Abstract
Background:
Everolimus is beneficial for patients with hormone receptor-positive and human epidermal growth factor receptor 2-negative (HR+/HER2−) advanced breast cancer (ABC). However, some patients developed drug resistance and the well-established predictor for everolimus efficacy was limited.
Objectives:
The study was designed to evaluate the efficacy of everolimus in different treatment lines and identify several clinicopathological markers to estimate everolimus efficacy in patients with HR+/HER2− ABC.
Design:
This was a retrospective and multicenter study.
Methods:
Between 2014 and 2022, more than 2000 patients with tumors who received everolimus were collected from multiple cancer centers in China (National Cancer Center, Chinese PLA General Hospital, Peking University Cancer Hospital & Institute). A training cohort and two validation cohorts were developed.
Results:
The training cohort included 338 patients. The median progression-free survival (PFS) for everolimus was 5.6 months, with an objective response rate of 25.1% and a clinical benefit rate of 54.4%. PFS was significantly worse from first-line (1L) to second-line (2L) to third-line (3L), with PFS1L for 13.5 months, PFS2L for 6.1 months, and PFS3L for 4.1 months (p = 2.9e−6, hazard ratio (HR) = 0.70, 95% confidence interval (CI) = 0.61–0.82). The clinicopathological characteristics, including post-1L everolimus treatment, Ki67 index of more than 40%, more than two metastatic sites at first recurrence, and receiving adjuvant chemotherapy, were independent risk factors for PFS. A predictive model for everolimus efficacy was established using these four factors. In the low-risk group, patients achieved a median PFS of 12.6 months, significantly longer compared to 2.7 months for those in the high-risk group (p = 2.4e−64, HR = 9.41, 95% CI = 7.05–12.56). The area under the curve was 0.96, 0.95, and 0.94 for 6-month, 1-year, and 3-year PFS, respectively. Internal validation cohort (PFS 18.4 vs 3.1 months, p = 3.6e−11, HR = 3.78, 95% CI = 2.49–5.74) and external validation cohort (PFS 13.5 vs 3.1 months, p = 2.9e−10, HR = 11.53, 95% CI = 4.68–28.37) confirmed its power for estimating clinical benefits of everolimus.
Conclusion:
A predictive model was successfully established to predict survival outcomes for everolimus in patients with HR+/HER2− ABC, which may provide references for the management of everolimus in Chinese patients with HR+/HER2− ABC.
Introduction
Everolimus, an extensively studied mammalian target of rapamycin (mTOR) inhibitor, is an effective treatment option for patients with hormone receptor-positive and human epidermal growth factor receptor 2-negative (HR+/HER2−) advanced breast cancer (ABC). Patients with HR+/HER2− ABC who developed disease progression to endocrine therapy can still derive survival benefits from everolimus-combined therapy.1–5 It exerts antitumor effects and reverses endocrine resistance, involving the molecular mechanism of the crosstalk between the phosphatidylinositol 3-kinase/Akt/mTOR signaling pathway and the estrogen receptor (ER) pathway.6–8 Moreover, emerging evidence suggested that everolimus could compromise the synthesis of cyclin D1 and cyclin-dependent kinase 4 (CDK4) proteins and restore the sensitivity to CDK4 and 6 (CDK4/6) inhibitors. 9 This provides the rationale that everolimus possesses the potential to become an optimal therapeutic option in the post-CDK4/6 inhibitor settings.
To date, everolimus has not been covered by medical insurance in China, which limits its wide popularity for patients with HR+/HER2− ABC. Given the considerable expense and potential toxicity, everolimus ought to be administered to patients who are responsive to the targeted drug in clinical practice. Such a phenomenon highlights the importance of exploring practical and cost-effective biomarkers that can reflect the clinical efficacy of everolimus, which can help patients who did not respond to everolimus to avoid unnecessary costs and toxic effects.
Regarding markers for everolimus efficacy, previous studies have conducted explorative analyses of genetic alternations and assessed the prognostic role in patients with HR+/HER2− ABC.10–12 However, they failed to identify practical biomarkers for the efficacy of everolimus. In this study, we aimed to explore associations of survival benefits from everolimus and clinicopathological characteristics using real-world and multicenter information and to develop a predictive model for everolimus efficacy in patients with HR+/HER2− ABC.
Methods
Data collection and inclusion criteria
We collected demographic and clinicopathologic data from the electronic medical records. Patients were from three cancer centers in China, including the National Cancer Center, Chinese PLA General Hospital, and Peking University Cancer Hospital and Institute. The collected information contained characteristics involving the Ki67 index, age, adjuvant therapy, number of metastatic organs at first relapse, treatment lines, and corresponding drug response to everolimus.
Eligible patients in the training cohort were identified by the following criteria. (1) Not male patients. (2) Patients who have positive status of ER or progesterone receptor (PgR). ER, PgR, and HER2 status were measured by immunohistochemistry (IHC) staining in accordance with American Society of Clinical Oncology recommendations. ER- or PgR-positive was defined as tumor cells with more than 1% ER or PgR. HER2 status was assessed by IHC score and fluorescence in situ hybridization (FISH), involving HER2-zero (IHC score 0), HER2-low (IHC score 1+ or 2+ with negative FISH), or HER2-enriched (IHC score 3+ or FISH amplification). (3) Patients who developed malignant metastases prior to everolimus therapy. (4) Patients were treated with everolimus for at least 6 weeks (the minimum time interval for follow-up). (5) Records of surgery, Ki67 index, first recurrence, medication duration, and drug response were available.
The validation cohort was established to confirm the efficacy of the predictive model. We randomly selected 40 patients per first line (1L), second line (2L), and third line (3L) from the training cohort to construct an internal validation cohort (namely cohort A). We collected an additional number of 51 patients to serve as the external validation cohort (namely cohort B). The reporting of this study conforms to the STROBE statement. 13
Drug efficacy
Drug response was assessed by imaging and physical examination using Response Evaluation Criteria in Solid Tumors 1.1. It could be categorized as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). The indicators of everolimus efficacy included clinical benefit rate (CBR), objective response rate (ORR), and progression-free survival (PFS). PFS was described as the period from drug initiation to PD or death. CBR was the percentage of target populations who had a CR, PR, or SD for greater than 6 months. ORR was the rate of patients who had a CR or PR to everolimus.
Statistical analysis
We applied the Chi-squared test to compare baseline characteristics of the eligible patients from different groups. Regarding correlation analyses, the Pearson method was used to analyze continuous variables, and the Kendall method was used to analyze categorical variables. Univariate Cox regression analysis was conducted to screen for variables associated with PFS. Multivariate Cox regression analysis was performed to further identify prognostic variables for model construction. Patients in the model were divided into the low-risk group and the high-risk group based on the percentile of 50%. The prognostic differences between the two groups were analyzed by the survfit function of the survival package. The area under the curve (AUC) was calculated by receiver operator characteristic (ROC) analysis in the pROC package (version 1.18.5). The Kaplan–Meier survival curve was plotted to vividly display survival differences between different groups. SPSS (version 29.0) and R software (version 4.3.2) were used for statistical analysis. A p value < 0.05 was considered statistically significant.
Results
Eligible patients of the training cohort
Between January 2014 and November 2022, 2518 tumor patients who were treated with everolimus were filtered from 3 cancer hospitals in China. Out of the total patients, 652 female patients of breast cancer who developed metastases were collected. Considering the crucial significance of the Ki67 index in the field of breast cancer, we excluded 197 patients with missing data on the Ki67 index. Of these, 21 patients were excluded due to the negative status of PgR and ER at either the primary or metastatic site, leading to a number of 434 patients with HR+ ABC. Three patients with brain metastasis were excluded according to exclusion criteria in pivotal clinical trials associated with everolimus. 1 Two patients with ovary metastasis were ruled out since it is somewhat obscure to distinguish between primary ovarian tumors. To minimize the negative effects of surgery type, we rejected 21 patients without any surgery for breast cancer, 17 patients with palliative surgery, and 1 patient with unknown time for surgery. To reduce the impact of irregular medication on everolimus, we precluded 52 patients who discontinued medication owing to intolerable toxicity, poor compliance, and other patient reasons. Finally, 338 patients were qualified and enrolled as the training cohort for ensuing analyses. The flowchart is shown in Supplemental Figure S1.
Baseline characteristics of patients in the training cohort
Baseline characteristics are shown in Table 1. For patients in the training cohort, the overall median age is 51 years, with 89.6% of patients younger than 65 years. A total of 21.3% of patients have a Ki67 index of more than 40%, which was determined as the optimal cut-off value for PFS benefits from everolimus in our previous study. 14 A majority of patients experienced one or two sites at the first recurrence (91.4%). Bone was the most common site of distant metastasis, accounting for 45% of patients. A substantial proportion of patients were administered adjuvant endocrine therapy (96.3%) and chemotherapy (94.1%) prior to everolimus treatment. The median disease-free survival (DFS) was 43.6 months, with 34.0% of patients more than 5 years. Regarding treatment lines of everolimus, 29.3%, 13.9%, and 56.8% of patients received everolimus as 1L, 2L, and 3L therapy, respectively.
Baseline characteristics of patients in the training cohort.
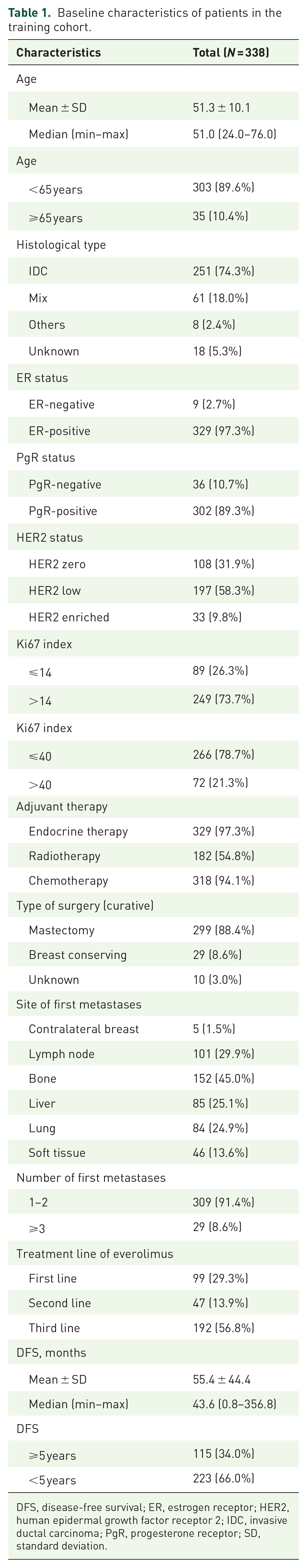
DFS, disease-free survival; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; PgR, progesterone receptor; SD, standard deviation.
Clinicopathological characteristics correlated to everolimus efficacy
The median follow-up duration was 72.2 months, with 331 patients reaching the endpoint of PFS. A total of 14 clinicopathological parameters were entered into the corresponding correlative analyses for everolimus efficacy. Five factors, involving age, Ki67 index, treatment line of everolimus, liver metastasis at first relapse, and receiving adjuvant chemotherapy, were significantly associated with PFS for everolimus, among which treatment line of everolimus was the most significant (Figure 1(a)). Patients who had a Ki67 index of more than 40% or received everolimus as post-1L treatment had a poorer response to everolimus (Figure 1(b)). A long DFS (>5 years) failed to show any correlation with either PFS or drug response on everolimus.

(a) Correlation between clinicopathological features and PFS for everolimus in efficacy-evaluable patients with HR+/HER2− ABC. Size of the circle, the number within the rectangles or the intensity of color represents the correlation coefficient. The red color represents positive correlation and the blue represents negative correlation. (b) Correlation between clinicopathological features and drug response for everolimus in patients with HR+/HER2− ABC. Response was analyzed as categorical variables. From patients developing PR to SD to PD, the assignment obtained gradually increases.
Clinical efficacy of everolimus in different treatment lines
The clinical outcome of patients treated with everolimus stratified by treatment lines is presented in Table 2.
Outcome of patients in the training cohort stratified by treatment lines of everolimus.
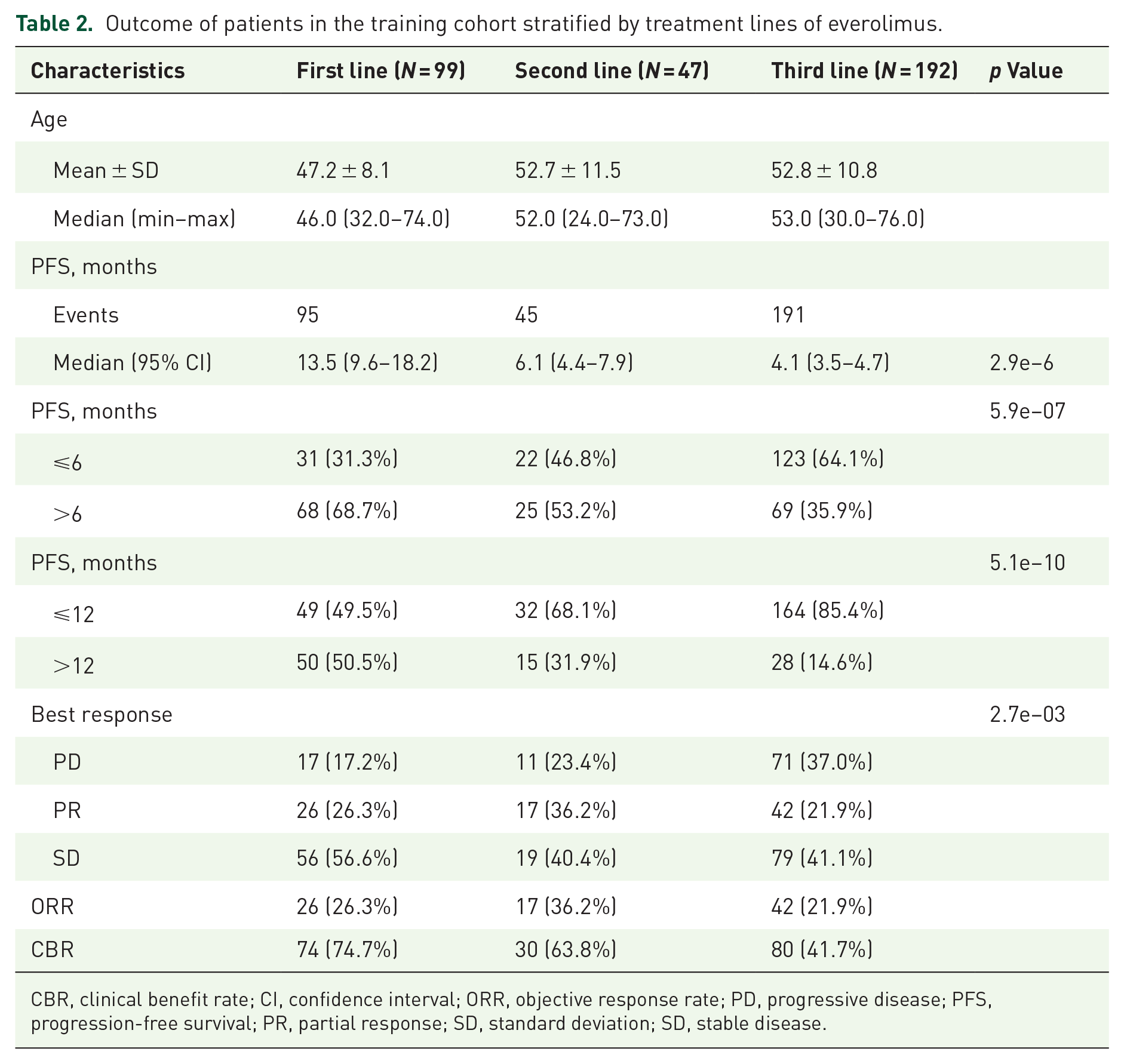
CBR, clinical benefit rate; CI, confidence interval; ORR, objective response rate; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, standard deviation; SD, stable disease.
No patients developed CR from everolimus in the study. The overall ORR was 25.1%, and the overall CBR was 54.4%. For 1L treatment, 26.3% of patients presented a favorable response to everolimus and 74.7% of patients managed to obtain clinical benefit from everolimus. The ORR2L, ORR3L reached 36.2%, 21.9% respectively. The CBR2L, CBR3L was 63.8%, 41.7%, respectively.
In the overall population, the median PFS was 5.6 months (range 4.9–6.3 months). The median PFS for 1L-, 2L-, and 3L-everolimus treatment was estimated to be 13.5, 6.1, and 4.1 months (Figure 2(a)). PFS for patients receiving everolimus was observed to worsen from 1L therapy to 2L therapy to 3L therapy (p < 0.0001, HR = 0.70, 95% CI = 0.61–0.82). Further subgroup analyses showed that 1L-everolimus therapy contributed to a profound improvement in PFS of 13.5 months, more than threefold that of 4.3 months for non-1L therapy (p < 0.0001, HR = 0.42, 95% CI = 0.33–0.54) (Figure 2(b)).

(a) In the training cohort, the Kaplan–Meier curve showed that PFS of efficacy-evaluable patients with HR+/HER2− ABC in different treatment lines differed, with PFS1L for 13.5 months, PFS2L for 6.1 months, and PFS3L for 4.1 months (p = 2.9e−6). (b) Kaplan–Meier curve showing PFS for patients with HR+/HER2− ABC receiving everolimus in 1L and non-1L. Patients in the training cohort who were treated with everolimus as 1L therapy had a significantly better PFS (p = 6.4e−12). p Value, HR, and 95% confidential internal are presented.
Construction of a predictive model for everolimus efficacy
We applied the univariate and multivariate Cox methods to investigate associations of 18 categorical variables and PFS for everolimus in the training cohort. The variables included age (65 years), Ki67 index (40%), DFS (5 years), HER2 status (HER2-zero, HER2-low, and HER2-enriched), HR status (ER+/PgR−, ER+/PgR+, and ER−/PgR+), treatment lines of everolimus (1L, 2L, and 3L), metastatic involvement at first relapse (lymph node, liver, lung, bone, breast, soft tissue, bone only, or visceral metastases only), number of metastatic sites at first recurrence (⩾3), and adjuvant therapy (endocrine therapy, radiotherapy, and chemotherapy).
Univariate Cox analysis revealed that post-1L treatment of everolimus, Ki67 index > 40, receiving adjuvant chemotherapy, liver metastasis at first relapse, and number of metastatic sites at first recurrence ⩾3 were significantly related to a shorter PFS (p < 0.05, Supplemental Figure S2). We further performed multivariate Cox analyses to analyze associations between these five factors and PFS. It found that post-1L treatment, Ki67 index > 40, number of metastatic sites at first recurrence ⩾3, and receiving adjuvant chemotherapy were independent risk factors for PFS. Liver metastasis at first relapse was not significantly associated with PFS in the multivariate Cox analyses (p = 0.65, Supplemental Figure S3).
Subsequently, we incorporated these four prognostic factors identified via multivariate Cox analyses to build a model for everolimus efficacy. Patients in the training cohort were segmented into the low-risk group and the high-risk group based on the median of the risk scores. The median PFS for patients in the low-risk group was 12.6 months, significantly longer than 2.7 months for patients in the high-risk group (p = 2.4e−64, HR = 9.41, 95% CI = 7.05–12.56) (Figure 3(a)). ROC analysis was used to test the specificity and sensitivity of the model. The AUC for 6-months, 1-year, and 3-year PFS was 0.96, 0.95, and 0.94, indicating its superior efficacy for predicting short-term and long-term survival for everolimus in patients with HR+/HER2− ABC (Figure 3(b) and (c)).

Construction of a model to predict survival outcome for everolimus therapy in patients with HR+/HER2− ABC. (a) In the training cohort, the Kaplan–Meier curve showed that PFS of patients with HR+/HER2− ABC in the high-risk group was 12.6 months, significantly longer than those who were in the low-risk group (p = 2.4e−64). p Value, HR, and 95% CI are presented. (b) The AUCs for 6-month PFS was 0.96. (c) The AUCs for 1-, and 3-year PFS were 0.95, and 0.94, respectively.
Validation of the predictive model for everolimus efficacy
From the aforesaid analyses, we can observe that the treatment line of everolimus displays predominant effects on PFS benefits for everolimus in patients enrolled in the training cohort. To balance the impact of the treatment line on the clinical benefits of everolimus, we randomly selected 40 patients from each treatment line (1L, 2L, and 3L) in the training cohort to establish the internal validation cohort (namely cohort A). An equal number of 60 patients were assigned to the low-risk group and the high-risk group. Patients of cohort A in the low-risk group achieved a median PFS of 18.4 months, significantly better compared to 3.1 months for patients in the high-risk group (p = 3.6e−11, HR = 3.78, 95% CI = 2.49–5.74). (Figure 4(a)). The AUC for 6-month, 1-year, and 3-year PFS was 0.84, 0.73, and 0.86, respectively (Figure 4(b) and (c)), suggesting a mediocre efficacy of the model for PFS benefits of everolimus.

Validation of the model for everolimus efficacy in patients with HR+/HER2− ABC. (a) In the internal validation cohort, the Kaplan–Meier curve showed that PFS of patients with HR+/HER2− ABC in the high-risk group was 18.4 months, significantly better as compared to 3.1 months for those who were in the low-risk group (p = 3.6e−11). (b) In the internal validation cohort, the AUCs for 6-month PFS was 0.84. (c) In the internal validation cohort, the AUCs for 1-, and 3-year PFS were 0.73, and 0.86, respectively. (d) In the external validation cohort, the Kaplan–Meier curve showed that PFS of patients with HR+/HER2− ABC in the high-risk group was 12.1 months, significantly better as compared to 2.3 months for those who were in the low-risk group (p = 2.9e−10). (e) In the external validation cohort, the AUCs for 6-month PFS was 0.94.
To further demonstrate the predictive merits of the model, we additionally collected 51 patients who had complete information about four parameters in the model to serve as the external validation cohort (namely cohort B). A total of 25 patients were distributed in the low-risk group, and 26 patients were in the high-risk group. Patients in the low-risk group had a better PFS of 12.1 versus 2.3 months for patients in the high-risk group (p = 2.9e−10, HR = 11.53, 95% CI = 4.68–28.37) (Figure 4(d)). The AUC for 6-month PFS was 0.94 (Figure 4(e)), showing high specificity and sensitivity of the model for short-term survival. Patients in cohort B who had PFS for more than 1 year were too less to conduct a ROC analysis. The main clinicopathologic characteristics of patients in the validation cohorts are presented in Table 3, Supplemental Tables S1 and S2.
Main characteristics and outcomes of patients in the validation cohorts.

CI, confidence interval; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, standard deviation; SD, stable disease.
Discussion
To the best of our knowledge, it represents the first attempt to establish a model for predicting survival benefits from everolimus in patients with HR+/HER2− ABC using clinicopathological characteristics collected from three cancer centers in a real-world setting.
Multiple studies have demonstrated the toleration and effectiveness of everolimus in patients with HR+ ABC.15–18 Among them, the BOLERO-2 study contributed to facilitating the use of everolimus in patients with HR+ ABC in clinical practice, which is widely accepted for comparison regarding studies related to everolimus for salvage therapy. 1 The overall CBR in the study was similar to the BOLERO-2 study (54.4% vs 51.3%), the ORR was higher than the BOLERO-2 study (25.1% vs 12.6%), the median PFS was shorter than the BOLERO-2 study (5.6 vs 7.8 months). PFS1L for everolimus in the study was 13.5 months, which was highly consistent with 14 months in the STEPAUT study 2 and was comparable to 10.1 months in the BRAWO study and 9.3 months in the EVEREXES study.17,18 To see, results from the study were generally consistent with previous studies.
Data for everolimus efficacy in various treatment lines were insufficient. In our study, we found that all patients who received everolimus as 1L, 2L, or 3L treatment derived clinical benefits and exhibited a good response to everolimus. Even for patients who were treated with everolimus as 3L or post-3L treatment, 41.7% of patients achieved CBR and 35.9% of patients had PFS of more than 6 months. Furthermore, we observed a progressive reduction in median PFS from 1L to 2L to 3L, with 13.5 months for 1L treatment, 6.1 months for 2L treatment, and 4.1 months for 3L or post-3L treatment. A single-arm study supported the results. It reported that survival benefits for everolimus in different treatment lines in patients with HR+ ABC differed. Specifically, PFS for patients with HR+ ABC that received everolimus as 1L, 2L, and 3L treatment was 14, 10.9, and 9.5 months, respectively. 2 In contrast, another study showed that PFS for HR+ patients who were treated with everolimus was maintained when comparing 1L-everolimus treatment to 2L or post-2L treatment. 17 Studies about PFS for everolimus in different treatment lines are expected.
Metastatic status of malignant tumors is a critical element for clinical staging, surveillance, and evaluation of therapeutic efficiency. 19 For patients with HR+/HER2− who have developed metastases, quality of life occupies an equally important position as extending survival. In the BOLERO-2 study, everolimus-based therapy led to prolonged PFS and tolerable toxicity for postmenopausal patients with HR+/HER2−, ABC, regardless of visceral or bone metastases. 1 Other studies showed that PFS was numerically better in patients with visceral metastases than in patients without visceral metastases but without significance.16,17 Previous studies have unraveled associations between PFS benefits for everolimus and metastatic involvements at baseline. From a different perspective, we paid attention to the metastatic site at the first recurrence and found that patients who experienced liver metastases at the first relapse had worse survival outcomes for everolimus in univariate Cox analysis, suggesting that liver metastases at the first relapse may play an impact in the efficacy of everolimus. It was consistent with a study specifically focusing on the Chinese population of patients with HR+/HER2− ABC, showing that liver metastasis was an independent risk factor for worse survival for everolimus-based treatment. 20 However, the subsequent multivariate analysis failed to show such statistical association after adjusting for other variables. From the perspective of statistics, it is typical and acceptable to see such a phenomenon. The variables included in multivariate analyses, which generally serve as potential confounding factors, can affect the original result of univariate Cox analyses. Considering the potent statistical significance between PFS and the variables for building the predictive model for everolimus, it is possible that the role of live metastasis in PFS was overshaded by other variables.
Ki67 index, as the most commonly used indicator for labeling the proliferative activity of tumor cells, plays a crucial role in the classification of HR+/HER2− breast cancer, therapeutic decision-making of neoadjuvant therapy, adjuvant therapy, salvage therapy, and prognostic assessment.21–23 It has been mentioned in numerous guidelines and consensus in the field of breast cancer due to its clinical significance.24,25 For clinical efficacy of everolimus, a small-scaled study showed that the Ki67 cut-off value of 35% could serve as a threshold for stratifying patients with HR+/HER2− ABC that could derive survival benefits from everolimus plus exemestane. 26 Similar to the previous study, we found that patients who had a Ki67 index of more than 40% developed shorter PFS and poor drug response for everolimus treatment in the advanced setting. Studies about the Ki67 index and everolimus efficacy remain scarce. More explorations ought to be conducted to demonstrate the associations.
In the study, we reported the treatment response and survival outcomes of everolimus in patients with HR+/HER2− ABC stratified by treatment lines of 1L, 2L, and 3L. More importantly, we identified four clinicopathological parameters associated with PFS for everolimus in a relatively large subset of patients with HR+/HER2− ABC, including post-1L treatment, Ki67 index of more than 40%, more than two metastatic sites at first relapse, and receiving adjuvant chemotherapy. We successfully used the parameters to develop a predictive model for the survival outcome of everolimus and confirmed its specificity and sensitivity in both the internal validation cohort and the external validation cohort.
The strengths of the study are worth mentioning. First, we successfully developed a predictive model for everolimus efficacy, based on a large-scaled sample size. Although some small-scale studies have explored correlations between different indicators and everolimus efficacy,20,26 we had a greater number of patients and evaluated more clinicopathologic factors. Second, the study was performed in a multicenter setting. Some single-institution studies have assessed everolimus efficacy for patients in different treatment lines, but our data are from more than one cancer center. This strengthens the generalizability and reliability of our findings. Third, survival outcomes including PFS, ORR, and CBR for everolimus as 1L, 2L, and 3L treatment were reported, which is more comprehensive than some existing studies.
There are also some limitations in the study. First, owing to its retrospective nature, caution should be exercised when interpreting the findings. Second, certain clinicopathological parameters, such as tumor-node-metastasis (TNM) staging, were not enrolled in the correlative analyses considering the large number of patients with missing information. Third, the study represented a large proportion of patients with HR+/HER2− ABC, but it did not exclude the small percentage of HR+/HER2-enriched patients. This might burden the results, though we conducted correlative analyses of HER2 status and survival outcomes and found no statistical associations. Fourth, the sample size for external validation was limited. Further evaluation of the predictive role of the model is favorably received in the future.
Conclusion
In the study, we successfully developed a model comprising post-1L everolimus treatment, Ki67 index of more than 40%, metastases of more than two sites at first recurrence, and receiving adjuvant chemotherapy, to predict inferior survival outcomes for everolimus in patients with HR+/HER2− ABC. It is conducive to the stratification of patients who can respond to everolimus, favoring the management of everolimus in the Chinese population of patients with HR+/HER2− ABC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241292256 – Supplemental material for Everolimus treatment in patients with hormone receptor-positive and human epidermal growth factor receptor 2-negative advanced breast cancer and a predictive model for its efficacy: a multicenter real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359241292256 for Everolimus treatment in patients with hormone receptor-positive and human epidermal growth factor receptor 2-negative advanced breast cancer and a predictive model for its efficacy: a multicenter real-world study by Yujing Tan, Zexi Peng, Hanfang Jiang, Fei Ma, Jiayu Wang, Pin Zhang, Qing Li, Xinzhu Tian, Yuhang Han, Danyang Ji, Binghe Xu, Weihong Zhao and Ying Fan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
