Abstract
Background:
Alpha-fetoprotein (AFP) and protein induced by vitamin K absence or antagonist-II (PIVKA-II) are classical tumor markers in clinical practice. However, the relationship between tumor markers and prognosis in hepatocellular carcinoma (HCC) patients receiving immune checkpoint inhibitor (ICI) therapy remains unclear.
Objectives:
This study aims to explore the prognostic value of AFP and PIVKA-II in HCC patients treated with ICIs.
Design:
This study was a single-center, retrospective investigation aimed to assess the prognostic value of AFP and PIVKA-II in HCC patients receiving immune checkpoint inhibitors.
Methods:
This retrospective study included HCC patients who received ICIs treatment at Wuhan Union Hospital between July 2020 and March 2024. Serum AFP and PIVKA-II levels were collected before treatment. Patients were stratified into two groups based on AFP ⩾ 400 μg/L (yes = 1, no = 0) and PIVKA-II ⩾ 40 mAU/mL (yes = 1, no = 0). A total of 61% (114/186) of patients scored ⩽ 1, while 39% (72/186) scored 2. The objective response rate (ORR) and disease control rate (DCR) were calculated for both groups. Kaplan–Meier survival curves and Cox regression models were used to analyze overall survival (OS) and progression-free survival (PFS). Receiver operating characteristic curves were generated to demonstrate the predictive ability of combined independent prognostic factors for long-term survival.
Results:
The cohort consisted of 186 patients, divided into the low-risk group (n = 114) and the high-risk group (n = 72). Among all patients, 34.4% (64/186) achieved complete or partial response. Concurrent elevation of AFP and PIVKA-II was inversely associated with ORR (p = 0.012). The high-risk group exhibited significantly shorter OS (adjusted HR: 2.226 (95% CI: 1.410–3.513); p < 0.001) compared to the low-risk group. The integrated model combining AFP, PIVKA-II, and Barcelona Clinic liver cancer stage demonstrated moderate to good predictive capability for long-term risk stratification, with time-dependent area under the curves of 0.78 (9-month), 0.68 (12-month), and 0.63 (15-month).
Conclusion:
Concurrent elevation of AFP and PIVKA-II is significantly associated with shorter survival outcomes in HCC patients following ICI therapy.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is a malignancy arising from hepatocytes, making up approximately 75%–85% of all primary liver cancer. 1 HCC is the fifth most common cancer worldwide and a leading cause of cancer-related mortality. 2 The disease often arises in the context of chronic liver disease and cirrhosis. Hepatitis B (HBV) and hepatitis C (HCV) virus infections are the leading causes of HCC worldwide, involved in roughly 50% of cases. 3 Additional risk factors, including heavy alcohol consumption and non-alcoholic fatty liver disease (NAFLD), also contribute to the complex etiology of HCC. The management strategy for patients with HCC is guided by clinical stage, most commonly assessed using the internationally recognized Barcelona Clinic Liver Cancer (BCLC) staging system. For patients diagnosed with early-stage HCC, the primary curative treatment options involve either surgical resection (partial or total hepatectomy) or liver transplantation. In patients diagnosed with intermediate-stage HCC, locoregional therapies, such as transarterial chemoembolization (TACE), represent the preferred treatment modality. For those with advanced-stage disease who are no longer candidates for curative or locoregional therapies, systemic therapy remains the sole treatment option capable of providing a survival benefit.4,5 Due to its insidious onset, approximately 80% of HCC patients are diagnosed at advanced stages.6,7 Moreover, the frequent recurrence of HCC remains a persistent clinical challenge. In recent years, immune checkpoint inhibitors (ICIs), such as anti-programmed cell death protein 1 (PD-1), anti-programmed death-ligand 1 (PD-L1), and anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), have emerged as promising therapeutic options for advanced HCC. 8 The immune system employs checkpoint molecules, such as CTLA-4 and PD-1, to maintain self-tolerance and prevent autoimmunity. PD-1 is a key inhibitory receptor on the surface of T cells. Its interaction with its ligand PD-L1 expressed by tumor cells suppresses T cell activity by transmitting an inhibitory signal that dampens T-cell-mediated tumor cell killing. Thus, blocking the PD-1/PD-L1 interaction can restore the functionality of effector T cells and reactivate a pre-existing immune response against cancer. 9 By competing with the co-stimulatory receptor CD28 for binding to B7 ligands (B7-1/B7-2) on antigen-presenting cells (APC), CTLA-4, which operates early in T-cell activation, suppresses both APC and effector T-cell function. This mechanism ultimately aids in the escape from immune surveillance. Inhibition of CTLA-4 serves to restore costimulatory signaling and augment T-cell activation, which in turn fosters greater recruitment of CD4+ and CD8+ T cells into the tumor microenvironment (TME). 10 It is noteworthy that HCC is known to upregulate CTLA-4 expression on CD4+ T cells. 11 Furthermore, biopsy specimens obtained from HCC patients frequently demonstrate overexpression of PD-L1. Thus, there exists a strong biological rationale for the use of ICI therapy in patients with advanced HCC. However, the objective response rate (ORR) remains suboptimal, and identifying potential responders to ICI-based therapies continues to be a critical unmet need in clinical practice.
Serum tumor biomarkers are biologically active substances, including products of aberrant gene expression, that reflect the presence of malignancies. Their detection plays a critical role in cancer diagnosis, staging, and prognostic evaluation. In non-small cell lung cancer (NSCLC) patients, reduction in common lung cancer biomarkers—including carcinoembryonic antigen (CEA) and cytokeratin fragment 19—has been identified as a reliable predictive marker for immunotherapy response. 12 Therefore, serum biomarkers in HCC may similarly play a critical role in prognosis prediction. Alpha-fetoprotein (AFP) and protein induced by vitamin K absence/antagonist-II (PIVKA-II) are classical tumor biomarkers widely utilized in clinical practice. AFP, a specific diagnostic marker for primary hepatocellular carcinoma, holds significant value in monitoring therapeutic efficacy and assessing prognosis. Its overexpression is associated with more aggressive tumor behavior.13,14 Notably, AFP has been reported as a reliable monitoring biomarker for atezolizumab/bevacizumab combination immunotherapy.15,16 In HCC patients, elevated AFP levels serve as a risk factor for tumor recurrence after living donor liver transplantation (LDLT) and indicate poor prognosis. 17 Furthermore, AFP remains a key component of traditional prognostic scoring systems for HCC patients undergoing TACE therapy. 18 PIVKA-II, also known as des-gamma-carboxy prothrombin (DCP), demonstrates significant clinical value in HCC screening, early diagnosis, and prognostic evaluation.19,20 It is recognized as a strong predictor of HCC recurrence in patients undergoing hepatic resection or ablation therapy. 21 Combined detection with AFP enhances the early diagnostic accuracy for HCC. However, the relationship between AFP/PIVKA-II and prognosis in HCC patients receiving ICI therapy remains unclear.
Based on the above research findings, our study addresses this critical gap by investigating whether pre-treatment levels of AFP and PIVKA-II predict treatment response and survival outcomes in HCC patients treated with ICIs. First, we analyzed the characteristics of 186 patients. Second, we performed survival analyses for overall survival (OS) and progression-free survival (PFS) based on the AFP and PIVKA-II group. Thirdly, we analyzed the prognostic factors related to OS and PFS. Finally, we integrated the independent prognostic factors and graphically demonstrated their predictive ability for long-term survival using receiver operating characteristic (ROC) curves.
Materials and methods
Approval for this retrospective cohort study was obtained from the local ethics committee and the institutional review board of Tongji Medical College, Huazhong University of Science and Technology (Institutional Review Board Number S188). The study was conducted in accordance with the ethical guidelines of the Declaration of Helsinki. Informed consent was waived by the ethics committee due to the retrospective design of the research. The reporting of this study conforms to the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) statement. 22 We have completed the TRIPOD checklist, which is provided as a Supplemental File.
Study design and patient selection
This study is a single-center, retrospective investigation. Patient data were retrieved from the hospital's electronic medical record system. The cohort comprised HCC patients consecutively enrolled for ICI therapy at Wuhan Union Hospital between July 2020 and March 2024. Inclusion criteria: (1) age ⩾ 18 years; (2) HCC diagnosis confirmed by histopathology or imaging according to the European Association for the Study of the Liver (EASL) guidelines 23 ; (3) BCLC stage B or C. Exclusion criteria: (1) absence of baseline imaging records prior to ICI therapy; (2) missing baseline serum AFP or PIVKA-II levels before ICI initiation; (3) lack of follow-up imaging assessments post-ICI treatment.
Procedures
Retrospective information was obtained from the medical records of the patients, including the following: demographic data (age, gender, body mass index (BMI, kg/m2), cardiovascular, hypertension, smoking history, alcohol consumption history, Eastern Cooperative Oncology Group (ECOG) status); date of first immune therapy; date of HCC recurrence and the last follow-up or death; Child-Pugh grade; laboratory data (AFP, PIVKA-II, neutrophil/lymphocyte ratio, platelet/lymphocyte ratio); pre-treatment imaging data (number of lesions, size of the largest lesion (cm), lymph node metastasis). The initial admission and discharge records of the patient provided all baseline data.
Follow-up and endpoints
Follow-up data collection integrated outpatient/inpatient clinical records and telephone follow-up assessments. The observation period continued until July 2024 or until patient death. OS was the primary outcome, and PFS was the secondary outcome. OS is defined as the period from ICI initiation to death from any cause or last follow-up. The duration of PFS is measured from the first ICI therapy to progressive disease (PD), death, or last follow-up. Treatment response was evaluated using computed tomography (CT) or magnetic resonance imaging (MRI) according to the modified Response Evaluation Criteria in Solid Tumors (mRECIST).24,25 Responses were stratified into four categories: complete response (CR), partial response (PR), stable disease (SD), and PD. The ORR represented CR/PR cases, and the DCR covered CR/PR/SD cohorts.
Statistical analysis
In this study, continuous variables with normal distribution were expressed as mean (SD) with standard deviations and compared using the t-test. Non-normally distributed variables were reported as median (IQR) and analyzed via the Mann–Whitney U-test. Categorical variables were summarized as numbers (%) and evaluated using Fisher's exact test or Pearson's chi-square test, as appropriate. Survival analyses for PFS and OS were performed using the Kaplan-Meier survival curves assessed by the log-rank test. Univariate and multivariate Cox regression models were employed to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) for PFS and OS, identifying independent prognostic factors. Subgroup analyses were visualized through forest plots to display the HRs for PFS and OS in each subgroup. Independent prognostic factors identified by Cox regression were integrated to construct a multivariable model. Time-dependent ROC curves were generated, and the area under the curve (AUC) was calculated to demonstrate the predictive ability of this model for long-term survival. Time points (9/12/15 months) were selected based on median OS. Decision curve analysis (DCA) was used to assess the net benefit of the model in clinical decision-making. Calibration curves were generated to evaluate the accuracy of the nomogram-predicted survival probabilities against actual observations. All analyses were conducted using SPSS version 27.0 (IBM, Chicago, IL, USA) and R statistical software version 4.5.0 (R Foundation for Statistical Computing, Vienna, Austria). A two-sided p-value < 0.05 was considered statistically significant.
Results
Patient characteristics
This analysis included 186 HCC patients treated with ICIs. Patients were stratified into low-risk (score ⩽ 1, n = 114) and high-risk (score 2, n = 72) groups based on predefined thresholds: AFP ⩾ 400 μg/L (yes = 1, no = 0) and PIVKA-II ⩾ 40 mAU/mL (yes = 1, no = 0). The baseline clinical characteristics of the cohort are summarized in Table 1. The median age of the cohort was 57.0 years (range, 28–84 years), with preserved liver function observed in the majority of patients: 89.8% (167/186) were classified as Child-Pugh A, and the remaining 18 patients as Child-Pugh B. Compared to the low-risk group, the high-risk group exhibited significantly larger maximum tumor diameter (10.0 cm (IQR 6.1–13.3), p < 0.001) and a higher proportion of BCLC stage C patients (68.1% vs 41.2%, p < 0.001).
Baseline characteristics of patients.

BCLC, Barcelona Clinic Liver Cancer; BMI, Body mass index; ECOG, Eastern Cooperative Oncology Group; NLR, Neutrophil to lymphocyte ratio; PLR, Platelet to lymphocyte ratio.
Tumor response
Tumor response outcomes for both groups are detailed in Table 2. The ORR was 20.8% in the high-risk group versus 43.0% in the low-risk group, and the DCR was 68.1% and 71.1%, respectively. Concurrent elevation of AFP and PIVKA-II was inversely associated with ORR (p = 0.012). However, no statistically significant differences were observed between the groups and DCR (p = 0.812).
Tumor response between the high-risk group and low-risk group.

CR, complete response; DCR, disease control rate; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
Significant values are given in bold.
Survival analysis
At the time of analysis, a total of 88 patient deaths had occurred. The detailed clinical outcomes of the cohort are provided in Supplemental Table S1. With a median follow-up of 14.2 months, the median OS for the entire cohort was 12.9 months (95% CI, 12.3–14.5), with 6-month, 1-year, and 18-month OS rates of 91.9%, 71.5%, and 54.3%, respectively. The median PFS was 8.4 months (95% CI, 6.1–9.9), with corresponding 6-month and 1-year PFS rates of 57.5% and 39.8%. The detailed data for median OS, median PFS, and survival rates for both groups are provided in Supplemental Table S2. Kaplan–Meier survival curves demonstrated significantly shorter OS (HR: 3.05 (95% CI: 1.97–4.71); p < 0.001) and PFS (HR: 1.53 (95% CI: 1.07–2.18); p = 0.021) in the high-risk group compared to the low-risk group (Figure 1(a) and (b)).

Kaplan–Meier curve of OS (a) and PFS (b) in the high-risk group (red) and low-risk group (blue). Analyses were conducted using log-rank test.
Cox regression analysis and subgroup analysis
In univariate Cox regression analysis, score (risk stratification) and BCLC stage emerged as potential prognostic factors for OS. For PFS, score (risk stratification), age, maximum tumor diameter, and BCLC stage were identified as candidate predictors. All significant covariates from univariate analysis were included in multivariate Cox regression models.
Multivariate Cox regression analysis revealed that the high-risk group (adjusted HR: 2.226 (95% CI: 1.410–3.513); p < 0.001) and BCLC stage C (adjusted HR: 2.715 (95% CI: 1.573–4.684); p < 0.001) were independently associated with significantly shorter OS (Table 3). Similarly, BCLC stage C demonstrated a strong independent correlation with reduced PFS (adjusted HR: 2.383 (95% CI: 1.550–3.664); p < 0.001; Table 4).
Univariate and multivariate Cox proportional hazards analyses for OS.

BCLC, Barcelona Clinic Liver Cancer; BMI, Body mass index; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; NLR, Neutrophil to lymphocyte ratio; OS, overall survival; PLR, Platelet to lymphocyte ratio.
Significant values are given in bold.
Univariate and multivariate Cox proportional hazards analyses for PFS.
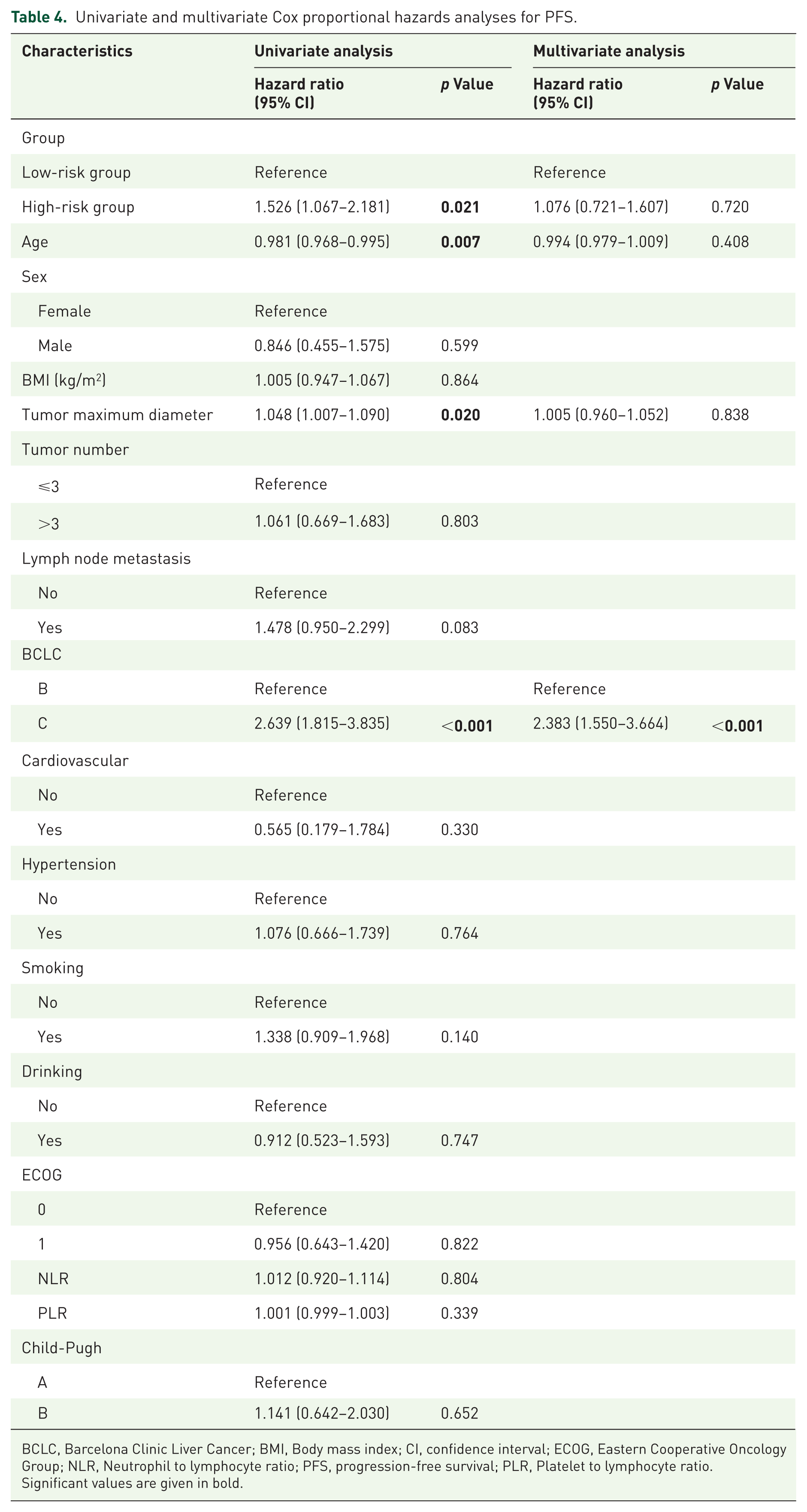
BCLC, Barcelona Clinic Liver Cancer; BMI, Body mass index; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; NLR, Neutrophil to lymphocyte ratio; PFS, progression-free survival; PLR, Platelet to lymphocyte ratio.
Significant values are given in bold.
We performed subgroup analyses, which demonstrated consistent stable outcomes in both PFS and OS. HRs for each subgroup were derived from univariate Cox regression models. In each PFS subgroup (Figure 2), in the male subgroup, ECOG status 0 cohort, lymph node metastasis-negative cohort, non-alcohol-consuming individuals, no smoking population, and non-hypertensive population, patients in the high-risk group exhibited significantly elevated progression risks compared to the low-risk group. In the OS subgroup analysis (Figure 3), with the exception of the female subgroup, lymph node metastasis-positive cohort, alcohol-consuming individuals, and Child-Pugh B subgroup, all other subgroups showed markedly higher mortality risks in the high-risk group versus the low-risk group.

Forest plot of subgroup analysis in progression-free survival between the high-risk group and low-risk group. Dashed line indicates hazard ratio of 1.

Forest plot of subgroup analysis in overall survival between the high-risk group and low-risk group. Dashed line indicates hazard ratio of 1.
The prognostic value of the independent prognostic factors
The multivariable model incorporated independent prognostic factors identified by Cox regression: AFP ⩾ 400 μg/L, PIVKA-II ⩾ 40 mAU/mL, and BCLC stage C. The ROC curves demonstrate the predictive ability of the multivariable model for long-term survival in HCC patients receiving ICI therapy (Figure 4(a)–(c)). The 9-month AUC was 0.78 (95% CI: 0.71–0.85), the 12-month AUC was 0.68 (95% CI: 0.58–0.78), and the 15-month AUC was 0.63 (95% CI: 0.47–0.79). DCA confirmed the clinical net benefit of the model, and calibration curves showed good agreement between predicted and observed survival probabilities (Figure 4(d)–(i)). These findings indicate that the multivariable predictive model combining the group and BCLC stage can forecast long-term survival, thereby significantly enhancing decision-making efficacy and providing clinicians with more reliable guidance for risk stratification and individualized treatment planning.
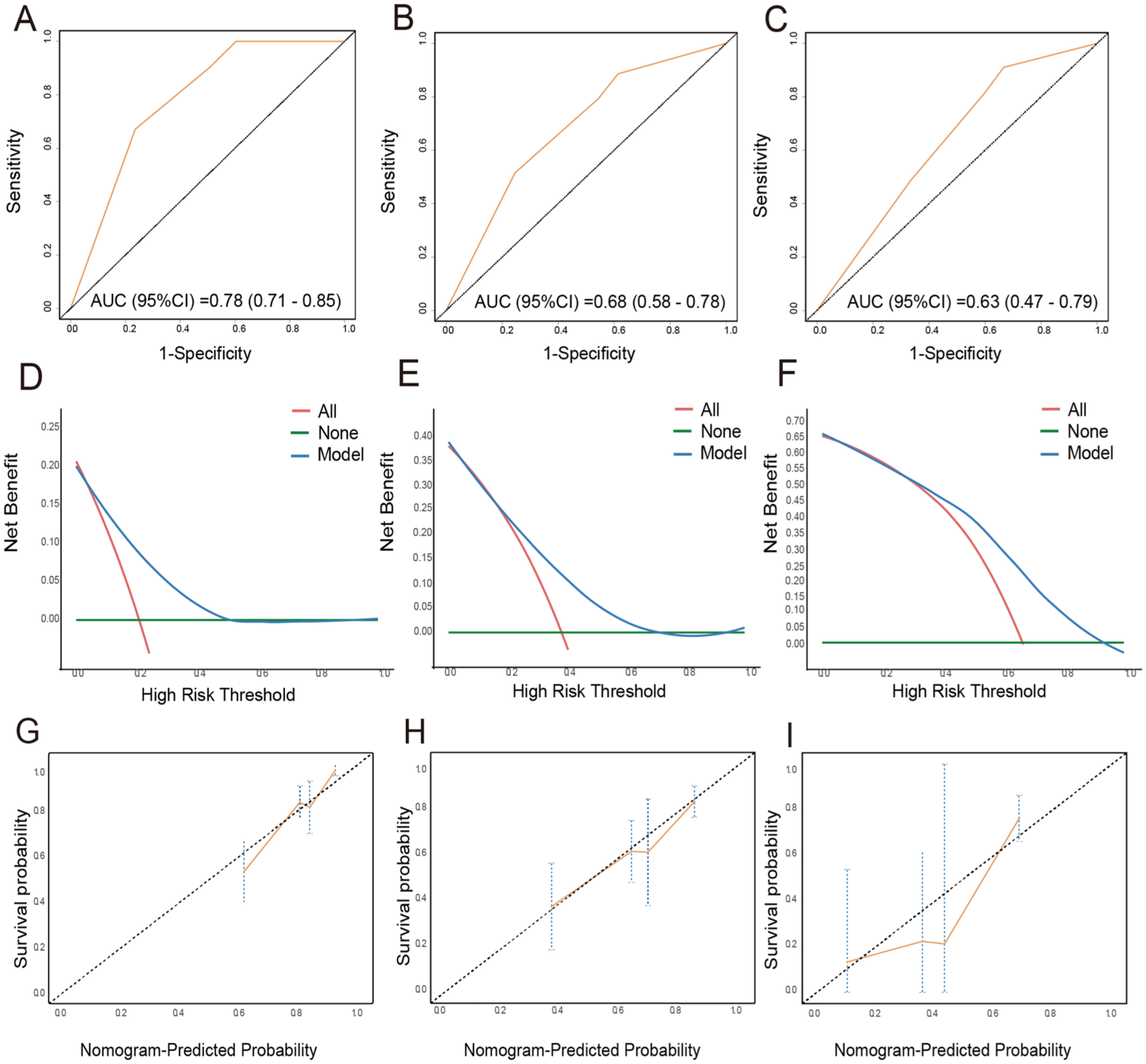
ROC curves of the multivariable predictive model combining the group and BCLC stage at 9-month survival rate (a), 12-month survival rate (b), and 15-month survival rate (c) for overall survival. DCA of the multivariable predictive model combining the group and BCLC stage at 9-month survival rate (d), 12-month survival rate (e), and 15-month survival rate (f) for overall survival. Calibration curve of the multivariable predictive model combining the group and BCLC stage at 9-month survival rate (g), 12-month survival rate (h), and 15-month survival rate (i) for overall survival.
Discussion
In this study, we demonstrated that HCC patients with elevated pretreatment serum AFP and PIVKA-II levels exhibited significantly poorer treatment responses and shorter survival durations following ICI therapy, which provided critical insights for identifying HCC patients less likely to benefit from ICIs, enabling clinicians to pursue timely therapeutic strategy modifications. To the best of our knowledge, this is the first study systematically investigating the association between AFP/PIVKA-II and both short-term efficacy (ORR/DCR) and long-term survival outcomes (PFS/OS) in ICI-treated HCC patients. Multivariable Cox regression and subgroup analyses were employed to delineate prognostic determinants. Three independent prognostic factors—AFP ⩾ 400 μg/L, PIVKA-II ⩾ 40 mAU/mL, and BCLC stage C—identified via Cox regression were incorporated into the final multivariable model. We generated ROC curves at 9/12/15 months to quantify the discriminative capacity of this integrated model for long-term survival prediction.
The potential of immunotherapy in advanced HCC is further highlighted by emerging strategies. A recent study by Karas et al. 26 demonstrated that the RORγt agonist AT7519 not only downregulated PD-1 and CTLA-4 expression in T helper 17 (Th17) lymphocytes but also significantly inhibited the proliferation, migration, and clonogenic capacity of HCC cells. Furthermore, AT7519 acted synergistically with the anti-PD-1 agent nivolumab, enhancing its antitumor efficacy against HCC. The finding suggests that combining AT7519 with either anti-PD-1 or anti-CTLA-4 regimens may provide additional therapeutic benefits. Dual blockade of PD-1 and CTLA-4 in B16 melanoma expands the infiltrating T cell population while diminishing regulatory T cells (Tregs) and myeloid-derived suppressor cells. 27 This combination strategy exerts a superior anti-tumor effect compared to either monotherapy, as antibodies targeting PD-1 and CTLA-4, whether alone or combined, induce distinct immunological profiles in vivo. 28 Collectively, these findings underscore the considerable therapeutic potential of ICI-based regimens in patients with advanced HCC.
The detection of serum biomarkers remains the preferred method for rapid clinical decision-making in primary hospitals due to its simplicity of operation, low cost, and high efficiency. Previous investigations have established AFP and PIVKA-II as predictive biomarkers for HCC recurrence and survival outcomes.29,30 Reductions in serum AFP and PIVKA-II levels have demonstrated utility in evaluating therapeutic response to hepatic arterial infusion chemotherapy (HAIC). 31 In patients with advanced HCC undergoing TACE, dynamic changes of AFP and PIVKA-II levels exhibit positive correlations with radiological response assessments. 32 Notably, patients achieving 50% reductions in AFP/PIVKA-II levels post-TACE show significantly improved 3-month prognoses. 33 These findings suggested that serum AFP and PIVKA-II levels may similarly correlate with treatment responsiveness in HCC patients receiving ICI therapy. Our study expands the clinical utility of these findings specifically to HCC patients receiving ICI therapy.
We employed AFP and PIVKA-II jointly to predict tumor survival prognosis because prior studies revealed no significant correlation between PIVKA-II and AFP, and PIVKA-II levels independently reflect tumor burden in HCC patients irrespective of AFP fluctuations.34 –37 This biological finding suggests that combined AFP/PIVKA-II assessment may exhibit complementary predictive capacity for ICI therapeutic efficacy, justifying their integration into a multivariable prognostic model. Cut-off values for AFP (400 μg/L) and PIVKA-II (40 mAU/mL) were based on clinical conventions. The underlying rationale may stem from the distinct mechanisms of origin between the two tumor biomarkers. AFP is a glycoprotein synthesized in the liver during the fetal stage, which declines rapidly to near-undetectable levels after birth. In healthy adults, AFP maintains minimal serum levels, but it becomes markedly elevated due to malignant hepatocytes regaining synthetic capacity.38,39 PIVKA-II is an aberrant byproduct generated through incomplete carboxylation of the prothrombin precursor during hepatic synthesis. Elevated serum PIVKA-II levels in HCC patients occur when hepatocytes undergo malignant transformation, downregulation of carboxylase gene expression, or intracellular accumulation of carboxylase inhibitors.40 –42 These mechanisms enable dual biomarker assessment to more precisely characterize tumor biological states. Furthermore, studies by Poté, Feng, et al. revealed through histopathological analysis that elevated AFP/PIVKA-II levels correlate with aggressive tumor features, including higher rates of microvascular invasion 43 and poor differentiation. 44 At the molecular level, AFP promotes tumor angiogenesis and may affect neoplastic proliferation and tissue growth. 13 These collective findings reinforce the biological rationale for the prognostic utility of these biomarkers in HCC. These studies further suggest that elevated AFP/PIVKA-II levels may serve as adverse prognostic factors, which aligns with our research findings. This consistency across independent cohorts underscores their potential utility in prognostic stratification frameworks.
In the multivariable Cox regression model, BCLC stage C emerged as an independent predictor for both OS and PFS. The BCLC classification system, endorsed by the EASL and American Association for the Study of Liver Diseases (AASLD) guidelines, serves as a critical framework for HCC patient stratification, therapeutic decision-making, and prognostic prediction. Consequently, BCLC staging demonstrates robust correlations with clinical outcomes in HCC patients.23,45 Additionally, we constructed a multivariable predictive model combining AFP, PIVKA-II, and BCLC stage to demonstrate their predictive ability for long-term survival. The model showed moderate to good predictive ability for overall survival (9-month AUC = 0.78, 95% CI: 0.71–0.85), suggesting its unique utility in long-term risk stratification. The integration of clinical stage with serum biomarkers significantly enhanced predictive accuracy beyond conventional staging systems. This clinically applicable stratification capability (AUC > 0.60 sustained over 15 months) may guide personalized surveillance intervals and adjuvant therapy allocation for HCC patients post-ICI treatment.
This study has several limitations. First, as a retrospective cohort study conducted at a single institution, the findings require validation through large-scale, prospective, multi-center investigations. However, the single-center design may also serve as a strength for ensuring consistent assessment for clinical and survival data during pre- and post-treatment phases. Second, this study has a limited sample size (n = 186), which may reduce the statistical associations between biomarkers and survival endpoints. Third, the number of female patients was limited compared to male patients, further investigation is needed with more female HCC patients undergoing immunotherapy.
Conclusion
In summary, our findings demonstrate that concurrent elevation of AFP and PIVKA-II is significantly associated with shorter survival outcomes in HCC patients following ICI therapy. Serial monitoring of serum AFP and PIVKA-II levels may aid in evaluating and predicting the prognostic of HCC patients treated with ICIs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251386801 – Supplemental material for Clinical significance of AFP and PIVKA-II for predicting prognosis in hepatocellular carcinoma patients treated with immune checkpoint inhibitors
Supplemental material, sj-docx-1-tam-10.1177_17588359251386801 for Clinical significance of AFP and PIVKA-II for predicting prognosis in hepatocellular carcinoma patients treated with immune checkpoint inhibitors by Shanshan Jiang, Yuyang Wang, Xiaona Fu, Jie Lou, Bingxin Gong, Weiwei Liu, Yi Li, Yuxin Sun, Chunfeng Liu, Zifang Song and Guofeng Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
