Abstract
Background:
Esophageal neuroendocrine carcinoma (ENEC) is a rare, aggressive malignancy with limited comparative data between Eastern and Western populations. Optimal management strategies remain unclear, and prognostic tools for risk stratification are lacking.
Objectives:
To analyze differences in clinicopathological features, treatment patterns, and survival outcomes between Chinese and American ENEC patients, and to develop a prognostic nomogram for unresectable disease.
Design:
A retrospective comparative cohort study.
Methods:
We analyzed 88 ENEC patients from a Chinese institution and 545 from the Surveillance, Epidemiology, and End Results (SEER) database. Demographic, tumor, treatment, and survival data were compared using Chi-square tests and Kaplan-Meier analysis. Cox regression identified prognostic factors for cancer-specific survival. A nomogram for unresectable ENEC was developed using SEER data and evaluated using C-index, calibration curves, and receiver operating characteristic analysis.
Results:
Significant population differences included age distribution (46–65 years: 51.1% Chinese vs 35.4% SEER, p < 0.001), tumor location (lower esophagus: 22.7% vs 62.2%), histology (small cell: 38.6% vs 76.9%), and metastatic presentation (M1: 28.4% vs 54.1%, all p < 0.01). Among non-metastatic patients, 77.8% Chinese underwent surgery versus 19.2% SEER (p < 0.001). Despite this treatment disparity, median survival was similar for surgical patients (48 vs 32 months, p = 0.93). Metastatic disease and age >65 were independent adverse prognostic factors in both cohorts. The Chinese cohort showed additional prognostic factors including tumor location and histology. The nomogram incorporating age, tumor location, N stage, M stage, and chemotherapy achieved a C-index of 0.725 with excellent calibration at 12 and 24 months.
Conclusion:
ENEC demonstrates distinct population-specific characteristics between Chinese and Western patients, with fundamental differences in treatment approaches but comparable surgical outcomes. The validated nomogram provides superior risk stratification for unresectable disease compared to traditional staging. These findings support population-tailored management strategies rather than universal treatment paradigms for ENEC.
Plain language summary
This study compares a rare but aggressive type of cancer called neuroendocrine carcinoma found in esophagus between Chinese and American patients. We analyzed data from 88 Chinese patients and 545 American patients to understand differences in disease features, treatments, and outcomes. We discovered significant differences between these groups. Chinese patients were generally younger and had tumors more often in the middle part of the food pipe, while American patients typically had tumors in the lower part. The cancer cells also looked different under microscopes in these two groups. Interestingly, Chinese doctors chose surgery much more often (about 78% of cases) than American doctors (only about 9% of cases) for patients whose cancer hadn’t spread. Despite this huge difference in treatment approach, patients who received surgery had similar survival times in both countries. We identified several factors that predict how long patients might survive, with cancer spread and patient age being important in both groups. For patients who couldn’t undergo surgery, we created a prediction tool that doctors can use to estimate survival chances and guide treatment decisions. Our findings suggest that carefully selected patients might benefit from surgery regardless of whether they’re from China or America, challenging current treatment approaches. This research helps doctors better understand this rare cancer and may improve how we treat patients from different backgrounds.
Keywords
Introduction
Esophageal neuroendocrine carcinoma (ENEC) represents a rare but highly aggressive subset of esophageal malignancies, characterized by neuroendocrine differentiation, rapid progression, and poor clinical outcomes. 1 With a global incidence of merely 0.04%–4.6% of all esophageal cancers, ENEC presents unique diagnostic and therapeutic challenges that remain inadequately addressed in current clinical practice guidelines. 2 The 2019 World Health Organization classification categorizes these tumors based on histomorphology and proliferation markers into well-differentiated neuroendocrine tumors (NET G1, G2, G3) and poorly differentiated neuroendocrine carcinomas (NEC), with the latter demonstrating particularly dismal prognosis with median survival often less than 12 months. 3
Despite significant advances in understanding more common esophageal malignancies, the molecular landscape, clinical behavior, and optimal management strategies for ENEC remain poorly defined. This knowledge deficit is particularly evident when comparing research outputs from Eastern and Western populations.4,5 Recent molecular profiling studies have suggested distinct genomic signatures between gastroenteropancreatic neuroendocrine neoplasms across different geographic regions, potentially reflecting differences in etiological factors, disease biology, and treatment responses.6,7 However, similar comparative analyses specifically for ENEC are conspicuously absent from the literature.
The epidemiological profile of ENEC exhibits notable variations across populations. While Western literature predominantly describes lower esophageal involvement with associations to Barrett’s esophagus and gastroesophageal reflux disease, reports from Asian countries suggest different anatomical predilections and potentially distinct risk factors.8,9 Two previous studies from China described a higher proportion of middle esophageal involvement and earlier tumor stage at diagnosis compared to Western cohorts, though these findings require validation in larger comparative analyses.10,11 These observed differences may reflect not only biological variability but also divergent screening practices, diagnostic capabilities, and healthcare access patterns.
Treatment approaches for ENEC remain heterogeneous and largely extrapolated from management paradigms for small cell lung cancer or non-neuroendocrine esophageal malignancies, rather than being evidence-based for this specific entity.12,13 Surgical resection, historically reserved for localized disease, shows variable utilization rates between Eastern and Western centers, with Asian institutions reporting higher rates of operative intervention.5,14 Meanwhile, the efficacy of systemic therapies, including platinum-based regimens, targeted agents, and immunotherapies, requires more rigorous evaluation across diverse patient populations.2,15
Prognostic stratification for ENEC patients remains challenging. Traditional staging systems designed for conventional esophageal cancers or small cell carcinomas may not adequately capture the unique biological behavior of ENEC. 16 Recent studies have attempted to identify reliable prognostic markers, including pathological features, tumor location, metastatic patterns, and molecular signatures, but findings have been inconsistent and rarely validated in independent cohorts.11,17 Furthermore, potential differences in prognostic factors between Eastern and Western populations have not been systematically investigated, leaving a significant gap in our understanding of this disease across different ethnic and geographic contexts.
Our study aims to address these critical knowledge gaps by conducting a comprehensive comparative analysis of demographic characteristics, clinicopathological features, treatment approaches, and prognostic factors between a Chinese ENEC cohort and patients from the Surveillance, Epidemiology, and End Results (SEER) database in the United States. We hypothesize that significant differences exist in disease presentation, management strategies, and prognostic determinants between these populations. Additionally, we seek to develop and validate a prognostic nomogram specifically for unresectable ENEC patients, a particularly vulnerable subgroup with limited therapeutic options and poorly characterized outcomes. This investigation not only enhances our understanding of geographical and ethnic variations in ENEC but also provides clinically relevant tools for risk stratification and treatment planning for this challenging malignancy.
Materials and methods
Study design and patient selection
This retrospective comparative study analyzed two distinct cohorts of patients with histologically confirmed primary ENEC: a Chinese cohort from a single academic institution and a Western cohort derived from a population-based cancer registry. The reporting of this study conforms to the STROBE statement (Supplemental File 1). 18
For the Chinese cohort, we identified patients diagnosed with primary ENEC at the First Affiliated Hospital of Zhejiang University between January 2007 and December 2022. Pathological diagnosis was established according to the 2019 World Health Organization classification criteria for neuroendocrine neoplasms, requiring characteristic morphological features (nested/trabecular growth pattern, salt-and-pepper chromatin) and positive immunohistochemical staining for at least two neuroendocrine markers (synaptophysin, chromogranin A, or CD56/NCAM) in >20% of tumor cells. 19 Proliferation index was assessed using Ki-67 immunostaining. Patients were excluded if they had secondary esophageal involvement from a non-esophageal primary NET, mixed histology with neuroendocrine component <30%, incomplete clinicopathological data, or loss to follow-up within 3 months of diagnosis without documented death.
The Western cohort was extracted from the SEER database (18 registries, November 2022 submission) using SEER★Stat software version 8.4.1 (https://seer.cancer.gov/seerstat/). To ensure temporal compatibility with the Chinese cohort while maintaining an adequate sample size, we restricted case selection to patients diagnosed between January 2004 and December 2020. Cases were identified using site codes C15.0–C15.9 (esophagus) combined with histology codes 8013/3 (large cell NEC), 8041/3 (small cell carcinoma), 8045/3 (combined small cell carcinoma), 8244/3 (mixed adenoneuroendocrine carcinoma), 8246/3 (NEC, NOS), and 8574/3 (adenocarcinoma with neuroendocrine differentiation) according to the International Classification of Diseases for Oncology, Third Edition (ICD-O-3).
Data collection and outcome measures
For the Chinese cohort, data were systematically extracted from electronic medical records by two independent investigators, with discrepancies resolved by consensus. Collected variables included demographic characteristics (age, sex), tumor-specific parameters (anatomical location, histological subtype, TNM classification according to the 8th edition of the American Joint Committee on Cancer staging system), presence and sites of distant metastases, treatment modalities (surgery, chemotherapy, radiotherapy), and survival outcomes. Pathological specimens were reviewed by two experienced pathologists specializing in gastrointestinal malignancies.
For the SEER cohort, we extracted comparable demographic, clinicopathological, treatment, and survival data using standardized SEER variables. Tumor location was categorized as upper third (cervical esophagus, C15.0), middle third (C15.1), lower third (C15.2, C15.5), overlapping (C15.8), or esophagus not otherwise specified (C15.9). TNM staging information was derived from the SEER-specific collaborative staging and AJCC staging variables. Metastatic sites were identified using relevant SEER site-specific metastasis codes.
The primary endpoint was cancer-specific survival (CSS), defined as the interval from initial diagnosis to death attributed to esophageal cancer or last follow-up. Patients who died from causes unrelated to esophageal cancer were censored at the date of death for CSS analysis.
Statistical analysis
Categorical variables were compared between cohorts using Pearson’s Chi-square test or Fisher’s exact test when appropriate. Survival curves were generated using the Kaplan-Meier method, with differences assessed by log-rank test. Median survival time with 95% confidence intervals (CI) was calculated for both cohorts and relevant subgroups.
Univariate Cox proportional hazards regression was performed to identify potential prognostic factors associated with CSS. Variables with p < 0.10 in univariate analysis or those with established clinical relevance were entered into multivariate Cox regression models using a backward stepwise selection procedure to identify independent prognostic factors. Hazard ratios (HR) with 95% CI were calculated for each variable. The proportional hazards assumption was verified using Schoenfeld residuals and log-minus-log survival plots.
All statistical analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, NY, USA) and R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria). Two-sided p values <0.05 were considered statistically significant.
Development and validation of prognostic nomogram
To enhance risk stratification for unresectable ENEC patients, we developed a prognostic nomogram using data from 250 non-surgically treated patients with complete clinical information from the SEER cohort. This unresectable cohort includes patients with metastatic (M1) disease and those with localized but unresectable tumors (e.g., advanced T4 or nodal involvement deemed inoperable per SEER treatment records). These patients were randomly divided into a training set (n = 167, 67%) and a validation set (n = 83, 33%) using computer-generated random numbers.
Variables were selected for the nomogram based on univariate and multivariate Cox regression analysis of the SEER training set (n = 167 unresectable patients), prioritizing both statistical associations and clinical relevance (e.g., established prognostic factors like age, tumor location, histology, N stage, M stage, and chemotherapy receipt). While some variables (e.g., histology) showed stronger significance in the Chinese cohort, inclusion was driven by SEER subgroup results and literature support to create a model superior to traditional TNM-based AJCC staging. The predictive performance of the nomogram was assessed using Harrell’s concordance index (C-index), with values ranging from 0.5 (no discrimination) to 1.0 (perfect discrimination). Calibration plots were generated to evaluate the agreement between predicted and observed survival probabilities at 12 and 24 months.
The model’s clinical utility was further evaluated using receiver operating characteristic curve analysis, with area under the curve (AUC) calculated for both the training and validation sets. Internal validation was performed using 1000 bootstrap resamples to assess potential model overfitting, while external validation was conducted using the validation set. Decision curve analysis was performed to determine the range of threshold probabilities for which the nomogram provides clinical net benefit.
The nomogram development and validation were performed using the “rms,” “survival,” “timeROC,” and “DecisionCurve” packages in R software (https://www.r-project.org/).
Results
Demographic and clinicopathological characteristics
Our analysis included 545 patients from the SEER database (Western cohort) and 88 patients from the First Affiliated Hospital of Zhejiang University (Chinese cohort) diagnosed with ENEC. Comprehensive demographic and clinicopathological characteristics are presented in Table 1.
Demographic and clinical characteristics of the SEER and Chinese cohort.
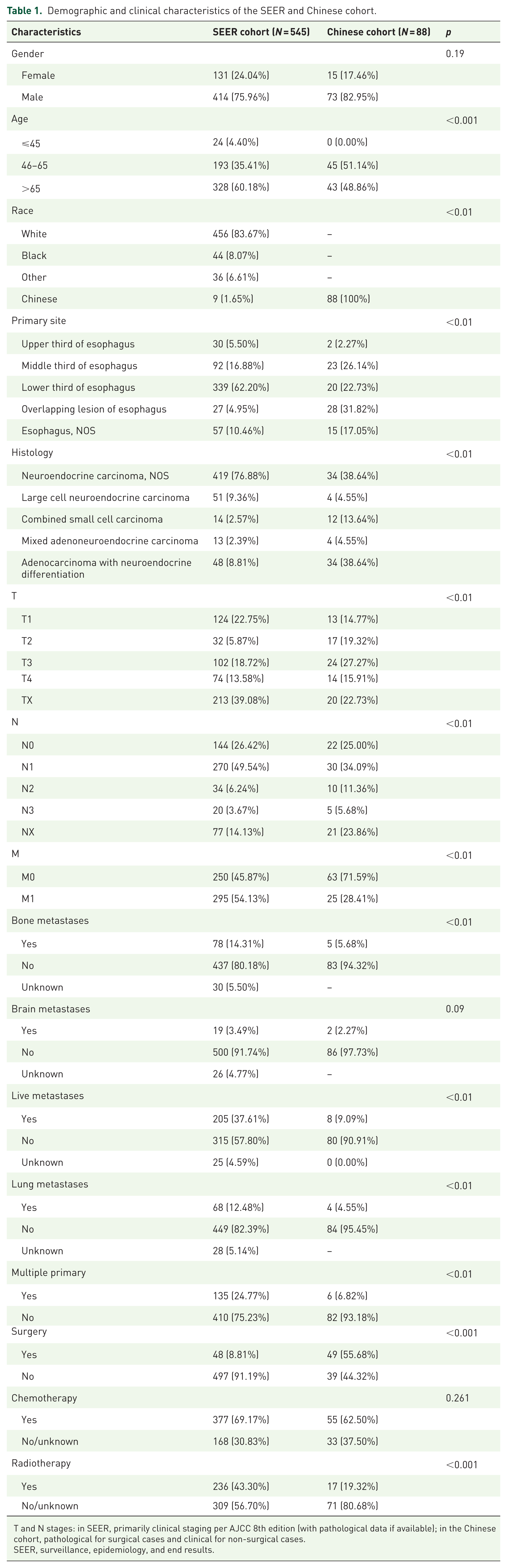
T and N stages: in SEER, primarily clinical staging per AJCC 8th edition (with pathological data if available); in the Chinese cohort, pathological for surgical cases and clinical for non-surgical cases.
SEER, surveillance, epidemiology, and end results.
The Chinese cohort demonstrated a male predominance (82.95% vs 75.96% in SEER, p = 0.19) and a significantly younger age distribution, with 51.14% of patients aged 46–65 years compared to 35.41% in the SEER cohort (p < 0.01). No Chinese patients were diagnosed at age ⩽45 years, while 4.40% of SEER patients fell into this category.
Striking differences were observed in tumor location between cohorts. Chinese patients exhibited a significantly higher proportion of middle esophageal tumors (26.14% vs 16.88%) and overlapping lesions (31.82% vs 4.95%), but substantially fewer lower esophageal tumors (22.73% vs 62.20%) compared to the SEER cohort (p < 0.01). This anatomical distribution divergence likely reflects distinct etiological factors between populations.
Histological classification revealed another marked disparity, with NEC, NOS (which predominantly includes small cell carcinoma cases), comprising 76.88% in the SEER cohort versus 38.64% in Chinese patients, while adenocarcinoma with neuroendocrine differentiation was significantly more prevalent in the Chinese cohort (38.64% vs 8.81% in SEER; p < 0.01). Mixed adenoneuroendocrine carcinoma and combined small cell carcinoma were more frequently diagnosed in Chinese patients (18.19% vs 4.96% in SEER).
Regarding disease extent, Chinese patients presented with earlier-stage disease across multiple parameters. The Chinese cohort showed higher proportions of T2 (19.32% vs 5.87%) and T3 (27.27% vs 18.72%) tumors compared to the SEER cohort (p < 0.01). Most notably, 71.59% of Chinese patients presented without distant metastases (M0) versus only 45.87% in the SEER cohort (p < 0.01). Specific metastatic sites also differed significantly between cohorts, with the SEER patients demonstrating higher rates of liver (37.61% vs 9.09%), lung (12.48% vs 4.55%), and bone (14.31% vs 5.68%) metastases (all p < 0.01).
Multiple primary tumors were significantly more common in the SEER cohort (24.77% vs 6.82% in Chinese patients, p < 0.01), suggesting potential differences in predisposition factors or surveillance practices.
Univariate and multivariate survival analyses
Univariate Cox proportional hazards regression analyses (Table 2) identified several factors associated with CSS in both cohorts. In the SEER cohort, advanced age, higher T stage, nodal involvement, distant metastasis, and presence of specific metastatic sites (bone, brain, liver, and lung) were significantly associated with poorer prognosis (all p < 0.05). For the Chinese cohort, older age, lower esophageal tumor location, non-NEC histology, advanced TNM stage, distant metastasis, and bone/liver metastases demonstrated significant negative prognostic impact.
Univariate analysis of patients in SEER and Chinese cohort.

HR, hazard ratios; NEC, neuroendocrine carcinoma; SEER, surveillance, epidemiology, and end results.
Multivariate Cox regression analysis (Table 3) revealed both shared and population-specific independent prognostic factors. In both cohorts, metastatic disease (M1) emerged as a powerful adverse prognostic indicator (SEER: HR = 1.96, 95% CI 1.44–2.67, p < 0.001; Chinese: HR = 49.74, 95% CI 21.75–113.75, p < 0.001), though with substantially greater magnitude in the Chinese cohort.
Multivariate analysis of patients in SEER and Chinese cohort.

CI, confidence interval; HR, hazard ratios; NEC, neuroendocrine carcinoma; SEER, surveillance, epidemiology, and end results.
Interestingly, younger age (46–65 years) conferred significantly better prognosis in both populations (SEER: HR = 0.69, 95% CI 0.57–0.85, p < 0.001; Chinese: HR = 0.29, 95% CI 0.13–0.66, p = 0.003). The Chinese cohort demonstrated more numerous independent prognostic factors, with tumor location, histology, T stage, N stage, and specific metastatic sites all independently affecting survival after multivariate adjustment.
Multiple primary tumors emerged as an independent adverse prognostic factor in the multivariate analysis of the Chinese cohort (HR = 0.13, 95% CI 0.03–0.60, p = 0.009), suggesting that patients without multiple primaries had better outcomes. This finding contrasted with the borderline significance observed in the SEER cohort (HR = 0.82, 95% CI 0.65–1.02, p = 0.075).
Comparative analysis of surgical intervention
A striking difference in initial treatment approach was observed between cohorts. Among non-metastatic patients (M0), 77.8% (49/63) of Chinese patients underwent surgical resection compared to only 19.2% (48/545) of the SEER cohort (p < 0.001), highlighting a fundamental divergence in therapeutic paradigms.
Despite this pronounced disparity in surgical utilization, median CSS for surgically treated patients was remarkably similar between cohorts (Chinese: 48 months, SEER: 32 months; HR = 0.97, 95% CI 0.53–1.78, p = 0.93; Figure 1(a)). Similarly, in non-surgically managed patients, median CSS showed no significant difference (Chinese: 6 months, SEER: 8 months; HR = 1.56, 95% CI 0.83–2.93, p = 0.17; Figure 1(b)).

Kaplan-Meier curve of cancer-specific survival of patients with (a) or without (b) surgery treatment. The prognostic value of T (c, d) and N (e, f) staging in SEER and Chinese cohorts, separately.
Subgroup analysis of surgically treated patients revealed consistent prognostic trends across both cohorts for T stage (Figure 1(c) and (d)) and N stage (Figure 1(e) and (f)), though the relatively small number of surgically treated patients in the Chinese cohort (n = 49) limits statistical power, and results should be interpreted with caution. Increasing T and N stages correlated with progressively worse prognosis. The prognostic impact of nodal status was particularly pronounced in the Chinese cohort (p = 0.041), where N0 patients achieved a median CSS of 35 months compared to just 12 months for N2 patients.
Prognostic nomogram for unresectable patients
To address the critical need for risk stratification in unresectable ENEC patients, we developed a prognostic nomogram using data from 250 non-surgically treated patients from the SEER cohort with complete clinical information. The training set (n = 167) and validation set (n = 83) demonstrated comparable baseline characteristics (data not shown).
The final nomogram incorporated five independent predictors: age, tumor location, N stage, M stage, and receipt of chemotherapy (Figure 2). The model demonstrated robust discriminative ability with a concordance index (C-index) of 0.725 (95% CI 0.648–0.802, p < 0.001) in predicting CSS.

Nomogram predicting the 12- and 24-month CSS for unresectable patients.
Performance metrics confirmed the nomogram’s accuracy across multiple endpoints. For 12-month CSS prediction, the AUC was 0.77 (95% CI 0.69–0.84) in the training set and 0.74 (95% CI 0.63–0.86) in the validation set. For 24-month CSS prediction, the model achieved even higher accuracy with AUC values of 0.82 (95% CI 0.74–0.90) and 0.87 (95% CI 0.76–0.98) in the training and validation sets, respectively.
Calibration curves demonstrated excellent agreement between predicted and observed survival probabilities at both 12 and 24 months in both the training and validation cohorts (Figure 3), confirming the nomogram’s reliability. Risk stratification using this model identified younger age (⩽45 years), upper esophageal tumor location, N3 nodal status, metastatic disease (M1), and non-receipt of chemotherapy as indicators of particularly poor prognosis in unresectable ENEC patients.

AUC of the training set (a) and validation set (b); 12- and 24-month cancer-specific survival according to the training set (c) and the validation set (d).
Discussion
This study presents one of the most comprehensive comparative analyses of ENEC between Eastern and Western populations to date. Our findings revealed significant differences in tumor characteristics, treatment approaches, and prognostic factors between Chinese and American patients.
The striking disparities in tumor location—with Chinese patients exhibiting more middle esophageal tumors (26.1% vs 16.9%) and fewer lower esophageal tumors (22.7% vs 62.2%)—likely reflect distinct etiological factors. Western populations experience higher rates of gastroesophageal reflux disease and Barrett’s esophagus affecting the distal esophagus, while in China, different dietary habits, environmental exposures, and genetic factors may contribute to the middle esophageal predominance. Similarly, the remarkable histological differences, with small cell carcinoma predominating in the SEER cohort (76.9% vs 38.6%) but adenocarcinoma with neuroendocrine differentiation more prevalent in the Chinese cohort (38.6% vs 8.8%), suggest both biological differences and variations in diagnostic practices across regions.
The earlier clinical presentation observed in Chinese patients, with 71.6% presenting without distant metastases compared to only 45.9% in the SEER cohort, could reflect differences in screening practices, healthcare access patterns, or potentially different biological behavior. This finding aligns with previous research suggesting more frequent endoscopic screening programs in Asian populations, resulting in earlier tumor detection.20,21
The most notable disparity was observed in the rates of surgical intervention, with 77.8% of non-metastatic Chinese patients undergoing surgery compared to merely 8.8% of SEER patients. This fundamental divergence in treatment philosophy reflects the more aggressive surgical approach traditionally favored in Asian countries for localized NETs, whereas Western practice patterns typically favor non-surgical approaches for small cell histologies based on extrapolation from small cell lung cancer management. Remarkably, despite this pronounced treatment disparity, median CSS for surgically treated patients was similar between cohorts (Chinese: 44 months, SEER: 30 months; p = 0.93), challenging conventional wisdom about optimal treatment strategies and suggesting that appropriately selected patients may benefit from surgery regardless of geographic origin.
Our multivariate analyses identified both shared and population-specific prognostic factors. In both cohorts, metastatic disease and age emerged as powerful independent predictors of survival. However, the substantially larger effect size for M1 disease in the Chinese cohort (HR = 49.74 vs 1.96 in SEER) suggests potential differences in biological behavior or post-progression treatment efficacy. The Chinese cohort demonstrated a wider array of independent prognostic factors, including tumor location, histological subtype, T stage, and N stage, possibly reflecting the higher proportion of surgical specimens, allowing for more precise pathological assessment. The observation that adenocarcinoma with neuroendocrine differentiation conferred better outcomes in the Chinese cohort challenges the view that all high-grade neuroendocrine neoplasms share similarly poor prognoses and suggests potential benefits from histology-specific treatment approaches.
The prognostic nomogram developed in this study addresses a critical need for accurate risk stratification in unresectable ENEC. With a C-index of 0.725 and excellent calibration, this tool integrates five readily available clinical variables—age, tumor location, N stage, M stage, and receipt of chemotherapy—ensuring practical applicability in diverse clinical settings. The model’s particularly strong performance in predicting 24-month survival (AUC 0.82–0.87) makes it valuable for identifying patients with unresectable disease who may achieve longer-term survival with appropriate management; however, external validation in independent datasets (e.g., non-SEER cohorts) is necessary before it can be widely applied in clinical practice.
Several limitations warrant consideration. The retrospective design introduces potential selection biases, including the unavoidable risk that patients selected for surgery may have had inherently better prognostic factors (e.g., younger age, fewer comorbidities, or earlier disease stages not fully captured in the data), which could confound treatment-outcome associations. Additionally, the criteria for surgical eligibility (e.g., based on T/N/M stage, overall physical condition, or institutional protocols) are not clearly delineated in the retrospective data, particularly in the SEER cohort, raising concerns about comparability between groups and amplifying the risk of selection bias in the surgical cohort. The marked imbalance in cohort sizes (SEER: n = 545; Chinese: n = 88) may compromise the robustness and generalizability of comparative statistical analyses, particularly by reducing power for detecting differences in subgroup analyses within the smaller Chinese cohort and potentially introducing type II errors; the relatively small number of surgically treated patients in the Chinese cohort (n = 49) further contributes to limited statistical power in subgroup analyses (e.g., stratified by T or N stage), potentially leading to unstable estimates and warranting cautious interpretation. Although methods such as propensity score matching or inverse probability weighting could theoretically mitigate these imbalances by adjusting for confounding variables and improving comparability between cohorts, these were not implemented in the current study due to limitations in the SEER database (e.g., missing data on key confounders like performance status and detailed treatment regimens) and the exploratory nature of this retrospective analysis. Future studies with larger, balanced cohorts, or prospective designs could incorporate such techniques to enhance validity. The SEER database lacks important clinical information, including performance status, comorbidities, and detailed treatment regimens (e.g., specific chemotherapy protocols or radiation doses), which hinders precise evaluation of treatment effects and may lead to incomplete interpretations of survival outcomes, as unobserved confounders could influence the observed associations between treatments and prognosis. Furthermore, the absence of molecular and genetic data, such as alterations in TP53 or RB1 genes, or biological markers like Ki-67 proliferation index, limits our ability to explore underlying biological differences between cohorts, despite recent studies emphasizing their prognostic and therapeutic relevance in NECs. Moreover, while the prognostic nomogram demonstrated robust internal validation within the SEER cohort, it lacks external validation using an independent dataset, which is essential to confirm its generalizability across diverse populations. Additionally, evolving diagnostic criteria over the study period may have influenced case classification, particularly for mixed neuroendocrine-non-neuroendocrine neoplasms. Given these limitations, our findings should be considered hypothesis-generating rather than definitive. Prospective, multicenter studies with standardized protocols are required to validate these observations and mitigate biases inherent to retrospective analyses.
Despite these limitations, our findings have important clinical implications. The substantial differences between Chinese and Western ENEC patients highlight the need for population-specific approaches. The relatively favorable outcomes in surgically treated patients suggest that surgical resection should be considered for appropriately selected patients regardless of histological subtype. The nomogram provides a practical tool for risk stratification in unresectable ENEC, potentially facilitating more informed treatment decisions.
Future research should focus on prospective validation in larger, multinational cohorts with standardized pathological assessment, as well as integration of genomic and biomarker analyses (e.g., TP53/RB1 mutations and Ki-67 index), across different ethnic groups. Such efforts may provide deeper insights into the biological mechanisms underlying observed differences and guide the development of more personalized management strategies for this rare but aggressive malignancy.
Conclusion
Our comparative analysis reveals significant heterogeneity in ENEC between Chinese and Western populations. Chinese patients present with distinctive features, including younger age, predominance of middle esophageal involvement, higher rates of adenocarcinoma with neuroendocrine differentiation, and earlier stage at diagnosis. The substantially higher utilization of surgical intervention in Chinese patients highlights a fundamental divergence in treatment philosophy. Notably, similar survival outcomes in surgically treated patients from both cohorts suggest potential benefit from surgical approaches in appropriately selected patients regardless of ethnicity. The prognostic nomogram developed for unresectable disease provides a practical tool that outperforms conventional staging systems, potentially enhancing clinical decision-making. Our findings indicate that management strategies should be contextualized to specific patient populations rather than universally extrapolated from small cell lung cancer paradigms. Future research incorporating molecular characterization across ethnic groups and dedicated clinical trials for ENEC-specific treatments may advance more personalized therapeutic approaches for this aggressive malignancy.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251375454 – Supplemental material for Population-specific characteristics and outcomes in esophageal neuroendocrine carcinoma: a Chinese-Western comparison with prognostic nomogram
Supplemental material, sj-docx-1-tam-10.1177_17588359251375454 for Population-specific characteristics and outcomes in esophageal neuroendocrine carcinoma: a Chinese-Western comparison with prognostic nomogram by Ai-Qing Li, Shu-hui Wang, Jun Li, Hong-Tan Chen and Zhen Liu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank the Surveillance, Epidemiology, and End Results (SEER) Program for database access and the medical staff at the First Affiliated Hospital of Zhejiang University for their contributions to patient care and data collection.
Declarations
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
