based on the VISION and PSMAfore trials has ushered in an era of theranostics whereby diagnostic imaging directs patient selection and personalized treatment planning.13,15 While current outcomes with beta‑emitting RLT demonstrate an absolute median survival improvement of 4 months (with 177Lu‑PSMA‑617 in the VISION trial), 11 these benefits are significant in the setting of heavily pretreated mCRPC and provide a foundation for further innovation.
Emerging next‑generation agents—including alpha‑emitting compounds such as 225Ac‑PSMA‑617 and 225Ac‑J591, as well as promising dual beta/Auger‑emitters like 161Tb—have the potential to further improve efficacy, particularly in patients with resistant or heterogeneous tumors. In parallel, combination regimens (such as with an ARPI, DNA damage repair pathway inhibitor, or immunotherapy) have shown some promise in early trials and may enhance outcomes compared to RLT monotherapy, and larger, randomized trials are needed to confirm these findings.
Looking ahead, the field will benefit from enhanced imaging techniques, advanced dosimetry, and personalized medicine approaches that allow clinicians to fine-tune therapy for each patient’s tumor biology. In addition, broadening the target portfolio beyond PSMA to include markers such as human kallikrein 2 (hK2)—an enzyme overexpressed in PCa and a promising diagnostic/prognostic biomarker—and six-transmembrane epithelial antigen of the prostate-2 (STEAP-2), which drives PCa progression and is highly expressed in aggressive disease, as well as FAP, holds the potential to address tumor heterogeneity and further expand the reach of RLT. Global efforts to standardize protocols, improve radionuclide production, and integrate multidisciplinary care will be essential if these therapies are to achieve widespread clinical impact.
In summary, RLT is reshaping the treatment landscape in advanced PCa and is at the forefront of precision oncology. By seamlessly integrating diagnostic imaging with targeted therapy, this strategy offers the promise of improved outcomes even for patients with limited treatment options. Over the next several years continued clinical trials, technological innovations, and strategic combination regimens are expected to further enhance the effectiveness and applicability of RLT, ensuring that more patients can benefit from this powerful therapy.
