Abstract
Background:
Chemoimmunotherapy has emerged as the standard first-line treatment for extensive-stage small cell lung cancer (ES-SCLC), improving survival outcomes. However, the role of prophylactic cranial irradiation (PCI) in the context of chemoimmunotherapy remains undefined.
Objectives:
This study aimed to evaluate the impact of PCI on overall survival (OS) in patients with ES-SCLC after chemoimmunotherapy administration.
Design:
Retrospective study.
Methods:
This retrospective analysis included 261 patients with ES-SCLC treated with first-line chemoimmunotherapy between January 2019 and December 2023. All patients underwent MRI scans to confirm the absence of brain metastases. After 1:2 propensity score matching (PSM), 46 and 81 patients were assigned to the PCI and observation groups, respectively. The primary endpoint was OS, with additional exploration of progression-free survival (PFS), the cumulative incidence of intracranial metastases, and intracranial progression-free survival (iPFS).
Results:
After PSM, the two groups were well-balanced in baseline characteristics. Survival analysis showed a median OS of 19.9 months (95% confidence interval (CI): 11.8–28.0) in the PCI group and 15.6 months (12.3–18.9) in the observation group, without a significant difference (hazard ratio (HR) = 0.763 (95% CI: 0.484–1.206), log-rank p = 0.265). PCI significantly reduced the risk of brain metastasis (Fine-Gray p = 0.002), with 1-year cumulative incidence rates of 13.8% (3.4%–24.2%) in the PCI group and 53.4% (41.3%–65.6%) in the observation group. Subgroup analysis showed that for ES-SCLC patients achieving a partial response to initial chemoimmunotherapy, the PCI group had longer median OS (25.7 months (95% CI: 15.4–36.1) vs 19.4 months (15.4–23.4); HR = 0.502 (0.284–0.886); log-rank p = 0.021).
Conclusion:
PCI did not improve OS in ES-SCLC patients receiving first-line chemoimmunotherapy, while it may confer a survival benefit for patients who achieve remission following chemoimmunotherapy. In addition, PCI significantly reduced the incidence of brain metastases. These findings warrant further randomized studies for verification.
Keywords
Introduction
Small cell lung cancer (SCLC) is a highly invasive, neuroendocrine-differentiated malignancy that comprises around 15% of lung cancers.1,2 According to the US Veterans Administration classification system, SCLC can be categorized into limited-stage SCLC (LS-SCLC) and extensive-stage SCLC (ES-SCLC), with approximately 70% of patients initially diagnosed with ES-SCLC and having a poor prognosis.3,4 Cytotoxic chemotherapy alone provides limited survival benefits for ES-SCLC, with a 5-year overall survival (OS) generally <3%.5,6 Following the era of immunotherapy, the 5-year OS for ES-SCLC has improved to nearly 15%, establishing chemoimmunotherapy as the preferred first-line treatment.7–9
Brain metastasis occurs frequently in SCLC, with up to 75% of patients developing cerebral metastases over the course of the disease, significantly impacting both quality of life and survival.10,11 The introduction of prophylactic cranial irradiation (PCI) was considered a landmark achievement in reducing the risk of brain metastases in SCLC.12,13 Early studies and meta-analyses showed that PCI could effectively improve survival in LS-SCLC,12,14 recommending PCI in several international guidelines for LS-SCLC with a response to initial systemic therapy, though practices vary globally.15–17
However, the role of PCI in ES-SCLC is debatable, as two large prospective phase III clinical trials in the ES-SCLC population yielded contradictory results. 18 The EORTC study showed that PCI reduced brain metastasis rate from 40.4% to 14.6% (p < 0.001) and prolonged median OS by 1.3 months in the PCI group 19 (HR = 0.68; p = 0.003). In contrast, a Japanese study by Takahashi et al. 20 demonstrated that while PCI lowered the rate of brain metastases, median OS was not statistically significantly different at 11.6 and 13.7 months (HR = 1.27; p = 0.094) for the PCI and control arms, respectively. Notably, the latter study emphasized routine MRI surveillance, fueling ongoing discussions around PCI’s efficacy. As MRI surveillance increasingly disseminates into oncological practice, the value of PCI for SCLC has faced further reconsideration. The latest meta-analysis revealed that when utilizing radiographic confirmation of the absence of brain metastases, PCI conferred no survival benefit for LS- and ES-SCLC. 21 Within the recent National Comprehensive Cancer Network guidelines, PCI is not strongly prioritized as routine management for ES-SCLC. 16
The emergence of immune checkpoint inhibitors has broken through the therapeutic conundrum in ES-SCLC. Atezolizumab combined with platinum-based doublet chemotherapy increased median OS from 10.3 to 12.3 months in ES-SCLC (HR = 0.76; p = 0.0154) and was the first to be approved by the US Food and Drug Administration as first-line treatment for ES-SCLC. 22 Subsequently, several large phase III trials have further validated the prognosis-improving effects of immunotherapy in ES-SCLC, with median OS ranging from 12.9 to 15.8 months in the chemoimmunotherapy arms.23–25 In addition, immune checkpoint inhibitors have also been shown to enhance drug penetration into the central nervous system. 26 A subset evaluation of the IMpower133 study found that atezolizumab improved time to intracranial progression (20.2 vs 10.5 months; p = 0.046). 22 Currently, the role of PCI in the setting of chemoimmunotherapy remains unclear. Given this, we aim to explore the role of PCI in the immunotherapeutic era through this propensity score-matched (PSM) study.
Materials and methods
Study design and participants
This was a retrospective, observational, PSM cohort study. The study was reported in accordance with the STROBE guidelines. 27 Patients initially diagnosed with ES-SCLC between January 18, 2019, and December 12, 2023, were reviewed. Eligible patients received first-line platinum-based chemotherapy plus immunotherapy. All enrolled patients underwent comprehensive laboratory and imaging evaluation, including crucial chest-enhanced CT, abdominal-enhanced CT, and cerebral contrast-enhanced MRI. Some patients received PCI after completing all cycles of chemoimmunotherapy, followed by continued immune checkpoint inhibitor maintenance therapy. Others only underwent immune checkpoint inhibitor maintenance therapy upon finishing the cycles of chemoimmunotherapy.
Specific inclusion criteria were as follows: histologically confirmed extensive-stage non-SCLC; Eastern Cooperative Oncology Group (ECOG) performance status of 0–2; age 18–75 years; receipt of at least two cycles of platinum-based doublet chemotherapy plus immunotherapy as first-line treatment with subsequent response evaluation; brain metastases excluded by enhanced contrast-enhanced MRI; no prior thoracic radiotherapy; absence of other cancer or significant comorbidities impacting life expectancy; availability of complete patient data through regular follow-up visits.
The study protocol was approved by the Ethics Committee of the Zhejiang Cancer Hospital (IRB No. 2023-432) and complied with ethical standards according to the Declaration of Helsinki. Consent waivers were granted in accordance with protocols approved by the institutional review boards at the participating institutions.
Treatment
Patients received four to six cycles of chemoimmunotherapy followed by immunotherapy maintenance every 3 weeks until disease progression or intolerance. Local thoracic or bone radiotherapy was permitted to improve quality of life. As an optional intervention strategy, PCI administration was determined through individualized decision-making that incorporated multiple factors, including patient preference, age, performance status, and socioeconomic considerations, with the final decision reached through shared decision-making between physicians and patients. For the PCI group, the prescribed dose was 25 Gy in 10 once-daily fractions. PCI was administered within 2–4 weeks after completion of chemoimmunotherapy cycles. Hippocampal Avoidance PCI (HA-PCI) was allowed.
Follow-up and endpoints
All patients underwent clinical and radiological evaluation every 6 weeks at the hospital along with telephone follow-ups after initial chemotherapy and immunotherapy, during which imaging results were recorded. Cerebral contrast-enhanced MRI was performed at an interval of 12 weeks. The date of the last follow-up was May 31, 2024. Treatment responses were assessed according to Response Evaluation Criteria in Solid Tumors version 1.1. Brain metastases during the course of the disease should be confirmed via enhanced MRI imaging. The primary endpoint was to determine OS (time from diagnosis to death). Secondary endpoints comprised the rate of brain metastases, intracranial progression-free survival (iPFS; interval from diagnosis to the progression of intracranial disease or death), and progression-free survival (PFS; duration from diagnosis to disease progression or death).
Statistical analysis
PSM was employed to balance potential confounding factors between the PCI and observation groups. Using 1:2 matching, the PCI group was matched to the observation group based on age, gender, ECOG performance status, smoking history, metastatic status, and response to initial therapy, with a caliper of 0.05 and greedy nearest-neighbor matching algorithm. Patients’ characteristics were described using median value and the interquartile range (IQR) for continuous non-normally distributed variables, while categorical variables were compared using Chi-squared or Fisher’s exact test. Survival curves were generated with the Kaplan-Meier method and compared using the log-rank test. Median follow-up was calculated from censored data. Hazard ratios (HR) and 95% confidence interval (CIs) were assessed using the Cox proportional hazards model (forward stepwise likelihood ratio method) for subgroup analyses. The cumulative incidence of brain metastases was calculated and compared between groups using the Fine and Gray method. Statistics and graphics were generated in SPSS (v27.0) or R (v4.4.1). p Values of <0.05 were considered statistically significant.
Results
Between January 18, 2019, and December 12, 2023, a retrospective review identified 863 treatment-naïve ES-SCLC patients treated at our center. In total, 602 patients who did not meet the inclusion criteria were excluded, leaving 261 patients who received 4–6 cycles of platinum-based chemotherapy plus immunotherapy. Subsequently, 49 patients received PCI, while 212 did not. Following PSM, 46 and 81 patients were assigned to the PCI and observation groups for further analysis (Figure 1). The demographic and baseline characteristics were well-balanced between matched groups (Table 1 and Supplemental Figure 1). After PSM, the median age was 63 years (IQR, 58–70). The cohort comprised predominantly male smokers, including 112 (88.2%) males and 93 (73.2%) current or former smokers. Thirty-six (28.3%) and 68 (53.5%) of 127 patients developed liver and multi-organ metastases, respectively. Of the 127 patients, 52 (40.9%) completed more than 4 cycles of platinum-based chemotherapy in combination with immunotherapy, while only 12 (9.4%) received thoracic radiotherapy. The median number of immunotherapy cycles completed in the matched cohort was 7 (IQR, 4–12). In the PSM cohort, the treatment responses included: complete response in 0 (0.0%) patients; partial response in 84 (66.1%) patients; stable disease in 41 (32.3%) patients; and progressive disease in 2 (1.6%) patients.

Trial profile.
Demographics of the population.
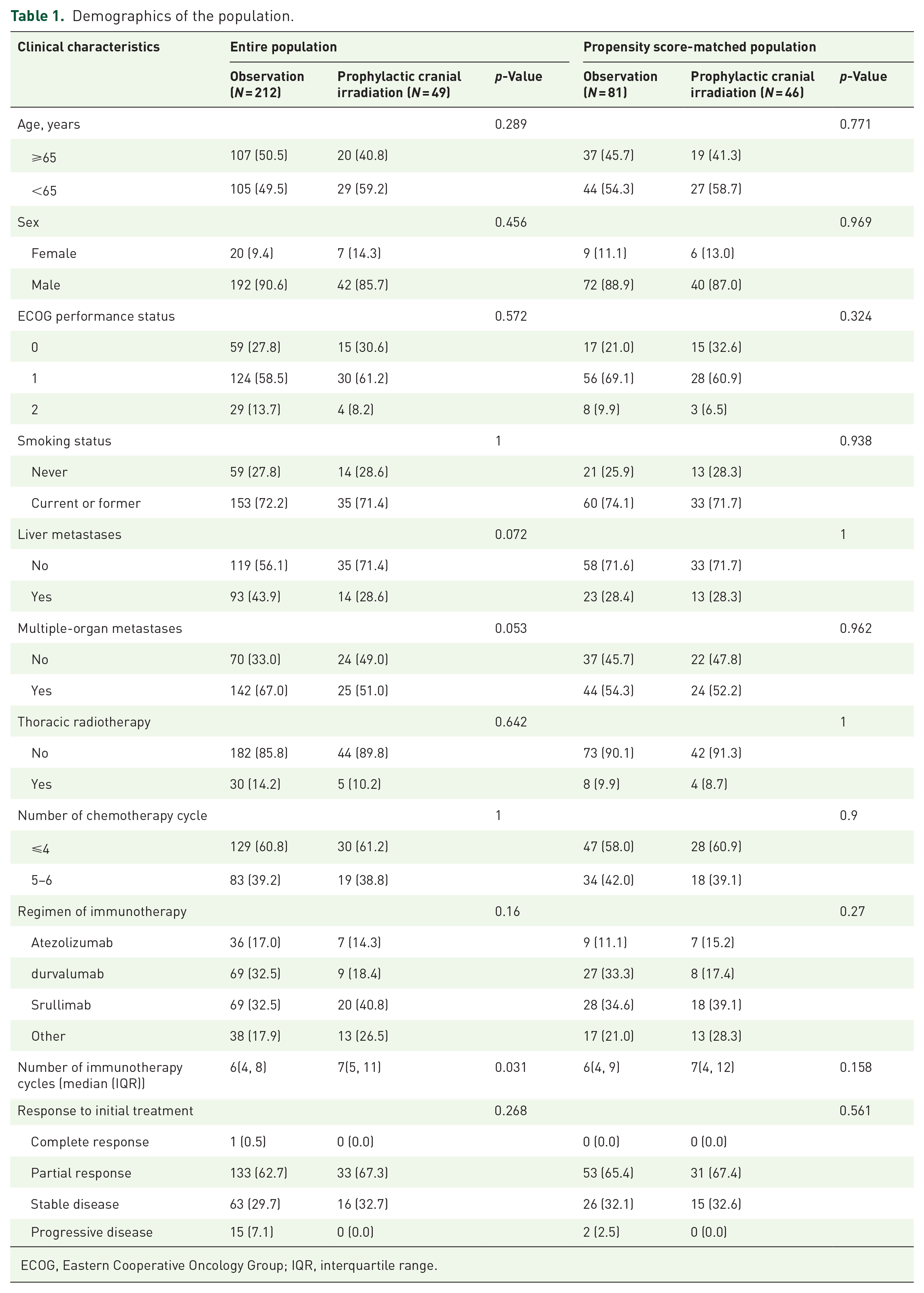
ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range.
As of the date of the last follow-up, the median follow-up was 24.9 months (IQR, 22.0–27.7) for the overall cohort and 23.6 months (IQR, 19.3–27.9) for matched cohorts. The median OS was 15.5 months (IQR, 13.5–17.4; Figure 2(a)) for the overall cohort and 16.6 months (IQR, 13.2–20.1) for matched cohorts. After PSM, 24 (52.2%) of 46 patients in the PCI group and 56 (69.1%) of 81 patients in the observation group had died. No significant difference between groups in OS was seen: median OS was 19.9 months (95% CI: 11.8–28.0) in the PCI group and 15.6 months (12.3–18.9) in the observation group (HR = 0.763 (95% CI: 0.484–1.206); log-rank p = 0.265; Figure 2(b)). The 1-year OS rates were 68.2% (95% CI: 57.4–79.0) in the PCI group and 60.0% (49.2–70.8) in the observation group, while the 2-year OS rates were 42.8% (24.6–61.0) and 27.5% (16.1–38.9), respectively. PFS did not differ significantly between the two groups: median PFS was 7.5 months (95% CI: 5.3–9.8) in the PCI group versus 6.9 months (5.6–8.2) in the observation group (HR = 0.768 (95% CI: 0.517–1.142); log-rank p = 0.207; Figure 2(c)).

The effect of PCI on the PSM cohort. (a) OS curves for the total population. (b) Kaplan-Meier curves of OS for PSM patients. (c) Kaplan-Meier curves of PFS for PSM patients. (d) Cumulative incidence curves of brain metastases for PSM patients. (e) Kaplan-Meier curves of intracranial PFS for PSM patients.
Post-matching, brain metastases were recorded in 8 (17.3%) of 46 patients in the PCI group versus 35 (43.2%) of 81 patients in the observation group. Counting death as a competing risk, the cumulative incidences of brain metastases at 6 and 12 months were 5.4% (95% CI: 0.7–11.5) and 13.8% (3.4–24.2), respectively, with the PCI group, versus 20.5% (11.9–29.2) and 53.4% (41.3–65.6) with observation group (Fine-Gray p = 0.002; Figure 2(d)). iPFS was significantly longer with PCI (35.4 months (95% CI: 26.9–43.9)) versus observation (17.4 months (95% CI: 8.9–25·9); HR = 0·318 (95% CI: 0.174–1.583); log-rank p = 0.002; Figure 2(e)).
Subgroup analyses were performed to identify potential ES-SCLC patients benefiting from PCI. Of note, in patients achieving the best response of PR to initial chemoimmunotherapy, median OS was significantly improved with the PCI group versus the observation group (25.7 months (95% CI: 15.4–36.1) vs 19.4 months (15.4–23.4); HR = 0.502 (0·284–0.886); log-rank p = 0.021; Figure 3(a)). The OS rates at 1 and 2 years were 86.3% (95% CI: 73.8–98.8) and 56.4% (34.4–78.4) in the PCI subgroup, compared to 69.2% (56.7–81.7) and 30.9% (15.6–46.2) in the observation subgroup. Nevertheless, in the subgroups of patients ⩽65 years old, those with ECOG 0–1 performance status, or current/former smokers, PCI did not confer statistically significant improvements in median OS versus the observation group (Figure 3(b)–(d)). Among patients without liver metastases or multi-organ metastases, median OS favored PCI over observation (30.8 months (95% CI: 14.3–47.3) vs 16.8 months (12.5–21.1) and not reached (NR) vs 14.9 months (10.0–19.8), respectively), though without statistical significance (log-rank p = 0.163; HR = 0.659 (0.377–0.162); and log-rank p = 0.059; HR = 0.433 (0.204–0.917); Figure 3(e) and (f)). Subgroup analyses revealed no significant survival benefit from PCI in patients stratified by either the number of chemotherapy cycles received or thoracic radiotherapy administration, with OS data across all predefined subgroups is depicted in Supplemental Figure 2.

Subgroup analysis of PCI in the PSM cohort. (a) Kaplan-Meier curves of OS for patients with treatment response. (b) Kaplan-Meier curves of OS for patients aged less than 65 years. (c) Kaplan-Meier curves of OS for patients with ECOG 0–1 performance status. (d) Kaplan-Meier curves of OS for smokers. (e) Kaplan-Meier curves of OS for patients without liver metastasis. (f) Kaplan-Meier curves of OS for patients without multiple-organ metastasis. Subgroups with a sample size of less than 15 patients in either study group are not shown.
The incidence and treatment of brain metastases in the overall cohort of 261 ES-SCLC patients is depicted in Figure 4(a). Of the 78 patients who developed brain metastases in the observation group, 58 (74.4%) subsequently received brain radiotherapy. Meanwhile, 6 (60.0%) of the 10 patients who developed brain metastases in the PCI group received brain radiotherapy. Modalities comprised whole-brain radiotherapy and stereotactic radiosurgery. Median OS was comparable between patients who received PCI (19.9 months (95% CI: 11.4–28.4)) and those in the observation group who developed brain metastases and received brain radiotherapy (19.3 months (16.2–22.4)), with no statistically significant difference (Log-rank p = 0.301, HR = 0.776 (0.484–1.246); Figure 4(b)). Importantly, in the patient cohort achieving PR, the median OS was significantly longer in the PCI group (28.8 months (95% CI: 18.6–40.0)) compared to the MRI monitoring plus salvage radiotherapy group (20.6 months (18.9–22.2); HR = 0.521 (95% CI: 0.287–0.947); p = 0.036; Figure 4(c)).
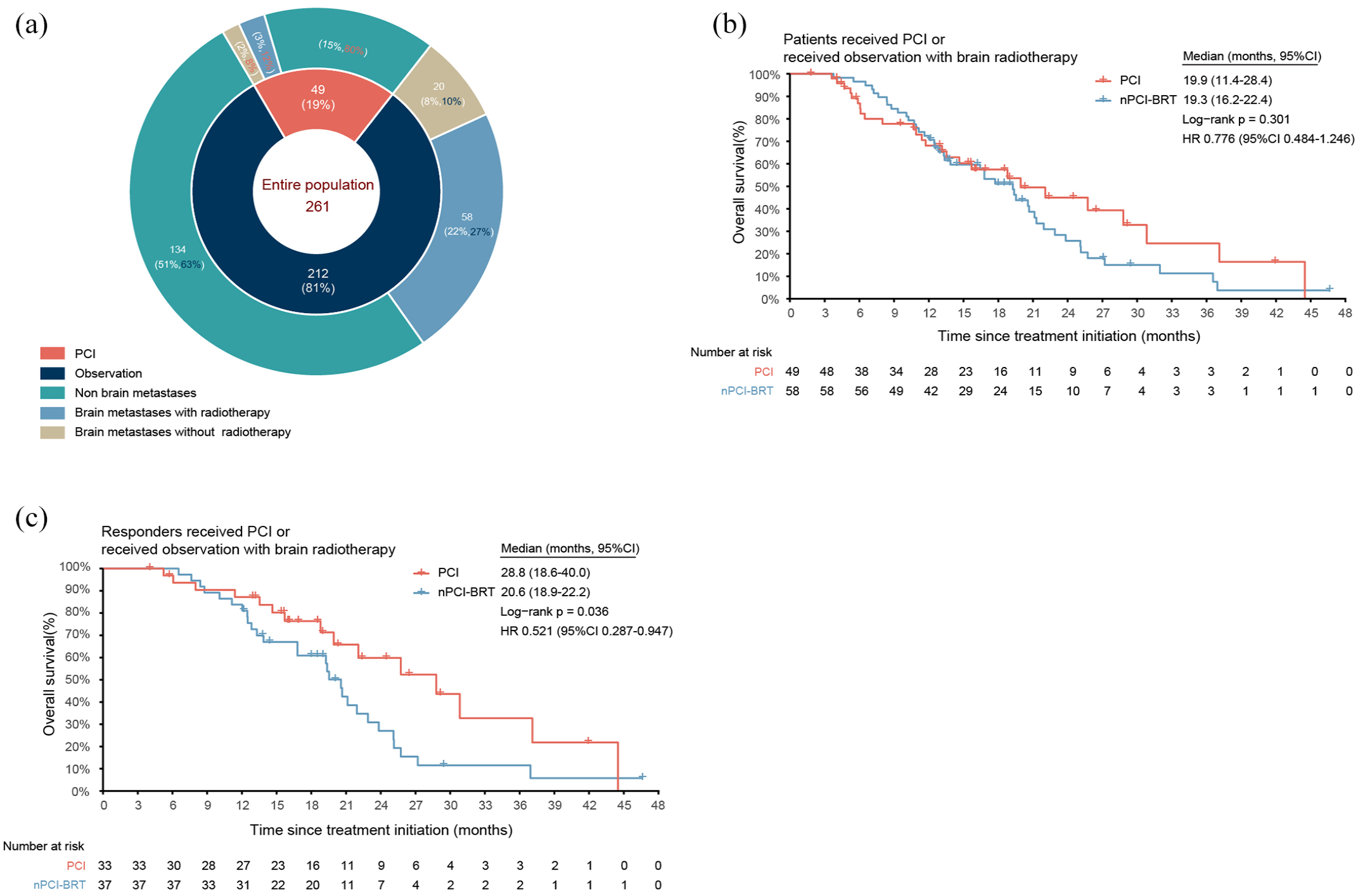
Comparative analysis of intervention patterns following chemotherapy combined with immunotherapy. (a) A pie chart showing the distribution of patients undergoing different management strategies. (b) Kaplan-Meier curves of OS for patients receiving PCI versus observation who developed brain metastases receiving radiotherapy. (c) Kaplan-Meier curves of OS for patients who achieved PR or CR receiving PCI versus observation combined with salvage radiotherapy.
Discussion
To our knowledge, this is the first study to investigate the benefit of PCI in ES-SCLC following first-line chemoimmunotherapy. By employing PSM to balance baseline characteristics between the PCI and observation cohorts, our analysis provides a more robust evaluation of outcomes.
In our study, PCI did not significantly prolong OS or PFS compared to observation, but we noted a 4.3-month median survival extension with PCI (19.9 vs 15.6 months). Importantly, all enrolled patients in our study underwent contrast-enhanced brain MRI at baseline and before PCI to exclude the presence of brain metastases. In contrast, earlier ES-SCLC studies, such as the representative EORTC trial, 19 did not require pre-PCI brain imaging, raising doubts regarding their outcomes. Subsequently, a phase III randomized controlled trial by Takahashi et al. 20 in a similar population but with mandatory pre-PCI brain MRI also reported negative results for PCI in improving OS. Therefore, the consensus is that any survival benefit PCI conferred upon patients without radiographic exclusion of brain metastases may have been therapeutic rather than prophylactic, as also corroborated in the meta-analysis of Gaebe et al. 21 Overall, our findings are broadly aligned with the literature. However, it is noteworthy that in the context of first-line chemoimmunotherapy, where the median OS was already 16.6 months in our study, the absolute 4.3-month OS advantage with PCI versus observation is still potentially clinically meaningful, warranting further randomized controlled trials to determine the role of PCI definitively.
Our data demonstrated that PCI significantly reduced the cumulative incidence of brain metastases by approximately 40% at 1 year, with median iPFS extended by around 18 months. This aligns with most prior studies,19,20,28 where PCI preventing cerebral dissemination was widely accepted in the chemotherapy-only era, irrespective of staging or MRI monitoring. For instance, Li et al. 28 ’s retrospective analysis of 190 LS-SCLC cases found that 1-year brain metastasis risk lowered by approximately 35% with PCI (37.7% vs 3.2%); the EORTC study of 286 ES-SCLC cases showed 1-year brain metastasis risk reduced by approximately 26% with PCI (40.4% vs 14.6%) 19 ; and Takahashi et al. 20 ’s study of 224 ES-SCLC cases found 1-year brain metastasis risk lowered by approximately 31% with PCI (59.0% vs 32.9%). Compared to these reports, the 1-year cumulative brain metastasis incidence reduction and projected iPFS extension observed in our study represent among the best historical data. This suggests potential interactions between PCI and immunotherapy beyond PCI’s role in cerebral metastasis prevention.
We further conducted subgroup analyses to identify potential populations preferentially benefiting from PCI. Importantly, our data showed that in patients who achieved a PR with initial chemoimmunotherapy, the PCI group experienced a clinically meaningful 6.3-month prolongation in median OS with statistical significance (OS 25.7 vs 19.4 months; HR = 0.502; p = 0.021). Patients with a favorable response to chemoimmunotherapy often exhibit an active immune microenvironment and prolonged survival, making them more susceptible to intracranial progression over time. Our findings suggest that robust treatment response may serve as an optimal selection criterion for PCI administration, as PCI not only reduces the risk of intracranial metastases but also demonstrates significant survival benefits in this selected population. Analyses of other negative studies did not suggest OS prolongation with PCI in first-line chemotherapy CR/PR patients, which may be attributable to the generally shorter OS of ES-SCLC in the chemotherapy-only era, as well as differences in therapeutic approaches. In addition, in our study, no clear survival benefits emerged in younger, better ECOG, or smoking subgroups. However, among those with lower tumor burden without hepatic metastases (26.0 vs 20.6 months) or solitary organ metastasis (21.2 vs 17.7 months), PCI appeared to confer greater median OS extensions, without statistical significance but with pronounced trends.
As MRI surveillance gradually became standard management for ES-SCLC patients, some researchers argued that PCI may lack cost-effectiveness,21,29,30 exemplified by Takahashi et al. 20 ’s phase III trial demonstrating no difference in survival with adequate MRI staging and observation. This comparison becomes even more obscure entering the immunotherapy era. 31 Those receiving MRI screening and salvage radiotherapy post-brain metastases represent current clinical practice pathways better. Our data showed median OS was similar between PCI and salvage radiotherapy groups under MRI monitoring (19.9 vs 19.3 months), suggesting PCI may not benefit all ES-SCLC in new treatment paradigms, especially with increasing stereotactic radiosurgery acceptance due to lower cognitive function and quality of life impacts. 32 Nonetheless, for the patient cohort with a good response to chemoimmunotherapy, PCI continued to confer a superior survival benefit compared to MRI monitoring combined with salvage radiotherapy (28.8 vs 20.6 months). Ongoing clinical trials such as SWOG 1827 MAVERICK (NCT04155034) and PRIMALung Study (NCT04790253) are prospectively comparing the impact of PCI versus MRI surveillance alone on survival outcomes in SCLC populations, with immunotherapy or adjuvant treatments allowed concurrently. More definitive answers will emerge from the results of these prospective clinical studies to potentially identify optimal PCI subgroups in the immunotherapy era.
As a retrospective single-center study with a limited sample size, the results are constrained in generalizability. Second, we did not analyze treatment-related adverse effects of PCI combined with immunotherapy, including potentially significant neurocognitive toxicity from PCI. Due to the small number of patients receiving PCI, we were also unable to independently compare differences between HA-PCI and non-HA-PCI. Furthermore, a lack of data characterizing distinct molecular subtypes of ES-SCLC precluded identifying potential subgroups that may benefit from PCI. Well-designed prospective multicenter studies with larger patient cohorts and more comprehensive clinical data are needed to validate the role of PCI in the context of immunotherapy.
Conclusion
This study found that PCI did not improve OS in ES-SCLC patients receiving first-line chemoimmunotherapy, while it may provide a survival advantage for patients who achieve remission after chemoimmunotherapy. Moreover, PCI significantly reduced the incidence of brain metastases. These findings warrant further randomized studies for validation.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251341158 – Supplemental material for Impact of prophylactic cranial irradiation on survival in extensive-stage small cell lung cancer receiving first-line chemoimmunotherapy: a propensity score-matched study
Supplemental material, sj-pdf-1-tam-10.1177_17588359251341158 for Impact of prophylactic cranial irradiation on survival in extensive-stage small cell lung cancer receiving first-line chemoimmunotherapy: a propensity score-matched study by Shichao Zhou, Wanchen Zhai, Qian Zhang, Hui Li and Yun Fan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251341158 – Supplemental material for Impact of prophylactic cranial irradiation on survival in extensive-stage small cell lung cancer receiving first-line chemoimmunotherapy: a propensity score-matched study
Supplemental material, sj-pdf-2-tam-10.1177_17588359251341158 for Impact of prophylactic cranial irradiation on survival in extensive-stage small cell lung cancer receiving first-line chemoimmunotherapy: a propensity score-matched study by Shichao Zhou, Wanchen Zhai, Qian Zhang, Hui Li and Yun Fan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
