Abstract
Background:
Afatinib’s efficacy and dose-adjustment strategies have been validated in clinical trials and increasingly supported by real-world evidence.
Objectives:
This study aimed to provide additional real-world insights into afatinib use and to assess the impact of initial dosing and subsequent dose modifications on treatment outcomes and tolerability in patients with advanced epidermal-growth factor receptor (EGFR)-mutant non-small cell lung cancer (NSCLC).
Design:
We conducted a multicenter retrospective analysis of patients with advanced EGFR-mutated NSCLC who received first-line afatinib between April 2018 and June 2022.
Methods:
Patients were categorized into four subgroups based on their starting and optimal afatinib doses. The primary endpoints were to analyze the association between afatinib dosing and time-to-treatment failure (TTF), overall survival (OS), and toxicity.
Results:
A total of 343 patients were included. The most common starting afatinib dose was 30 mg (58.6%), followed by 40 mg (39.9%). The optimal dose maintained during treatment was 30 mg in 62.1% of patients, 40 mg in 33.2%, and 20 mg in 4.7%. Dose reductions due to toxicity occurred in 23.6% of cases. Regarding four subgroups: 53.1% of patients started and remained on <40 mg, 25.4% started and maintained 40 mg, 14.6% started at 40 mg but de-escalated to <40 mg, and 7.0% started <40 mg and escalated to 40 mg. After a median follow-up of 36.8 months, the median OS for all patients was 31.3 months (95% confidence interval: 29.3–33.1). Multivariate Cox regression analysis identified smoking status, Eastern Cooperative Oncology Group performance status, and EGFR mutation subtype as independent prognostic factors for OS. Notably, patients who initiated treatment at 40 mg but de-escalated to <40 mg had the longest median TTF and OS. In terms of safety, the highest incidence of adverse events was observed in this group, followed by those who started and remained at <40 mg.
Conclusion:
In this real-world cohort, flexible afatinib dosing—either by initiating at 40 mg followed by dose reduction or starting at <40 mg with later escalation—was associated with favorable survival outcomes and manageable tolerability. These findings support the use of individualized afatinib dosing strategies to optimize both efficacy and safety in patients with EGFR-mutant NSCLC.
Introduction
Lung cancer is the leading cause of cancer-related mortality worldwide. It is one of the most prevalent cancers, associated with high morbidity and mortality among newly diagnosed patients in Vietnam. 1 According to the WHO classification, there are two main histologic groups including small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC, accounting for approximately 85%). 2 Adenocarcinoma and squamous cell carcinoma are the two most common histological types of NSCLC. 2 In terms of race, epidermal-growth factor receptor (EGFR) and ALK driver mutations are more common in Asian than non-Asian populations,3–7 and these patients benefit from treatment with receptor tyrosine kinase-targeted inhibitors therapy. 8
Afatinib is a second-generation tyrosine kinase inhibitor (TKI) approved for the treatment of NSCLC harboring EGFR mutations, both globally and in Vietnam. Its efficacy has been well established through clinical trials and supported by real-world evidence.9–12
The most commonly reported adverse events (AEs) of afatinib involve the skin and mucosa. These AEs can significantly impact patients’ quality of life and often necessitate dose adjustments to optimize both efficacy and tolerability. Higher afatinib plasma concentrations are associated with an increased frequency and severity of AEs, often necessitating dose adjustments to improve tolerability.13,14 A study by Wind et al. 15 showed that age, ethnicity, smoking status, and hepatic function did not significantly affect the pharmacokinetics of afatinib, whereas female patients and those with low body weight exhibited increased drug exposure. 15
During clinical practice in Vietnam, many patients either begin treatment at a lower dose or start at 40 mg but require dose reductions during therapy due to toxicities. This is often influenced by factors such as female sex, poor general condition, low body weight, or the presence of comorbidities. Dose adjustments are made throughout the treatment course to determine the most tolerable and effective dose for each individual patient. This was also observed in the RealGiDo study, a large real-world investigation that assessed the impact of afatinib dose adjustments in routine clinical practice. Dose reductions were primarily prompted by adverse drug reactions and occurred more frequently in female patients, individuals of East Asian descent, and those with low body weight. 16
In our study, we analyzed the data across nine major hospitals specialized in lung cancer treatment in Vietnam. It aims to provide insights into real-world afatinib use and to evaluate the impact of starting dose and dose modifications on treatment outcomes and tolerability in patients with advanced-stage EGFR-mutant NSCLC.
Patients and methods
Patients
This retrospective analysis included advanced-stage NSCLC patients treated with first-line afatinib from April 2018 to June 2022. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines (Supplemental File). 17 The eligibility criteria were: (1) age over 18 years old; (2) histological diagnosis confirmed of NSCLC; (3) Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1, 2 or 3; and (4) confirmation of EGFR mutations (include common mutations (Del 19, L858R) and other uncommon EGFR mutations detected in the pretreatment biopsy specimens, mostly by polymerase chain reaction (PCR) methods or next-generation sequencing). Patients were excluded if they had severe hepatic impairment (Child-Pugh C), renal dysfunction with an estimated glomerular filtration rate below 15 mL/min/1.73 m2, significant comorbid conditions, other active malignancies, or a de novo T790M mutation.
Patients were to receive afatinib at nine Vietnamese cancer centers, including Bach Mai Hospital, Vietnam National Cancer Hospital, Ho Chi Minh City Oncology Hospital, Cho Ray Hospital, Thong Nhat Hospital, National Lung Hospital, 108 Military Central Hospital, Hanoi Medical University Hospital, and Hanoi Oncology Hospital. The physician determined the initial dose of afatinib based on patient age and ECOG PS. The assessment of treatment-related toxicity occurred every month using clinical tests and laboratory blood work. The clinical physician should evaluate dose adjustment for patients who begin with a 40-mg dose and develop severe toxicities. Dose adjustments due to toxicity were recorded within the first 6 months of treatment. Patients who show good tolerance to doses below 40 mg may receive a dose escalation to 40 mg. The optimal dose is defined as the dose adjusted based on AEs and maintained as the long-term dose throughout the course of treatment. It was determined primarily according to tolerability, rather than efficacy, and reflects the dose at which the patient experienced manageable toxicity without requiring further reduction or treatment discontinuation. The response to therapy was assessed every 3 months using abdominal-pelvic and/or chest computed tomography. If patients experienced deteriorating clinical symptoms, imaging evaluations were conducted promptly by clinical physicians. Afatinib was given until disease progression or occurrence of intolerable toxic effects.
Outcomes and evaluation
All patients were followed up until October 15, 2024, with endpoints assessing the association between afatinib dosing and time-to-treatment failure (TTF), overall survival (OS), and toxicity. TTF was defined as the duration from the initiation of treatment to discontinuation due to progression, death, or intolerable toxicity. OS was defined as the time interval between the date of starting afatinib and the date of death from any cause or last follow-up. The tumor response was divided into complete response, partial response, stable disease, and progressive disease according to RECIST 1.1. 18 AEs were assessed by retrospective chart review of laboratory tests and physical examination data during treatment and graded according to the National Cancer Institute Common Terminology Criteria for AEs (version 4.03; http://www.oncology.tv/SymptomManagement/NationalCancerInstituteUpdatesCTCAEtov403.aspx).
Information obtained from the medical records included patient’s characteristics (age, gender), smoking status, PS, and tumor characteristics (histological type, EGFR mutations). The patient’s PS was assessed according to the ECOG score.
Statistical analysis
The data were analyzed using R statistical software (version 4.1.0; The R Foundation for Statistical Computing, Vienna, Austria; http://www.r-project.org, accessed on May 18, 2021). Categorical variables were represented as percentages, and continuous variables were represented as a mean ± standard deviation or a mean with a range depending on the normal distribution of the variables. Chi-square and Fisher’s exact tests were used to assess the difference between categorical variables. Survival estimates were calculated using the Kaplan–Meier method. In subgroup analyses, survival was compared using the log-rank test, and hazard ratio was estimated by Cox regression. All variables included in the univariate analysis were subsequently entered into a multivariate Cox regression model to identify independent prognostic factors. A backward stepwise selection method was applied for variable selection. All statistical analyses were tested independently, and a statistically significant threshold was defined with a p-value of <0.05.
Results
Between April 2018 and June 2022, 343 patients with NSCLC harboring EGFR mutations were treated with afatinib (Figure 1).

Flowchart of the patients included in the study.
Patient characteristics
Patient characteristics are summarized in our previous report. 11 In the overall cohort, the mean age at the diagnosis was 63.2 ± 9.7 years, and 56.6% of patients were male. Most patients had a good PS at the time of diagnosis, with 93.0% having an ECOG PS of 0–1. The predominant histological type was adenocarcinoma (98%). Regarding common EGFR mutations in 251 patients (73.2%), we observed the Del19 mutation in 46.9% patients and L858R mutation in 26.2% patients. In our study, 87 patients (25.4%) had brain metastasis at the time of diagnosis.
We classified patients into four subgroups based on the starting and optimal doses of afatinib: those who started and remained on 40 mg; those who started at 40 mg but had their dose reduced to less than 40 mg; those who started and remained on a dose below 40 mg; and those who started below 40 mg but had their dose increased to 40 mg. Detailed patient characteristics for each subgroup are detailed in Table 1. There were no significant differences in baseline characteristics among these groups. In terms of treatment outcomes, although there was a trend toward higher overall response rates in the subgroup receiving a starting dose of 40 mg with an optimal dose less than 40 mg, no statistically significant differences were observed among the four subgroups (Supplemental Table 1).
Characteristics of patients according to subgroups of afatinib doses.

n (%).
Pearson’s Chi-squared test; Fisher’s exact test.
CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; PS, performance status.
Survival outcomes
The median follow-up duration was 36.8 months (95% confidence interval (CI): 34.3–39.1 months). With 210 events recorded, the median OS (mOS) for all patients was 31.3 months (95% CI: 29.3–33.1; Figure 2(a)). Univariate OS analysis based on patient characteristics revealed significant differences in mOS related to ECOG PS, smoking history, dose adjustments due to toxicity, and EGFR mutation type (p < 0.05; Table 2). The mOS was significantly longer in patients with ECOG PS 0–1 compared to those with ECOG PS ⩽2 (31.5 vs 17.8 months, p < 0.001). Non-smokers and former smokers had a significantly higher mOS compared to current smokers (32.5 vs 25.8 months, p = 0.0004). Patients with common EGFR mutations had a longer mOS than those with uncommon EGFR mutations (32.3 vs 27.6 months, p = 0.004; Figure 2(b)). Interestingly, patients who required dose adjustments due to afatinib-related toxicity had significantly longer mOS compared to those who did not require dose modifications (35.5 vs 30.1 months, p = 0.031). However, there were no significant differences in OS among subgroups based on different starting or optimal doses of afatinib.
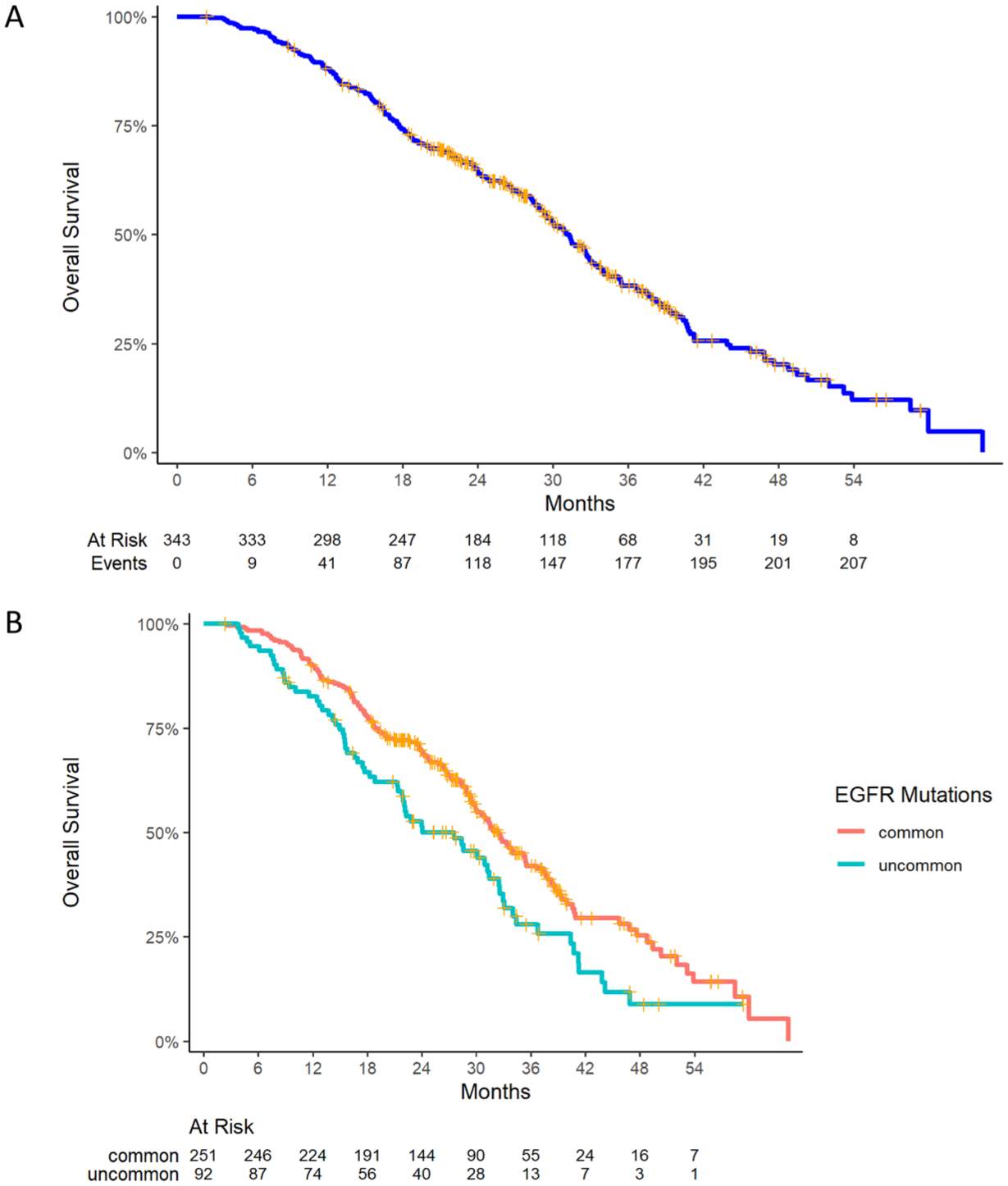
Kaplan–Meier estimates of OS. (a) All patients. (b) Subgroups by EGFR mutations.
Univariate analyses of OS according to subgroups.

Log-rank test. Bold values indicate statistical significance (p < 0.05).
CI, confidence interval; CNS, central nervous system; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; OS, overall survival; PS, performance status.
Similar to previous findings on median TTF (mTTF) reported in our earlier study, 11 patients with common EGFR mutations (Del19 or L858R) had significantly longer OS compared to those with uncommon EGFR mutations (32.4 vs 27.6 months, p = 0.004). However, there was no significant difference in OS between patients with EGFR Del19 and those with L858R (31.7 vs 35.4 months, p = 0.565). In multivariate Cox regression analysis, smoking status, ECOG PS, and EGFR mutation type were identified as independent prognostic factors for OS (Table 3).
Multivariate analyses of OS according to subgroups.
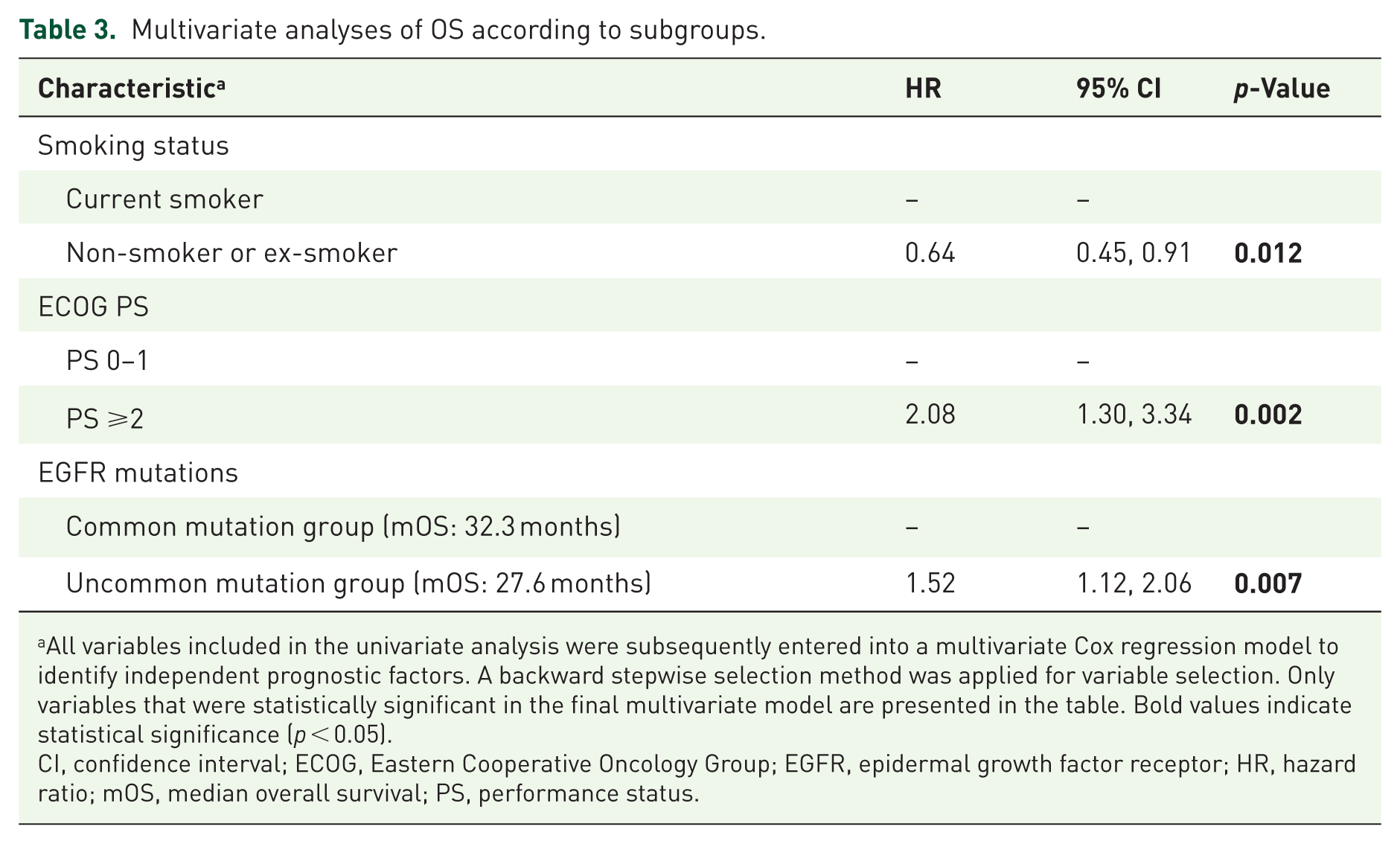
All variables included in the univariate analysis were subsequently entered into a multivariate Cox regression model to identify independent prognostic factors. A backward stepwise selection method was applied for variable selection. Only variables that were statistically significant in the final multivariate model are presented in the table. Bold values indicate statistical significance (p < 0.05).
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor; HR, hazard ratio; mOS, median overall survival; PS, performance status.
Our previous report indicated that the optimal dose and dose reductions of afatinib were independent factors affecting mTTF. 11 We further analyzed mTTF and mOS in relation to the four subgroups defined by starting and optimal doses of afatinib. For statistical comparison, the group with a starting dose of 40 mg and an optimal dose of 40 mg was used as the reference. p-Values for all subgroup comparisons are reported in Tables 4 and 5. Interestingly, patients who started with 40 mg but had an optimal dose <40 mg demonstrated a prolonged mTTF of 21.5 months, with a significant difference compared to the other three subgroups (p < 0.05). Conversely, patients who received both a starting and optimal dose of 40 mg had the shortest mTTF at 13.4 months (Table 4, Figure 3(a)). Regarding OS, patients who started at 40 mg but had an optimal dose <40 mg and those who started at <40 mg but reached an optimal dose of 40 mg had significantly longer mOS (37.9 and 38.3 months, respectively; Table 5, Figure 3(b)).
TTF according to starting and optimal doses.
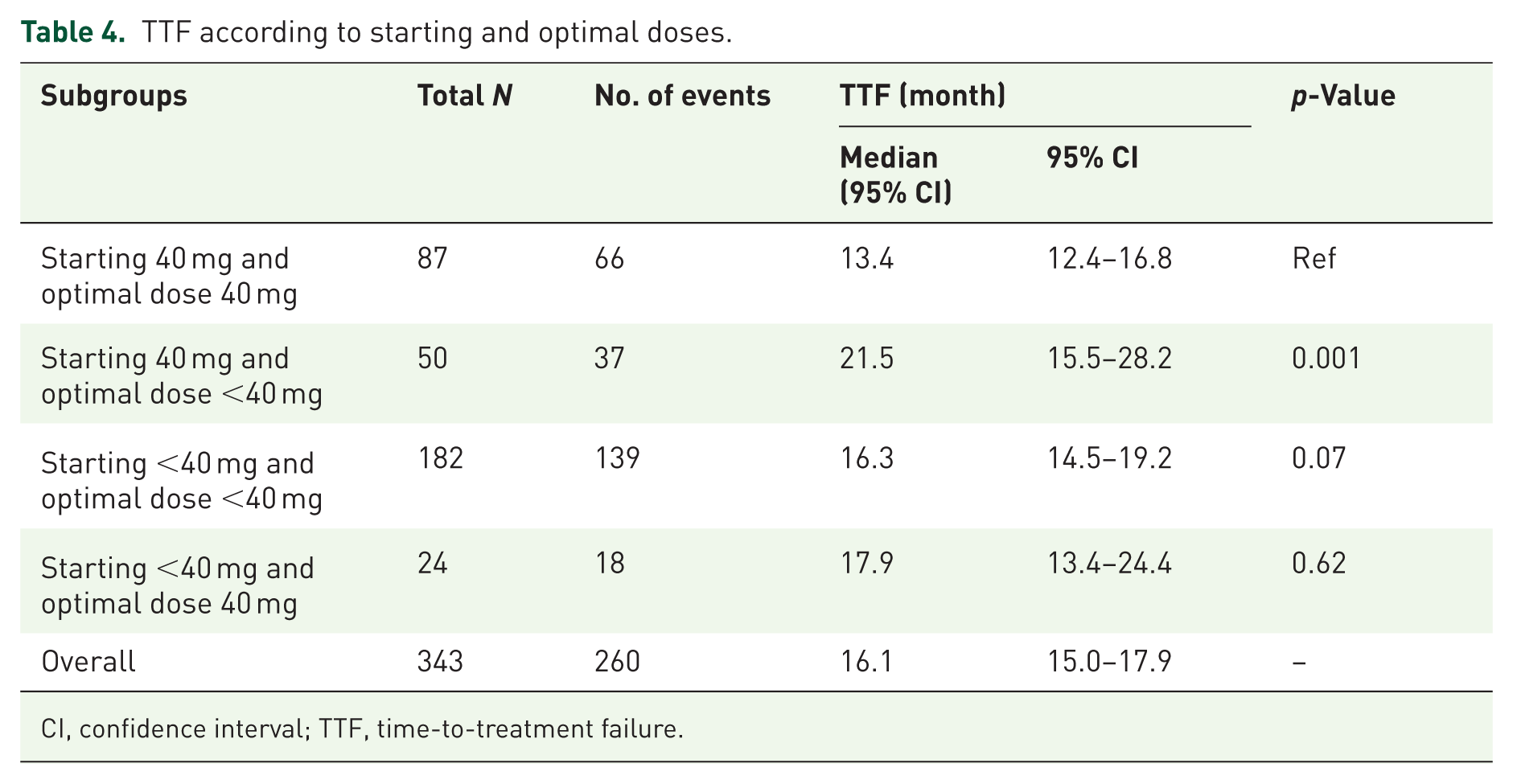
CI, confidence interval; TTF, time-to-treatment failure.
OS according to starting and optimal doses.

CI, confidence interval; NR, not reached; OS, overall survival.
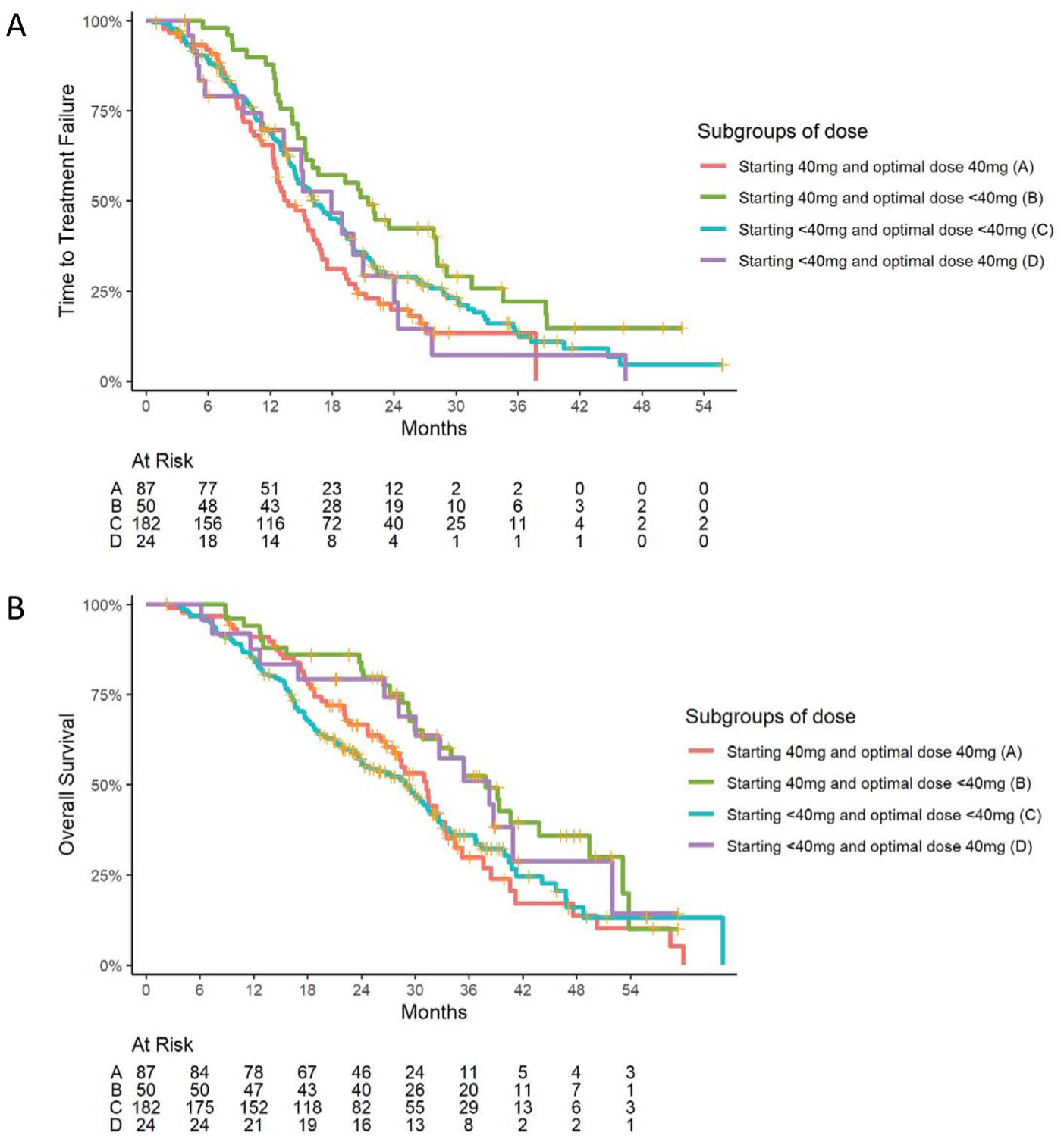
TTF (panel (a)) and overall survival (panel (b)) in relation to the four subgroups defined by starting and optimal doses of afatinib.
Toxicity
Treatment-related AEs of any grades were reported in 287 patients (83.7%). The five most common AEs were diarrhea, rash, paronychia, stomatitis, and dry skin. Grade 3 AEs were observed in 37 patients (10.8%), while no patients experienced grade 4 or 5 AEs. Dose reductions or interruptions due to AEs occurred in 13.6% of patients, and treatment discontinuation due to AEs occurred in four patients. Table 6 presents the most frequent treatment-related AEs according to starting and optimal dose subgroups. Overall, the incidence of AEs across all grades, as well as grade 3 AEs, was highest in the group that started at 40 mg but had an optimal dose <40 mg, followed by those who started and remained at <40 mg. Patients who started at <40 mg but had an optimal dose of 40 mg exhibited the most favorable safety profile in terms of toxicity. Regarding the five most common AEs, significant differences were observed in the incidence of paronychia and diarrhea among the four subgroups. The group with a starting dose of 40 mg and an optimal dose <40 mg exhibited the highest toxicity rates (p < 0.05).
Most common treatment-related AEs.

n (%).
Pearson’s Chi-squared test; Fisher’s exact test.
There was no grade 4 AE.
AE, adverse event; GOT, Glutamate Oxaloacetate Transaminase; GPT, Glutamate Pyruvate Transaminase.
The most common AEs according to optimal dose and starting dose subgroups are presented in Supplemental Tables 2 and 3, respectively. Grade 3 AEs were most frequently observed in patients who started treatment at 40 mg. Notably, a substantial proportion of these toxicities were also observed in the subgroup of patients with an optimal dose <40 mg, indicating that patients initiated treatment at 40 mg, experienced high-grade AEs, and subsequently underwent dose reduction. These findings support the real-world pattern of early onset toxicity prompting dose titration.
Discussion
In this real-world study, we analyzed data from 343 patients with advanced or metastatic NSCLC harboring EGFR mutations who were treated with first-line afatinib in Vietnam. The results are useful for the discussion of the most appropriate dosing strategies in the real-world practice. In comparison with other global real-world studies on afatinib, the baseline characteristics of our Vietnamese cohort were largely similar. The median age ranged between 60 and 65 years, and over 80% of patients had an ECOG PS of 0–1, reflecting a population with preserved functional status. However, a notable difference is that our study included 26.8% of patients with uncommon EGFR mutations, whereas some large-scale real-world studies of afatinib focused exclusively on patients harboring common mutations (exon 19 deletion or L858R).16,19
The recommended dose of afatinib for the initiation of treatment is 40 mg/day, but only 39.9% of patients received this dose at the initiation of the treatment, with the majority (58.6%) of patients receiving 30 mg. Interestingly, the objective response rate was significantly higher in the group receiving the full starting dose (83.9% vs 74.3%, p = 0.034), which may indicate that a higher initial dose may lead to earlier tumor shrinkage. 11 However, there was no statistically significant difference in mTTF between patients who were initiated at 40 mg and those who were initiated at lower doses (16.7 vs 16.9 months), which means that the long-term results may not be influenced by the dose at which the treatment was started but by the dose adjustment according to the tolerance of the patient. 11
Our study also emphasizes the importance of dose optimization during therapy. Within the first month of treatment, dose adjustments were made in 18.1% of patients based on tolerability, without compromising efficacy (mTTF 19.4 vs 16.1 months, p = 0.192). More notably, patients who experienced dose reductions due to toxicity had significantly longer mTTF (22.0 vs 15.7 months, p < 0.001). These findings support the concept of personalized dosing, in which adjusting afatinib based on patient tolerance may lead to improved outcomes—similar to the observations reported in the Japanese study by Nakamura et al., 20 where individualized dose titration resulted in extended mTTF and fewer AEs.
Importantly, 81 patients (23.6%) required dose reductions due to treatment-related toxicity. Surprisingly, patients who had dose reductions experienced significantly longer mTTF than those who did not (22.0 vs 15.7 months, p < 0.001). The international RealGiDo study showed that the mTTF and time to progression (TTP) were not impacted by a reduced starting dose or dose modification. 16 Similar findings were reported in studies from Asia, where early dose reductions due to toxicity did not compromise, and even improved, treatment duration due to better tolerability. A study by Nakamura et al. 20 showed a low-dose afatinib maintenance treatment with prolonged median progression-free survival (PFS) of 11.8 months. A study by Minegishi et al. 21 showed that old patients who underwent dose modification had significantly longer median PFS and OS compared to those who did not receive a dose adjustment. A study by Poh et al. 22 showed patients on lower doses of afatinib were twice as likely to achieve an objective response (odds ratio = 2.64; 95% CI: 1.20–5.83; p = 0.016), and although they had a numerically longer mTTF, the difference was not statistically significant. A post hoc analyses of the randomized LUX-Lung 3 and 6 trials showed that the median PFS was similar between patients who reduced their dose during the first 6 months and those who did not. 23 This is also true for Chinese subgroups from the LUX-Lung clinical trials and in real-world data from Taiwan.19,24
At a median follow-up of 26.2 months, the mOS for all patients was 31.3 months (95% CI: 29.3–33.1). Consistent with previous findings, patients with ECOG PS 0–1 had significantly longer OS than those with PS ⩾2 (31.5 vs 17.8 months, p < 0.001), and non-smokers or former smokers lived longer than current smokers (32.5 vs 25.8 months, p = 0.0004). Regarding EGFR mutation type, patients with common mutations (Del19 or L858R) had significantly longer OS than those with uncommon mutations (32.4 vs 27.6 months, p = 0.004). No significant difference was found between Del19 and L858R subtypes (31.7 vs 35.4 months, p = 0.565). Multivariate analysis confirmed ECOG PS, smoking status, and EGFR mutation subtype as independent predictors of OS. Importantly, patients who experienced dose reductions due to afatinib-related toxicity had significantly longer OS compared to those who did not (35.5 vs 30.1 months, p = 0.031), reaffirming that toxicity-guided dose reductions are not detrimental and may even prolong survival.
Patients were divided into four subgroups based on starting and optimal doses of afatinib. The subgroup that began at 40 mg but required titration to less than 40 mg achieved the longest mTTF at 21.5 months. The mTTF of patients who maintained a constant 40-mg dose was the shortest at 13.4 months. The two subgroups that underwent flexible titration achieved the best survival results for OS because they either received a dose reduction from 40 mg or an increase to 40 mg. To the best of our knowledge, the research presents the first recorded attempt to divide patients into four distinct subgroups for personalized treatment approaches. We acknowledge that this subgroup categorization is based primarily on how the results were analyzed rather than on pre-defined clinical criteria. Therefore, while the classification offers valuable insights into dose adjustment patterns and their association with outcomes, it should be interpreted as an exploratory analytical approach rather than a definitive clinical guideline.
Although survivor bias is a potential concern—where patients with longer survival may have more opportunities for dose adjustments—we note that most dose reductions occurred within the first month of afatinib treatment. In particular, the subgroup with the most favorable outcomes (both OS and TTF) consisted of patients who initiated treatment at 40 mg, experienced early dose reduction to 30 mg, and then tolerated this dose well throughout the treatment course, with minimal further adjustments. This pattern suggests that the impact of survivor bias in this setting is likely limited.
The research methodology holds special value because no real-world data exist to study dose adjustments in this level of detail. The four subgroups created by stratifying patients according to their initial dose and optimal dose enable researchers to study the distinct effects of dose modifications on treatment results. Our findings could significantly contribute to personalized treatment strategies in NSCLC, particularly in terms of dose titration and its impact on patient outcomes such as TTF and OS.
Among 87 patients with brain metastases, 14 (16.1%) received gamma knife radiosurgery and 24 (27.6%) received whole-brain radiotherapy in addition to afatinib. Brain metastases are generally associated with poor prognosis due to limited drug penetration across the blood–brain barrier. Nevertheless, dose reduction did not adversely affect mTTF in this subgroup, suggesting that afatinib retains intracranial activity, likely due to its better central nervous system penetration compared to first-generation EGFR-TKIs.
The most common AEs were skin and mucosal toxicities and grade ⩾3 toxicities were rare (3%–5%), and no cases of grade 4 toxicity was observed, indicating that afatinib is relatively safe in real-world clinical settings. The results of this study are lower compared to clinical trial findings,9,12 but are consistent with other real-world studies.19,22,24 Among the subgroups, those starting at 40 mg and titrating down had the highest incidence of all-grade and grade 3 AEs, while patients who started at <40 mg and titrated up to 40 mg had the most favorable safety profile. This is evident because toxicity was observed throughout the treatment. Patients who started at a 40-mg dose frequently experienced toxicity, which led to dose reduction to an optimal level of <40 mg. Consequently, this group had the highest incidence of AEs. According to a post hoc analyses of the randomized LUX-Lung 3 and 6 trials, reducing the dose of afatinib was linked to a lower incidence of treatment-related AEs and tended to occur more frequently in patients with higher plasma drug levels. On day 43, the geometric mean plasma concentration in patients who stepped down to 30 mg was 23.3 ng/mL, while it was 22.8 ng/mL in those who continued on 40 mg. 23 In our study, statistically significant differences were observed in paronychia and diarrhea rates, further supporting the strategy of starting lower and titrating up in selected patients. The results showed that patients who received a tolerable dose less than 40 mg experienced fewer and less severe AEs, which supports the importance of dose personalization.
Our subgroup analysis based on both starting and optimal doses provides further insight into the clinical feasibility of dose titration. The observation that grade 3–4 AEs were most common in patients who started at 40 mg and eventually reduced to an optimal dose below 40 mg suggests that toxicity typically occurred early in the treatment course. This highlights the utility of afatinib dose adjustment in real-world settings, where individualized dosing can help manage toxicity while maintaining therapeutic benefit. Such findings align with the rationale for flexible dose titration in EGFR-mutated NSCLC, especially in Asian populations known to experience higher rates of toxicity with standard-dose afatinib.
Most treatment-related toxicities in our cohort occurred within the first 1–2 months after afatinib initiation, with common AEs including diarrhea, rash, and mucositis. These were typically managed with supportive care, temporary interruption, or dose reduction, allowing continued treatment in the majority of patients. This emphasizes the importance of early monitoring and individualized dose adjustment to optimize tolerability. Although concomitant therapies may have influenced toxicity profiles or outcomes, detailed evaluation was not feasible due to the retrospective nature of the study and incomplete documentation of concurrent medications. We acknowledge this as a limitation and suggest that future prospective studies explore these aspects more comprehensively.
Overall, our results show that starting the treatment at lower doses or decreasing the dose of afatinib due to toxicity does not affect the treatment results. On the contrary, patients who had dose reductions tended to have longer mTTF and OS, which may be due to longer treatment duration and better adherence. Our data suggest that the dosing of afatinib should be individualized, and that the importance of flexibility according to tolerance should be stressed. Starting at 40 mg is acceptable for fit patients to maximize early tumor response, but subsequent dose reduction can sustain or even enhance long-term results and minimize toxicity. On the other hand, a lower starting dose with up-titration may be more advantageous for frailer patients or those with comorbidities to get good results with less side effects.
A limitation of our study is its retrospective design, where the starting dose selection was based on clinical decisions made by the physician. This introduces the potential for selection bias, as well as unmeasured confounding factors (e.g., patient frailty, comorbidities, or physician preferences) that may have influenced both dosing decisions and treatment outcomes. In our cohort, the initial afatinib dose was individualized, primarily based on patient age and ECOG PS. Patients with better PS (0–1) and younger age typically received the standard 40-mg dose, while those with impaired general condition or older age were often started on a lower dose to reduce early AEs. Moreover, the timing of AEs relative to dose adjustments was not captured, limiting our ability to assess toxicity specifically at the optimal dose. This real-world heterogeneity, while reflecting clinical practice, limits the ability to make definitive causal inferences about the optimal starting dose.
Another important limitation is the lack of plasma drug level monitoring. As this study focused on dose adjustment and tolerability, the absence of pharmacokinetic data limits our ability to directly correlate afatinib exposure with toxicity or efficacy. Future prospective studies with therapeutic drug monitoring may help clarify this relationship.
Additionally, the distribution of patients into the four dose-based subgroups was not uniform, limiting direct comparisons between groups. There is a need for real-world studies or clinical trials with larger datasets from the four subgroups, or meta-analysis from existing studies, to draw more objective and robust conclusions.
Conclusion
In this real-world cohort of EGFR-mutant NSCLC patients, flexible afatinib dosing—either starting at 40 mg with subsequent dose reduction or starting at <40 mg with escalation—was associated with improved survival and manageable tolerability. Dose adjustments due to toxicity were not detrimental and may be beneficial. These findings support individualized afatinib dosing strategies to optimize both efficacy and safety. In clinical practice, an initial dose of 40 mg may be considered for patients with good PS and minimal comorbidities, whereas a starting dose of <40 mg may be more appropriate for frail patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251382665 – Supplemental material for Optimizing afatinib dosage: enhancing treatment outcomes and minimizing toxicities in advanced EGFR-mutated non-small cell lung cancer patients in Vietnam
Supplemental material, sj-docx-1-tam-10.1177_17588359251382665 for Optimizing afatinib dosage: enhancing treatment outcomes and minimizing toxicities in advanced EGFR-mutated non-small cell lung cancer patients in Vietnam by Cam Phuong Pham, Hung Kien Do, Anh Tu Do, Tuan Khoi Nguyen, Thi Anh Thu Hoang, Tuan Anh Le, Dinh Thy Hao Vuong, Dac Nhan Tam Nguyen, Van Khiem Dang, Thi Oanh Nguyen, Van Luan Pham, Minh Hai Nguyen, Thi Thai Hoa Nguyen, Thi Bich Phuong Nguyen, Ha Thanh Vu, Thi Thuy Hang Nguyen, Van Thai Pham, Le Huy Trinh, Khac Dung Nguyen, Hoang Gia Nguyen, Cong Minh Truong, Tran Minh Chau Pham and Van Tai Nguyen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251382665 – Supplemental material for Optimizing afatinib dosage: enhancing treatment outcomes and minimizing toxicities in advanced EGFR-mutated non-small cell lung cancer patients in Vietnam
Supplemental material, sj-docx-2-tam-10.1177_17588359251382665 for Optimizing afatinib dosage: enhancing treatment outcomes and minimizing toxicities in advanced EGFR-mutated non-small cell lung cancer patients in Vietnam by Cam Phuong Pham, Hung Kien Do, Anh Tu Do, Tuan Khoi Nguyen, Thi Anh Thu Hoang, Tuan Anh Le, Dinh Thy Hao Vuong, Dac Nhan Tam Nguyen, Van Khiem Dang, Thi Oanh Nguyen, Van Luan Pham, Minh Hai Nguyen, Thi Thai Hoa Nguyen, Thi Bich Phuong Nguyen, Ha Thanh Vu, Thi Thuy Hang Nguyen, Van Thai Pham, Le Huy Trinh, Khac Dung Nguyen, Hoang Gia Nguyen, Cong Minh Truong, Tran Minh Chau Pham and Van Tai Nguyen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
