Abstract
Chimeric antigen receptor T-cell (CART) therapy is an efficacious immunotherapy with known multi-organ toxicities, including gastrointestinal adverse events (GI-AEs). Eosinophilic colitis (EoC) is the inflammation of the intestine with diffuse eosinophilic infiltration. We present the case of a 66-year-old male who presented with diarrhea and biopsy-proven EoC two months after CART therapy for recurrent multiple myeloma (MM) and achieved a favorable response following corticosteroids and infliximab. A 66-year-old male with a past medical history of MM presented with watery stools 5–6 times per day. The patient was diagnosed with MM 10 years ago and achieved remission following an autologous stem cell transplant and maintenance chemotherapy. Three years ago, the patient developed recurrent MM, received CART therapy, and achieved cancer remission. Two months following CART therapy, he presented to the local emergency department (ED) for several weeks of diarrhea with a negative infectious workup. This disease course was associated with several ED visits and hospital admissions. He was started on budesonide without a significant response. Subsequent colonoscopy and resultant histology were consistent with EoC. The patient was started on an IV steroid with infliximab and a prednisone taper for refractory EoC. Following his third dose of infliximab and completing his prednisone taper, he reported a return to baseline symptomatically. CART is an immunotherapy associated with GI-AEs and requires corticosteroids or other immunosuppressants in select cases. EoC has been associated with cancer and cancer therapy and may require biological agents. Early recognition and treatment of immunotherapy toxicities are essential for successful management of gastrointestinal adverse events.
Introduction
Chimeric antigen receptor T-cell (CART) therapy is a novel immunotherapy used to treat recurrent lymphoma, leukemia, and multiple myeloma (MM). Ciltacabtagene autoleuc (Carvykti) is a BCMA-targeting CART therapy for the treatment of MM. In clinical trials, 98% of participants treated with ciltacabtagene autoleucel elicited a clinical response, with 80% of participants developing a stringent complete response. Despite high efficacy, it can cause multi-organ toxicities, such as cytokine release syndrome, increased risk of infection, neurological side effects (altered mental status, numbness, and tingling), anemia, thrombocytopenia, and/or leukopenia. 1 CART-related gastrointestinal adverse events can also occur in up to 15% of patients with inflammation in the GI tract that is associated with significant morbidities and mortalities. 2 Certain medical management strategies have been reported for CART toxicities, such as IL-6 inhibitors (tocilizumab) and corticosteroids. 3
Eosinophilic colitis (EoC) is a rare condition characterized by inflammation of the intestine due to diffuse eosinophilic infiltration on histology. 4 Primary EoC is extremely uncommon, but secondary EoC has been associated with a variety of conditions including allergic (atopy, asthma, allergic rhinitis, hypereosinophilic syndrome, food allergies, and protein hypersensitive enteropathy), autoimmune (Churg-Strauss syndrome), gastrointestinal infections (especially parasites), inflammatory bowel disease, celiac disease, medications (NSAIDs, clozapine, and carbamazepine), allogenic bone marrow transplantation, malignancies, and chemotherapy agents (tacrolimus).5,6 EoC has a nonspecific presentation including nausea, vomiting, diarrhea, abdominal pain, anorexia, and weight loss. Mainstay therapy includes corticosteroids and additional treatments include avoidance of allergen/trigger, elemental diet, mast cell stabilizers, leukotriene receptor antagonists, anti-IgE monoclonal antibodies, anti-IL5 antibodies, and TNF-alpha inhibitors. 7
We present the case of a 66-year-old male who presented with grade 3 diarrhea based on the Common Terminology Criteria for Adverse Events (CTCAE) and biopsy-proven EoC two months after CART therapy for recurrent MM and achieved a favorable response following treatment with corticosteroids and infliximab.
Case description
A 66-year-old male with a past medical history of MM presented to our center with a complaint of watery stools 5–6 times per day (CTCAE grade 2). The patient was diagnosed with IgG Kappa, ISS Stage 1, Durie-Salmon Stage IIIa MM ten years ago and began induction chemotherapy with bortezomib, cyclophosphamide, and dexamethasone one month following his diagnosis which resulted in inadequate tumor response. Two months following his diagnosis, the patient received carfilzomib, lenalidomide, and dexamethasone which resulted in partial tumor response. He then received an autologous stem cell transplant and maintenance chemotherapy with lenalidomide five months following his diagnosis and achieved cancer remission for six years. The patient discontinued carfilzomib four years ago due to chemotherapy-induced cardiomyopathy and was subsequently switched to daratumumab for maintenance therapy. Three years ago, the patient was found to have recurrent MM and received elotuzumab, pomalidomide, and dexamethasone for two years followed by CART therapy with ciltacabtagene autoleucel five months ago, and has achieved cancer remission since.
Two months following CART therapy, he presented to the local emergency department (ED) with complaint of several weeks of grade 2 diarrhea complicated by weight loss and electrolyte abnormalities with negative infectious workup including a gastrointestinal panel for 22 pathogens. This disease course was associated with several ED visits and hospital admissions (Figure 1). He was started on cholestyramine, diphenoxylate/atropine, and budesonide for his diarrhea (positive stool lactoferrin) without much response. Subsequently, he had a colonoscopy with resultant histology revealing the presence of eosinophils and apoptotic bodies in the ileum and throughout the colon, consistent with eosinophilic enterocolitis (C-reactive protein (CRP) was within normal limits—2.07). Despite ongoing budesonide and added mesalamine, he continued to have significant symptoms and inflammation (significantly elevated CRP—12.62 and sedimentation rate—32 one month following endoscopy) including CTCAE grade 3 diarrhea and weight loss and began receiving home health treatment with IV fluids and electrolyte replacement. The patient was then started on IV steroid with infliximab and a prednisone taper for refractory EoC. His symptoms improved to 3–5 semi-solid bowel movements per day. Following his third dose of infliximab and completing his prednisone taper, he reported a resolution of his bowel and constitutional symptoms.
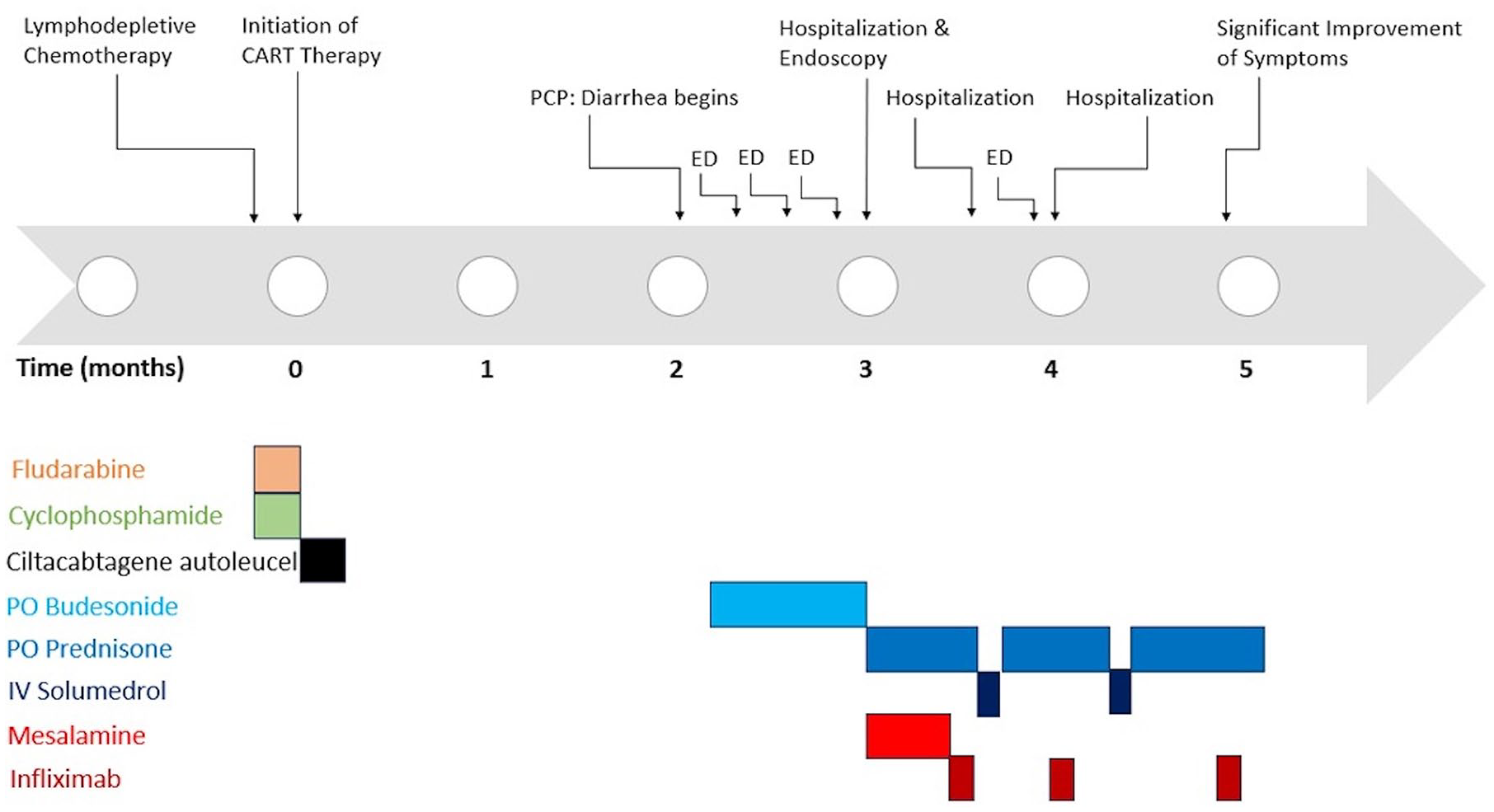
Timeline of patient’s chimeric antigen T-cell receptor therapy and onset of illness with subsequent treatment.
Discussion
CART therapy has been associated with gastrointestinal adverse effects (GI-AEs) in patients with cancer. Up to 15% of patients receiving CART experienced GI-AEs including diarrhea, nausea, vomiting, abdominal pain/distension, fever, and bloody stools. Current common practice for management of moderate to severe CART toxicity includes corticosteroids and off-label use of IL-6 inhibitors (tocilizumab). 2 Even after initial improvement following treatment with IL-6 antagonists and corticosteroids, 24% of patients experienced recurrence of gastrointestinal symptoms. Given the overall low volume of patients receiving CART and low prevalence of refractory CART-related GI-AEs, there are limited data to assess therapeutic strategies for treatment failure or inadequate response to first-line treatment of CART-related GI-AEs in this same study. 2
On the other hand, EoC is the rarest of gastrointestinal diseases with a prevalence of 2.1 per 100,000 individuals. 8 EoC has been associated with a variety of conditions including cancer and cancer therapy and managed with biologic agents. In a study of the cancer population with EoC, over half had hematologic malignancy. CART therapy has not necessarily been associated with eosinophilia or eosinophilic gastrointestinal diseases; however, elevated eosinophil levels have been correlated to improved efficacy of CART therapy in cancer patients. 9 The median duration between chemotherapy and EoC onset was six months, 76% of participants were symptomatic, and 15% of patients required hospitalization. Diarrhea was more prevalent in patients who received chemotherapy (85% vs 42%) and the median duration of EoC symptoms was significantly shorter in patients with cancer (30 days) than in patients without cancer (240 days). Most patients (88%) had normal colonoscopy findings. The primary treatment for EoC is anti-inflammatory agents such as corticosteroids, which reduce eosinophilic growth factors. 5 TNF-alpha inhibitors have been shown in several case studies to successfully treat severe or refractory EoC.10 –14
The CART toxicity profile is believed to be the result of an off-target effect of CART, which is largely reversible and resolves with appropriate treatment. 3 Gastrointestinal symptoms are likely a result of cytokines acting on gastrointestinal mucosa, leading to impaired barrier function and progression of mucositis. 15 TNF-alpha is produced by CAR T-cells upon tumor recognition and is one of the main cytokines involved in endothelial activation; thus, TNF-alpha blockade may have therapeutic potential for treating CART toxicity. 16 TNF-alpha is also implicated in the pathogenesis of eosinophilic esophagitis by promoting cytokine release which may play a similar role in EoC.11,17
Conclusion
CART is an immunotherapy that has been associated with gastrointestinal adverse events and required corticosteroids or other potent immunosuppressants on select cases. Early recognition and prompt treatment are essential for successful management of these gastrointestinal adverse events.
