Abstract
Background:
Immune checkpoint inhibitors (ICIs) have shown efficacy in endometrial cancer (EC); however, their efficacy varies according to mismatch repair (MMR) status. Notably, even among patients with MMR-deficient (MMRd) or microsatellite instability-high (MSI-H) tumors, approximately one-third exhibit primary resistance to ICI monotherapy.
Objectives:
We aimed to characterize dissimilarities in the tumor immune microenvironment of ICI-treated MMRd/MSI-H versus MMR-proficient (MMRp)/microsatellite stable (MSS) EC, and to identify mechanisms of resistance.
Design:
Adults with histologically confirmed advanced or recurrent EC treated with ICIs in 6 French comprehensive cancer centers were included. Patients without available archival formalin-fixed paraffin-embedded primary tumor samples were excluded. Clinical data were collected retrospectively.
Methods:
Patients were classified as ICI-Responders or Non-Responders based on best objective response. A seven-color multi-immunofluorescence staining (CD20, CD4, CD8, FoxP3, CD68, CK, and DAPI) was performed on sections from archival formalin-fixed paraffin-embedded primary tumors. Cell densities and spatial proximity were analyzed using inForm software. T/B lymphoid aggregates (LA) and tertiary lymphoid structures (TLS) were separately quantified. Microsatellite status, presence of LA/TLS, and immune cell densities were correlated to response to treatment.
Results:
Twenty-one MMRd/MSI-H and 12 MMRp/MSS tumors were analyzed. We observed more MMRd/MSI-H tumors with LA/TLS compared to MMRp/MSS cases: 81% versus 17%, p ⩽ 0.001. There were more CD8+ T effector cells in the vicinity of B cells in MMRd/MSI-H tumors compared to MMRp/MSS tumors (1.26 (0–3.40) vs 0.49 (0–1.86), p = 0.017), suggesting cooperation between CD8+ T cells and B cells in MMRd/MSI-H tumors. No differences were shown in terms of the presence of LA/TLS and the subsequent response to ICI in EC (p = 0.400). Using a multivariate logistic regression model, we found that a low density of CD68+ tumor-associated macrophages (TAMs) in the stroma, was associated with response to ICI in EC (odds ratio = 11.67, 95% CI (1.69–237.45), p = 0.033) and showed good accuracy in predicting response to ICI in the whole cohort (AUC = 0.75, 95% CI (0.59–0.91)).
Conclusion:
We characterize the immune landscape in EC patients treated with ICIs. Distinct immune infiltrate patterns were observed in MMRd/MSI-H and MMRp/MSS tumors. A significant negative association between TAM density and ICI response was shown.
Plain language summary
Immune checkpoint inhibitors (ICIs) are promising treatments for endometrial cancer (EC), particularly in patients with specific genetic profiles, such as mismatch repair-deficient or microsatellite instability-high (MMRd/MSI-H) tumors. However, these treatments do not work for everyone, and many patients with MMRd/MSI-H tumors show resistance to ICIs. This study analyzed differences in the immune environment of EC tumors with and without these genetic markers to understand why some patients respond better to ICIs. Tumor samples from 33 patients treated with ICIs were studied using advanced staining techniques to visualize immune cells. Researchers also measured the densities of various immune cells, including T cells, B cells, and tumor-associated macrophages (TAMs), and examined their interactions within the tumors. The study found that MMRd/MSI-H tumors had more immune structures, such as lymphoid aggregates and tertiary lymphoid structures, compared to other types of EC. These tumors also showed more collaboration between two types of immune cells: CD8+ T cells (which attack cancer cells) and B cells. However, the presence of these structures did not predict whether patients would respond to ICI treatment. Interestingly, the researchers discovered that a lower number of TAMs, which can suppress the immune response, was strongly linked to better responses to ICIs. This finding suggests that TAM density could serve as a potential biomarker to predict which patients might benefit most from this therapy. Overall, this study highlights differences in immune environments between EC tumor types and identifies TAMs as a potential target to improve ICI efficacy. This research may help refine treatment strategies and improve outcomes for patients with endometrial cancer.
Introduction
Endometrial cancer (EC) is the most common gynecological cancer in Western countries. 1 While most patients with localized disease have an excellent prognosis, a subset of patients presents recurrent and/or metastatic disease without treatment of curative intent. While carboplatin and paclitaxel have traditionally served as the established first-line therapy for advanced and recurrent EC, trials investigating the addition of immune checkpoint inhibitors (ICIs) have been successful. In five randomized trials, the ICIs dostarlimab, durvalumab, atezolizumab, and pembrolizumab improved progression-free survival (PFS) when added individually to carboplatin and paclitaxel, with a trend for overall survival (OS) benefit in all-comers for some ICIs.2 –6 Greatest PFS benefits were observed in those trials among those with mismatch repair deficient (MMRd).2,3,6 –8 Although the importance of the use of ICIs in EC is now established, some questions remain. Identification of the best combination in MMR-proficient (MMRp)/microsatellite stable (MSS) tumors (chemotherapy, anti-angiogenic therapy, and/or alternative therapies) still needs to be identified. 5 Despite their ICIs-sensitive profile, 29%–36.1% of MMRd/microsatellite instability-high (MSI-H) will progress under ICI alone as reported in the Garnet and Keynote-158 trials9,10 and may require combination therapies. MMRd/MSI-H subtypes are characterized by a high tumor mutation burden, 11 increased infiltration by both T cells and B cells, and higher expression of both PD-1 and PD-L1, 12 which could explain a better clinical response when treated with anti-PD-1/PD-L1 compared to MMRp/MSS patients. Indeed, tumor-infiltrating B and T cells can organize into tertiary lymphoid structures (TLS) in EC 13 and these structures have been shown to be associated with response to ICIs in other solid tumors.14 –16 In contrast, CD68+ tumor-associated macrophages (TAMs) and CD4+ regulatory T cells (Tregs), which were found to be associated with EC aggressivity,17 –20 could contribute to the limited efficacy of ICI in some patients.21 –23 Herein, our objectives were first to characterize pre-treatment EC samples, followed by analyses of dissimilarities in the tumor immune microenvironment of ICI-treated MMRd/MSI-H versus MMRp/MSS EC, and finally to identify possible mechanisms of resistance in EC, using in situ multispectral immunofluorescence (IF) tumor tissue staining and digital image analysis.
Methods
Patient cohort and tumor samples
We included all patients reported by treating physicians from ARCAGY-GINECO (National Investigators Group for Ovarian and Breast Cancer Studies in France) as having endometrial cancer (EC), regardless of histologic subtype. This was based on declarative data and may not be exhaustive. Patients were identified through analysis of their own center’s database from October 2020 to May 2021. Eligible patients were 18 years of age or older and had received immunotherapy (anti-PD-1, anti-PD-L1, anti-CTLA-4, or others) alone or in combination in the recurrent setting for a histologically confirmed advanced-stage EC. Patients with no primary tumor samples available or lost to follow-up were excluded. Archival formalin-fixed paraffin-embedded (FFPE) primary tumor samples were collected prospectively. All the available clinicopathologic information was retrieved retrospectively from the clinical records for the patients included. This included age, histology, initial FIGO stage, number of previous lines, MMRd/MSI-H status, p53 status, follow-up information for recurrence, and vital status. As per standard clinical practice, MMRd/MSI-H status was determined in priority on the basis of immunohistochemistry (IHC). In case of ambiguous results of IHC (lack of positive internal control, heterogeneous loss of MMR protein expression), the MMRd/MSI-H status was assessed by PCR/NGS.24,25 Patients were classified as ICI-Responders (R; complete or partial response as a best overall response—BOR) or Nonresponders (NR; stable or progressive disease as BOR) according to local assessment. The best overall response was defined as the best response recorded from the start of the treatment until disease progression/recurrence. The objective response was evaluated according to the RECIST 1.1 criteria.
Seven-color multiplex IF tissue staining
A hematoxylin and eosin (H&E) stained slide of each sample was examined by a trained pathologist to confirm tissue quality, select samples for multiplexed-IF (multi-IF) staining, and annotate the tumor area. Seven-color sequential multi-IF staining was performed with the BOND RX stainer (Leica Microsystems, Buffalo Grove, IL, USA) using previously validated antibody (Ab) panels, and a tonsil section was included in each staining batch to inspect the overall staining fidelity for all markers. After deparaffinization, rehydration, and antigen retrieval, 4 µm FFPE tumor sections were sequentially stained with each primary antibody, followed by OPAL-HRP secondary antibody incubation, and then revealed with tyramide signal amplification and OPAL fluorophores (Akoya Biosciences, Marlborough, MA, USA) and the same cycle was reproduced until staining with the last Ab of the panel (Supplemental Table 1). Slides were counter-stained with spectral DAPI (Akoya Biosciences), and cover slips were mounted using Prolong Gold medium (Invitrogen, Paisley, UK). Whole slides were imaged at a 20× magnification using the Vectra Polaris multispectral scanner (Akoya Biosciences).
Analysis of multi-IF digital images
Digital images were visualized with the Phenochart viewer (Akoya Biosciences) and representative regions of interest (ROIs; median 10, range (3–35) depending on the tumor surface) were selected by one researcher blinded to the study endpoint. After spectral unmixing using the synthetic library of the inForm software (Akoya Biosciences), tissue and cell segmentation were performed for the other multi-IF panel to classify tumors, followed by cell phenotyping. A machine learning algorithm was trained using user-specified tissue annotations and the epithelial marker signal to accurately segment tumor tissue, stromal tissue, and background. Individual cells were segmented using the nuclear DAPI signal. The algorithm was trained on one-third of the selected ROIs and validated on the remaining ROIs. These ROIs, consisting of one to two per tumor sample, were chosen to be representative of the entire cohort prior to initiating batch analysis across all slides and all ROIs (median 10, range (3–35) depending on the tumor surface). After manual inspection, the algorithm was again optimized, if necessary, until correct segmentation and phenotyping (>80% of cells correctly identified). Different phenotypes were analyzed: CD20+ as B cells, CD68+ as TAMs, CD4+/CD68–/FOXP3– as CD4 helper T cells, CD4+/CD68–/FOXP3+ as CD4 Tregs, CD8+/FOXP3– as CD8 T cells, and CD8+/FOXP3+ T cells. Finally, the phenotyping data were exported from inForm, and tabulated reports including cell densities and spatial analyses (count within radius—20 µm) were obtained using the R package phenoptr or PhenoptrReports (Akoya Biosciences). TLS were defined as dense aggregates of CD20+ cells (B-follicle) adjacent to a T cell (CD4+) rich areas on whole slides. Lymphoid aggregates (LA) were dense clusters of lymphocytes with a lack of segregated B-follicle and T-cell zones. LA/TLS were considered together as they represent different stages of TLS maturation and are both associated with good prognosis in EC.26,27
Statistical analysis
Statistical analyses were performed using R programming language v4.0.2 (R Foundation, University of Auckland, Auckland, New Zealand). Nonparametric statistical tests were used. Survival analysis included Kaplan-Meier estimators and likelihood ratio (LR) tests due to the small sample size. Duration of response was defined as the time from onset of response to progression or death due to any reason, whichever occurs earlier. Tests p-values <0.05 were statistically significant. No statistical comparison was performed to compare response rate and survival between MMRd/MSI-H and MMRp/MSS tumors due to the heterogeneity in therapies received. The initial phase involved a comparison of immune cell density and the presence of LA/TLS between MMRd/MSI-H and MMRp/MSS tumors. In the second phase, immune cell density and the presence of LA/TLS were compared between R and NR in the whole cohort. Multivariate logistic regression models were generated using logistic regression with the least absolute shrinkage and selection operator (LASSO) penalty for response to therapy. This model allows both variable selection and regression coefficient estimation by maximizing the log-likelihood function. This model is used in cases of several potential markers to test the model relative to the number of observations. LASSO regression analysis was applied to microsatellite status, presence of LA and TLS, and density of immune cells with p-values <0.05, as estimated through univariable Wilcoxon tests. Tenfold cross-validation was utilized to determine the optimal value of penalty parameter λ within 1 standard deviation (SD) from the minimum (λ = 0.15). When comparing a cohort of 8 responder patients to 24 nonresponder patients, and considering a multiple correlation coefficient (R) of 0.5, with a type I error rate (α) of 0.05 and a type II error rate (β) of 0.20, the minimum detectable odds ratio (OR) per 1 SD increase in the continuous explanatory variable is 5.695. This indicates that our study was sufficiently powered to detect large effect sizes but not more modest associations.
This study was conducted according to the REMARK (recommendations for tumor marker prognostic studies, Supplemental File 1) criteria. 40
Results
Patients’ characteristics
A total of 33 advanced EC patients pretreated with ICIs from 2016 to 2021 in 6 French Cancer Centers were included. Patients’ characteristics are described in Table 1. The mean age was 64 years ± SD 9.41. Patients with initial FIGO stages IV comprised more than 80% of the population. Twenty-one MMRd/MSI-H and 12 MMRp/MSS tumors were analyzed. All patients with MMRd/MSI-H tumor and MLH-1 promoter methylation status assessment (data missing—not tested—in seven cases) displayed a hypermethylation profile, suggesting a somatic origin of MMR deficiency. p53 IHC status was not available in most MMRp/MSS tumors (10/12, 83%). When receiving ICI, all patients were in the relapse setting. Every patient received monoclonal antibodies targeting PD-1/PD-L1, most of them as monotherapy (58%). Most patients whose tumors were MMRd/MSI-H received anti-PD-1/PD-L1 antibodies monotherapy (67%) while half of patients with MMRp/MSS tumors received Pembrolizumab + Lenvatinib (50%) (Table 1). The BOR rate was 26%, 95% CI (13–46) versus 25%, 95% CI (9–53) in MMRd/MSI-H and MMRp/MSS groups, respectively. Median duration of response was 34.5 months (range: 8.0–57.0) and 18 months (range: 16.0–24.0) in MMRd/MSI-H and MMRp/MSS groups, respectively (Figure 1). Median follow-up was 16 months 95% CI (7–21). Median PFS was 3.5, 95% CI (3–not reached) versus 6.5 months 95% CI (2.0–not reached) in the MMRd/MSI-H and MMRp/MSS group, respectively, with, as expected, a tail of curve PFS benefit in the MMRd/MSI-H group (Supplemental Figure 1(A)). Median OS was not reached in the MMRd/MSI-H group 95% CI (16.0–not reached) and was of 7.5 months in the MMRp/MSS group 95% CI (5.0–not reached) (Supplemental Figure 1(B)).
Clinical and demographic characteristics of the patients.
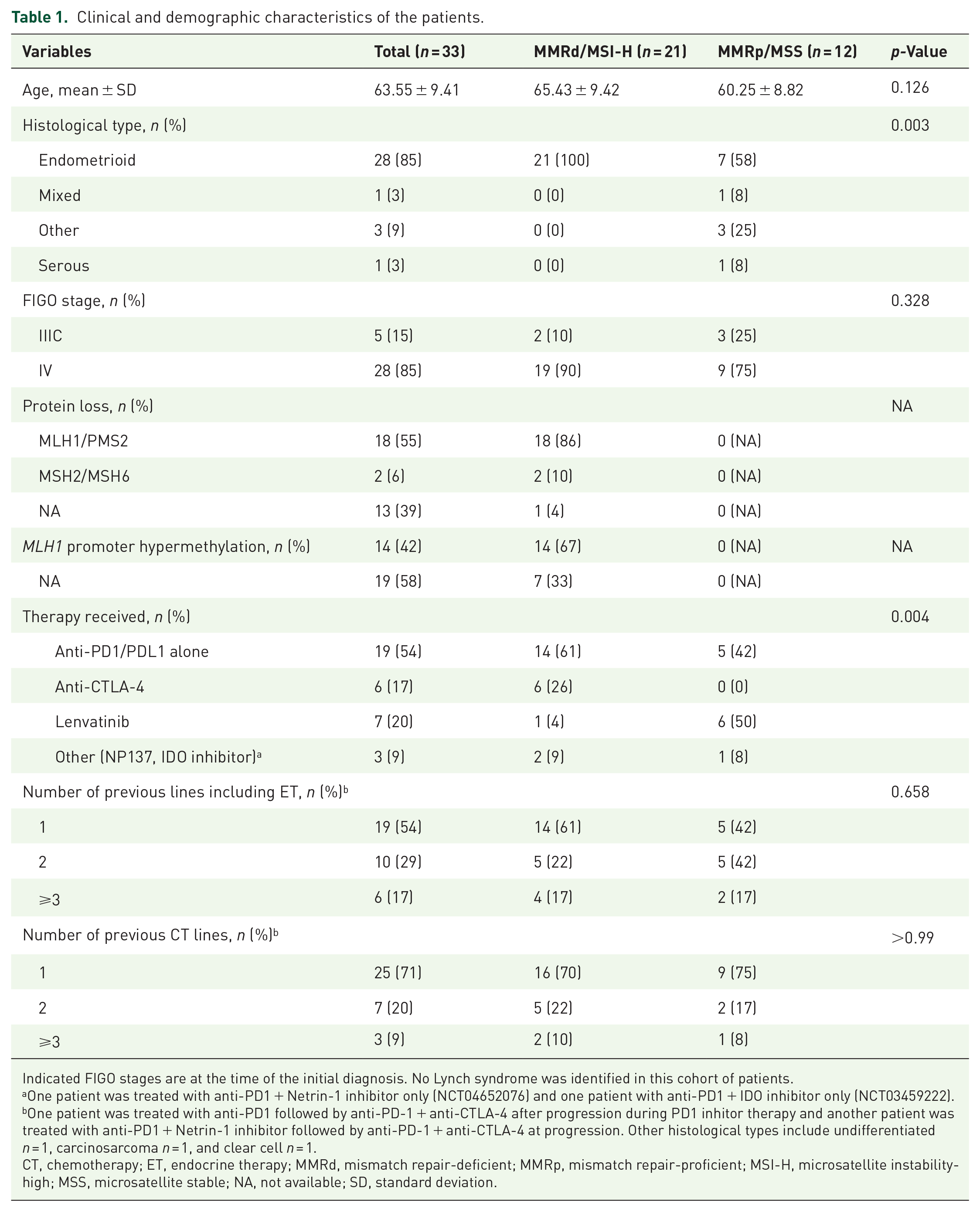
Indicated FIGO stages are at the time of the initial diagnosis. No Lynch syndrome was identified in this cohort of patients.
One patient was treated with anti-PD1 + Netrin-1 inhibitor only (NCT04652076) and one patient with anti-PD1 + IDO inhibitor only (NCT03459222).
One patient was treated with anti-PD1 followed by anti-PD-1 + anti-CTLA-4 after progression during PD1 inhitor therapy and another patient was treated with anti-PD1 + Netrin-1 inhibitor followed by anti-PD-1 + anti-CTLA-4 at progression. Other histological types include undifferentiated n = 1, carcinosarcoma n = 1, and clear cell n = 1.
CT, chemotherapy; ET, endocrine therapy; MMRd, mismatch repair-deficient; MMRp, mismatch repair-proficient; MSI-H, microsatellite instability-high; MSS, microsatellite stable; NA, not available; SD, standard deviation.

Swimmer plot of treatment and disease course.
EC tumor immune infiltrate characteristics
By using a 7-color multi-IF staining (CD20, CD4, CD8, FoxP3, CD68, panCK, and DAPI), we characterized, for each tumor sample collected at tumor diagnosis, the presence of LA/TLS (Figure 2(a)), and the density of CD20+ B cells, CD4+ T helper (Th) cells (CD4+FOXP3−), Tregs (CD4+FOXP3+), CD8+ T cells (Teffs; CD8+FOXP3−), CD8+FOXP3+ T cells, and CD68+ TAMs both in the stroma and the tumor islets (Figure 2(a) and (b)). Immune cells were mostly located in the stroma of EC with a majority of CD4+ Th cells (mean: 654 cells/mm2 ± SD 429) and TAMs (620 cells/mm2 ± SD 452) followed by CD8+ Teffs cells (383 cells/mm2 ± SD 326) (Figure 2(b)). Within tumor islets, CD8+ Teffs cells were the most prevalent immune cell subtype (94 cells/mm2 ± SD 136) (Figure 2(b)). B cells and Tregs were scarce within tumors, but their occurrence was higher in the stroma and variable based on individual patients (Figure 2(b)).

Density of immune cells in endometrial cancer in the tumor and the stroma. (a) Multi-IF staining displaying a tumor sample with a sparse distribution of immune cells. (b) Illustration of the segmentation of the tumor slide into tumor islets (red), stroma (green), empty regions (yellow), and non-specific staining areas (blue). (c) Tumor sample with lymphoid aggregate. (d) Tumor sample featuring a TLS and intra-epithelial CD8+ T cells (in green). (e) Number of cells/mm2 (i.e., the density) of B cells, TAMs, CD4+ Th cells, CD4+ Tregs, CD8+ T cells (Teff), and CD8+/FOXP3+ T cells both in the stroma (left) and within tumor islets (right).
LA/TLS are present in the vast majority of MMRd/MSI-H tumors
In response to the differing efficacy of ICI in EC molecular subtypes, we investigated the tumor immune profile based on EC genomic status. MMRd/MSI-H tumors displayed higher densities of both B cells and CD4+ Th cells in the stroma compared to MMRp/MSS tumors (236 cells/mm2 ± SD 374 vs 166 cells/mm2 ± SD 423, p = 0.027 for B cells, and 804 cells/mm2 ± SD 430 vs 391 cells/mm2 ± SD 284, p = 0.007 for CD4+ Th cells). (Figure 3(a)). In line with the variations noted among B cells and CD4+ Th cells, which together form LA/TLS, we observed more MMRd/MSI-H tumors with LA/TLS compared to MMRp/MSS cases: 81% versus 17% respectively, p < 0.001 (Figure 3(b)). To gain a deeper insight into cell interactions involving LA/TLS and other cells, we examined the cellular environment within a 20 µm radius around B and CD4+ Th cells (Supplemental Table 2). There were more TAMs (1.23 ± SD 0.95 vs 0.62 ± SD 0.54, p = 0.041) and CD8+ Teffs cells (1.45 ± SD 1.05 vs 0.62 ± SD 0.57, p = 0.017) in the vicinity of B cells in MMRd/MSI-H tumors compared to MMRp/MSS tumors, respectively suggesting cooperation between TAM and B cells and between CD8+ T cells and B cells in MMRd/MSI-H tumors (Supplemental Table 2). In summary, a higher presence of LA/TLS and increased proximity between B and CD8+ T cells are observed in MMRd/MSI-H EC, potentially elucidating a higher sensitivity to ICIs.14 –16
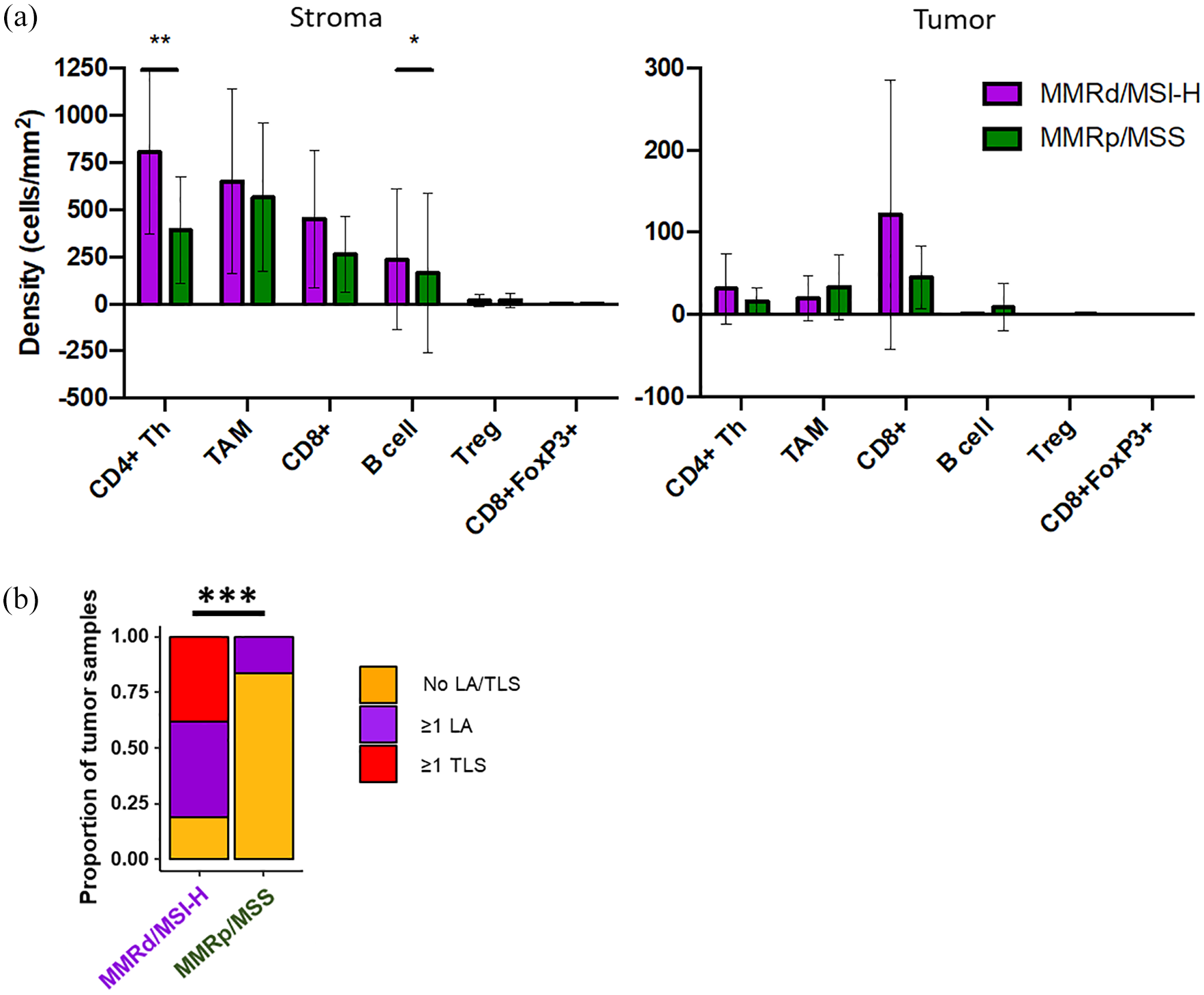
A greater percentage of MMRd/MSI-H tumors exhibit LA/TLS in comparison to MMRp/MSS tumors. (a) Number of cells/mm2 (i.e., the density) of B cells, TAMs, CD4+ Th cells, CD4+ Tregs, CD8+ T cells (Teff), and CD8+/FOXP3+ T cells both in the stroma (left) and within tumor islets (right) in MMRd/MSI-H (magenta) and MMRp/MSS (dark green) tumors. (b) Proportion of MMRd/MSI-H (magenta)/MMRp/MSS (dark green) samples with (⩾1 LA in the sample or ⩾1 TLS in the sample) and without TLS (no TLS in the sample).
Resistance to ICIs is associated with a significant abundance of TAMs in the stroma in EC
We then categorized patients according to their response to treatment. Eight responder (R) patients (25%) were compared to 24 NR patients (75%). One patient was excluded from the analysis as she received two different ICI regimens with opposite responses. R patients’ tumors had less TAMs (357 cells/mm2 ± SD 117 vs 706 cells/mm2 ± SD 498 in the stroma, p = 0.043; and 15 cells/mm2 ± SD 31 vs 28 cells/mm2 ± SD 33 in tumor islets, p = 0.028) and less CD4+ Th cells in tumor islets (13 cells/mm2 ± SD 17 vs 30 cells/mm2 ± SD 40, p = 0.037) (Figure 4(a)). No differences were shown in terms of the presence of LA/TLS and the subsequent response (p = 0.400) (Figure 4(b)). To identify significant and independent biomarkers associated with response, we used a multivariate logistic regression model featuring LASSO. This model encompassed all density biomarkers correlated significantly with ICI response in univariate analysis, alongside the presence of LA/TLS (a recognized predictive biomarker) and microsatellite status. We found that the density of TAM in the stroma was the only independent factor associated with response. A low density of TAM in the stroma, below the median, was associated with response (OR = 11.67, 95% CI (1.69–237.45), p = 0.033. A low density of TAMs in the stroma showed good accuracy in predicting response to ICI in the whole cohort (area under curve – AUC = 0.75, 95% CI = 0.59–0.91) but also in MMRd/MSI-H tumors (AUC = 0.70, 95% CI = 0.47–0.93) and in MMRp/MSS tumors (AUC = 0.83, 95% CI = 0.67–0.99) (Figure 4(c)). A low proportion of TAMs in the stroma did not show an association with PFS (HR = 0.59 (0.25–1.38), p = 0.2) but exhibited a significant correlation with OS (HR = 0.27 (0.10–0.77), p = 0.009 LR test) (Figure 4(d)). As an illustration, Figure 4(e) shows one NR EC patient with a high number of TAMs in the stroma and an R patient with few TAMs in the stroma (Figure 4(e)).

Resistance to immune checkpoint inhibitors is associated with a significant abundance of TAMs in the stroma in EC. (a) Number of cells/mm2 (i.e., the density) of B cells, TAMs, CD4+ Th cells, CD4+ Tregs, CD8+ T cells (Teff), and CD8+/FOXP3+ T cells both in the stroma (left) and within tumor islets (right) in responders (R) (yellow) and NR (blue). Mean and standard deviations are displayed. (b) Proportion of R (yellow) and NR (blue) samples with (⩾1 LA in the sample or ⩾1 TLS in the sample) and without TLS (no TLS in the sample). p-Values <0.05 were considered significant, with stars corresponding to *p < 0.05. If no stars are indicated, no statistically significant difference was found. (c) ROC curves representing the area under the curve for the model considering a high/low proportion of TAMs in the stroma in the whole (blue), the MMRd/MSI-H (magenta), and MMRp/MSS (dark green) cohorts. (d) Survival curves representing the PFS (left) and OS (right) probability of patients presenting a low proportion of TAMs in the stroma (yellow) compared to others (blue). p-Values are calculated using a likelihood ratio test. p-Values <0.05 were considered significant, with stars corresponding to *p < 0.05, **p < 0.01. If no stars are indicated, there was no statistically significant difference. (e) Multiplex IF staining showing one NR EC patient with a high number of TAMs in the stroma and an R patient with few TAMs in the stroma.
Discussion
The landscape of immune cell infiltration within the tumor microenvironment has emerged as a crucial determinant of response to ICIs in various cancer types. 28 In this study, we investigated the immune characteristics of EC patients treated with ICIs, using in situ multispectral IF tumor tissue staining and digital image analysis, aiming to unravel the intricate relationship between tumor immune infiltrate, LA/TLS, and TAMs in influencing ICI response.
Our investigation into the immune profile based on genomic status highlighted significant differences between MMRd/MSI-H and MMRp/MSS tumors. MMRd/MSI-H tumors exhibited elevated densities of B cells and CD4+ Th cells within the stromal compartment, corroborating previous studies indicating a more inflamed microenvironment in MSI-H tumors as expected due to a higher number of predicted neoantigens.29,30 Moreover, the increased prevalence of LA/TLS in MMRd/MSI-H tumors underscores their potential role in facilitating immune cell interactions and immune priming of T cells with anti-tumor activity. 31 This corroborates the increased proximity between B and CD8+ T cells observed in MMRd/MSI-H compared to MMRp/MSS EC. Those observations align with emerging evidence linking LA/TLS with favorable ICI responses, indicating a potential biomarker for therapeutic efficacy in EC.14 –16 On the other hand, we do not report any significant differences in terms of effector T cell density. Differences in infiltrating immune cells, with higher CD8+ immune cells and lower CD68+ TAMs, have been noted between Lynch syndrome (LS)-associated with sporadic MMRd/MSI-H EC.30,32 This could be explained by the fact that no LS-associated MMRd/MSI-H tumors were included in this cohort.
The frequency of patients harboring TLS is rather low, ranging from 19% in the PORTEC trial in localized first-line therapy compared to a quarter of cases for structured LA in cohorts including patients whose disease had progressed. 13 Tumor infiltration by TAMs, identified by the expression of CD68, was similar in both molecular subgroups but was lower in R compared to NR, implicating TAMs in mediating immune resistance. This finding is in line with recent studies that have highlighted the role of TAMs in fostering an immunosuppressive microenvironment, thereby limiting the efficacy of ICIs.33,34 Interestingly, MMRd/MSI-H tumors displayed a higher proportion of TAMs in the vicinity of B cells compared to MMRp/MSS tumors, potentially explaining the low response rate reported in our MMRd/MSI-H cohort compared to the literature. While TAMs have been recognized as crucial for the development of high endothelial venules and TLS formation, 35 some studies indicate a potential inhibitory effect on B cell proliferation 36 and downregulation of germinal center reaction. 37 This might provide insight into the adverse impact of a high macrophage density within TLS on cancer patients’ survival. 38 In addition, it is noteworthy that these TAMs could display characteristics similar to the M2-like phenotype. We can then hypothesize that targeting TAMs in patients in combination with conventional ICIs might increase survival. While our findings demonstrate a significant association between low TAM density and improved response to ICI therapy, it is important to consider that this observation may be influenced by underlying tumor-intrinsic factors. Oncogenic signaling pathways could shape both the immune microenvironment and treatment responsiveness independently. These factors may confound the observed association and highlight the need for integrative analyses that combine immune profiling with tumor-intrinsic characteristics to better understand the drivers of ICI response.
Despite the insights gained, our study has limitations. The small sample size and retrospective nature of the analysis warrant a cautious interpretation of the results and could explain why TLS are not associated with response to ICI in this cohort. Due to sample size constraints, we were unable to perform subgroup analyses to identify the respective impact of the immune parameters according to the type of therapy received. In addition, the lack of availability of p53 IHC status in a subset of tumors could introduce confounding variables. p53 IHC status was not available in most of the cases mainly because the sample collection was performed before the implementation of molecular classification in routine. In addition, the specimens of tumors utilized for examination were procured at the time of tumor diagnosis and exclusively comprised tissues from the endometrial site. Despite patients undergoing at least one treatment regimen between the collection of tumor samples and ICI initiation, we assert that focusing solely on endometrial samples helped mitigate potential bias associated with variations in immune composition across different anatomical sites (such as the liver, endometrium, and lymph nodes). Finally, we were unable to address the potential added value of having access to the immune milieu to identify relevant ICI combinations. Indeed, we did not have serial samples from the same patients treated with both ICI monotherapy and combination therapy. And, in our cohort, only one patient exhibited a nonresponse followed by a response to combination treatment. This question would require larger studies with serial sampling to be adequately explored. Future studies with larger cohorts and prospective designs are needed to validate our findings and explore the underlying mechanisms driving the observed associations.
Conclusion
Our study provides a comprehensive characterization of the immune landscape in EC patients treated with ICIs. The distinct immune infiltrate patterns observed in MMRd/MSI-H and MMRp/MSS tumors, coupled with the significant association between TAM density and ICI response, offer valuable insights into the determinants of ICI sensitivity and resistance. Further exploration of TAM-targeted strategies may hold the key to enhancing ICI efficacy and improving outcomes in EC patients. However, the clinical implementation of TAM-targeting strategies aiming at depleting or repolarizing TAMs has been limited to date, mostly due to the high heterogeneity and plasticity of TAMs. 39 Enhancing our understanding of TAM biology, differentiating markers, and interactions with immune and non-immune cells is essential for developing successful anticancer treatments.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251347364 – Supplemental material for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy
Supplemental material, sj-docx-1-tam-10.1177_17588359251347364 for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy by Olivia Le Saux, Renaud Sabatier, Isabelle Treilleux, Léa-Isabelle Renaud, Pierre-Emmanuel Brachet, Alejandra Martinez, Jean-Sébastien Frenel, Cyril Abdeddaim, Justine Berthet, Sarah Barrin, Amélie Colombe-Vermorel, Laetitia Odeyer, Alexandra Lainé, Christophe Caux, Bertrand Dubois and Isabelle Ray-Coquard in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251347364 – Supplemental material for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy
Supplemental material, sj-docx-2-tam-10.1177_17588359251347364 for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy by Olivia Le Saux, Renaud Sabatier, Isabelle Treilleux, Léa-Isabelle Renaud, Pierre-Emmanuel Brachet, Alejandra Martinez, Jean-Sébastien Frenel, Cyril Abdeddaim, Justine Berthet, Sarah Barrin, Amélie Colombe-Vermorel, Laetitia Odeyer, Alexandra Lainé, Christophe Caux, Bertrand Dubois and Isabelle Ray-Coquard in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251347364 – Supplemental material for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy
Supplemental material, sj-docx-3-tam-10.1177_17588359251347364 for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy by Olivia Le Saux, Renaud Sabatier, Isabelle Treilleux, Léa-Isabelle Renaud, Pierre-Emmanuel Brachet, Alejandra Martinez, Jean-Sébastien Frenel, Cyril Abdeddaim, Justine Berthet, Sarah Barrin, Amélie Colombe-Vermorel, Laetitia Odeyer, Alexandra Lainé, Christophe Caux, Bertrand Dubois and Isabelle Ray-Coquard in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359251347364 – Supplemental material for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy
Supplemental material, sj-docx-4-tam-10.1177_17588359251347364 for Immune landscape and TAM density in endometrial cancer: implications for immune checkpoint inhibitors efficacy by Olivia Le Saux, Renaud Sabatier, Isabelle Treilleux, Léa-Isabelle Renaud, Pierre-Emmanuel Brachet, Alejandra Martinez, Jean-Sébastien Frenel, Cyril Abdeddaim, Justine Berthet, Sarah Barrin, Amélie Colombe-Vermorel, Laetitia Odeyer, Alexandra Lainé, Christophe Caux, Bertrand Dubois and Isabelle Ray-Coquard in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
