Abstract
Background:
The role of postmastectomy radiotherapy (PMRT) in breast cancer patients achieving pathologically negative lymph nodes (ypN0) after neoadjuvant chemotherapy (NAC) remains controversial due to heterogeneous recurrence risks and lack of prospective evidence.
Objectives:
To evaluate the effects of PMRT in ypN0 patients after NAC.
Design:
Multicenter retrospective study.
Methods:
Data of 624 breast cancer patients with ypN0 was assessed to establish a recurrence risk score model based on a disease-free survival (DFS) rate-based multivariate Cox model. Moreover, the locoregional control (LRC), DFS, and overall survival (OS) rates in PMRT and non-PMRT patients were calculated using the Kaplan–Meier method.
Results:
All patients received a median of four NAC cycles, followed by mastectomy and axillary lymph node dissection; moreover, 257 (41.2%) patients underwent PMRT. Over a median follow-up duration of 74 months, the 5-year LRC, DFS, and OS rates for all patients were 96.6%, 90.1%, and 95.7%, respectively. The differences in the LRC, DFS, and OS rates between PMRT and non-PMRT patients were nonsignificant in the univariate and multivariate analyses. By using our recurrence risk score model based on four factors (i.e., age, clinical N stage, NAC cycle number, and pathological tumor stage after NAC), we stratified the patients into low- and high-risk groups; their 5-year rates of LRC (98.8% vs 93.9%), DFS (95.1% vs 83.8%), and OS (98.4% vs 92.4%) were significantly different (all p < 0.05). PMRT improved LRC (97.6% vs 90.8%, p = 0.027) but not DFS or OS in high-risk patients and had no benefit in low-risk patients.
Conclusion:
Our recurrence risk score model effectively distinguished ypN0 patients with different recurrence risk stratifications. PMRT improved LRC but not DFS or OS in high-risk patients and low-risk patients did not benefit from PMRT.
Keywords
Introduction
Postmastectomy radiotherapy (PMRT) and neoadjuvant chemotherapy (NAC) are an integral part of the multidisciplinary treatment of locally advanced breast cancer. Conventionally, NAC is used for treating locally advanced breast cancer, and recommendations for PMRT after NAC are based on the initial tumor and nodal stages, regardless of the treatment response. 1 With widespread use of NAC in early-stage breast cancer and established prognostic significance of a favorable treatment response to NAC, PMRT is typically indicated on the basis of the patient’s initial and post-NAC pathological stages.
Pathological lymph node stage is the most crucial locoregional recurrence (LRR) predictor. The LRR rates are 0%–12% in patients with all pathologically negative lymph nodes (ypN0), 6%–21% with one to three pathologically positive lymph nodes (ypN1), and 11%–27.5% with more than four pathologically positive lymph nodes (ypN2-3).2,3 PMRT can significantly reduce LRR and increase disease-free survival (DFS) in patients with ypN1, and in patients with ypN2-3, it can increase both DFS and overall survival (OS). 4 However, the role of PMRT in ypN0 breast cancer remains debatable because it may not have any survival or locoregional control (LRC) benefit.4 –6
Breast cancer patients with ypN0 demonstrate a heterogeneous disease because they present with a wide variety of primary tumor sizes and axillary lymph node metastasis before NAC. According to the recommendations of National Comprehensive Cancer Network guidelines, 7 post-NAC PMRT should be recommended to patients based on initial clinical and pathological stages and biological tumor features. Therefore, establishing a risk model with clinically available factors may aid in distinguishing the prognoses of patients with ypN0, guiding clinical treatment recommendations.
In this multicenter retrospective study, we determined the survival benefits of PMRT in breast cancer patients with ypN0 after NAC and mastectomy. By using identified risk factors, we constructed a risk scoring model to distinguish patients into low-, and high-risk groups. Finally, we assessed the survival benefits of PMRT in patients at different risk levels.
Patients and methods
Patients
We retrospectively reviewed the data of breast cancer patients with ypN0 after NAC and mastectomy from 10 grade A tertiary hospitals in China between 2000 and 2014. The inclusion criteria were as follows: breast cancer at clinical tumor stage (cT) 1–3, clinical nodal stage (cN) 0–1, and clinical metastasis stage (cM) 0; undergoing total mastectomy and axillary dissection with negative lymph nodes after NAC; and pathological data including tumor size, lymphovascular invasion (LVI), estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) status available. The exclusion criteria were as follows: relapse within 2 months or loss to follow-up within 6 months of mastectomy (Figure 1).
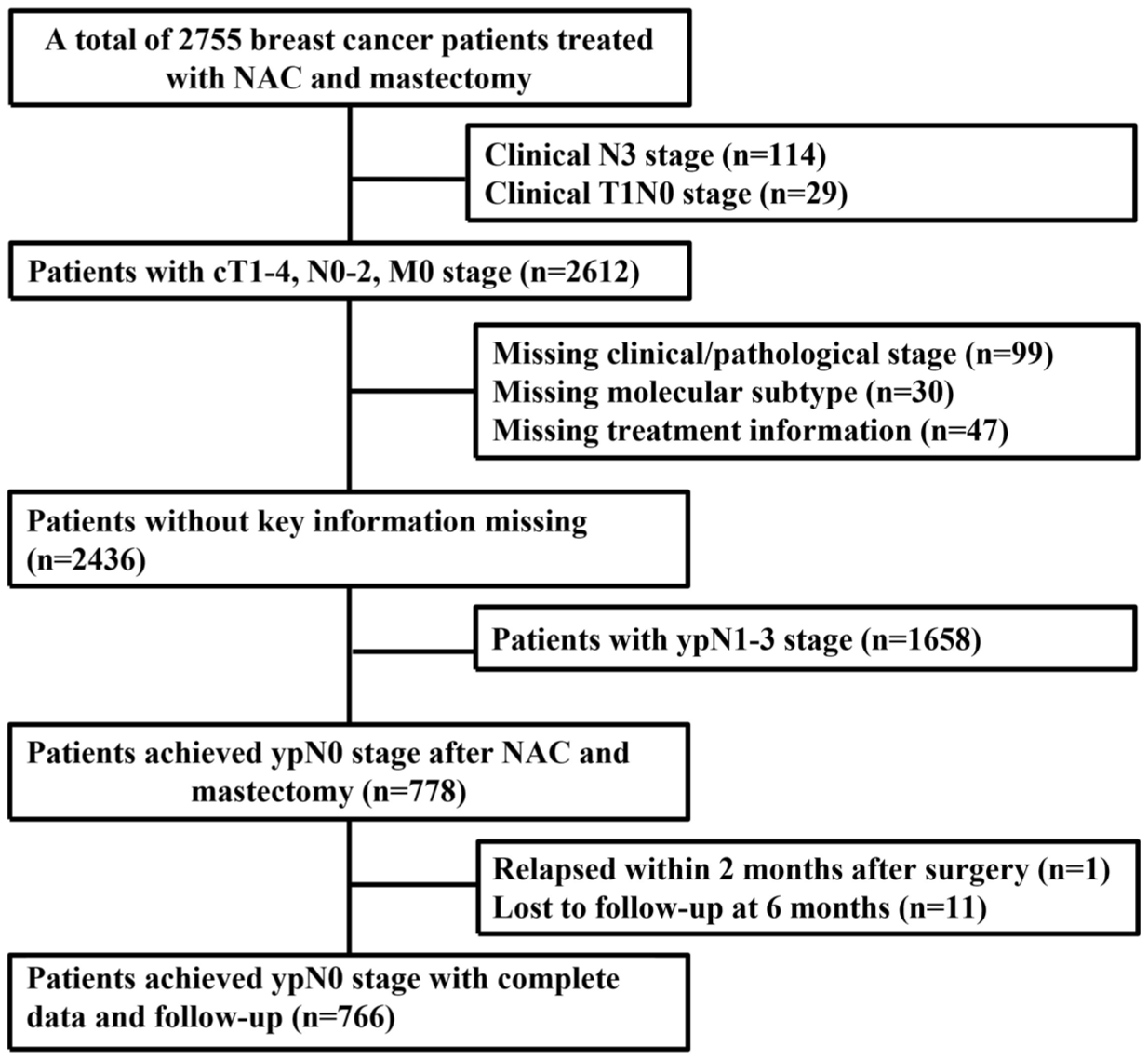
Patient recruitment flowchart.
cT was determined through breast ultrasonography, magnetic resonance imaging, or both. cN was confirmed through needle biopsy, ultrasonography, or chest computed tomography before treatment.
This study was approved by the Cancer Hospital of Chinese Academy of Medical Sciences (approval number: 15-057/984), and the requirement for informed consent was waived. The reporting of this study conforms to the STROBE statement (see Supplemental Materials A). 8
Treatment
Systemic therapy
Before 2009, 2–4 NAC cycles and 1–8 cycles of adjuvant chemotherapy were administered. From 2009, 6–8 NAC cycles were administered since it provided a higher pathologic complete response (pCR) rate in the randomized trial. 9 All patients received a median of four NAC cycles (range, 1–8 cycles), and 452 (72.4%) patients received a median of two adjuvant chemotherapy cycles (range, 1–8 cycles). Consequently, patients received a median of six total chemotherapy cycles (range, 1–12 cycles). Regarding the NAC regimen, a regimen combining anthracycline with taxanes was administered to 398 (63.8%) patients, an anthracycline-based regimen to 91 (14.6%) patients, a regimen combining taxanes with platinum to 44 (7.1%) patients, a taxane-based regimen to 27 (4.3%) patients, and other regimens to 53 (8.5%) patients; the regimen administered to 11 (1.8%) patients remained unknown.
Of the 380 patients with positive ER and/or PR, 345 (90.8%) received endocrine therapy. Of the 203 patients with positive HER2, 75 (37.0%) received anti-HER2 targeted therapy.
Surgery
All patients underwent total mastectomy and axillary lymph node dissection with negative margins and ypN0. The median (range) number of removed axillary lymph nodes was 18 (9–48).
Postmastectomy radiotherapy
PMRT was recommended after surgery and chemotherapy to patients with high-risk factors, including age <50 years, cT3, ER, and PR negativity. In total, 257 (41.2%) patients received PMRT, whereas 367 (58.8%) did not. Of the patients who underwent PMRT, 207 (80.5%) received chest wall and supra/infraclavicular nodal irradiation, 39 (15.2%) received chest wall irradiation alone, 7 (2.7%) received chest wall plus supra/infraclavicular nodal and internal mammary nodal irradiation, 3 (1.2%) received chest wall plus supra/infraclavicular and axilla nodal irradiation, and 1 (0.4%) received chest wall and internal mammary nodal irradiation. Of the 257 PMRT patients, 246 (95.7%) received a median (range) radiation dose of 50 (36–60) Gray in a median (range) fractions of 25 (18–30), whereas 11 (4.3%) patients a radiation dose of 40–43.5 Gray in 15 or 16 fractions.
Outcomes
LRC was measured until recurrences of ipsilateral chest wall and/or ipsilateral axillary, supraclavicular, infraclavicular, or internal mammary lymph nodes or the last follow-up. We recorded all LRR events regardless of whether they occurred before, simultaneously with, or after distant metastasis. DFS was measured from the date of definitive surgery until the date of LRR, distant metastasis, death, or final follow-up. OS was calculated from the date of definitive surgery until the date of death or final follow-up.
Statistical analysis
All patient characteristics are presented as frequencies and percentages. The Chi-square test or Fisher exact test was used to compare the clinical and pathological characteristics between PMRT and non-PMRT patients. To reduce potential selection bias due to the retrospective nature of the study, propensity score matching (PSM) was performed using a 1:1 nearest-neighbor matching algorithm without replacement. Matching variables included age, clinical T stage, clinical N stage, grade, LVI, molecular subtype/trastuzumab, pathological tumor stage after NAC (ypT stage), NAC cycles, adjuvant chemotherapy, and endocrine therapy. LRC, DFS, and OS rates were estimated using the Kaplan–Meier method. The log-rank test was used to identify the prognostic factors associated with the LRC, DFS, and OS rates. Restricted cubic spline was used to examine the assumption of NAC cycle number as continuous variables related to the effects on DFS. Age, clinical T stage, clinical N stage, grade, LVI, molecular subtype/trastuzumab, ypT stage, NAC cycles, and adjuvant chemotherapy were included in our multivariate Cox models to determine the prognostic factors associated with LRC, DFS, and OS rates. p < 0.05 was considered to indicate statistical significance. Since delaying PMRT was proved to increase risk of distant metastases and reduce OS, 10 indicating that PMRT affected not only local recurrence but also distant metastases, recurrence risk scores were assigned proportionally to the estimates of the relative contributions of the independent prognostic factors in the multivariate model of DFS. Next, a decision tree analysis was performed to determine the optimal cutoff points according to the effects of recurrence risk scores. Finally, the entire cohort was stratified into low-, and high-risk groups. Time-dependent receiver operating characteristic analysis was performed at 60 months to assess model discrimination. All analyses were performed using SPSS (version 26.0; IBM Corp., Armonk, NY, USA) and R software (version 4.1.2; http://www.r-project.org/).
Results
Patient characteristics
In total, 624 patients were included in this study; Table 1 presents the clinical and pathological characteristics of the entire cohort, as well as those of PMRT and non-PMRT patients. The median (range) age was 48 (20–78) years. Moreover, 219 (35.1%) patients were at cN0, 405 (64.9%) at cN1. Compared with non-PMRT patients, PMRT patients were significantly younger; in addition, significantly more PMRT patients had cT2-3 and cN1 cancer; received more than four NAC cycles; had ER and PR negative and HER2-positive receiving Trastuzumab therapy. Among the 74 cN0 patients in the PMRT group, 44 (59.4%) had cT3 tumors at diagnosis, and 28 (93.3%) of the remaining cT2 patients had ypT2–3 disease after NAC.
Clinical and pathological characteristics of entire cohort, PMRT and non-PMRT patients, and low-risk and high-risk groups.

ET, hormone therapy; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; LVI, lymphovascular invasion; NAC, neoadjuvant chemotherapy; PMRT, postmastectomy radiotherapy; Trastuzumab−, without trastuzumab; Trastuzumab+, with trastuzumab; ypT stage, pathological tumor stage after NAC.
LRC, DFS, and OS outcomes
Over a median (range) follow-up duration of 74 (6–207) months, the 5-year cumulative LRC, DFS, and OS rates for the entire cohort was 96.6%, 90.1%, and 95.7%, respectively. Among all patients, 21 (3.4%) developed LRR (10 (1.6%) in supra/infraclavicular regions, 9 (1.4%) in chest wall, 3 (0.5%) in internal mammary chain, 1 (0.2%) in axilla), 64 (10.3%) developed distant metastasis, and 42 (6.7%) died (38 (90.5%) died of breast cancer and 4 (9.5%) died of other causes).
Prognostic factors related to LRC, DFS, and OS rates
Table 2 presents the results of our univariate analyses of prognostic factors associated with LRC, DFS, or OS. Age and LVI were the risk factors for LRC rates. Age, cN stage, and NAC cycle number were the risk factors for DFS rates (Supplemental Figure 1). The hazard ratio of DFS increased with the increase of 1–4 NAC cycles and tended to be stable at 5–8 NAC cycles (Supplemental Figure 2). Finally, ypT stage was the risk factor for OS rates.
Univariate analysis of prognostic factors in patients with ypN0 (n = 624).
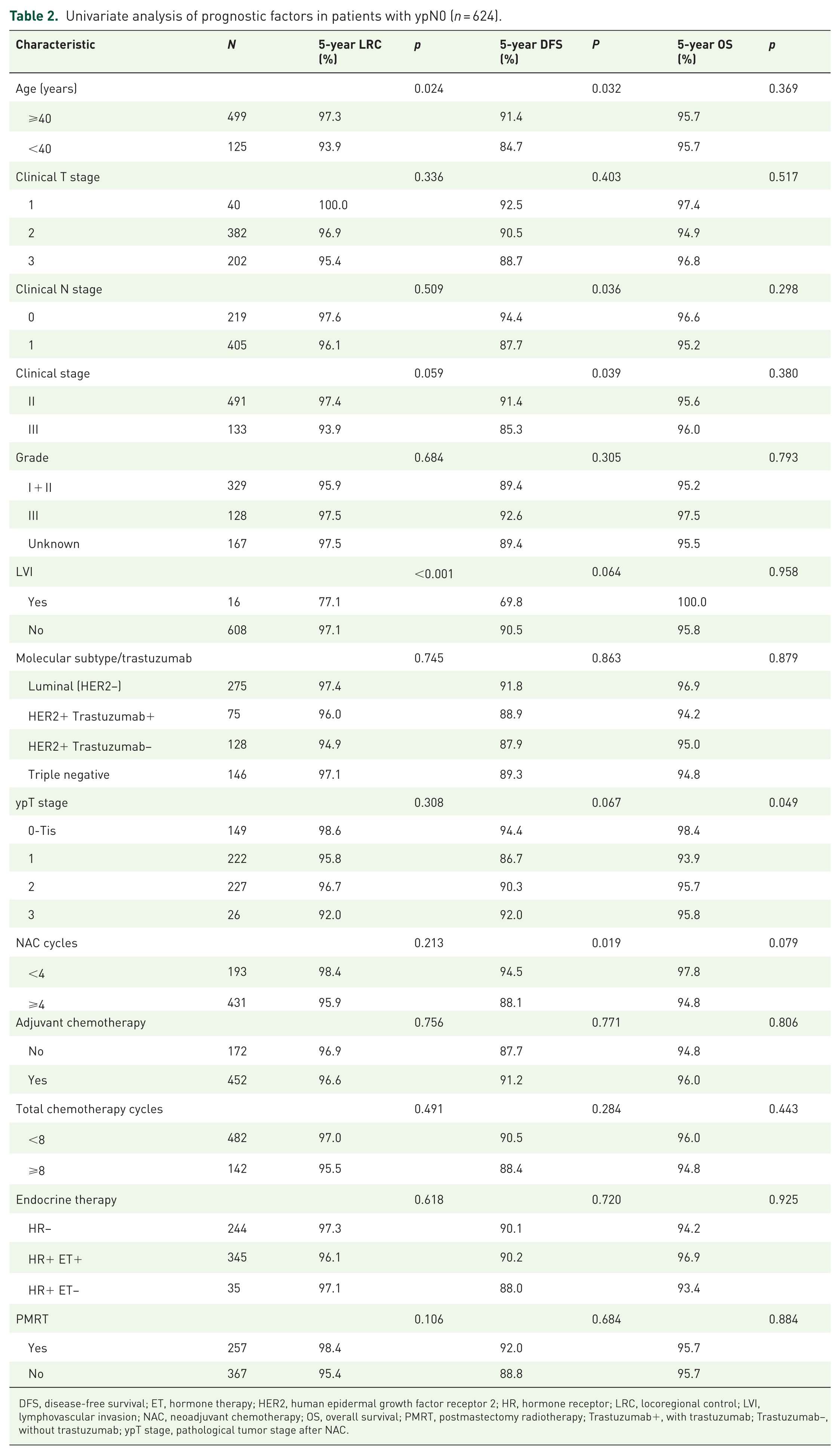
DFS, disease-free survival; ET, hormone therapy; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; LRC, locoregional control; LVI, lymphovascular invasion; NAC, neoadjuvant chemotherapy; OS, overall survival; PMRT, postmastectomy radiotherapy; Trastuzumab+, with trastuzumab; Trastuzumab−, without trastuzumab; ypT stage, pathological tumor stage after NAC.
Table 3 presents the results of multivariable analyses for LRC, DFS, and OS rates. Age and LVI were significantly associated with LRC rates. Moreover, cN stage, ypT stage, and NAC cycle number ⩾4 were significantly associated with DFS rates, whereas age had borderline significance. Finally, NAC cycle number ⩾4 and ypT stage were significantly associated with OS rates.
Multivariate analysis of prognostic factors in patients with ypN0 (n = 624).
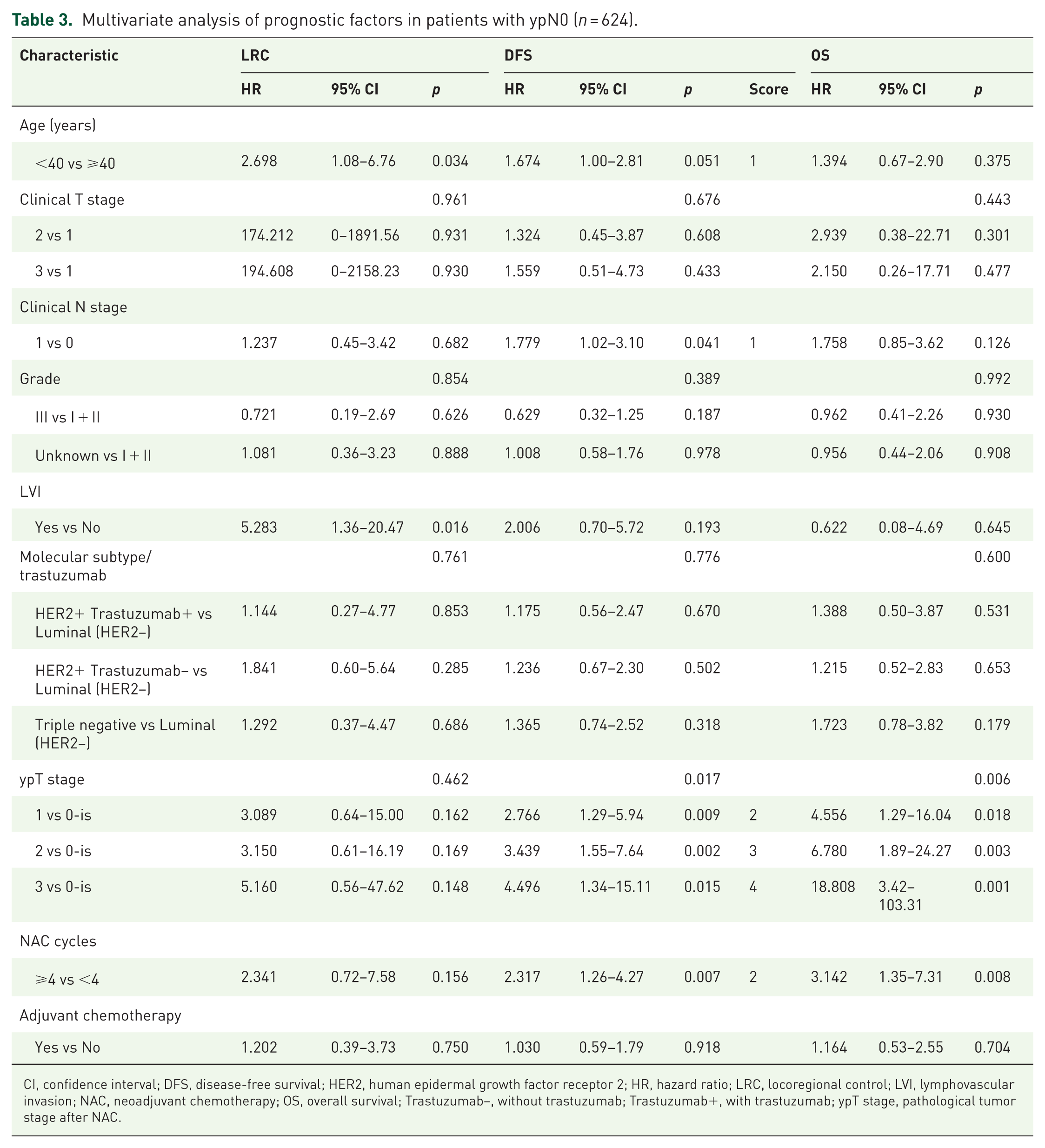
CI, confidence interval; DFS, disease-free survival; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; LRC, locoregional control; LVI, lymphovascular invasion; NAC, neoadjuvant chemotherapy; OS, overall survival; Trastuzumab−, without trastuzumab; Trastuzumab+, with trastuzumab; ypT stage, pathological tumor stage after NAC.
HER2-targeted therapy was included in the univariable and multivariable analyses as part of the molecular subtype variable, but was not identified as an independent prognostic factor for LRC, DFS, or OS. In HER2-positive patients, no significant differences in survival outcomes were observed between those who received targeted therapy and those who did not. PMRT also did not confer survival benefit in either subgroup (Supplemental Figure 3). Further subgroup analyses across molecular subtypes showed that PMRT did not improve LRC, DFS, or OS in either low- or high-risk patients in any subtype (HR-positive/HER2-negative, HER2-positive, or triple negative; Supplemental Figure 4).
Recurrence risk score model and risk group classification
On the basis of the DFS rate-based multivariate model, our recurrence risk score model consisted of four factors, with possible total scores ranging from 0 to 8 points: 1 point each for age <40 years and cN1; 2 points each for NAC cycle number ⩾4, and ypT1; 3 points for ypT2; and 4 points for ypT3 (Table 3; Supplemental Table 2). On the basis of the DFS rates, we identified 4 points as cutoffs for recurrence risk scores in our decision tree analysis. Next, we used these cutoffs to stratify patients into two risk groups (Figure 2(a)): low-risk group (0–4 points; n = 343 (55.0%)), and high-risk group (5–8 points; n = 281 (45.0%)). Clinical and pathological characteristics of the low-, and high-risk patients were presented in Table 1.

Decision tree analysis (a) according to the effect of recurrence risk score on DFS and comparison of LRC (b), DFS (c), and OS (d) between low-, and high-risk patients with ypN0.
As shown in Figure 2, the low- and high-risk groups had significantly different 5-year LRC rates (98.8% vs 93.9%), 5-year DFS rates (95.1% vs 83.8%), and 5-year OS rates (98.4% vs 92.4%; all p < 0.05). At 60 months, the model showed good discrimination with an AUC of 0.666, a sensitivity of 0.730, and a specificity of 0.602 (Supplemental Figure 5).
Impact of PMRT
In the entire cohort, PMRT and non-PMRT patients demonstrated nonsignificant differences in 5-year LRC rates (98.4% vs 95.4%, p = 0.106), 5-year DFS rates (92.0% vs 88.8%, p = 0.684), and 5-year OS rates (95.7% vs 95.7%, p = 0.884; Figure 3).

Comparison of LRC, DFS, and OS rates between PMRT and non-PMRT ypN0 patients in the entire cohort (a1, a2, and a3, respectively), low-risk group (b1, b2, and b3, respectively), and high-risk group (c1, c2, and c3, respectively).
There were 129 (37.6%) PMRT patients in the low-risk group and 128 (45.6%) in the high-risk group. PMRT was observed to improve LRC (97.6% vs 90.8%, p = 0.027) but not DFS (87.3% vs 81.0%, p = 0.409) or OS (94.0% vs 91.0%, p = 0.266) in high-risk group. However, PMRT has no significant effect on the LRC (99.2% vs 98.6%, p = 0.888), DFS (96.7% vs 94.2%, p = 0.739), and OS (97.3% vs 99.0%, p = 0.111) rates of patients in low-risk group (Figure 3).
To further account for baseline imbalances, PSM analysis was conducted. As shown in Supplemental Table 3, there were no significant differences in baseline characteristics between the PMRT and non-PMRT groups (both n = 224) after matching. In the matched cohort, PMRT was not associated with improvements in 5-year LRC rates (98.3% vs 95.2%, p = 0.117), 5-year DFS rates (92.99% vs 86.9%, p = 0.0093), and 5-year OS rates (96.6% vs 95.33%, p = 0.192; Supplemental Figure 6). After stratification, PMRT remained ineffective in improving survival outcomes in the low-risk group, while in the high-risk group, PMRT significantly improved LRC (98.7% vs 92.1%, p = 0.025) but not DFS (87.5% vs 79.8%, p = 0.151) or OS (94.6% vs 91.6%, p = 0.062).
To evaluate the potential impact of treatment heterogeneity across the long study period, subgroup analysis based on treatment era (before 2010 vs 2010 or later) was performed. The number of NAC cycles, total chemotherapy cycles, and endocrine therapy use were similar between the two periods. Patients treated after 2010 were more likely to receive HER2-targeted therapy (16.0% vs 11.2%, p < 0.001) and NAC regimen combining anthracycline with taxanes (71.3% vs 52.4%, p = 0.02). No significant differences were observed in LRC, DFS, or OS between patients treated before and after 2010. Moreover, PMRT was not associated with improved survival in either time periods (Supplemental Figure 7). After stratification, PMRT consistently improved LRC in high-risk patients but not DFS or OS, and remained ineffective in low-risk patients regardless of treatment periods (Supplemental Figure 8).
Among the 257 patients receiving PMRT, 39 (15.2%) received chest wall irradiation only, while 207 (80.5%) received chest wall and regional nodal irradiation (RNI). No statistically significant differences were observed in 5-year LRC, DSF, or OS between the two groups (Supplemental Figure 9).
Discussion
NAC is being increasingly used to treat breast cancer because it leads to favorable tumor response, facilitates down-staging, and enables breast conservation. Because the response to NAC cannot facilitate decision-making related to PMRT administration, whether PMRT provides benefits in patients with ypN0 remains unclear. In the current study, patients with ypN0 after NAC and mastectomy demonstrated low relapse incidence and favorable survival outcomes. We stratified ypN0 patients with significant survival differences into low-, and high-risk groups based on four factors including age, cN stage, NAC cycle number, and ypT stage. Our results indicated that PMRT improved LRC but not DFS or OS in high-risk patients, while PMRT did not increase LRC or survival rates across the entire cohort group or in the low-risk group.
In general, breast cancer patients with ypN0 after NAC had favorable survival outcomes. These patients typically demonstrate 5-year LRC, DFS, and OS rates of 91.9%–97.7%, 77.1%–92.3%, and 89.3%–96.1%, respectively.5,6,11 –17 Le Scodan et al. 5 reported that in a cohort treated with NAC and mastectomy, 5-year LRC, DFS, and OS rates patients with ypN0 were 94.6%, 81.7%, and 90.8%, respectively; this result is in line with the current results. Cho et al. 13 reported that, in patients at advanced cT and cN who achieved ypN0, the 5-year LRC, DFS, and OS rates were 91.9%, 77.1%, and 89.3%, respectively. In their prospective study, de Wild et al. 15 categorized cT1–2N0-1 patients with ypN0 after NAC and mastectomy into a non-PMRT low-risk group; their 5-year LRC rate was as high as 97.7%. In NSABP B-17 and B-27 trials, the 10-year LRC rate was 89%–100% in non-PMRT patients with ypN0. 3
The role of PMRT in patients with ypN0 after NAC remains controversial because relevant data are scant (Supplemental Table 1),5,6,11,12,16 –18 without any prospective evidence being available. In the present study, PMRT was not associated with LRC, DFS, or OS rates in either the entire cohort or the low-risk group, while PMRT improved LRC but not DFS or OS in high-risk patients. Similar to the present study, Huang et al. 4 observed that PMRT significantly improved LRC rates but did not affect DFS and OS rates in the subgroup analysis of cT1-4N1-2 patients with ypN0 after NAC. The inclusion of 21% of cN2 patients increased the relapse risk of the study population, which were more likely to benefit from PMRT. However, the authors did not report the subgroup characteristics of patients with ypN0. A meta-analysis including studies on patients with ypN0 over 2004–2019 reported that PMRT was associated with a significant increase in LRC rate but a nonsignificant improvement in DFS and OS rates, particularly in patients with stage III breast cancer. 19 Studies including cT1-4N0-3 patients with ypN0 after NAC have confirmed that PMRT does not increase LRC, DFS, or OS rates in either stage II or III patients.5,11 Ren et al. 20 found PMRT improved DFS rates in clinical node-positive patients with ypN0 but not in clinical node-negative patients. After excluding the low-risk cN0 patients, a study on 142 cT1-2N1 patients with ypN0 after NAC and mastectomy reported significant differences in DFS rate between PMRT and non-PMRT groups. 12 Because we included a large number of cN0 patients, our entire cohort had a relatively lower relapse risk and was less likely to achieve survival benefit. In a 2003–2011 NCDB study on cT1-3N1M0 patients after NAC, subgroup analysis of patients with ypN0 after mastectomy demonstrated that PMRT significantly increased 5-year OS rates. 21 In another 2004–2017 NCDB study, 14,690 cT1-4N1-3M0 patients with ypN0 did not gain 10-year OS improvement from PMRT. Subgroup analysis demonstrated that PMRT may improve OS rates in cT3-4, ypT2-3, or cN3 patients. 22 Although cT4 and cN2-3 patients were included in the later study, pCR rates were similar in both the earlier and later studies; this was probably because of more advanced systemic therapeutic modalities. 23 Advances in systemic therapy, such as newer chemotherapeutic regimens, targeted therapy for HER2-positive disease, and endocrine therapy for ER-positive disease, which further reduced relapse risk, were employed in the latter years.9,24 In long-term studies, detailed staging workup in the latter years has increased the accuracy of clinical and pathological staging, thus influencing characteristic definition and PMRT recommendation efficacy in the study population. Because the heterogeneity among patients with ypN0 was large, the varied clinical and pathological features should be taken into consideration for further identification of high-risk patients that benefit from PMRT.
To identify high-risk patients with ypN0 that may benefit from PMRT, we developed a recurrence risk score model by combining the prognostic factors related to DFS, including age, cN stage, NAC cycle number, and ypT stage. However, studies on risk factors for survival in patients with ypN0 are limited, and their results are inconsistent: patients of a younger age, 11 with LVI, 13 or with advanced ypT5,11,25 have been reported to have poor DFS. In their study, Ma et al. recruited 1108 cT1-4N0-3 breast cancer patients, of whom 33.6% achieved ypN0 after NAC. The authors used risk factors for LRR, including histological classification, LVI, ypT, ypN, ER status, and Ki-67 expression, to construct a nomogram based on risk scores to stratify patients into low- and high-risk groups. Their results indicated that PMRT was strongly associated with improved LRC and survival in the high-risk group but not in the low-risk group. 26 In contrast to the current study, which only included patients with ypN0, the aforementioned study included all patients who received NAC, increasing the population heterogeneity. Nevertheless, consistent with the current study, the nomogram of LRC confirmed the importance of pathological staging; however, the nomogram considered molecular subtype, which was not considered in the current study. With advancements in systemic therapy, more patients with HER2-positive or triple-negative cancer are achieving ypN0 or even ypCR than those with other subtypes.27,28 De Wild et al. 15 reported that the percentage of HER2-positive (32.1% vs 15% vs 9%) or triple-negative (24% vs 9% vs 11%) cancer patients achieving ypN0 was higher than that of those achieving ypN1 or ypN2-3. Compared with pathological node-positive patients, patients with ypN0 demonstrated minimized differences in DFS and OS between molecular subtypes. 29 Thus, in the ypN0 cohort, systemic therapy improved survival in patients with a molecular subtype associated with poor biological behavior, resulting in higher rate of ypCR, a proven predictor of improved survival, 27 thus weakening the correlation between molecular subtype and DFS.
A major strength of this study is that we incorporated NAC cycle number, which is often overlooked, into our recurrence risk score model. For the first time, we demonstrated that NAC cycle number is a prognostic factor for DFS of ypN0 patients. Notably, we found that patients receiving less than four NAC cycles had better survival. Patients with fewer NAC cycles to reach ypN0 means an early response to NAC. Therefore, it is reasonable that early NAC response was associated with better survival outcomes. Besides, our patients with ypN0 received a median of only four NAC cycles implying that these patients may have had a low risk initially.
This study also has some limitations. First, PMRT was not a randomized variable; this may have introduced a selection bias. Because the characteristics were distributed unevenly between the two groups, PMRT was used preferentially in high-risk patients. We initially developed our recurrence risk score model to identify patients with high recurrence risk and noted LRC improvement of PMRT in high-risk group. Besides, we performed a PSM analysis, which showed balanced baseline characteristics and consistent survival outcomes with the main findings. Overall failure and mortality rates increase as the follow-up duration increases; therefore, our follow-up duration of 5 years may have been insufficient to evaluate the impact of PMRT on survival. Second, the proportion of HER2-positive patients receiving anti-HER2 targeted therapy is relatively small; and both neoadjuvant and adjuvant systemic therapies have significantly evolved, leading to a further decrease in recurrence risk, diminishing the incremental benefit afforded by PMRT, while subgroup analysis showed that HER2-targeted therapy did not significantly affect survival outcomes, or the impact of PMRT. Third, clinical nodal staging in most patients was based on physical examination and imaging (mainly axillary ultrasound or MRI). Axillary ultrasound, a widely accepted non-invasive tool, has shown a sensitivity of 70%, negative predictive value of 84%, and positive predictive value of 56% for detecting significant nodal disease in early breast cancer. 30 The lack of pathological confirmation may have caused false-positive staging in some cN1 patients. To better reflect real-world practice and avoid underrepresentation of low-risk cases, both cN0 and cN1 patients were included. Finally, our patients received a median of four NAC cycles along with axillary nodal dissection in the ypN0 cohort in contrast to the current clinical practice, where six to eight NAC cycles along with sentinel node biopsy are performed frequently in patients with ypN0. At that time, sentinel node biopsy after NAC was not widely accepted due to limited evidence and concerns about false-negative rates. Our patient population may thus represent a low-risk group in the ypN0 cohort. Therefore, our results should be cautiously extrapolated to other ypN0 populations in the context of modern treatment practices, and further validation in contemporary cohorts is needed.
The results related to the effects of PMRT on patients with ypN0 have varied among different studies because of heterogeneity in the patient populations, differences in irradiated volume, and differences in systemic therapy regimens. Improvements in systemic therapy efficacy have led to increases in the ypCR rate in patients with ypN0, further improving their DFS rate.5,17 Thus, advances in systemic therapy alone might be adequate for treating patients with ypN0, and they may not require PMRT. To the best of our knowledge, this is the largest retrospective study to investigate the impact of PMRT in patients with ypN0. Considering the strong heterogeneity among patients with ypN0, the recurrence risk score model was developed for further risk-based patient stratification. PMRT led to no survival benefit in the entire cohort or in any risk group, only LRC improvement in high-risk patients, indicating the requirement for further research. The ongoing NSABP B-51/RTOG 1304 phase III trial, enrolling cT1-3N1 patients with ypN0 after NAC and mastectomy or lumpectomy and randomizing them into a RNI or non-RNI group, is expected to clarify whether PMRT increases recurrence-free interval among patients with invasive breast cancer. 31
Conclusion
In the current study, cT1-3N0-1 breast cancer patients with ypN0 after NAC and mastectomy demonstrated favorable survival outcomes; moreover, PMRT did not demonstrate any survival benefit. By using recurrence risk score model combined with age, cN stage, NAC cycle number, and ypT stage, we further stratified patients with ypN0 into three risk groups based on their risk levels. PMRT improved LRC but not DFS or OS in high-risk patients and had no benefit in low-risk patients. As such, ypN0 patients after NAC and mastectomy might benefit from PMRT in high-risk group; this result warrants further prospective verification before clinical application.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251367347 – Supplemental material for A recurrence risk score model evaluating effects of postmastectomy radiotherapy in breast cancer patients with pathologically negative lymph nodes after neoadjuvant chemotherapy: a multicenter, retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359251367347 for A recurrence risk score model evaluating effects of postmastectomy radiotherapy in breast cancer patients with pathologically negative lymph nodes after neoadjuvant chemotherapy: a multicenter, retrospective study by Dan-Qiong Wang, Zhou Huang, Hong-Fen Wu, Dong-Xing Shen, Hao Jing, Hui Fang, Li Zhu, Xiao-Bo Huang, Liang-Fang Shen, Mei Shi, Jia-Yi Chen, Min Liu, Jing Cheng, Ye-Xiong Li, Jian Tie, Yu Tang and Shu-Lian Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251367347 – Supplemental material for A recurrence risk score model evaluating effects of postmastectomy radiotherapy in breast cancer patients with pathologically negative lymph nodes after neoadjuvant chemotherapy: a multicenter, retrospective study
Supplemental material, sj-docx-2-tam-10.1177_17588359251367347 for A recurrence risk score model evaluating effects of postmastectomy radiotherapy in breast cancer patients with pathologically negative lymph nodes after neoadjuvant chemotherapy: a multicenter, retrospective study by Dan-Qiong Wang, Zhou Huang, Hong-Fen Wu, Dong-Xing Shen, Hao Jing, Hui Fang, Li Zhu, Xiao-Bo Huang, Liang-Fang Shen, Mei Shi, Jia-Yi Chen, Min Liu, Jing Cheng, Ye-Xiong Li, Jian Tie, Yu Tang and Shu-Lian Wang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None.
Author’s note
Statistician: Dan-Qiong Wang, Department of Radiation Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, 17 Panjiayuan Nanli, Chaoyang District, Beijing 100021, China. E-mail:
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
