Abstract
Breast cancer (BC) remains a leading global health challenge, characterized by significant heterogeneity that complicates its detection, diagnosis, and management. The integration of imaging biomarkers and radiomics into clinical workflows has revolutionized early detection, risk stratification, and personalized treatment strategies. Established modalities, such as mammography and magnetic resonance imaging, in conjunction with biomarkers like hormone receptor status, continue to play a pivotal role in guiding therapeutic decisions. Simultaneously, advancements in radiomics and artificial intelligence (AI) have enabled the extraction and analysis of high-dimensional imaging data, offering novel insights into tumor biology and predicting treatment outcomes. This review explores the synergy of imaging biomarkers, radiomics, and AI, emphasizing their potential to transform BC care through enhanced precision and optimized patient outcomes.
Keywords
Introduction
Breast cancer (BC) remains a major global health issue, accounting for nearly 30% of cancers in women and contributing to significant morbidity and mortality worldwide. 1 In 2021, the United States reported approximately 284,200 new BC cases and 43,600 deaths. 2 Early diagnosis and timely intervention are essential to improving survival rates and reducing progression, particularly in high-risk groups.3,4 Current diagnostic tools like mammography and tissue biopsy have limitations—mammography may miss tumors in dense breast tissue5,6 and biopsies, while informative, are invasive and may not capture full tumor heterogeneity.7,8 These challenges highlight the need for biomarkers that provide precise, noninvasive insights into tumor biology and prognosis. 9 Traditional markers such as estrogen receptor (ER), progesterone receptor (PR), and HER2 have helped classify BC and guide therapy,10 –12 improving outcomes through tailored treatment.13,14 However, more biomarkers are needed to address tumor variability and treatment resistance. 15 Personalized medicine enables better therapy targeting, improves prognosis, and minimizes side effects by leveraging biomarker profiles.16 –18 It has proven effective in managing hormone receptor-positive and HER2-positive BC through tailored interventions19,20 and supports ongoing monitoring for treatment response or resistance21,22 while also enhancing quality of life by avoiding overtreatment.19,23 In addition, biomarker and genetic profiling can identify high-risk individuals, enabling earlier preventive strategies.24,25
Epidemiology and BC screening
BC is the second most common cancer globally after lung cancer and the fourth leading cause of cancer-related deaths worldwide.27,28 In 2022, 2.3 million people were diagnosed with BC, resulting in 666,130 deaths. The global incidence and mortality rates stand at 46.8 and 12.7 per 10,000, respectively. 28 Among women, BC is the second leading cause of cancer death, with its burden varying widely by geography and socioeconomic status.
While BC incidence is higher in developed countries—due to lifestyle factors and widespread screening—mortality is disproportionately greater in less developed regions, where limited access to diagnostics and treatment worsens outcomes. High-income countries report better 5-year survival rates, thanks to early detection and comprehensive care. Risk factors include obesity, alcohol consumption, smoking, early menarche, late menopause, delayed childbirth, and hormone therapy use. 29
Asia and Europe have the highest BC prevalence, with incidence rates of 42.9 and 24.3 per 10,000 and mortality rates of 47.3% and 21.7%, respectively. 27 In North America, incidence and prevalence are 13.3% and 16.3%, with a lower mortality rate of 7.5%. In 2021, about 4 million U.S. women were living with BC, and the 5-year survival rate was 91.2%.28,30
Early detection significantly improves survival, with mammography as the gold standard. 31 However, it is less effective in women with dense breast tissue, leading to false positives and missed diagnoses. Magnetic resonance imaging (MRI) is recommended for high-risk groups, such as BRCA1/2 carriers, women with dense breasts, and Black women—who are more prone to aggressive subtypes like triple-negative BC. 32
Emerging technologies like abbreviated MRI, contrast-enhanced spectral mammography (CESM), elastography, and positron emission mammography offer improved sensitivity and diagnostic precision.33 –36 Yet, disparities persist: non-Hispanic Black women in the United States have a 40% higher mortality rate than non-Hispanic White women, despite similar incidence rates.30,37 These inequities are tied to systemic racism, socioeconomic barriers, and limited healthcare access—especially in South Central and Mid-Atlantic states.37,38 Addressing them requires targeted interventions, including improved access, support services, and inclusive clinical trials. 38
Cancer risk and personalized screening
Advancements in BC screening have significantly improved outcomes and life expectancy, but equitable access to resources remains critical for maximizing benefits, and unfortunately, as it often represents a challenge. Personalized screening protocols are essential to stratify women based on individual risk levels, categorized as high (>20% lifetime risk), intermediate (15%–20%), and low (<15%).27,31 Dr. Dana Ataya highlights the importance of accurately assessing risk to tailor screening plans to patient needs, particularly for high-risk groups. 27
BC risk factors span genetic, hormonal, reproductive, and lifestyle domains. Genetic predisposition is one of the most significant, and mutations in tumor suppressor genes, among which BRCA1/2, TP53, and PTEN (Table 1, Figure 1) account for approximately 25% of hereditary BC cases.39 –41 These high-penetrance genes indeed confer a significant risk of developing BC, particularly among populations like Ashkenazi Jews, who have elevated risk due to founder mutations like BRCA1 185delAG and BRCA 6174delT. 42 Moderate-penetrance genes such as CHEK2 and ATM also contribute, underscoring the complexity of genetic influences.39,41 Beyond genetics, additional factors such as breast density, family history, early menarche, late menopause, and lifestyle choices (e.g., smoking, alcohol intake, physical activity) further stratify risk profiles.29,41
Genetic mutations and associated BC risks.

This table compares high-penetrance and moderate-penetrance genes, highlighting their contributions to BC susceptibility.
BC, breast cancer.

Heatmap displaying the distribution of genetic mutations linked to BC, categorized by penetrance levels: low, moderate, and high. Each cell represents a specific gene, with color intensity denoting the degree of penetrance. Darker shades indicate a stronger association between the gene and its corresponding penetrance category, emphasizing genes with higher susceptibility to BC.
Several predictive models integrate these risk factors to refine personalized screening (Table 2). The Gail Model, widely used for population-level assessments, estimates 5-year and lifetime BC risk based on age, reproductive history, and first-degree relatives with BC. However, it excludes second-degree relatives and genetic factors, limiting its utility for high-risk individuals.27,43 The Tyrer-Cuzick Model incorporates a broader range of variables, including genetic predisposition, lifestyle factors, and hormonal exposures, offering a more comprehensive risk evaluation. 44 Similarly, BOADICEA, the first polygenic risk prediction model, leverages genetic information, family history, and BC pathology to calculate risk. 43 BRCAPRO instead focuses specifically on estimating the likelihood of carrying BRCA mutations but lacks consideration of non-genetic factors like lifestyle and breast density.45,46
Overview of risk assessment models.
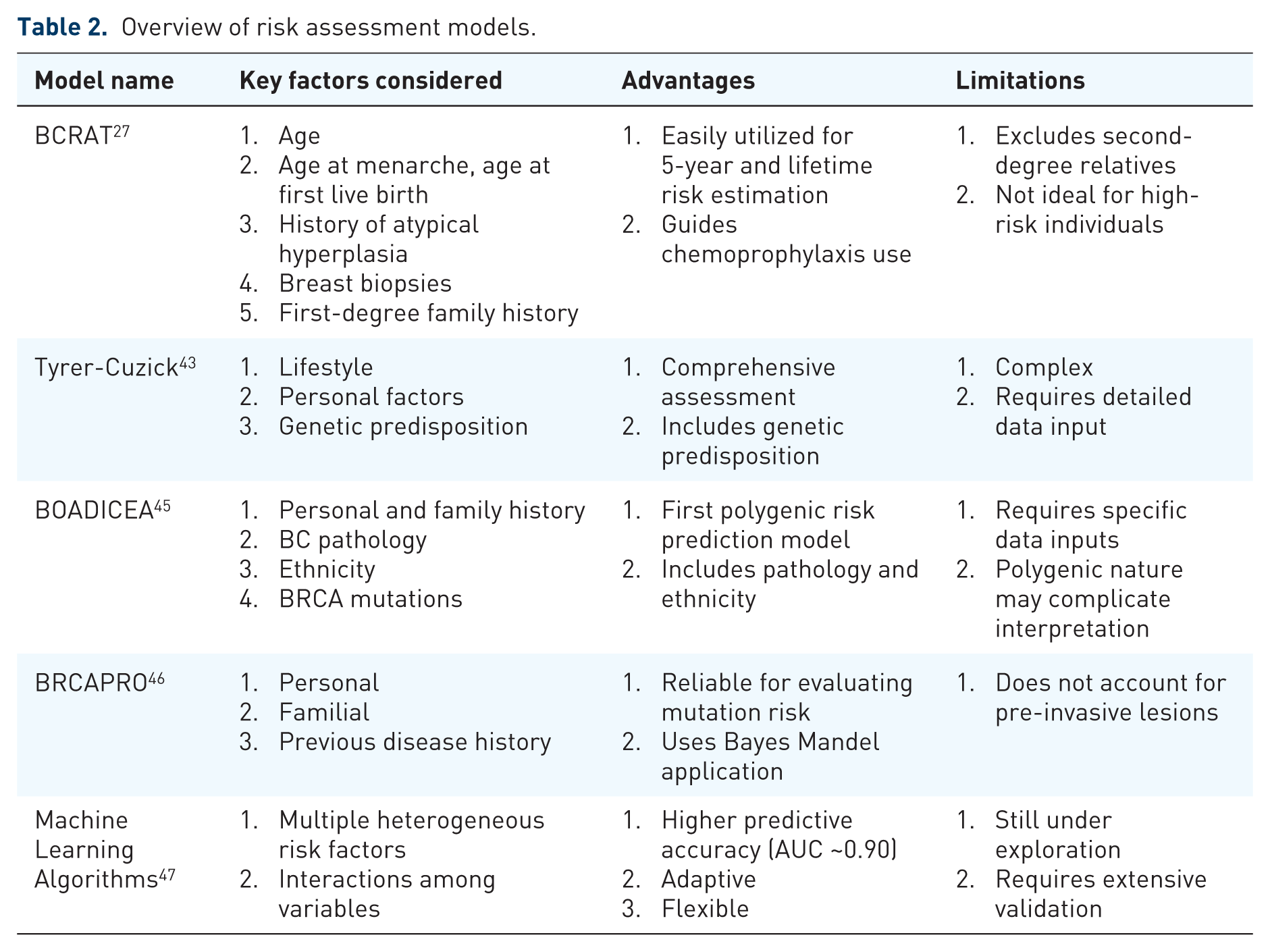
This table outlines the key advantages, limitations, and ideal applications of various risk assessment models.
Machine learning (ML) algorithms are emerging as highly promising and transformative tools in BC risk prediction. These computational models analyze complex interactions among heterogeneous risk factors, outperforming traditional methods in accuracy. Studies comparing ML approaches to the Gail and BOADICEA models already demonstrate marked improvements in predictive accuracy, with AUC values increasing from below 0.65 to approximately 0.90. 43 Techniques like Adaptive Boosting and Random Forest have shown particular promise in enhancing risk stratification 43 (Table 3).
Comparison of traditional and ML risk prediction models.

This table highlights performance differences between traditional models and ML algorithms in predicting BC risk.
BC, breast cancer; ML, machine learning; RF, radiomic features.
In addition, high-risk women, such as BRCA mutation carriers or those with dense breast tissue, benefit from advanced imaging techniques like MRI, which offers superior sensitivity for early detection.31,32,45 MRI screening is especially recommended for individuals with syndromic predispositions, such as Li-Fraumeni or Cowden syndrome, and for Black women, who face higher prevalence rates of aggressive subtypes like triple-negative BC.24,39 Meanwhile, intermediate-risk individuals may benefit from combining mammography with supplemental tools like ultrasound or abbreviated MRI40,41 (Table 4).
Risk categorization and screening recommendations.

This table summarizes risk categories and their corresponding screening protocols to guide clinical decision-making.
MRI, magnetic resonance imaging.
Reproductive and hormonal factors also play a significant role in risk assessment. Early menarche, late menopause, nulliparity, and prolonged use of hormone replacement therapy increase BC risk. Pregnancy-related factors, including gestational age at delivery and preeclampsia, have been linked to varying risk levels, further emphasizing the need for tailored approaches. 29 Also, lifestyle factors such as obesity, physical inactivity, and alcohol consumption exacerbate risk, while vitamin D levels and sleep duration may provide protective effects. 24
Personalized screening, therefore, not only enhances detection but also minimizes unnecessary interventions. By focusing on tailored strategies, healthcare providers can improve early detection rates while reducing false positives and psychological distress. For instance, MRI screenings are conducted at accredited facilities to ensure precision and reduce variability in imaging quality. 24 Emerging technologies, such as optical imaging and electrical impedance tomography, hold promise as supplementary tools to refine screening accuracy further.40,42
Despite advancements, significant disparities persist in BC outcomes across geographic, racial, and socioeconomic lines. Black women, for example, experience a 40% higher mortality rate than White women, driven by systemic inequities, delayed diagnoses, and limited access to advanced screening techniques.27,39,49,50 Geographic factors, including proximity to specialized care and healthcare facility density, also influence outcomes, though socioeconomic factors often play a more significant role. 43 Addressing these disparities requires targeted interventions, such as establishing screening programs in underserved areas and promoting equitable participation in clinical trials. 43
Personalized screening strategies incorporating genetic, hormonal, and lifestyle factors offer the potential to revolutionize BC detection and prevention. Integrating advanced predictive models and emerging technologies with equitable access can bridge existing gaps and optimize outcomes for diverse patient populations.
Breast imaging biomarkers
There are many substantial biomarkers for BC, including receptor, protein, and imaging biomarkers, each of which has its benefit. Some crucial receptor biomarkers for BC are ERs and PRs. 51 ER-positive BC indicates that the tumor has ERs that encourage estrogen to bind to the tumor, and PR-positive BC indicates that the tumor has PRs that encourage progesterone to bind to the tumor. 51 Each of these biomarkers encourages growth and division of the tumor and influences which treatment will be most beneficial for the patient. 51 These are tested in the tumor through immunohistochemistry, where a sample of the tumor is biopsied and stained to identify the presence of these proteins or receptors. 51
Some protein-based biomarkers are the HER2, programmed death-ligand 1 (PD-L1), and trophoblast cell surface antigen 2 (TROP2).52,53 HER2 expression shows the presence of the HER2 protein in the BC Tumor, which, if present in excess, encourages the growth and division of the BC cells. 52 PD-L1 is a protein that, if present in the cancer cells, will prevent the body’s T cells from attacking the cancer and allowing it to grow and divide without problem. 53 TROP2 is a glycoprotein that, when overexpressed in cancer cells, encouraged the tumor to heal itself, grow, and invade other parts of the body. 54 Each of these in an over-abundance in the tumor encourages the tumor to live and thrive within the body. All three of these proteins are identifiable through immunohistochemistry testing.52 –54
In contrast to receptor and protein biomarkers, imaging biomarkers are defined as objective and measurable characteristics from medical imaging that reflect the presence or progression of disease, such as BC, or a patient’s response to therapy. 55 These biomarkers play a pivotal role in screening, diagnosis, and prognosis by allowing the identification of specific tumor features noninvasively, contributing to earlier detection and characterization. 55 Imaging techniques used to detect these biomarkers include mammography, ultrasound, and MRI. Among these, MRI presents high sensitivity, specificity, and positive predictive value in detecting breast abnormalities, followed by mammography and ultrasound.56,57 While MRI alone has the highest sensitivity, studies show that combining multiple modalities, such as positron emission tomography (PET)/CT scans alongside mammography and ultrasound, enhances diagnostic accuracy and enables comprehensive evaluation of breast lesions. 27
Breast imaging biomarkers can be categorized as qualitative, quantitative, or ordinal. Qualitative biomarkers include visual characteristics such as tumor shape, margins, and calcification patterns, which can be identified through mammography and aid in distinguishing benign from malignant lesions. 58 For example, irregular shapes or spiculated margins are often associated with malignancy. 59 Quantitative biomarkers provide measurable parameters, such as tumor size or metabolic activity, with MRI and PET scans offering detailed information on tumor volume and metabolic tumor activity. This data can help assess tumor aggressiveness and monitor treatment response. 60 Ordinal biomarkers, like breast density and background parenchymal enhancement (BPE), provide ranked or categorical data. Higher breast density, as assessed on mammography, is associated with increased BC risk and can complicate imaging interpretation, thus requiring additional screening for more accurate diagnosis.61,62
Several specific imaging biomarkers have proven valuable in BC assessment. Microcalcifications, detectable through mammography, are calcium deposits that may indicate ductal carcinoma in situ (DCIS) or invasive ductal carcinoma (IDC). 63 Their pattern, size, and distribution provide critical insights, often differentiating benign from malignant processes. Similarly, breast density is another significant biomarker; women with denser breast tissue face a higher risk of BC, as well as reduced sensitivity of mammography in detecting tumors within dense tissue. 64 High breast density can signal a need for supplemental screening, like an MRI, to ensure a thorough assessment. 65 In addition, tumor vascularity and texture are biomarkers assessed by MRI, where contrast enhancement patterns reveal blood flow and tissue heterogeneity, aiding in the differentiation between tumor types and the prediction of treatment responses. 60 MRI has been shown to have a 92% sensitivity and 70% specificity with its ability to type BC. However, MRI does have a higher false-positive rate with approximately 83% of patients obtaining an initial false positive.66,67 This imposes a serious downside to the imaging mechanism as a reliable diagnostic tool.
Beyond diagnosis, imaging biomarkers are instrumental in predicting treatment efficacy. For instance, fludeoxyglucose-positron emission tomography (FDG-PET) combined with CT allows for accurate identification of active tumors versus post-treatment scarring. The level of FDG uptake, or FDG avidity, provides an objective measure that can track treatment response, especially in chemotherapy. 68 Another promising technique is texture analysis on MRI scans, which evaluates gray-level intensity variations within the tumor. Studies have shown that high BPE is linked to an increased risk of recurrence and may also serve as a predictive marker for hormone therapy response.69,70 Research by King et al., 71 for example, demonstrated an association between BPE and serum estrogen levels, suggesting BPE’s utility as a marker for estrogen-driven BC risk.
Clinically, imaging biomarkers have already shown substantial benefits, but their full integration into practice is currently under development. Emerging research focuses on combining these biomarkers with artificial intelligence (AI) to streamline BC diagnosis. A recent study, 72 for example, demonstrates how deep learning models trained on large imaging datasets can enhance diagnostic accuracy and reduce interpretation time, though further validation is needed before a systemic clinical adoption. As technology advances, imaging biomarkers, alongside AI support, hold promise for enabling rapid, precise, and personalized BC care.
Radiomics in BC imaging
Radiomics is a rapidly growing field in medical imaging that enables the extraction of quantitative features from modalities, among which are MRI, CT, and PET scans. These features include tumor volume, shape, texture, and FDG uptake, offering clinicians a detailed view of the tumor’s characteristics beyond conventional imaging.73,74 By mathematically analyzing image data, radiomics provides a deeper understanding of tumor biology, facilitating more precise diagnostics and tailored treatment strategies. The radiomics workflow includes segmentation, processing, feature extraction, and deep learning (Figure 2).

Workflow of the Radiomics Process. The figure outlines the key steps involved in radiomics analysis, including segmentation, image processing, feature extraction, and deep learning integration. Each step contributes to the comprehensive evaluation of imaging data for advanced diagnostic and predictive insights.
Segmentation, the initial step, identifies regions or volumes of interest in either two-dimensional (2D) or three-dimensional (3D) imaging. 75 Accurate segmentation ensures that meaningful data from the tumor area are processed. In the next step, images undergo processing through algorithms designed to quantify pixel spacing, gray-level intensities, and histogram bins, translating the raw imaging data into measurable variables. 75 Subsequently, radiomic features (RFs) are extracted according to standardized guidelines developed by the Image Biomarker Standardization Initiative, ensuring consistency across studies and applications. 75 The final stage involves incorporating these features into ML and deep learning systems. These systems use data points, such as individual image pixels, to identify patterns and improve the accuracy of diagnosis, prediction, and prognosis. 75
This figure is created by the authors with the data summarized from Van Timmeren et al. 75
The integration of radiomics with AI has significantly expanded its clinical potential. AI systems, particularly those employing deep learning, analyze imaging data to identify patterns linked to specific clinical outcomes. 76 Over time, these systems progress from recognizing simple patterns to understanding complex tumor characteristics, eventually providing nuanced insights into tumor behavior. 76 For instance, AI can identify risk factors or subtle imaging features that may not be immediately apparent to clinicians, offering preliminary diagnostic support and aiding in treatment planning. 77
Radiomics has proven particularly beneficial in BC management, where it assists in distinguishing between benign and malignant lesions, subtyping tumors, and correlating imaging features with clinicopathological information. 78 Key RFs, as texture, shape, and intensity, play pivotal roles in this process. Texture, reflecting tumor heterogeneity, is a vital biomarker for prognosis and treatment selection, as tumor subtypes often exhibit characteristic textures.64,79 Shape provides quantitative insights into tumor aggressiveness; for example, irregular shapes and compactness indices can help differentiate malignant from benign tumors, directly informing prognosis at the time of diagnosis. 80 Intensity, which measures the relative normalcy or abnormality of a mass, serves as a critical indicator of malignancy and potential patient outcomes. 81 Together, these features offer a noninvasive approach to improve diagnosis, assess prognosis, predict therapeutic responses, and guide treatment planning. 82
One of the most promising aspects of radiomics is its ability to integrate with ML, and AI to enhance the predictive accuracy of cancer diagnostics. By creating centralized databases, radiomics feeds AI systems with labeled data, enabling algorithms to further learn and improve, and identify imaging patterns linked to specific diagnoses. 75 Studies have demonstrated that machine-learning models significantly outperform traditional diagnostic tools. For example, a meta-analysis by Fu et al.82,83 evaluated ultrasound-based radiomics in BC diagnosis, achieving sensitivity and specificity rates of 78% and 77%, respectively. Another study by Haghshomar et al. 84 explored AI’s utility in liver malignancy diagnosis, reporting a predictive accuracy of 84.3%. These findings highlight the transformative potential of AI-radiomics integration in different areas of oncology, though further research is required to validate clinical applicability and optimize performance.
In BC care, radiomics and AI can streamline the diagnostic process, particularly for aggressive and complex tumors, ensuring patients receive timely and effective treatment. Radiomics can also enhance early detection and personalized therapy, leveraging imaging biomarkers to tailor interventions to individual tumor characteristics. While the field is still evolving, current evidence highlights its promise in advancing precision medicine and improving patient outcomes through innovative diagnostic and prognostic tools.76,83,84
Radiomics of DCIS
DCIS encompasses a diverse spectrum of epithelial proliferations confined to the milk ducts and is frequently detected through BC screening programs. 85 Over recent decades, the incidence of DCIS has risen sharply, now comprising 20%–25% of all BC diagnoses, largely due to widespread screening efforts. 86
Although DCIS is often categorized alongside invasive BCs as “stage 0” under the National Comprehensive Cancer Network guidelines, this classification is debated. Biologically, DCIS lacks the capacity to invade or metastasize, challenging its designation as a true malignancy. 85 It is recognized as a potential precursor to IDC, though its pathogenesis is not as well characterized. 87 The clinical interpretation of DCIS has evolved over time: initially regarded as a critical target for early intervention due to its perceived role as a direct precursor to invasive cancer, 87 more recent data suggest that only about 40% of DCIS cases actually progress to invasive disease. 88
As BC treatment options become increasingly tailored, it is vital for clinicians to access reliable prognostic tools to guide management decisions. 89 Histological evaluation of DCIS involves multiple classification systems based on architecture and nuclear features, with nuclear grade—defined by features such as cellular proliferation and mitotic activity—playing a particularly prominent role.90,91 The most widely used system adopts a three-tier classification of nuclear grade. 92
High-grade and intermediate-grade DCIS are the most common subtypes, comprising approximately 42% and 43% of cases, respectively, while low-grade DCIS is less frequent (14%). 92 Nuclear grade serves as a crucial prognostic indicator, informing assessments of progression risk and eligibility for active surveillance.92,93 It also correlates with imaging characteristics; for instance, high-grade DCIS typically shows more prominent enhancement on MRI, while low-grade lesions are more likely to be visible on ultrasound. 92 Radiomics studies have shown that features extracted from mammography and MRI often reflect underlying nuclear grade. However, since nuclear grade is usually confirmed via histopathology at biopsy, the utility of imaging for this purpose remains somewhat limited in current practice.92,93
The term radiomics was first introduced 94 in 2012 to describe the process of deriving quantitative features from medical images that may capture underlying biological heterogeneity. The radiomics hypothesis posits that differences in genomic profiles may translate into phenotypic variability detectable through imaging, with potential implications for prognosis. 95 RFs—which represent subvisual image characteristics such as texture, shape, and intensity distribution—can be extracted from modalities like CT, MRI, and mammography. These noninvasive assessments can be repeated over time and provide rich, in vivo data for characterizing tumors. 93 Radiomics supports a precision medicine framework by allowing disease monitoring and treatment response prediction. A wide array of RFs can be computed using open-source or commercial platforms, encompassing descriptors such as tumor margins, pixel intensity patterns, and textural complexity.93,96
In DCIS, both mammography and MRI have shown potential to reveal features aligned with histopathologic findings, especially nuclear grade.92,97 High-grade DCIS tends to present with striking enhancement and fine linear calcifications, whereas low-grade lesions are more subtle and often best visualized via ultrasound. 98 Radiomics, by analyzing these imaging traits quantitatively, provides a tool to noninvasively assess tumor heterogeneity and morphology.99,100 This could enable clinicians to better stratify risk and make more personalized treatment decisions. Emerging ML-based radiomics models have demonstrated initial success in differentiating aggressive from indolent DCIS and in forecasting recurrence risk.99 –101
Given the potential of DCIS to evolve into invasive carcinoma, surgical excision remains the standard of care. However, the stable incidence of invasive BC despite the increased detection of DCIS suggests possible overtreatment. 99 Conventional imaging techniques remain central to diagnosis, but their limitations are well recognized: suboptimal sensitivity, discomfort from biopsies, and delayed results. 82
While pathology remains the cornerstone of diagnosis, imaging biomarkers may offer adjunctive value, particularly by helping to avoid overtreatment in low-risk patients.
Recent developments in radiomics have improved our ability to predict outcomes such as disease-free survival (DFS) in BC using noninvasive methods. By extracting and analyzing high-dimensional imaging data, radiomics can provide a comprehensive profile of tumor biology and heterogeneity. A meta-analysis encompassing 13 studies and over 5000 patients found that radiomics models achieved a C-index of 0.763 in training sets and 0.702 in validation cohorts for DFS prediction. 103 Incorporating clinical and pathological data further improved prediction accuracy, raising the validation C-index to 0.840. Radiomics features derived from preoperative ultrasound have also been shown to correlate with the expression of molecular markers, supporting more tailored approaches in managing DCIS. 104
Challenges and limitations of biomarkers and radiomics in BC
Integrating biomarkers and radiomics into routine clinical practice for BC diagnosis and treatment has been transformative, but several challenges and limitations still challenge their full adoption. These issues span technical variability, standardization gaps, data diversity, and ethical and regulatory hurdles, all of which require careful consideration to bridge the gap between research and clinical implementation.
Variability in imaging protocols
One of the most significant challenges lies in the inconsistency of imaging protocols across institutions and worldwide. As highlighted in recent bibliometric analysis, 105 variations in equipment calibration, imaging techniques (e.g., MRI, CT, and ultrasound), and patient preparation directly impact RFs, challenging data standardization.106,107 In particular, scanner model variations can significantly affect feature extraction and reproducibility, especially in MRI studies, which include many more parameters than ultrasound or mammography. 105 These inconsistencies reduce the reproducibility and generalizability of radiomic models, posing a major obstacle to clinical integration. Gomez et al. 108 pointed out how such variability affects diagnostic accuracy and treatment decisions, emphasizing the need for consistent imaging protocols. Tariq et al. also highlighted how inconsistent imaging protocols lead to heterogeneity in data, making it difficult to develop and validate robust AI models. This variability affects the generalizability of AI tools, as models trained on one protocol may not perform well on data from another institution. 109 Thériault-Lauzier et al. further elaborated on this challenge, noting that different manufacturers use different acquisition parameters and post-processing approaches, which can lead to variability in imaging protocols. This variability not only affects algorithm performance but also complicates the standardization of image quality across institutions. 100 Collaborative efforts like the Quantitative Imaging Biomarkers Alliance (QIBA) aim to address these challenges by developing standardized imaging processes and ensuring uniformity in feature extraction and analysis. 110
Lack of standardized data practices
Beyond imaging protocols, the absence of universally accepted methodologies for RF extraction is another significant barrier. Different segmentation techniques, as manual, semi-automated, or fully automated, can yield vastly different RFs, complicating the validation and reproducibility of findings across studies,111,112 particularly noting that while automated methods are suitable for extensive data analysis, they perform optimally only when lesions have well-defined margins. 113 This inconsistency extends to biomarker testing, where variations in assay techniques and reporting formats further hinder comparability. 114
Elahi and Nazari 113 point out that Radiomics, coupled with AI, can provide Quantitative imaging biomarkers that can improve specificity and sensitivity across MRI, mammography, CESM, DBT, and ultrasound. Although challenges such as reproducibility and data integration remain, the potential to reduce unnecessary biopsies and enhance early cancer detection could transform clinical practice. Standardization efforts through initiatives like the Quantitative Imaging Network and QIBA are urgently needed to align data acquisition, processing, and analysis, fostering reliable radiomics research and clinical applications.78,115 Nandita et al. suggested establishing a standard methodology based on metrology for validating image acquisition and analysis methods. This approach aims to reduce variability and improve clinical workflows. 116
Small and homogeneous datasets
Radiomic models frequently rely on small, homogeneous datasets for training, limiting their applicability to diverse clinical populations. This lack of diversity undermines the external validity of these models, raising concerns about their generalizability and therefore applicability in real-world settings.115,117 Developing large, diverse datasets through multi-institutional collaborations and data-sharing initiatives by emphasizing the importance of multicenter validation using large-scale cohorts or data from open databases is essential to address this limitation. 78 Expanding repositories would not only improve model robustness but also mitigate biases and enhance the accuracy of AI-driven applications in BC care. 118
Ethical and regulatory challenges
The use of AI and radiomics in BC screening introduces a plethora of advantages as well as critical ethical challenges, particularly regarding patient privacy and data security. Handling large datasets inherently risks breaches of sensitive health information, necessitating prompt and stringent regulations. 119 Algorithmic bias remains another pressing issue. Training AI models on unrepresentative datasets can also accentuate healthcare disparities, resulting in suboptimal care to underrepresented groups. 120 Ethical concerns are further compounded by the “black box” nature of many AI algorithms, which lack transparency in their decision-making processes. This opacity can undermine trust among clinicians and patients, posing significant barriers to adoption. 121
From a regulatory standpoint, existing guidelines for AI-based tools are still evolving and often fail to address the intricacies of radiomics. For instance, the FDA’s current framework for post-marketing surveillance is not robust enough to ensure the long-term safety and efficacy of AI tools in diverse clinical environments. 122 Establishing comprehensive regulations that include rigorous validation, real-world monitoring, and transparency requirements is vital for addressing these challenges.123,124
Limitations of imaging modalities
Imaging modalities themselves present some inherent limitations. Elahi and Nazari 113 comprehensively outline these challenges, noting that image quality significantly affects high-level feature reproducibility, particularly in ultrasound-based radiomics studies. Wu et al. 105 corroborate this by discussing how variations in imaging protocols and post-processing for image normalization can reduce the reproducibility of BC radiomic studies.
Thériault-Lauzier et al. proposed using AI-based techniques to address heterogeneity in image quality. This could potentially help normalize data from different protocols. 125 While MRI provides high sensitivity for BC detection, it also generates higher false-positive rates, leading to unnecessary biopsies. 126 Mammography, though widely used, has limited sensitivity in dense breast tissue, which can obscure lesions and delay diagnosis. 127 Radiomics could significantly enhance the utility of these modalities, but only if standardized imaging protocols and data harmonization efforts are implemented across clinical centers worldwide. 112
Gupta et al. emphasized the importance of community-driven, open-source tools like MONAI Deploy to facilitate standardization across institutions. This study also recommended compatibility with existing standards like DICOM, HL7, and Fast Healthcare Interoperability Resources to ease implementation across diverse institutions. The implementation of AI in medical imaging also necessitates significant infrastructure adaptations. 128
In addition, Hong et al. discussed the need for advanced computational capabilities to support complex AI methodologies like federated learning and digital twins. Gupta et al. 128 suggested the use of cloud technologies to provide scalable computational resources. Thériault-Lauzier et al. 125 proposed the PACS-AI platform as a solution for integrating AI models into existing clinical workflows. Bizzo et al. 129 emphasized the importance of post-deployment monitoring infrastructure to ensure proper and safe use of AI tools.
The development of open-source tools like MONAI Deploy (Gupta et al.) and PACS-AI (Thériault-Lauzier et al.) aims to provide standardized infrastructure for AI implementation. Thériault-Lauzier et al. 125 also proposed the use of containerized microservices to enhance scalability and maintainability of AI systems.
Therefore, the integration of biomarkers and radiomics into BC care entails interconnected challenges that currently prevent their full adoption. Addressing these barriers requires synergistic efforts to standardize imaging protocols, develop diverse and comprehensive datasets, establish robust regulatory frameworks, and tackle ethical considerations. Collaborative initiatives, international consortia, and expanded data-sharing efforts are critical to realizing the potential of radiomics and AI in improving BC diagnosis, treatment, and patient outcomes.
Conclusions and future perspectives
BC continues to be a significant global health challenge, contributing substantially to morbidity and mortality worldwide. Despite remarkable advancements in screening, diagnosis, and treatment, the complexity and heterogeneity of the disease demand continuous innovation to improve patient outcomes. This review highlights the transformative potential of biomarkers, radiomics, and AI in enabling personalized BC care. By integrating these tools, clinicians can achieve earlier detection, more precise risk stratification, and tailored therapeutic interventions, ultimately enhancing survival rates and quality of life.130,131
Advancements in biomarkers and radiomics
Biomarkers have revolutionized the understanding and management of BC by offering critical insights into tumor biology, prognosis, and therapeutic response. Established markers, such as ER, PR, and HER2, have long guided treatment decisions. However, the evolving molecular landscape of BC necessitates the identification of additional biomarkers to capture its diverse subtypes and complexities. This evolution has paved the way for personalized medicine, allowing therapies to be tailored to the distinct molecular characteristics of each tumor.132,133 Similarly, radiomics has emerged as a promising tool in BC imaging, enabling the extraction and analysis of quantitative features from MRI, CT, and mammography. These features, encompassing tumor texture, shape, and intensity, provide noninvasive biomarkers for predicting disease progression, subtyping tumors, and assessing treatment response. Integration of radiomics with AI offers the potential to enhance diagnostic accuracy, streamline workflows, and uncover hidden patterns in imaging data, significantly advancing precision medicine.134,135
The role of AI and multi-omics integration
The convergence of AI, ML, and multi-omics techniques represents a significant and promising frontier in BC research. By analyzing large, multi-dimensional datasets, AI algorithms can uncover intricate relationships between genomic, proteomic, and radiomic markers, offering deeper insights into tumor behavior and treatment outcomes. ML models, for instance, have demonstrated their ability to predict therapeutic responses with unprecedented accuracy, guiding clinicians toward more effective and individualized treatment strategies.136,137
Multi-omics approaches that combine genomics, transcriptomics, proteomics, and radiomics hold immense promise for refining risk stratification and optimizing treatment selection. Understanding the interplay between these data layers can provide a holistic view of cancer biology, enabling the development of multi-dimensional biomarkers that capture the heterogeneity of BC.95,138
Challenges and future directions
Despite these advancements, several challenges must be addressed to fully integrate biomarkers and radiomics into clinical practice. Variability in imaging protocols and the lack of standardized methodologies remain significant barriers to reproducibility and generalizability. Standardizing data acquisition, segmentation techniques, and feature extraction processes is essential to ensure consistency across institutions and studies.139,140 Collaborative initiatives, such as the QIBA, have made progress in this area, but more work is needed to harmonize imaging practices globally. 141 The reliance on small, homogeneous datasets for training radiomic and AI models limits their applicability to diverse patient populations. Expanding datasets through multi-institutional collaborations and international consortia can improve model robustness and mitigate biases, ensuring equitable application of these technologies.142,143
Ethical and regulatory challenges also require attention. Ensuring patient privacy and data security is paramount when handling large datasets, while addressing algorithmic bias is critical to prevent disparities in care. Transparent AI models that clinicians and patients can understand are essential to build trust and promote adoption. Regulatory frameworks must evolve to include rigorous validation processes for AI-driven technologies, ensuring safety, effectiveness, and equity.144,145
Future research priorities
Moving forward, validating radiomics in larger, multi-institutional studies is a priority to establish its clinical utility. Combining radiomics with multi-omics data, including genomic and transcriptomic information, represents a promising avenue for developing more precise risk assessment tools. In addition, research should focus on integrating imaging biomarkers with real-time patient monitoring systems to enhance longitudinal care and early detection.146,147
Efforts should also prioritize addressing healthcare disparities. Ensuring access to advanced screening and treatment options for underserved populations is critical to reducing inequities in BC outcomes. Strategies such as increasing clinical trial participation among diverse groups and developing culturally appropriate care models are vital for achieving this goal. 82
Although significant progress has been made in BC diagnosis and treatment, achieving equitable, precise, and personalized care remains an ongoing challenge. Biomarkers, advanced imaging modalities, and AI-driven solutions hold the key to transforming BC management, offering earlier detection, tailored therapies, and improved prognoses. By promoting and strengthening collaboration among researchers, clinicians, and policymakers and addressing existing challenges, the healthcare community can continue to advance toward the goal of truly personalized medicine. These efforts will not only enhance survival rates and quality of life but also reduce the global burden of BC.
Declarations
Footnotes
Acknowledgements
We thank Dana Ataya for the opportunity to learn from a global Leader in Medicine. We are grateful to be part of MedNews Week. This manuscript benefited from the use of OpenAI’s language model, ChatGPT, for assistance with language editing. All outputs were reviewed and verified by the authors. Some figures in this manuscript were created with ![]() .
.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Competing interests
The authors declare that there is no conflict of interest.
Availability of data and materials
No patient data were directly utilized in this study.
