Abstract
Background:
Approximately half of the patients with advanced pancreatic ductal adenocarcinoma (PDAC) receive subsequent lines of chemotherapy. Recently, the liposomal irinotecan (nal-IRI) plus 5-fluorouracil/leucovorin (5-FU/LV) regimen is recommended as subsequent lines of chemotherapy. However, little is known about the predictive factors for the nal-IRI + 5-FU/LV regimen, especially in patients with previous irinotecan (IRI) exposure.
Objectives:
We investigated the predictive factors associated with nal-IRI + 5-FU/LV treatment in patients with PDAC.
Design:
Multicenter, retrospective cohort study.
Methods:
This study included patients with advanced PDAC who received the nal-IRI + 5-FU/LV regimen for palliative purposes.
Results:
Overall, 268 patients were treated with nal-IRI + 5-FU/LV. The median overall survival (OS) was 7.9 months (95% confidence interval (CI): 7.0–8.8), while the median progression-free survival (PFS) was 2.6 months (95% CI: 1.9–3.2). An albumin level of<4.0 g/dL, neutrophil-to-lymphocyte ratio (NLR) of ⩾3.5, liver or peritoneal metastasis, and a history of >3 lines of palliative chemotherapy were associated with worse OS. An NLR of ⩾3.5 and liver metastasis were significant predictive factors for worse PFS. Previous exposure to IRI was not a significant predictor. Patients without prior IRI (no-IRI) treatment showed relatively longer OS and PFS compared to IRI responders and nonresponders, but these differences were not significant when compared specifically to the responders (OS: 8.8 vs 8.1 months, p = 0.388; PFS: 3.6 vs 2.6 months, p = 0.126).
Conclusion:
An NLR of ⩾3.5 and liver metastasis were associated with worse PFS. Prior IRI exposure was not a significant predictive factor for OS and PFS, especially in IRI responders.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is an extremely aggressive cancer characterized by poor survival rates and is anticipated to rank as the second most common cause of deaths from cancer by 2030.1,2 Due to its aggressive characteristics, the absence of distinctive symptoms, and the lack of efficient screening methods, PDAC is often diagnosed at an advanced stage. Consequently, most patients with PDAC receive chemotherapy, and approximately half of them undergo second or subsequent lines of chemotherapy. 3
The current first-line chemotherapy options include FOLFIRINOX, a combination of 5-fluorouracil (5-FU), leucovorin, irinotecan (IRI), and oxaliplatin, or gemcitabine plus albumin-bound (nab) paclitaxel. The choice of subsequent chemotherapy is determined based on the response to initial treatment. According to the National Comprehensive Cancer Network guidelines, liposomal irinotecan (nal-IRI) in combination with 5-fluorouracil/leucovorin (5-FU/LV) is recommended for patients with advanced PDAC who have previously received gemcitabine-based chemotherapy. 4 However, FOLFIRINOX is commonly used in patients with various PDAC treatment status, including those who received neoadjuvant, adjuvant, and palliative therapy.5–8 This raises the question of whether the efficacy of nal-IRI + 5-FU/LV regimen is influenced by prior exposure to FOLFIRINOX.
A subgroup analysis of the NAPOLI-1 trial revealed that the benefits of overall survival (OS) and progression-free survival (PFS) was absent in patients who had been previously exposed to IRI compared with IRI-naïve patients. 9 By contrast, a previous retrospective study based on a nationwide database, which included 675 patients, reported that prior exposure to IRI was not a significant predictor of treatment outcomes for the nal-IRI + 5-FU/LV regimen. 10
Therefore, a comprehensive analysis of several factors, such as the patient’s clinical and tumor characteristics as well as chemotherapy history, should be carried out to identify the predictive factors associated with nal-IRI + 5-FU/LV treatment. This multicenter, retrospective, observational study aimed to investigate the predictive factors associated with nal-IRI + 5-FU/LV treatment in patients with PDAC.
Materials and methods
Study population
In this retrospective, observational study, we reviewed the electronic medical records obtained from four academic tertiary hospitals. This study was approved by the Institutional Review Board of each participating centers (Yonsei Medical Center, 4-2022-0776; Gachon University Gil Medical Center, GDIRB2022-227; Konkuk University Medical Center, KUMC 2022-08-018-001) and was carried out in accordance with the Declaration of Helsinki. The study included patients with histologically diagnosed or cytologically confirmed PDAC who received nal-IRI + 5-FU/LV treatment with a palliative intent between November 2017 and May 2022. The disease stage included recurred, locally advanced, or metastatic setting. The cutoff date for data analysis was December 31, 2022. The reporting of this study conforms to the statement of the STROBE (Supplemental Method S1). 11
Procedures
The nal-IRI + 5-FU/LV was administered following the protocol used in the NAPOLI-1 trial (nal-IRI 70 mg/m2 IRI free base over 90 min, followed by LV 400 mg/m2, and then 5-FU 2400 mg/m2 over 46 h, every 2 weeks). 12 The schedule and dose modification of chemotherapy were determined based on the patient’s condition and the physician’s discretion. The nal-IRI + 5-FU/LV regimen continued until disease progression, unacceptable toxicity, or patient refusal.
Variables
The baseline data including gender, age, Eastern Cooperative Oncology Group (ECOG) performance status, primary tumor site, site of metastatic lesions, the number of measurable metastatic sites, and tumor stage at initial diagnosis were obtained from the electronic medical records. The number of previous lines of palliative chemotherapy, previous first-line palliative chemotherapeutic agents, previous exposure to IRI, and previous surgical resection before nal-IRI + 5-FU/LV initiation was also assessed. A subgroup analysis was performed based on previous exposure and response to IRI. Based on their response to prior IRI, the patients were further categorized into the no-exposure, responder, and nonresponder groups. The responsiveness was determined based on the response to the initial four cycles of IRI-containing chemotherapy and assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 13 The responder group included patients who demonstrated a response, such as complete response (CR), partial response (PR), or stable disease (SD), while the nonresponder group comprised those who did not exhibit a response.
The laboratory results including carbohydrate antigen 19-9 (CA 19-9) levels, neutrophil count, lymphocyte count, neutrophil-to-lymphocyte ratio (NLR), albumin levels, and total bilirubin levels were evaluated within 30 days before nal-IRI + 5-FU/LV initiation.
Clinical outcomes
To evaluate the effectiveness of the chemotherapy, contrast-enhanced computed tomography and serum CA 19-9 tests were performed at intervals of 8–12 weeks. The treatment responses, assessed in accordance with RECIST version 1.1, were documented by radiologists specifically designated for this purpose. The final decision about the treatment response was then made separately by the attending physicians. The adverse events were assessed at each clinic visit and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. 14
To estimate OS and PFS, the dates of death and the last follow-up were reviewed. Data on survival and follow-up were collected through December 2022. OS was calculated from the initiation of nal-IRI + 5-FU/LV treatment into the date of the last follow-up or death. PFS was determined from the initiation of nal-IRI + 5-FU/LV treatment until the date of last follow-up, death, disease progression, or before the clinical cutoff date, whichever occurred first. Patients who did not experience disease progression or death were censored at the date of the last tumor assessment. At the time of the last follow-up, patients who did not fulfill the criteria for progression or death were censored. The objective response rate (ORR) was characterized as the proportion of patients attaining CR or PR. The disease control rate (DCR) was determined as the percentage of patients achieving CR, PR, or SD.
UGT1A1 polymorphisms
Based on their UGT1A1 phenotype, the patients were divided into three groups to investigate the association between nal-IRI + 5-FU/LV treatment and UGT1A1 polymorphisms. An intermediate metabolizer (IM) is a person who has a heterozygous genotype for one decreased function allele (UGT1A1 *1/*28 or *1/*6). A poor metabolizer (PM) is defined as an individual who carries two reduced function alleles (UGT1A1 *28/*28, *6/*6, or *6/*28). 15 A person with an extensive metabolizer (EM) genotype, such as a UGT1A1 *1/*1, is shown to carry two normal function alleles. UGT1A1 testing was conducted among patients who consented to undergo the screening.
Statistical analysis
Descriptive statistics was presented as the median (interquartile range) or number (%) where applicable. The Kaplan–Meier method was used for survival analysis, and the log-rank test was used for comparisons. Subgroup analysis was performed according to IRI exposure, IRI response, and UGT1A1 polymorphism. The Cox regression model was utilized for conducting univariate analyses of OS and PFS. The multivariable Cox regression models were adjusted for variables that were considered significant in the univariable analysis. The Student’s t-test or Mann–Whitney test was used for continuous variables, while the Chi-squared test or Fisher’s exact test was used for categorical variables to compare differences between the two groups. The three groups were subjected to comparison based on UGT1A1 polymorphism using the Kruskal–Wallis test. Subsequently, the Mann–Whitney test with Bonferroni correction was applied for further analysis.
A p value of less than 0.05 on both sides was deemed significant. The statistical program SPSS 24.0 (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses.
Results
Patient characteristics
After screening the study participants, only 268 received nal-IRI + 5-FU/LV treatment (Figure 1). Table 1 displays the baseline characteristics of the study population. The median age was 62 years (interquartile range: 54–68 years), and 153 patients (57.1%) were men. A total of 251 (93.7%) patients exhibited an ECOG performance status of 0 or 1, with only five patients (1.9%) having an ECOG performance status of 2. Patients with no information in the electronic medical records are indicated as unknown. In the initial presentation, 154 patients (57.5%) were diagnosed with metastatic PDAC. The liver (39.9%), distant lymph nodes (20.9%), and peritoneum (13.8%) were the most frequently found metastatic sites. Surgical resection with a curative intent was carried out in 87 patients (32.5%). Most patients (50.4%) underwent nal-IRI + 5-FU/LV as third-line palliative treatment. Only three patients (1.1%) were administered nal-IRI + 5-FU/LV as first-line treatment. FOLFIRINOX and gemcitabine plus nab-paclitaxel were the most commonly selected first-line palliative chemotherapeutic regimens (89.2%). Meanwhile, 179 patients (66.8%) had a history of IRI exposure, and all patients with IRI exposure received the FOLFIRINOX regimen.

Flow chart of the patient selection process.
Baseline characteristics of patients with pancreatic cancer treated with nal-IRI + 5-FU/LV.

Values are expressed as numbers (%) or median (interquartile range).
Some patients had multiple metastatic sites and are listed in more than one group.
Others including the brain, bone, and stomach metastasis.
Patients received neoadjuvant, adjuvant, or locally advanced treatment, but were not previously treated for metastatic disease.
Including exposure to FOLFIRINOX as neoadjuvant and adjuvant treatment.
CA 19-9, carbohydrate antigen 19-9; ECOG, Eastern Cooperative Oncology Group; 5-FU/LV, 5-fluorouracil/leucovorin; nal-IRI, liposomal irinotecan; NLR, neutrophil-to-lymphocyte ratio.
Clinical outcomes
The therapeutic outcomes are outlined in Table 2. A total of two (0.7%), 14 (5.2%), 112 (41.8%), and 140 (52.3%) patients receiving nal-IRI + 5-FU/LV demonstrated CR, PR, SD, and progressive disease (PD), respectively. The ORR was 6.0%, while the DCR was 47.8%. Of the 268 patients, 122 (45.5%) received subsequent chemotherapy after experiencing PD on nal-IRI + 5-FU/LV regimen. The median PFS and OS were 2.6 months (95% confidence intervals (CI): 1.9–3.2 months) and 7.9 months (95% CI: 7.0–8.8 months), respectively (Figures 2 and 3).
Clinical outcomes.
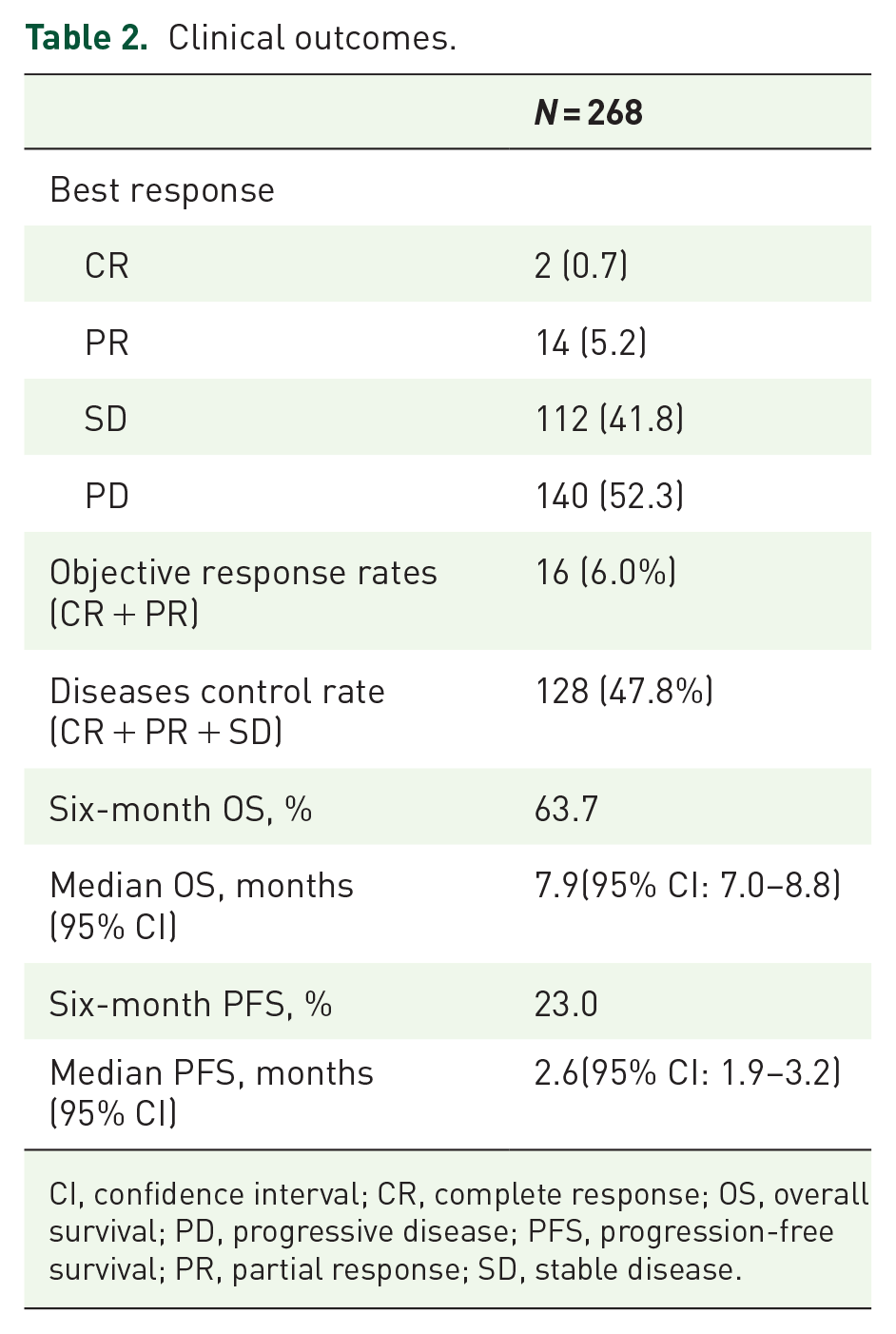
CI, confidence interval; CR, complete response; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.

Survival outcomes with nal-IRI + 5-FU/LV.
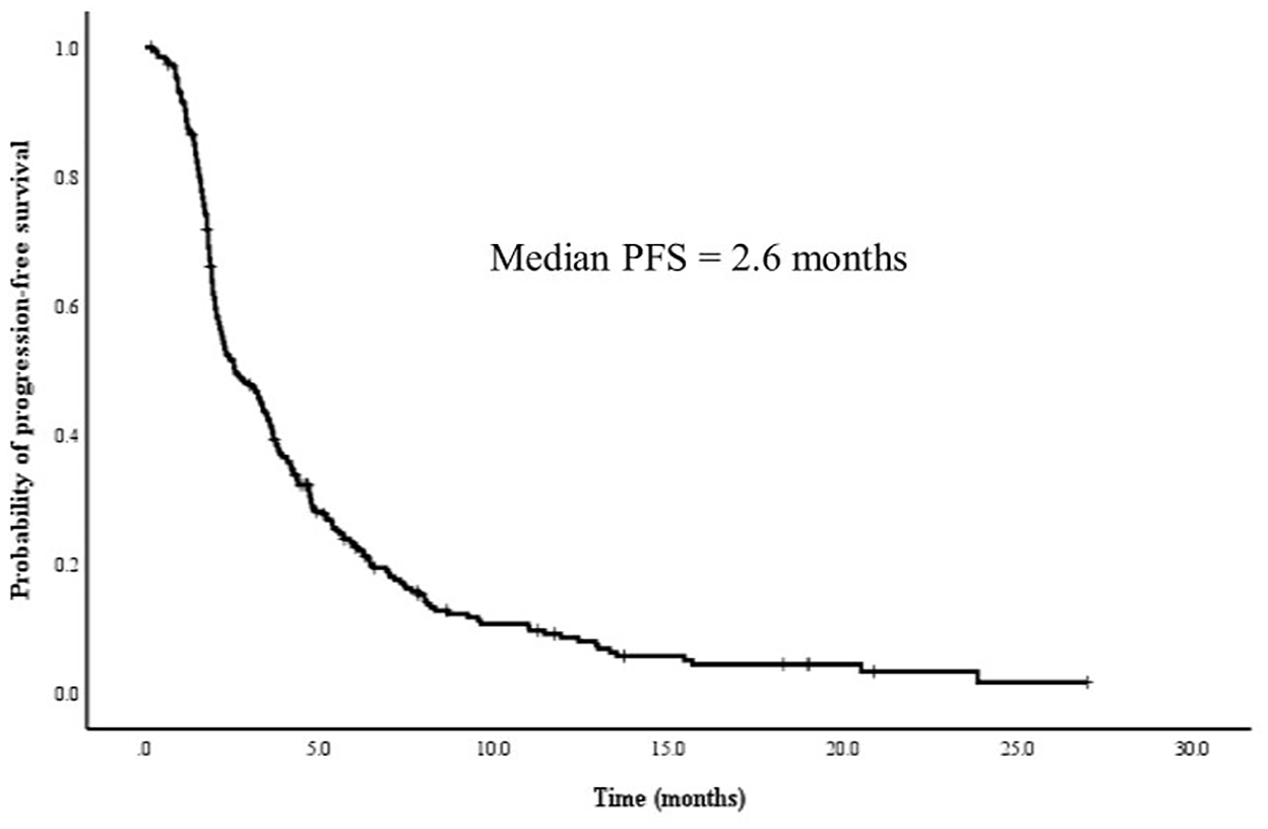
Progression-free survival outcomes with nal-IRI + 5-FU/LV.
Predictive factors for survival outcomes
To ascertain predictive factors influencing survival outcomes, a multivariable analysis was performed using a Cox proportional hazard model (Table 3). Patients with a low albumin level (albumin < 4.0 g/dL) showed worse OS (hazard ratio (HR): 1.46, 95% CI: 1.05–2.05, p = 0.027) compared with other patients (albumin ⩾ 4.0 g/dL). The presence of peritoneum metastases and a history of receiving ⩾3 lines of palliative chemotherapy were significantly associated with worse OS (HR: 1.54, 95% CI: 1.00–2.38, p = 0.049; HR: 1.73, 95% CI: 1.22–2.46, p = 0.002, respectively). However, albumin levels, peritoneum metastases, and a history of receiving ⩾3 lines of palliative chemotherapy were not significant predictive factors for PFS. Patients with high NLR (NLR ⩾ 3.5) demonstrated shorter PFS (HR: 1.92, 95% CI: 1.43–2.58, p < 0.001) and OS (HR: 2.34, 95% CI: 1.64–3.34, p < 0.001) in comparison to other patients (NLR < 3.5). The presence of liver metastases was significantly associated with worse PFS (HR: 1.55, 95% CI: 1.18–2.03, p = 0.001) and OS (HR: 1.55, 95% CI: 1.12–2.16, p = 0.008).
Cox regression analysis of survival outcomes.

CI, confidence interval; HR, hazard ratio; OS, overall survival; PFS, progression-free survival.
Previous exposure to IRI and ECOG performance status were not significant predictors of OS and PFS in the multivariable analysis.
We also conducted subgroup analyses for the populations receiving FOLFIRINOX or gemcitabine plus nab-paclitaxel in the first line to decrease the heterogeneity of the study populations (Supplemental Table S2). The Cox regression analysis still showed that NLR of ⩾3.5 and the presence of liver metastasis were associated with worse PFS and OS in the multivariable analysis.
Patients administered nal-IRI + 5-FU/LV, who had previously experienced progression after four cycles of prior IRI-based chemotherapy, exhibited notably shorter OS and PFS compared to those who had not been exposed to any IRI-based chemotherapy (OS: 5.9 vs 8.8 months, p = 0.021; PFS: 1.8 vs 3.6 months, p = 0.012) and when compared with patients who responded after four cycles of prior IRI-based chemotherapy (OS: 5.9 vs 8.1 months, p = 0.049; PFS: 1.8 vs 2.6 months, p = 0.057) (Figure 4(a) and (b)). Patients who did not receive prior IRI treatment exhibited the longer OS and PFS. However, even in the 4-cycle responders with a history of prior IRI treatment, no significant difference was found in the median OS and PFS compared with patients who had not received previous IRI treatment (OS: 8.1 vs 8.8 months, p = 0.388; PFS: 2.6 vs 3.6 months, p = 0.126). Furthermore, we performed additional analysis among 14-cycle responders, 14-cycle nonresponders, and no-exposures (Supplemental Figure S1). We defined a cutoff level of 14 cycles based on the median PFS value of patients who received FOLFIRINOX as first-line chemotherapy. There was also no significant difference between 14-cycle responders and no-exposure in the median OS and PFS (OS: 7.8 vs 8.8 months, p = 0.280; PFS: 2.6 vs 3.6 months, p = 0.177). Regarding survival outcomes between 14-cycle responders and 14-cycle non-responders, there was no significant difference (OS: 7.8 vs 7.3 months, p = 0.912; PFS: 2.6 vs 2.1 months, p = 0.418). To evaluate the relationship between the previous exposure to IRI and the benefit from subsequent treatment with nal-IRI + 5-FU/LV, we classified patients into non progressor to IRI (IRI-sensitive), progressors with clinical benefit (IRI-resistant), and progressors with PD at first response assessment (and IRI-refractory). Our study included 2, 136, and 40 patients in the IRI-sensitive, IRI-resistant, and IRI-refractory groups, respectively. Consequently, due to the limited sample size of the IRI-sensitive group, only the IRI-resistant and IRI-refractory groups had sufficient sample sizes for analysis in this study. IRI-resistant group showed the trend of better OS and PFS compared to the IRI-refractory group (OS: 8.0 vs 5.9 months, p = 0.055; PFS: 2.6 vs 1.8 months, p = 0.052, Supplemental Figures S2 and S3).

Survival outcomes with nal-IRI + 5-FU/LV based on prior irinotecan (IRI) response: overall survival (a) and PFS (b). Patients were classified based on whether their disease had responded to prior IRI treatment (IRI responder), had not responded to prior IRI treatment (IRI non-responder), or had not previously received any IRI chemotherapy (no-IRI).
Adverse events
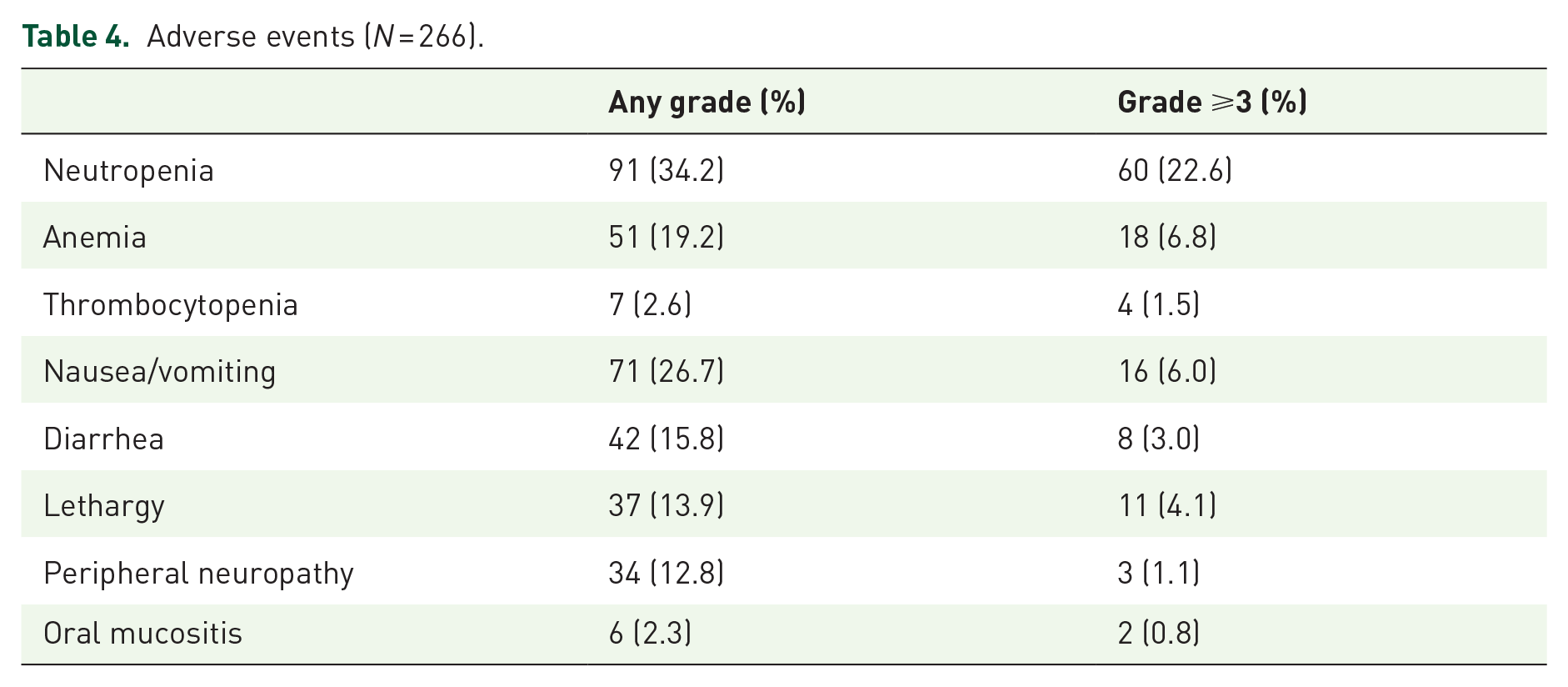
Table 4 provides a list of adverse events observed during the treatment with nal-IRI plus 5-FU/LV. Two patients with missing values were excluded, leaving 266 patients for analysis. Adverse events of any grade were noted in 194 (72.9%) patients, with grade 3–4 adverse events observed in 94 (35.3%) patients. The most prevalent adverse events included neutropenia (34.2%) and nausea/vomiting (26.7%). Grade 3–4 adverse events were particularly common in cases of neutropenia (22.6%).
Adverse events (N = 266).
Impact of UGT1A1 polymorphism on survival outcomes and adverse events
UGT1A1 genotype testing was conducted in 79 (29.5%) patients. In their midst, 24 (30.4%) were classified as EMs, 45 (57.0%) were classified as IMs, and 10 (12.7%) were classified as PMs. Among the IMs, 26.6% (21/79) and 30.4% (24/79) carried the UGT1A1 *1/*6 and *1/*28 genotypes, respectively. The most common genotype among the PMs was UGT1A1 *6/*28 (7.6%, 6/79), followed by UGT1A1 *6/*6 (5.1%, 4/79).
To assess the impact of UGT1A1 on both survival outcomes and adverse events, a subgroup analysis was carried out. Patients with EM plus IM experienced significantly longer OS and PFS compared with patients with PM (OS: 8.8 vs 3.4 months, p = 0.003; PFS: 2.8 vs 2.0 months, p = 0.054, Figures 5 and 6). To account for potential confounding factors influencing survival outcomes, a multivariate analysis was conducted using a Cox regression model (Supplemental Table S1). However, only NLR emerged as a significant predictive factor for OS and PFS. Meanwhile, patients with high NLR (NLR ⩾ 3.5) had shorter OS (HR: 4.28, 95% CI: 2.14–8.55, p < 0.001) and PFS (HR: 2.50, 95% CI: 1.39–4.49, p = 0.002) compared with other patients (NLR < 3.5). Patients with a history of receiving ⩾3 lines of palliative chemotherapy had significantly shorter PFS (HR: 4.27, 95% CI: 1.43–12.75, p = 0.009) than those with a history of receiving < 3 lines of palliative chemotherapy. However, UGT1A1 status did not emerge as a significant predictor in the multivariate analysis using the Cox regression model even though it appeared significant in the univariate analysis.
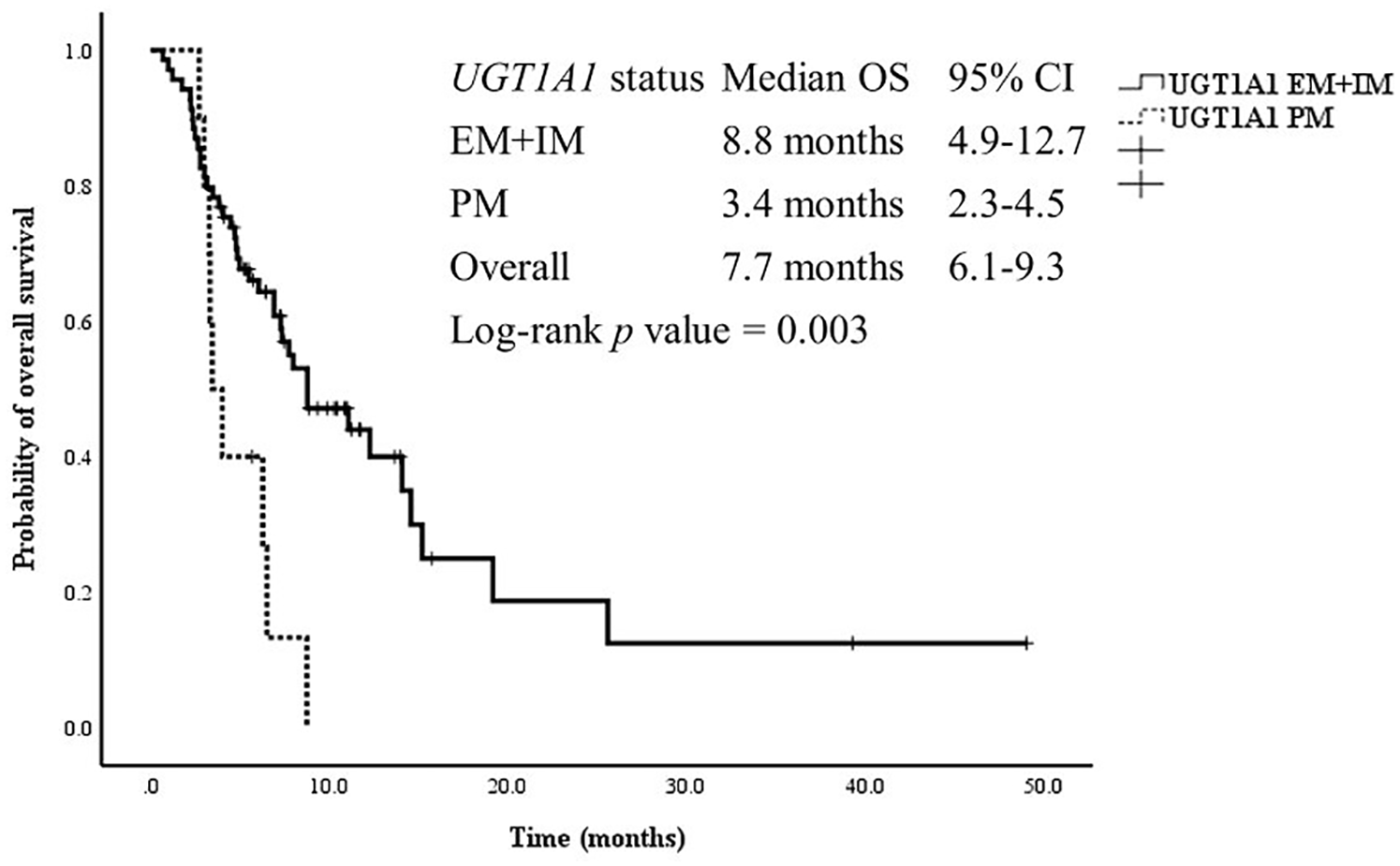
Survival outcomes with nal-IRI + 5-FU/LV according to UGT1A1 status (N = 79).

PFS outcomes with nal-IRI + 5-FU/LV according to UGT1A1 status (N = 79).
The adverse events of UGT1A1 EM plus IM and PM were evaluated (Table 5). However, one patient with missing values was excluded, leaving 78 patients for analysis. Diarrhea was more prevalent in the PM group than in the EM plus IM group (50% vs 7.4%, p = 0.002). Analysis of grade 3–4 adverse events was not possible due to the limited number of patients.
Adverse events according to UGT1A1 phenotype (N = 78).

Discussion
With the recent improvement in the survival rates of patients with PDAC, the number of patients receiving second- or third-line chemotherapy also increased. Notably, a significant surge was observed in the utilization of nal-IRI + 5-FU/LV in subsequent chemotherapy. Consequently, there is a growing need for real-world data regarding clinical outcomes and predictive factors for treatment responsiveness. In this study, the observed PFS and OS were 2.6 and 7.9 months, respectively. These results align closely with those reported in the NAPOLI-1 trial and other previous real-world studies, which indicated a median PFS of 2.0–3.5 months and a median OS of 4.4–9.4 months.16–19 In terms of the predictors of OS and PFS, an NLR of ⩾3.5 and liver metastasis were associated with worse PFS. The NLR is a widely recognized marker of systemic inflammatory response and functions as a predictive factor in cancer treatment. Elevated levels of neutrophil can lead to the formation of compartments containing vascular endothelial growth factors and contribute to tumor angiogenesis. 20 Conversely, lymphocytopenia can negatively affect immune surveillance and lymphocyte-mediated immune responses.21,22 In the meta-analysis, high NLR was significantly associated with worse OS in pancreatic cancer.21,23 The thresholds used for NLR varied between >5 and >3, reflecting a combination of tumor-related and host-related genetic and environmental factors. Further prospective studies are required to establish a clear and robust correlation between NLR and survival outcomes.
In this study, patients who did not previously receive IRI treatment showed longer OS and PFS than poor responders to IRI. No significant difference was observed in the median OS and PFS between responders and those who had not previously received IRI treatment. Several studies reported poor survival outcomes with nal-IRI + 5-FU/LV treatment in patients who previously received conventional IRI treatment, such as the FOLFIRINOX regimen.9,19 Subgroup analysis of the NAPOLI trial reported that the improvement in ORR and CA 19-9 response following nal-IRI + 5-FU/LV treatment was observed only in patients who had not previously received IRI treatment. 9 Another multicenter center study using real-world data from 86 Asian patients treated with nal-IRI + 5-FU/LV showed reduced survival outcomes in the subgroup who had previously received IRI-based chemotherapy. 19 However, a recent retrospective study, using a nationwide Flatiron Health electronic record-derived database from 675 patients with metastatic PDAC treated with nal-IRI + 5-FU/LV, reported that prior IRI exposure was not a significant predictor of patient outcomes. 10 Among the studies mentioned earlier, the NAPOLI-I subgroup analysis had limitations due to the absence of multivariable analysis, and the Asian study had a relatively small sample size. Therefore, a multivariable analysis was conducted to investigate the relationship between prior IRI exposure and the survival outcomes of patients with PDAC treated with nal-IRI + 5-FU/LV.
In this study, prior IRI exposure was not a significant predictor for survival outcomes in the multivariable analysis. Previous studies have reported that patients who had a favorable prior response to IRI-containing chemotherapy showed longer OS and PFS compared with those who progressed earlier.24,25 Thus, we further categorized patients into three groups based on their response to prior IRI exposure status: no-exposure group, responder group, and nonresponder group. The no-exposure group exhibited longer survival outcomes compared to the other two groups. However, this difference was not statistically significant in OS or PFS analyses when compared specifically to the responder group. Interestingly, the no-exposure group received nal-IRI + 5-FU/LV earlier in their treatment course (e.g., first or second line) compared to the responder group. This earlier administration could potentially explain the observed longer survival outcomes in the no-exposure group. Furthermore, when analyzing patients whose response to prior IRI treatment was sustained after 14 treatment cycles (Supplemental Figures S1), no significant difference was observed in the survival outcomes between IRI responders and nonresponders. This suggests that nal-IRI + 5-FU/LV could be administered as subsequent chemotherapy for patients with a sustained response to prior IRI treatment.
Here are two potential reasons why patients may benefit from nal-IRI even after experiencing disease progression following IRI treatment are as follows: First, studies have shown that nal-IRI therapy exposes SN-38 (the active metabolite of irinotecan) in the body through a different mechanism compared to IRI. This difference may help overcome resistance to irinotecan observed in small-cell lung cancer models. 26 In previous study, nal-IRI demonstrated antitumor activity in tumors that progressed following treatment with nonliposomal irinotecan. Even though the tumors did not respond to nonliposomal irinotecan, mice treated with nal-IRI experienced significant tumor shrinkage. This suggests that the limited antitumor activity of irinotecan may be due to inadequate drug delivery rather than resistance to topoisomerase 1 inhibition. Therefore, sustained delivery of SN-38 through nal-IRI could enhance antitumor efficacy. The liposomal formulation of nal-IRI alters drug pharmacokinetics, resulting in increased drug exposure to the tumor. Second, patients who responded well to prior irinotecan treatment are likely to be in better health and to have survived long enough to receive additional treatments.
The relationship between UGT1A1 polymorphism and treatment outcomes or adverse events in patients with PDAC treated with nal-IRI + 5-FU/LV is not well established. Among the Taiwan patient cohort treated with nal-IRI + 5-FU/LV, patients with homozygosity or compound heterozygosity for UGT1A1*6 or UGT1A1*28 showed a significantly higher incidence of neutropenia and diarrhea compared with those with single heterozygosity or wild-type UGT1A1. 27 In a Japanese cohort study conducted, 54 patients treated with nal-IRI + 5-FU/LV, no significant difference was found between the wild-type and single heterozygous groups (UGT1A1*1/*6 or *1/*28). 28 Herein, a subgroup analysis was conducted in patients who were examined for UGT1A1. Patients treated with EM plus IM had significantly longer OS and PFS compared with those with PM. However, in our multivariate analysis using the Cox regression model, UGT1A1 was not a significant predictor of survival outcomes. Diarrhea was more frequent in the PM group than in the EM plus IM group. In contrast to the Japanese study, no significant difference was observed in the occurrence of neutropenia between the PM and EM plus IM groups. The absence of a difference could be attributed to the smaller sample size in the PM group. To validate the impact of UGT1A1 status on the efficacy and safety of nal-IRI + 5-FU/LV treatment, a larger prospective study is necessary.
In this study, the incidence of adverse events was lower in patients treated with nal-IRI + 5-FU/LV treatment than in those treated with FOLFIRINOX. Considering the tolerable safety profile of nal-IRI + 5-FU/LV treatment, it could be used as an alternative treatment option for patients who cannot tolerate FOLFIRINOX due to its toxicity. We suggest the nal-IRI + 5-FU/LV regimen as a potential alternative to FOLFIRINOX for patients with the following characteristics: those precluded from receiving FOLFIRINOX due to performance status (e.g., ECOG 2) or toxicity concerns, those who need to discontinue FOLFIRINOX due to neutropenia or peripheral neuropathy, or those at high risk of neutropenia/diarrhea due to UGT1A1 polymorphism (PMs).
This study has several limitations owing to its retrospective nature. First, dose modifications were made at the discretion of the physicians rather than following a predefined protocol, as observed in prospective randomized trials. This could introduce the risk of potential selection bias, which we attempted to address through the multicenter study design. Second, in this study, the rates of many toxicities are lower than the ones found in NAPOLI-1 study. Our study differs from the NAPOL-1 study in its inclusion criteria for the adverse event analysis. While NAPOL-1 included all patients receiving at least one dose of the drug, our analysis is restricted to patients who received at least three cycles of chemotherapy. We suspect that this may be one of the reasons for the discrepancy. However, the interpretation of adverse event rates in this study requires caution due to several limitations. The retrospective nature of the study design may introduce selection bias and lack of missing data including adverse events. The use of prophylactic treatments (e.g., antiemetic premedication, steroids, atropine, and G-CSF prophylaxis) and drug modifications were also at the discretion of the medical staff, potentially leading to variability in patient management. Lastly, the relatively small size of the UGT1A1 PM group poses limitations on the explanation of the subgroup analysis. To validate the influence of UGT1A1 status on the efficacy and safety of nal-IRI + 5-FU/LV treatment, we plan to carry out a more extensive prospective study.
Conclusion
We investigated the predictors of survival outcomes in patients treated with nal-IRI + 5-FU/LV. Our study distinguishes itself from other research studies by using real-world data with a substantial patient sample size and incorporating UGT1A1 genetic factor analysis. An NLR of ⩾3.5 and the presence of liver metastasis were associated with poorer OS and PFS. Meanwhile, prior exposure to IRI did not emerge as a significant predictive factor for OS and PFS, especially among patients who had exhibited a prolonged response to FOLFIRINOX treatment.
Supplemental Material
sj-docx-4-tam-10.1177_17588359241279688 – Supplemental material for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study
Supplemental material, sj-docx-4-tam-10.1177_17588359241279688 for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study by Jiyoung Keum, Hee Seung Lee, Chan Su Park, Jeehoon Kim, Wonjoon Jang, Kyung In Shin, Huapyong Kang, Sang Hoon Lee, Jung Hyun Jo, Sung Ill Jang, Moon Jae Chung, Jeong Youp Park, Seung Woo Park, Jae Hee Cho and Seungmin Bang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-5-tam-10.1177_17588359241279688 – Supplemental material for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study
Supplemental material, sj-docx-5-tam-10.1177_17588359241279688 for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study by Jiyoung Keum, Hee Seung Lee, Chan Su Park, Jeehoon Kim, Wonjoon Jang, Kyung In Shin, Huapyong Kang, Sang Hoon Lee, Jung Hyun Jo, Sung Ill Jang, Moon Jae Chung, Jeong Youp Park, Seung Woo Park, Jae Hee Cho and Seungmin Bang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-1-tam-10.1177_17588359241279688 – Supplemental material for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study
Supplemental material, sj-jpg-1-tam-10.1177_17588359241279688 for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study by Jiyoung Keum, Hee Seung Lee, Chan Su Park, Jeehoon Kim, Wonjoon Jang, Kyung In Shin, Huapyong Kang, Sang Hoon Lee, Jung Hyun Jo, Sung Ill Jang, Moon Jae Chung, Jeong Youp Park, Seung Woo Park, Jae Hee Cho and Seungmin Bang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359241279688 – Supplemental material for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study
Supplemental material, sj-jpg-2-tam-10.1177_17588359241279688 for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study by Jiyoung Keum, Hee Seung Lee, Chan Su Park, Jeehoon Kim, Wonjoon Jang, Kyung In Shin, Huapyong Kang, Sang Hoon Lee, Jung Hyun Jo, Sung Ill Jang, Moon Jae Chung, Jeong Youp Park, Seung Woo Park, Jae Hee Cho and Seungmin Bang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359241279688 – Supplemental material for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study
Supplemental material, sj-jpg-3-tam-10.1177_17588359241279688 for Survival predictors in patients with pancreatic cancer on liposomal irinotecan plus fluorouracil/leucovorin: a multicenter observational study by Jiyoung Keum, Hee Seung Lee, Chan Su Park, Jeehoon Kim, Wonjoon Jang, Kyung In Shin, Huapyong Kang, Sang Hoon Lee, Jung Hyun Jo, Sung Ill Jang, Moon Jae Chung, Jeong Youp Park, Seung Woo Park, Jae Hee Cho and Seungmin Bang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
