Abstract
Background:
In gastric cancer (GC), the dissemination of neoplastic cells (NCs) in the peritoneal cavity is related to disease progression and poor prognosis. Elimination of NC through chemotherapy is needed to achieve better outcomes before conversion surgery.
Objectives:
The objective of this study was to evaluate the impact of NC CD44+/CD326+ levels through flow cytometry (FC) on peritoneal lavage (PL) fluid as a response indicator for conversion surgery.
Methods:
Patients with GC and NCs in the peritoneal cavity with or without peritoneal carcinomatosis (PC) and ascites were evaluated via minimally invasive staging. The PLs of patients were analyzed by FC to quantify NCs. All patients were treated with repeated intraperitoneal perfusion normothermic chemotherapy (RIPPENC). Patients who had negative NCs or reduced NCs were referred for conversion surgery.
Results:
Thirty patients were enrolled in this study and divided into three groups. In the first group, 20 patients with positive cytology (C+) and/or PC with a PC index (PCI) ⩽6 were treated with RIPPENC. Otherwise, six patients with C+ and PC with a PCI >7 and four patients with C+, ascites, and a PCI ranging from 15 to 22 were treated with palliative RIPPENC. The percentage of CD44+/CD326+ cells was correlated with the number of RIPPENC cycles and resections. The median follow-up time was 14.8 months. The overall median survival since the first RIPPENC was 14.6 months among those who did not undergo resection and 22.6 months among those who underwent resection (p = 0.001). Moreover, we observed a correlation between the percentage of CD44+/CD326+ cells in the PL region and patient survival.
Conclusion:
The use of FC to identify PL CD44+/CD326+ cell levels may be an important innovative biomarker for determining the presence of NCs, directly affecting the success of RIPPENC for conversion surgery.
Keywords
Introduction
Gastric cancer (GC) is the fifth most common cancer and the fifth most common cause of cancer-related death, accounting for almost 659,800 deaths worldwide. 1 Positive cytology (C+) and peritoneal carcinomatosis (PC) in GC patients were classified as metastatic disease or stage IV disease according to the American Joint Committee on Cancer Staging (8th edition)2,3 and the Japanese Gastric Cancer Treatment Guidelines (6th edition). 4 This dissemination pattern is associated with poor prognosis, with median overall survival (OS) rates of 4–8 months.5–7
Up to 36% of patients with GC that appear localized upon imaging will have PC or C+ on staging laparoscopy. Peritoneal metastatic disease is an important target for prolonging survival in patients with stage IV GC.8,9 The incidence of positive peritoneal cytology in patients with GC varies in the literature and may range from 4% to 41%.10,11 Studies have shown that the presence of positive metastatic cells has a negative effect on survival in patients who undergo gastrectomy.12–15 As such, a positive cytology result is a powerful independent predictor of survival. Knowing cytological status before surgery allows the use of a neoadjuvant treatment strategy with chemotherapy, and in cases where the cytological status becomes negative, curative surgery can be performed.
Traditionally, cytological evaluation of peritoneal fluid has been performed with the Pap test, which has low sensitivity and poor predictive value. 16 Other techniques, such as immunohistochemical (IHC) analysis 17 and reverse transcriptase-polymerase chain reaction (RT-PCR), have been used to improve the detection sensitivity for neoplastic cells (NCs), which are strongly correlated with peritoneal disease. However, these other techniques have not yet been demonstrated to have sufficient sensitivity. Although RT-PCR appears to be a superior detection method to other methods, it still has limitations related to its cost, time delay, and quality of its results.10,17–19
Flow cytometry (FC) is an established method for detecting the expression patterns of various antigens, especially in cells of hematological origin, and is widely used in the diagnosis of hematological neoplasms. 20 Recently, many researchers have applied FC to detect disseminated NCs via the mAb EpCAM (CD326), which is widely overexpressed in a variety of neoplasms21–23 and is also found in fluid samples obtained from the pleural or peritoneal cavities.24–29 In GC, EpCAM has strong clinical implications: +CD44/+EpCAM gastric tumor stem cells are highly tumorigenic and are associated with TNM stage and metastatic disease. 30 Strong immunostaining of EpCAM in both primary tumors and metastatic lymph nodes in GC patients was observed. 31 In addition, single-cell in situ immunofluorescence of cells separated from ascites and peritoneal lavage (PL) successfully identified EpCAM+/CD45− tumor cells, 32 and patients with peritoneal metastasis (PM) presented a high tumor‒leukocyte ratio of EpCAM+/CD45− compared with patients without PM. 33 Furthermore, peritoneal metastatic lesions highly express EpCAM, 34 which makes their presence in the peritoneal cavity of GC patients an important potential biomarker of prognosis and treatment/response assessment.
In Japan, repeated intraperitoneal chemotherapy (RIPEC) combined with systemic treatment was introduced for the treatment of GC patients with peritoneal disease, which resulted in improved survival.35–41 Recent studies have demonstrated that RIPEC is safe and effective for patients with PM, 42 and the discovery of biomarkers would be ideal for identifying patient populations in which surgery could result in a real survival benefit. Sugarbaker, in a recently published comment, suggested the use of neoadjuvant intraperitoneal chemotherapy (IPC) in combination with multiple cycles of intraperitoneal paclitaxel (PTX) as an alternative for the treatment of PMs from GC. 43
The primary objective of this study was to evaluate the impact of the levels of CD44+/CD326 cells in PL fluid and the presence of PC quantified through the use of the peritoneal carcinomatosis index (PCI) and to evaluate changes in the response to repeated intraperitoneal perfusion normothermic chemotherapy (RIPPENC) treatment, comparing the values of FC, PCI, and the need for RIPPENC cycles. The secondary objectives were to verify the impact of RIPPENC treatment on OS measured at the time of the first RIPPENC procedure and the response rates and complications from RIPPENC and gastrectomy. Clinical, pathological, and treatment variables were analyzed for associations with the results/outcomes.
Methods
Patients
This was a nonrandomized prospective cohort study. The study was conducted from June 2019 to June 2022 at the National Cancer Institute, Brazil. Patients aged 18 years or older who had advanced (stage IV) gastric adenocarcinoma were eligible. Additional inclusion criteria included a performance status <2 (Eastern Cooperative Group), a white blood cell count ⩾3000/µL, an absolute neutrophil count ⩾1500/L, a platelet count ⩾100,000/L, a serum creatinine ⩽1.5 mg/dL, and aspartate transaminase and alanine transaminase levels ⩽5 times the upper limit of normal. Patients were considered ineligible if they presented other malignancies or distant metastasis or had been subjected to previous treatment. The presence of ascites was not considered an exclusion criterion. The study was approved by the Ethics Committee of the National Cancer Institute (CAAE 25619913.9.0000.5274), and informed consent was obtained from each patient. The reporting of this study conforms to the REMARK statement 44 (Supplemental File 1).
Diagnostic
For diagnostic purposes, a trocar is placed in the umbilical scar through which the optics of the video laparoscopy equipment are introduced, and a trocar is placed in each of the bilateral upper mid-abdominal quadrants. Afterward, we staged the cavity by assessing the presence of a gastric tumor, the presence of ascites, and the presence of PC via PCI and performed a PL sample and biopsies of any suspicious lesions. PCI scoring was performed to identify disease extent, helping to assess the operability and prognosis of patients.45,46 To verify the positive cytology of the peritoneal cavity, ~100 to 300 mL of phosphate-buffered saline (PBS) solution was used to wash the cavity. The washing was evaluated for positive cytology throughout the counting of CD44+/326+ tumor cells through FC and for the presence of PCs during laparoscopy. The trocars remained in the patients through which the treatment was performed, as did the subsequent counts of tumor cells in the peritoneal cavity. Three groups were created based on the following criteria: the first group was composed of patients with positive cytology (C+) and/or PC with a PCI ⩽6; the second group was composed of patients with C+ tumors and a PC with a PCI >7; and the third group was composed of patients with C+, ascites, and a PCI ranging from 15 to 22.
Peritoneal cytology analysis
To assess the cytological status of the PL fluid, we used FC analysis. FC is a methodology that allows for multiparametric analysis of cellular content in a liquid medium. The high sensitivity and specificity of this method allow quick and safe results. Using a panel of specific monoclonal antibodies, it is possible to identify and quantify metastatic tumor cells. Research into the CD326 molecule associated with the expression of the CD44 molecule provides important information for the prognosis of patients with GC. To search for CD326+ epithelial tumor cells, samples obtained during surgical procedures were collected in sterile vials and sent to the Immunology Laboratory for immediate processing. In the laboratory, the samples were prepared and labeled with specific monoclonal antibodies capable of identifying and allowing the quantification of CD326+/CD44+ epithelial tumor cells (a panel of monoclonal antibodies used in the identification and quantification of epithelial tumor cells CD44 (FITC)/CD326 (APC)/CD45 (V500)/7AAD; Figure 1(a) and (b)). Initially, the samples obtained from the PL were centrifuged (5 min, 2000 RPM, RT) and then resuspended in 200 µL of PBS solution. The cells were subsequently labeled via incubation with CD326+/CD44+ antibodies (5 µL of anti-CD326 (BD Biosciences 347200, San Diego, CA, USA) plus 10 µL of anti-CD44 (BD Biosciences 347943)) for 15 min at RT. After labeling, 2 mL of PBS was added to the cells, and a new centrifugation (5 min, 2000 RPM, RT) was performed, followed by incubation with 10 µL of 7AAD (BD Bioscience) for 10 min. The labeled samples were acquired on a FACSCanto II flow cytometer (BD Bioscience) with the FACS DIVA data acquisition program (BD Bioscience). The data analysis was performed via INFINICITY software (Cytognos, Salamanca, Spain). The percentage of CD44+/CD326+ cells among 1 million cells analyzed via PL was calculated via FC and used to determine the number of RIPPENC cycles each patient should receive.
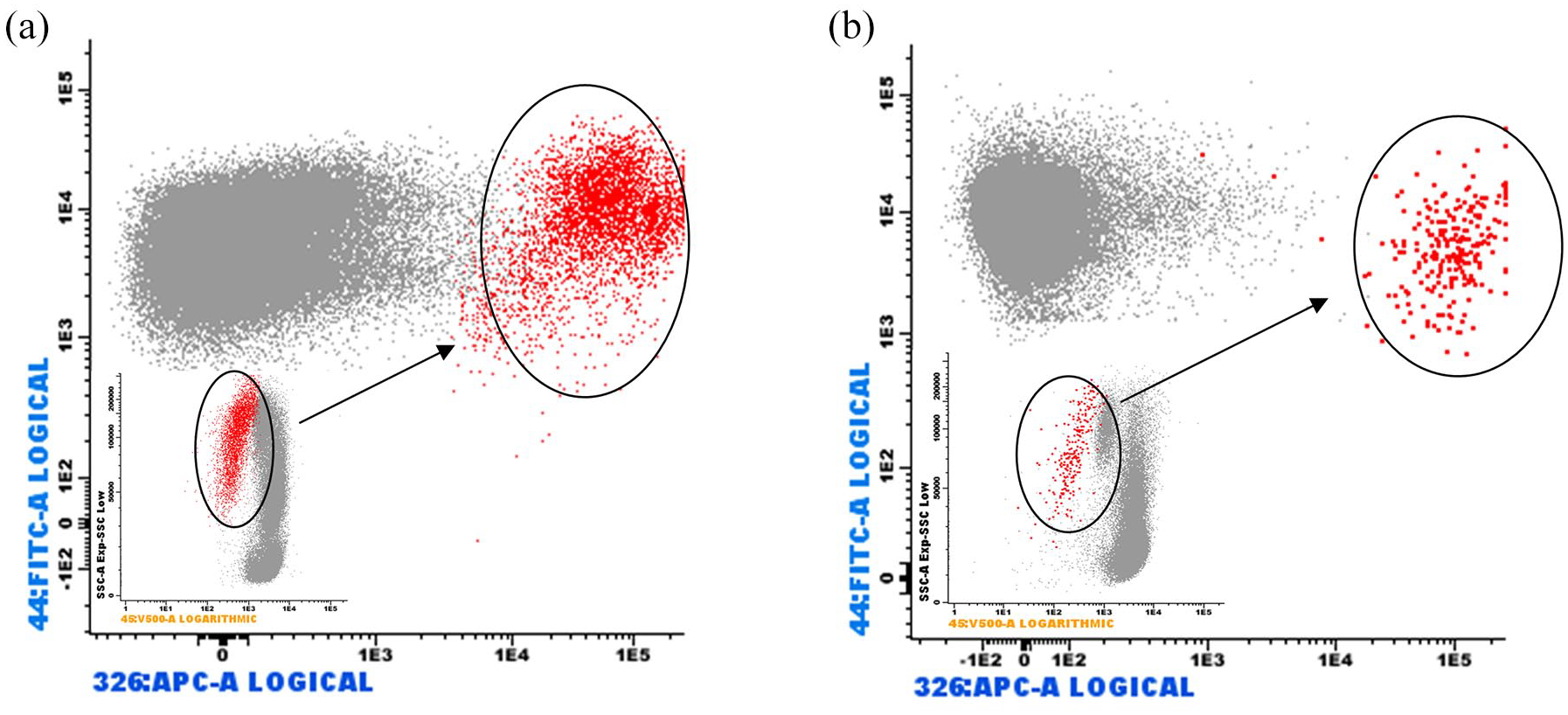
(a) Ascitic fluid—population of CD44+/CD326+ tumor cells located on CD45. (b) Peritoneal lavage—population of CD44+/CD326+ tumor cells located on CD45.
Treatment design
The design of this study is shown in Figure 2. The patients were subjected to laparoscopic video IPC associated with systemic neoadjuvant therapy. Once staging was completed, for the administration of the RIPPENC, an infusion catheter and a drainage catheter were introduced through the trocars. The catheters are connected to the perfusion set. To perform perfusion, we used a cardioplegia pump similar to that used in heart surgery.

Study design.
For RIPPENC, patients received 40 mg/m2 PTX diluted in crystalloid perfusate and were administered for 1 h. Concurrently, systemic neoadjuvant chemotherapy was administered. Oxaliplatin was administered intravenously (IV) at 130 mg/m2 on Day 1, and capecitabine was administered orally at 1000 mg/m2 for 12–12 h for 14 consecutive days, followed by 7 days of rest. This IPC regimen was administered to negative peritoneal washings. During each procedure, PL was evaluated for the presence of CD44+/CD326+ tumor stem cells. In addition, computed tomography was carried out after the procedure to investigate the dissemination of the tumor. Patients with negative peritoneal washings and without imaging evidence of distant metastases underwent tumor resection.
Adjuvant systemic chemotherapy with capecitabine and oxaliplatin (CAPOX) was administered as follows: oxaliplatin IV was administered at 130 mg/m2 on Day 1, and capecitabine was administered at 1000 mg/m2 for 12–12 h for 14 consecutive days, followed by 7 days of rest. This regimen was planned after resection for up to eight cycles. After the completion of study-related treatment, the subjects were followed until death occurred or until the date of loss to follow-up.
Tumor response assessment and statistical analysis
Complications were graded according to a commonly accepted classification system observed during hospitalization or within 30 days of the procedure. 47 Toxicity was classified according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0.
The study was designed with Bayesian interim monitoring plans for OS and toxicity.48,49 Patient characteristics were summarized via descriptive statistics, such as medians (ranges) for continuous variables and frequencies (percentages) for categorical variables. The OS time in months was calculated from the date of the first repeated normothermic IPC infusion until the date of death or censoring (in cases of loss to follow-up or study closure). OS probabilities were estimated via the Kaplan‒Meier method. 50 All the statistical analyses were performed via IBM SPSS software (version 24; IBM Inc; Chicago; IIL, USA).
Results
Patient characteristics and treatment
We evaluated 30 patients from the National Institute of Cancer, Brazil, from June 2019 to June 2022. Patients were divided into three groups according to differences in tumor stage, and all patients were treated with the same therapeutic regimen.
The clinicopathological characteristics and treatment frequencies are presented in Table 1. All patients had peritoneal cytology and PCI data evaluated during initial staging laparoscopy before the administration of RIPPENC. A total of 72 RIPPENC treatments were performed, with an average of 2 cycles per patient (range 1–7); 56.6% of patients underwent surgical resection, and 53.3% underwent postoperative chemotherapy. The majority of the patients (14) underwent 1 cycle of RIPPENC, followed by 5 with 2 cycles, 1 with 3 cycles, 7 with 4 cycles, 2 with 5 cycles, and 1 with 7 cycles. Among the 20 patients who underwent curative RIPPENC, 17 underwent conversion surgery (resection), of whom 16 underwent postresection chemotherapy (one patient died from COVID-19 during treatment).
Simple frequency distributions according to clinicopathological characteristics and treatment.
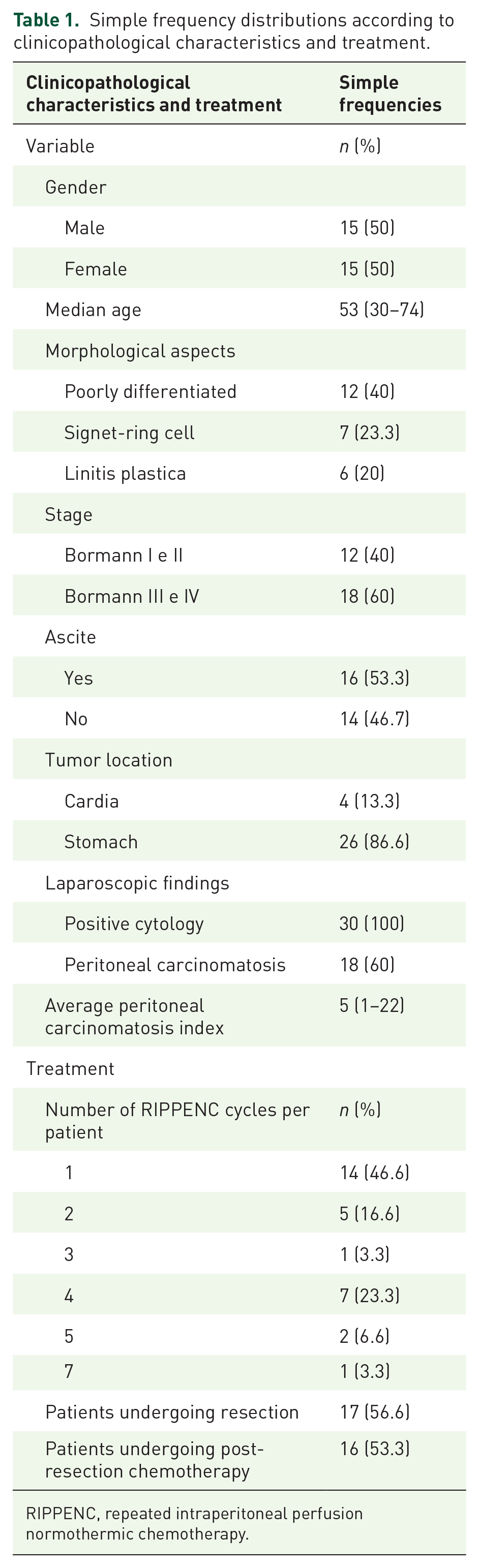
RIPPENC, repeated intraperitoneal perfusion normothermic chemotherapy.
Phenotypic analysis of PL fluid by FC reveals the number of CD44+/CD326+ cells as a biomarker of response
Phenotypic analysis of PL fluid via FC revealed an initial correlation between the percentage of CD44+/CD236+ cells, which are tumor markers, and the clinicopathological characteristics of the patients. There was a correlation between the percentage of neoplastic CD44+/CD236+ cells identified by FC in the peritoneal fluid and the PC status, as well as the presence of ascites. We also observed a correlation between the percentage of cells and the number of cycles necessary for negative PL cytology, as well as the disappearance of PC.
Table 2 shows the correlation of the PC status and the presence of ascites with the percentage of cells in the PL and IPC cycles needed to obtain the response. Thirty patients were treated, and 72 RIPPENC procedures were performed, with an average of 2 perfusions per patient, ranging from 1 to 7. We observed that among the 20 patients with positive cytology (C+) and/or PC with a PCI ⩽6, the average percentage of cells was 0.1088 (ranging from 0.001 to 0.49), with an average of 2.25 cycles of RIPPENC (ranging from 1 to 7). Sixteen of these patients achieved negative cytology results and underwent resection. On the other hand, among the six patients with C+ and PC with a PCI >7, the mean percentage of cells was greater, at 3.5875 (range 0.045–13.2), and no patients underwent resection. Among the four patients with ascites with PCIs ranging from 15 to 22, the mean percentage of cells was the highest at 5.581 (ranging from 0.514 to 16.7), and one patient underwent resection.
Distributions of the percentage of CD44+/CD236+ cells with the characteristics of the impairment pattern and average number of cycles required for negative cytology for each group.
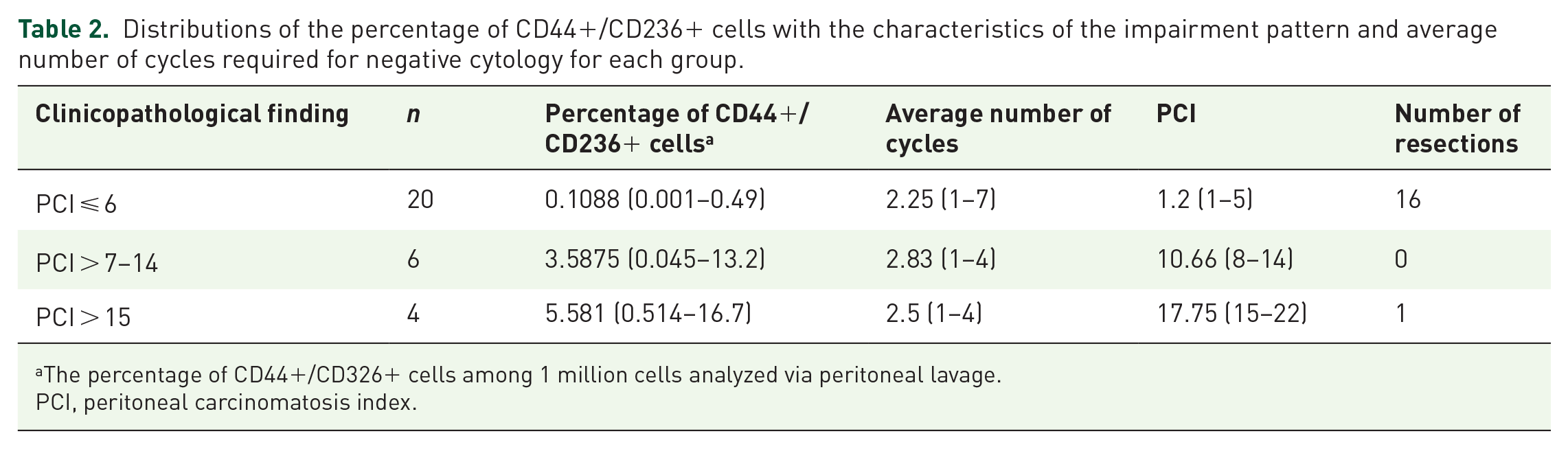
The percentage of CD44+/CD326+ cells among 1 million cells analyzed via peritoneal lavage.
PCI, peritoneal carcinomatosis index.
The RIPPENC strategy allows for gastrectomy with no complications
No complications related to the RIPPENC procedure were observed during the perioperative or postoperative period. A low rate of adverse effects was observed, and the treatment was administered in the day clinic. The number of RIPPENCs depended on the tumor stage.
We observed adverse effects in 17 of the 30 patients (56%) treated with RIPPENC. Eleven patients had a grade 1 adverse effect, five patients had a grade 2 effect, and only one had a grade 3 effect. The most frequent toxicities were nausea, vomiting, asthenia, peripheral neuropathy, diarrhea, neutropenia, thrombocytopenia, and alopecia.
Postoperative complications occurred in 4 of the 17 patients who underwent resection (23.5%), and one died—unrelated to the surgery but due to severe lung infection due to COVID-19. Table 3 reports the complications observed and the frequency of occurrence.
Postoperative complications within 30 days.

NET, nasoenteral tube.
Seventeen patients (56.6%) underwent resection after conversion. Gastrectomy was performed when the peritoneal cytology results became negative, and the disappearance or significant reduction of PC was confirmed via laparoscopy. The same chemotherapy regimen was restarted after surgery. Locoregional treatment with RIPPENC was safe and presented no complications.
The use of biomarkers by FC and RIPPENC improved the survival of patients who underwent gastrectomy
The response rate of the 30 patients enrolled in this study was 73.33%, where 22 patients reached negative cytology after RIPPENC treatment. The overall median survival (OS) time since the first RIPPENC was performed was 14.6 months (95% confidence interval (CI), 8.9–20.3; Figure 3). We observed a significant increase in survival in patients who were able to undergo surgical resection after RIPPENC. The overall median survival of the 17 patients who underwent surgical resection was 22.6 months (95% CI, 13.2–32.1), with 88.2% (±7.8) of the patients alive at 1 year and 51.3% (±12.4) at 2 years. In patients who did not undergo surgical resection, the median OS was 10.0 months (95% CI, 5.3–14.6), with 30.8% (±12.8) of patients alive at 1 year and 7.7% (±7.4) at 2 years (p = 0.001; Figure 4).

The median OS time since the first RIPPENC was 14.6 months (95% CI, 8.9–20.3).

Comparison of overall survival according to surgical resection.
Discussion
Combined chemotherapy with intraperitoneal PTX and systemic chemotherapy followed by gastrectomy is a promising strategy for advanced GC patients with PMs. The administration of intraperitoneal taxane via repeated infusions, similar to RIPEC treatment, is a reasonable strategy for controlling peritoneal lesions. The combination of PTX and IPC for the treatment of peritoneal disease in patients has significant efficacy with acceptable toxicity and is considered a treatment option for patients with metastatic GC due to its strong antitumor activity and excellent response rate; thus, PTX is more suitable for induction chemotherapy for conversion surgery.13–15,35–42
However, ensuring that the patient is correctly assessed for a negative PM status or the presence of malignant cells is a bottleneck in conversion surgery success. Thus, the objective of this study was to use FC as a tracking tool for identifying the correct number of neoplastic CD44+/CD236+ cells to achieve negative PL via neoadjuvant RIPPENC through a minimally invasive approach in patients with GC with positive cytology and/or PC. We evaluated the clinical response to chemotherapy through the presence of NCs in the PL fluid, the presence of a peritoneal implant during video laparoscopy, the percentage of NCs by FC, and the measurement of the PCI.
As a mostly noninvasive tool, liquid biopsy has become an important method in clinical practice for the treatment of oncologic patients owing to its potential to detect biomarkers such as tumor-derived DNA, circulating tumor DNA, proteins, and other biomarkers in human fluids, such as blood, saliva, and ascites/PL. In this regard, molecular alterations in these important biofluids accessed by liquid biopsies (LB) in advanced GC, diagnosis, prognosis, and treatment/response assessment are promising. 51
Papanicolaou and May–Grünwald–Giemsa staining are among the most common cytological examinations used in the clinic to investigate malignant cells present in several biofluids. However, this staining method involves multiple steps that may interfere with the yield of the cells analyzed and therefore decrease the sensitivity of this strategy. Thus, the choice in this study to use a more sensitive tool, such as cytometry, to identify CD44+/CD236+ NCs in patients with PL and ascites was determined in the correct tracking of patient response to RIPPENC. This approach directly affects the management of patients’ treatment, helping guide the IPC strategy to perform conversion therapy, and resulting in excellent results for the OS of patients with GC.
Induction chemotherapy with RIPPENC was used in 30 patients with advanced GC, and we evaluated the safety of this regimen. We performed resection surgery on 17 patients (56.6%) treated with RIPPENC and obtained promising results in terms of efficacy and safety. The outcomes tended to be better in patients with a low PCI and a lower percentage of cells in the PL who underwent gastric resection.
The ideal period of chemotherapy for obtaining impacts on PL cytology in patients initially diagnosed with GC has not yet been established. Aizawa et al. 13 reported one to five cycles of preoperative chemotherapy and reported negative cytology results in 48.9% of patients. In Yasufuku et al.’s 14 study, two or three cycles of preoperative chemotherapy were necessary to ensure that the PL cytology was negative. Kim et al. 15 studied the response of 18 patients who underwent conversion surgery; the mean survival time of patients who received curative conversion therapy was 37 months, and the 3-year survival rate was 50%.
The best time to perform resection of the primary tumor was based on studies by Kitayama and Ishigama, who performed resection after the disappearance or reduction of PMs, preventing the appearance of new metastases, bleeding, and stenosis determined by progression of the primary tumor. The absence of NCs in the cavity and the reduction or disappearance of peritoneal lesions in the absence of progression of distant metastatic disease, combined with good clinical conditions and good PS, are considered the best times to perform resection.4,17
Our treatment strategy was conversion therapy after the neoplastic CD44+/CD236+ cells became negative in the PL fluid and after the PC disappeared or was reduced. This procedure is easy to perform, safe, and effective. Repeated peritoneal videolaparoscopy (VLPs) make it possible to observe the clinical response to chemotherapy and evaluate the response to treatment safely and efficiently way and are useful in selecting candidates for conversion surgery.
In this study, RIPPENC was well tolerated, with a low rate of complications and a short hospital stay. Furthermore, we found a median OS of 20.3 months for patients with positive cytology and GC metastasis limited to the peritoneum who underwent treatment and who underwent resection. The present study is the first to use this strategy of RIPPENC for GC and the first to describe the repeated use of RIPPENC to eradicate metastasis of this type of tumor in the peritoneum before gastric resection.
The safety and efficacy of this therapy were demonstrated in the present study, and it was possible to prolong the survival of GC patients with PC and positive cytology for NCs. In this regard, we observed that the percentage of CD44+/CD236+ cells in the PL was correlated with the PCI, indicating the need for additional RIPPENC cycles. Even in patients without PC, depending on the percentage of cells in the PL, RIPPENC should be performed more than once until the NC count is negative, which is the criterion for performing gastric resection. In addition, we observed that the smaller the number of CD44+/CD236+ cells was, the greater the number of conversion therapies performed. However, in patients with ascites, which shows a varied range of positive cells but an average level of conversion, the RIPPENC procedure is still useful as a palliative tool for the treatment of these patients.
In our study, we demonstrated the advantage of repeated VLP infusions, allowing us to perform a macroscopic assessment of macroscopic peritoneal disease and collect PL fluid to analyze the presence of NCs by FC during each procedure. We did not observe adhesions in the ports or between loops in the VLP or even during gastric resection surgery. The infusion set proved to be safe and easy to use, and no complications were observed. The procedure and hospitalization time were short. The treatment proved to be easy to perform, safe, practical, low cost, and low risk for the patient and the health care team, suggesting that the application of RIPPENC in the neoadjuvant setting is safe and associated with low morbidity.
Saito et al. 42 evaluated the efficacy and tolerability of induction chemotherapy in combination with intraperitoneal TX and systemic S-1/oxaliplatin in 44 patients with GC with PC. The average number of cycles was 16 (1–48). The OS rate was 79.5% at 1 year, and that of median survival time was 25.8 months. Gastrectomy was performed in 20 (45%) patients who showed a macroscopic reduction in PC and an OS of 100% at 1 year; it was concluded that intraperitoneal PTX is highly effective and recommended as induction chemotherapy for patients with PC of GC. 42
Sugarbaker, 43 in a recent commentary, concluded that the chemotherapy response and hyperthermic intraperitoneal chemotherapy (HIPEC) were successful enough to generate widespread interest in the surgical treatment of PM; however, the effectiveness of HIPEC may be more limited than suggested by its current application. On this subject, the use of RIPPENC overcomes some limitations of the HIPEC approach related to its hyperthermic nature, which limits the number of applications, morbidity, and mortality. The use of normothermic drug delivery in the RIPPENC approach enables several cycles of IPC with fewer adverse effects. This characteristic also enables more effective tracking of NCs through the use of FC to identify CD44+/CD236+ cells, which can be useful biomarkers. Thus, the use of multiple cycles of intraperitoneal PTX as an alternative for patients with PM of GC and for patients whose PM responds, conversion surgery is indicated; Saito et al. reported increased survival after conversion surgery. Taken together, RIPPENC IPC associated with FC identification of CD44+/CD236+ cells has good potential for treating GC patients with PC.
Conclusion
The use of FC to identify PL CD44+/CD326+ cell levels may be an important innovative biomarker for determining the presence of NCs, directly affecting the success of RIPPENC for conversion surgery. Our results, although they are based on a small number of cases, suggest that repeated laparoscopic IPC combined with systemic and oral chemotherapy may represent a new tool for treating patients with advanced GC, allowing resection to be performed when the patient presents a response to induction therapy. Gastric resection after locoregional treatment with RIPPENC via PTX chemotherapy via a minimally invasive route combined with CAPOX is safe and promising and can prolong the survival of patients with GC who present with peritoneal disease. However, comparative studies are needed to clarify the survival benefits and clinical significance of conversion gastrectomy.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251337480 – Supplemental material for Impact of CD44/326 peritoneal cells as response indicators in advanced gastric cancer patients receiving repeated intraperitoneal perfusion normothermic chemotherapy
Supplemental material, sj-docx-1-tam-10.1177_17588359251337480 for Impact of CD44/326 peritoneal cells as response indicators in advanced gastric cancer patients receiving repeated intraperitoneal perfusion normothermic chemotherapy by Odilon de Souza Filho, Reinaldo Rondinelli, Antônio Carlos Acetta, Sérgio Bertolace, Alexandre Palladino, Cristiano Duque Guedes, Claudia Cristine Rocha Vieira, Luis Claudio Santos Thuler, Claudia Diniz, Everton Cruz dos Santos and Eliana Abdelhay in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
