Abstract
Background:
Esophagogastric and pancreaticobiliary cancers are associated with chronic blood loss, poor nutrition, and surgical interventions that interfere with iron absorption. Patients with these cancers often have a higher incidence of chemotherapy-induced anemia (CIA) than patients with other malignancies.
Objectives:
To investigate the efficacy of intravenous iron or erythropoietin-stimulating agents (ESA) for CIA treatment in patients with esophagogastric or pancreaticobiliary cancer.
Design:
Retrospective, comparative chart review of patients with esophagogastric or pancreaticobiliary cancer who received ferric carboxymaltose (FCM), or darbepoetin alfa (DA), and myelosuppressive chemotherapy at Chungbuk National University Hospital between June 2018 and December 2022.
Methods:
To assess the efficacy of FCM or DA over time, data on hemoglobin (Hb) levels were collected from the time of administration of FCM or DA (baseline) until 6 months post-baseline, when available.
Results:
In total, 214 patients (124 in the FCM and 90 in the DA group) were included in the analysis. The FCM group had a higher maximum Hb level and Hb changes for 3 months (mean ± standard deviation) following FCM or DA administration from baseline than the DA group (11.3 ± 1.5 versus 10.9 ± 1.2 g/dL, p = 0.02 and 2.0 ± 1.4 versus 1.5 ± 1.1 g/dL, p = 0.004, respectively). The FCM group had a higher proportion of Hb responders than the DA group (83.9% versus 68.9%, p = 0.013). Based on multivariable analysis, only the CIA treatment group was a significant factor for Hb response (odds ratio = 2.06, 95% confidence interval = 1.05–4.06, p = 0.036).
Conclusion:
Both FCM and DA are effective, and FCM showed a higher Hb response than DA for CIA treatment in patients with esophagogastric or pancreaticobiliary cancer. Therefore, further randomized controlled trials should determine the optimal treatment for CIA in patients with these cancers undergoing myelosuppressive chemotherapy.
Introduction
Anemia is prevalent in patients with cancer, particularly in patients receiving cytotoxic chemotherapy.1–5 The prevalence of anemia in patients with cancer varies, ranging from 20% to 60% and reaching 60–90% in those undergoing chemotherapy.4,5 It is associated with decreased quality of life, efficacy of cancer treatments, and overall survival.6,7 Evaluating the causes of anemia in patients with cancer can be challenging because they are often multifactorial, including factors such as tumor bleeding, nutritional deficiencies, iron deficiency, renal insufficiency, metastatic bone marrow infiltration, chemotherapy-induced myelosuppression, or a combination of these factors.1–3
Current therapeutic options for chemotherapy-induced anemia (CIA) include red blood cell (RBC) transfusions, erythropoietin-stimulating agent (ESA) therapy, or iron supplements.1–3 In clinical practice, RBC transfusion and/or ESAs are commonly performed to treat CIA. Although RBC transfusions can increase hemoglobin (Hb) levels rapidly and, thus, a rapid improvement in anemia-related symptoms, their effects are temporary and can be associated with transfusion-related adverse events. ESA therapy stimulates erythropoiesis, providing a gradual improvement in CIA, and helping to avoid RBC transfusions. However, given their association with increased thrombotic events and possible shortened time to tumor progression, ESAs should not be used when the treatment intent is curative. Therefore, treatment guidelines for CIA aim to prevent RBC transfusions and minimize ESA dosage.
Iron deficiency anemia (IDA) is defined as depleted iron stores (absolute IDA) or limited mobilization of iron from adequately filled iron stores (functional IDA).1–3,8–10 In patients with cancer, absolute IDA is mainly caused by bleeding or insufficient intestinal iron resorption. Most importantly, in patients with cancer, iron homeostasis is often impaired via the release of proinflammatory cytokines and upregulation of hepcidin, the main regulator of iron uptake and release. Increased hepcidin levels result in insufficient iron supply due to the internalization of ferroportin, the most important transmembrane channel for the export of iron from erythrocytes and macrophages into the circulation.8–10,11 Iron supplements can be administered orally or intravenously. Although oral iron supplementation is suitable for most patients with IDA, many cancer patients with CIA do not respond to or tolerate it well. Consequently, the National Comprehensive Cancer Network (NCCN) and European Society for Medical Oncology (ESMO) guidelines recommend intravenous (IV) iron supplementation, either alone or in conjunction with ESA therapy, for CIA treatment.1,2
Esophagogastric and pancreaticobiliary cancers are associated with chronic blood loss, poor nutrition, and surgical interventions that interfere with iron absorption. Additionally, most systemic anticancer therapies for these cancer types involve myelosuppressive cytotoxic chemotherapeutic agents. Therefore, the incidence of CIA in patients with these cancers is higher than in those with other malignancies.12–16
This study aimed to investigate the efficacy of IV iron supplementation or ESA therapy in CIA treatment in patients with esophagogastric or pancreaticobiliary cancer undergoing chemotherapy.
Materials and methods
Study design and population
In our Medical Oncology Division, ferric carboxymaltose (FCM) as an IV iron supplement and darbepoetin alfa (DA) as an ESA are administered according to the decisions of individual clinicians for CIA treatment in patients with esophagogastric or pancreaticobiliary cancer undergoing chemotherapy. We conducted a retrospective study by reviewing the medical records of all consecutive patients diagnosed with esophagogastric or pancreaticobiliary cancer, who underwent chemotherapy and received FCM or DA for CIA treatment at Chungbuk National University Hospital between June 2018 and December 2022. We excluded patients who had not been followed up for Hb levels within 3 months following FCM or DA administration, and those who had received CIA treatment other than FCM or DA within 6 months.
This study was reviewed and approved by the Institutional Review Board of Chungbuk National University Hospital (IRB No. CBNUH 2023-03-039-001). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement (Supplemental File).
Data collection
This study retrospectively used data routinely collected during clinical practice at Chungbuk National University Hospital. Data were collected by extracting the information from electronic medical records. Demographic and disease characteristics included age, sex (male or female), Eastern Cooperative Oncology Group (ECOG) performance status, primary tumor site (esophagogastric cancer, including esophageal and gastric cancer, or pancreaticobiliary cancer, including pancreatic and biliary tract cancer), stage (I–III or IV), the intent of chemotherapy (neoadjuvant, adjuvant, or palliative), and chemotherapy regimen of cytotoxic agents (single, doublet, or triplet). To evaluate the efficacy of FCM or DA over time, Hb levels and packed red cell (PRC) transfusions from the time of administration of FCM or DA (baseline) until 6 months post-baseline were recorded when available.
Statistical analysis
The data are reported as numbers and percentages for categorical variables and mean and standard deviation (SD) for numerical variables. The Chi-square test was used to compare percentages, and Student’s t-test was used to compare mean values. The maximum Hb level, maximum change in Hb level, and proportion of patients showing an Hb response within 3 months of the baseline were also analyzed. The definition of Hb response following CIA treatment is unclear. Several previous studies have defined it as a Hb increase of ⩾2.0 g/dL or Hb of ⩾12.0 g/dL,17,18 whereas recent studies have defined it as a Hb increase of ⩾1.0 g/dL or Hb of ⩾11.0 g/dL.19,20 In the present study, Hb response was defined as a Hb increase of ⩾1.0 g/dL or a Hb of ⩾11.0 g/dL within 3 months following CIA treatment without an RBC transfusion. The proportion of patients receiving a PRC transfusion within 6 months of the baseline and the time to PRC transfusion were also analyzed.
An exploratory analysis of the clinical factors predicting the Hb response was conducted using univariable and multivariable analyses. Only the factors with a significance level of less than 0.1 in the univariable analysis were considered for the multivariable analysis. Odds ratios (ORs) and their corresponding 95% confidence intervals (CIs) were calculated using logistic regression analysis.
All statistical analyses were two-sided, and a p < 0.05 was considered statistically significant. The SPSS 21 software was used for the statistical analysis (IBM®, Chicago, IL, USA).
Results
Patients
We retrospectively reviewed 261 consecutive patients with esophagogastric or pancreaticobiliary cancer who received FCM or DA for CIA treatment between 1 June 2018 and 31 December 2022. A total of 214 patients (124 in the FCM and 90 in the DA group) were included in this analysis (Figure 1).
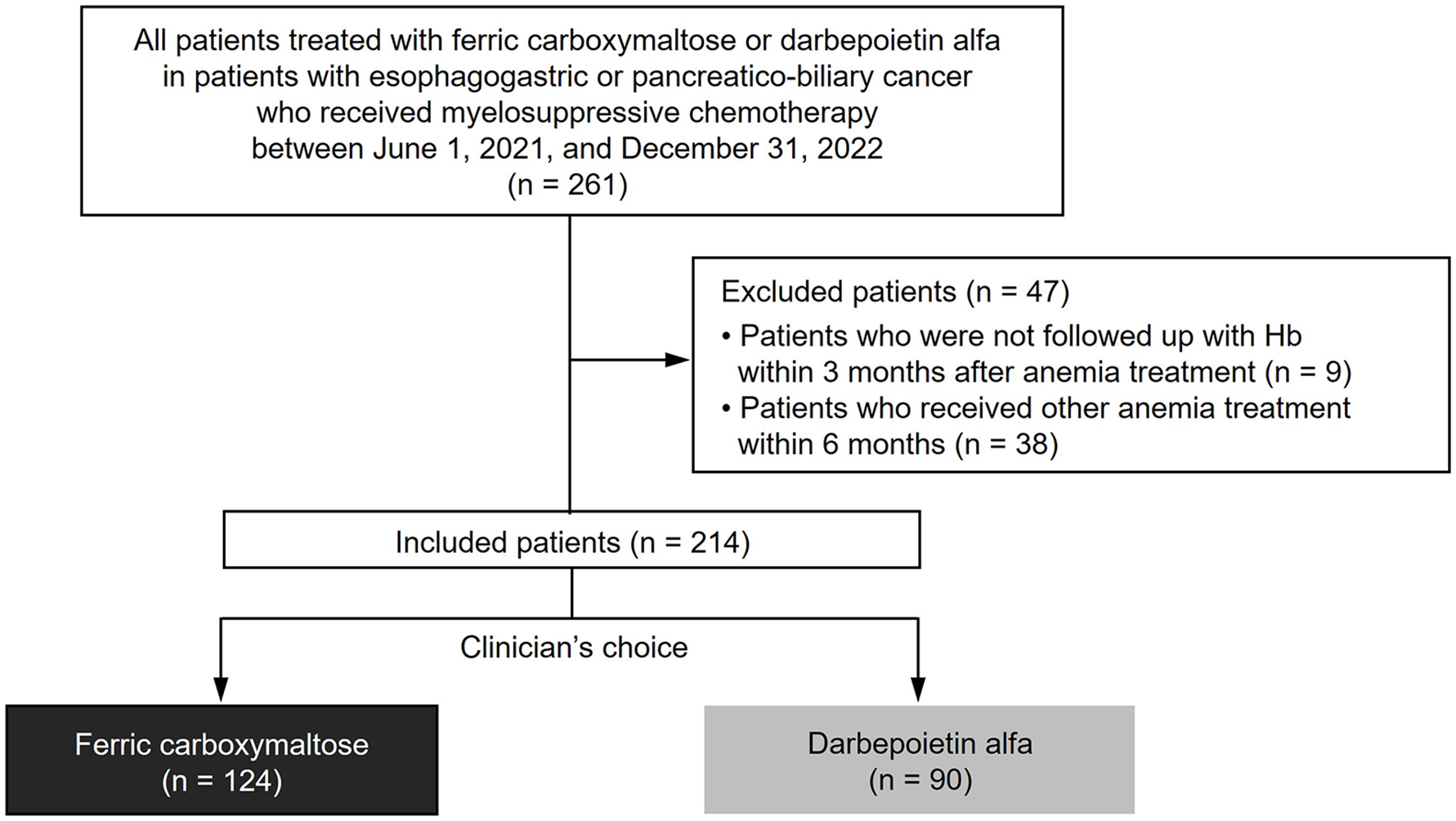
Study population and patient identification.
Baseline patient and tumor characteristics are presented in Table 1. No significant differences were observed in age, sex, ECOG performance status, or chemotherapy regimen of cytotoxic agents between the FCM and DA groups. In the FCM group, significantly more patients received chemotherapy for esophagogastric cancer (66.1% versus 45.6%, p = 0.003), were at stage I–III (53.2% versus 13.3%, p < 0.0001), and had neoadjuvant/adjuvant intent (52.4% versus 13.3%, p < 0.0001) and a less intensive regimen (single-agent 36.6% versus 18.9%, triplet 16.1% versus 28.9%, p = 0.008) compared to those in the DA group. The baseline Hb levels (mean ± SD) exhibited no difference before CIA treatment between the FCM and DA groups (9.3 ± 1.2 versus 9.4 ± 0.9 g/dL, p = 0.453).
Patient characteristics.

Chemotherapy regimens composed of a single cytotoxic agent included 5-fluorouracil, capecitabine, S-1, paclitaxel, irinotecan, and gemcitabine.
Chemotherapy regimens composed of double cytotoxic agents included XELOX (capecitabine and oxaliplatin), FOLFOX (5-fluorouracil, oxaliplatin, and leucovorin), XP (capecitabine and cisplatin), FP (5-fluorouracil and cisplatin), DP (docetaxel and cisplatin), IP (irinotecan and cisplatin), FOLFIRI (5-fluorouracil, irinotecan, and leucovorin), nal-IRI/5-FU/LV (nanoliposomal irinotecan, 5-fluorouracil, and leucovorin), GA (gemcitabine and nab-paclitaxel), GP (gemcitabine and cisplatin), and GX (gemcitabine and capecitabine).
Chemotherapy regimens composed of triplet cytotoxic agents included FOLFIRINOX (5-fluorouracil, oxaliplatin, irinotecan, and leucovorin), AGP (nab-paclitaxel, gemcitabine, and cisplatin), OIS (oxaliplatin, irinotecan, and S-1), and PIE (etoposide, ifosfamide, and cisplatin).
DA, darbepoetin alfa; ECOG, Eastern Cooperative Oncology Group performance status; FCM, ferric carboxymaltose; Hb, hemoglobin; SD, standard deviation.
Efficacy
The Hb response in the CIA treatment group for 3 months after FCM or DA treatment is shown in Figure 2. The maximum Hb level (mean ± SD) was higher in the FCM group than in the DA group [11.3 ± 1.5 versus 10.9 ± 1.2 g/dL, p = 0.02, Figure 2(a)]. Maximum Hb changes from baseline (mean ± SD) were also higher in the FCM group than in the DA group [2.0 ± 1.4 versus 1.5 ± 1.1 g/dL, p = 0.004, Figure 2(b)]. When Hb response was defined as a Hb increase of ⩾1.0 g/dL or Hb of ⩾11.0 g/dL, the proportion of Hb responders was significantly higher in the FCM group than that in the DA group [83.9% versus 68.9%, p = 0.013, Figure 2(c)]. Additionally, when Hb response was defined as a Hb increase of ⩾2.0 g/dL or Hb of ⩾12.0 g/dL, the proportion of Hb responders was also significantly higher in the FCM group than that in the DA group (50.0% versus 35.6%, p = 0.036). In the subgroup analysis, no significant difference was observed in the maximum Hb level, maximum Hb changes from baseline for 3 months, and proportion of Hb responders according to age, sex, ECOG performance status, primary tumor site, stage, intent of chemotherapy, or chemotherapy regimen of cytotoxic agents (Supplemental Table 1, Table 2). In the subgroup analysis stratified by tumor type and chemotherapy regimen of cytotoxic agents, no significant difference was observed in the maximum Hb level, and maximum Hb changes from baseline for 3 months (Supplemental Table 2).

Hb responses by treatment group. (a) Maximum Hb level. (b) Maximum Hb change. (C) Hb responder.
Univariable and multivariable analyses of hemoglobin response.
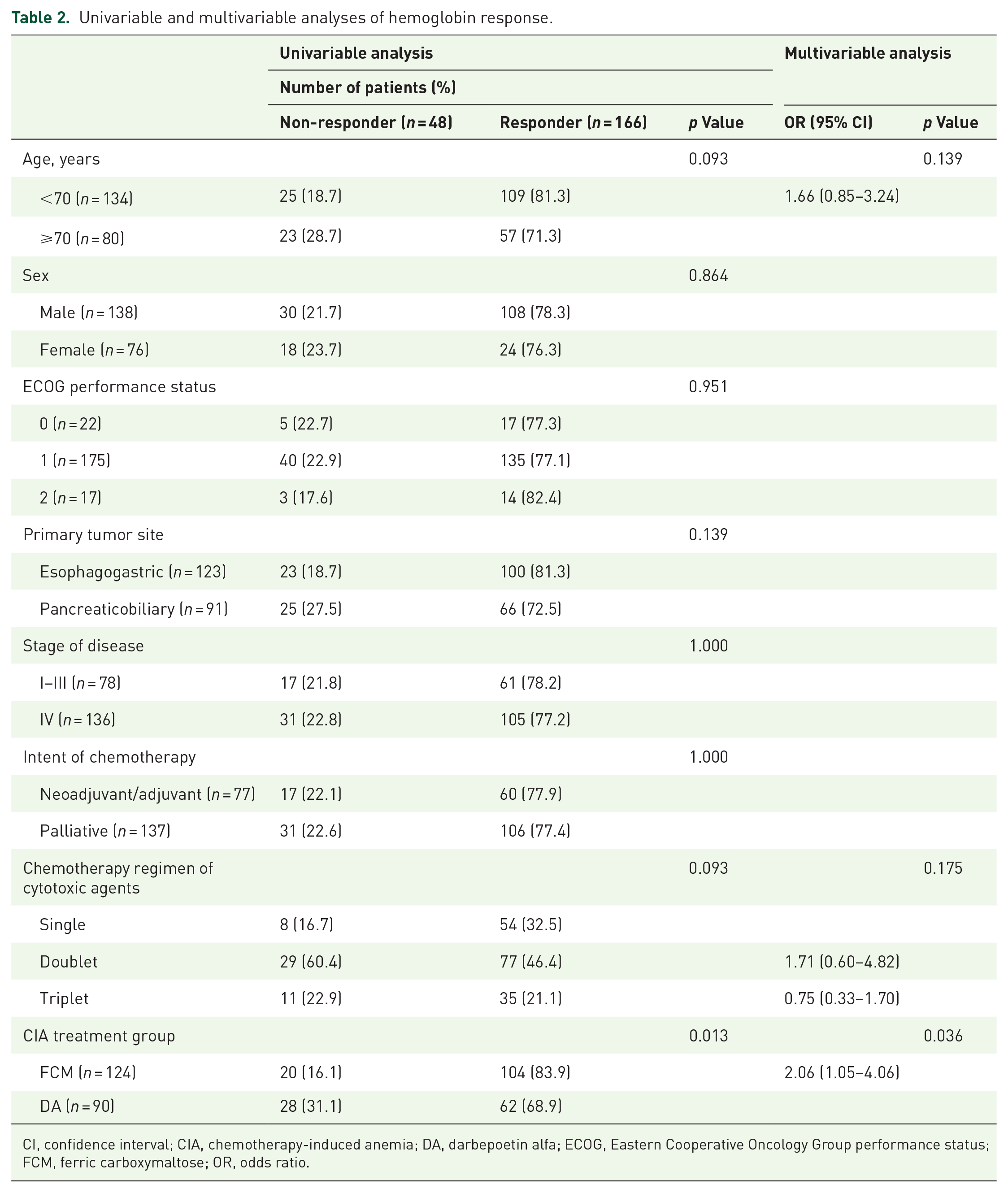
CI, confidence interval; CIA, chemotherapy-induced anemia; DA, darbepoetin alfa; ECOG, Eastern Cooperative Oncology Group performance status; FCM, ferric carboxymaltose; OR, odds ratio.
After FCM or DA administration, Hb levels increased steadily from baseline until 6 months in both the treatment groups. Hb levels at 1, 2, 3, 4, 5, and 6 months from baseline were significantly higher in the FCM group than those in the DA group (all p < 0.05) (Figure 3).

Hb changes over time by treatment group.
No significant differences were observed between the FCM and DA groups in the percentage of patients who received PRC transfusions (15.3% versus 18.9%, p = 0.579) or median time to transfusion (515.8 versus 565.9 days, p = 0.545).
Univariable and multivariable analyses for Hb response
Age (<70 versus ⩾70 years), sex (male versus female), ECOG performance status (0 versus 1 versus 2), primary tumor site (esophagogastric versus pancreaticobiliary), stage (I–III versus IV), intent of chemotherapy (neoadjuvant/adjuvant versus palliative), chemotherapy regimen of cytotoxic agents (single versus doublet versus triplet), and CIA treatment group (FCM versus DA) were compared between the Hb non-responders and responders (Table 2).
Patients under 70 years of age and those treated with less intensive single cytotoxic agents included higher Hb responders than those aged 70 years or older and those treated with more intensive chemotherapy regimens, with marginal statistical significance (all p = 0.093). Patients who received FCM as a CIA treatment had a significantly higher proportion of Hb responders compared with those who received DA (83.9% versus 68.9%, p = 0.013). Multiple logistic regression analysis indicated that the CIA treatment group was the only significant factor for Hb response (OR = 2.06, 95% CI: 1.05–4.06, p = 0.036).
Discussion
Although this study is a retrospective comparative study and provides preliminary data from a single institution, it is the first to present the efficacy of CIA treatment in patients with esophagogastric or pancreaticobiliary cancer. Both FCM and DA were effective for CIA in these patients; however, FCM showed a higher Hb response than DA, and this response was sustained for 6 months.
Most previous studies on CIA treatment included patients with both hematologic and solid malignancies or cancer patients with all types of solid malignancies.1–3 Gastrointestinal cancer, a tumor type with a high risk for absolute or relative iron deficiency due to tumor bleeding, nutritional deficiencies, and poor intestinal absorption, is particularly vulnerable. While there have been studies on perioperative treatment of anemia or CIA in colorectal cancer,21–23 no studies have focused on patients with upper gastrointestinal, pancreatic, or biliary tract cancer. Patients with esophagogastric or pancreaticobiliary tract cancer who receive chemotherapy often exhibit more pronounced symptoms, such as poor oral intake, nausea, vomiting, and abdominal pain, and a higher degree of inflammation due to aggressive features than those with colorectal cancer. Therefore, the present study, which analyzed the efficacy of CIA treatment in patients with esophagogastric and pancreaticobiliary tract cancers, is meaningful.
Current treatment guidelines for CIA do not recommend using ESA in patients receiving chemotherapy with curative intent due to concerns about shortened survival.1–3 Therefore, patients undergoing palliative chemotherapy may be candidates for ESA therapy. Within our study population, the FCM group exhibited a significantly higher proportion of patients with esophagogastric cancer, stage I–III cancer, and those receiving neoadjuvant/adjuvant chemotherapy and a less intensive regimen than the DA group. The aggressive nature of pancreaticobiliary cancer was reflected by a high proportion of stage IV patients receiving palliative chemotherapy with more intensive regimens, whereas patients with esophagogastric cancer had a high proportion of stage II–III patients receiving neoadjuvant or adjuvant chemotherapy with less intensive regimens. Consequently, notable differences were observed in baseline characteristics between the FCM and DA groups. These differences in baseline characteristics may have influenced the difference in CIA treatment efficacy between the two groups. However, no difference was observed in baseline Hb levels between the two groups, and to minimize the influence of underlying diseases while assessing the efficacy of CIA treatment, we analyzed Hb levels over a relatively short period of 12 weeks. Additionally, in our subgroup analysis, no significant difference was observed in Hb response according to primary tumor site (esophagogastric versus pancreaticobiliary), stage (I–III versus IV), intent of chemotherapy (neoadjuvant/adjuvant versus palliative), or cytotoxic chemotherapy regimen (single versus doublet versus triplet) (Table 2, Supplemental Tables 1 and 2).
Cancer-associated thromboembolism is highly consequential for patients with cancer and is associated with worsened survival. 24 The risk of cancer-associated thromboembolism is largely influenced by the underlying type of cancer, with the highest thrombotic risk observed in pancreatic and gastric cancers.25–27 The Khorana score for predicting cancer-associated thromboembolism defined pancreatic and gastric cancers as having very high-risk scores. 27 Moreover, platinum-based chemotherapies, which are the most important anticancer drugs for esophagogastric and pancreaticobiliary cancer, have also been associated with cancer-associated thromboembolism. 28 Therefore, it is significant that in our study, FCM is more effective than DA for thrombotic adverse events in patients with esophagogastric or pancreaticobiliary cancer.
Supplementing ESA therapy with IV iron provides the necessary iron support for ESA-driven erythropoiesis. Studies have shown that concurrent IV iron supplementation improves not only the Hb response and reduces the need for transfusions, but also the quality of life and facilitates reductions of ESA dose, all without the additional risk of thromboembolism beyond that associated with ESAs alone.17,18,29 However, recent several studies have assessed the efficacy of IV iron as a monotherapy for CIA; these studies have consistently demonstrated increases in Hb levels and/or notable reductions in transfusion rates.19,20,30–33 One example is the IRON-CLAD study, which showed that IV iron supplementation, specifically in the form of FCM monotherapy, allowed most patients receiving chemotherapy to maintain Hb levels within 0.5 g/dL of baseline, and was significantly more effective in doing so when compared with a placebo. 30 The PROFOUND trial, demonstrated that iron isomaltoside and oral iron produced comparable sustained increases in Hb concentration. 31 Iron isomaltoside was better tolerated than oral iron and significantly reduced fatigue.
Several IV iron formulations are available on the market, with recommended options including low-molecular-weight iron dextran, ferric gluconate, iron sucrose, FCM, and ferumoxytol. FCM (Ferinject™; Vifor Pharma, Glattbrugg, Switzerland) is a stable colloidal solution of nanoparticles, consisting of a polynuclear iron (III)-(oxyhydr)oxide core stabilized by carboxymaltose. This formulation allows for slow and prolonged release of iron and is administered as a single high-dose infusion of 1000 mg of iron over 15 min. 34 Based on extensive experience in clinical trials and real-world settings, FCM is an effective and well-tolerated treatment for rapidly replenishing iron stores and correcting anemia in patients with IDA of various etiologies. 34 FCM was proven effective in patients with active malignancies and IDA (n = 420) and in those with hematological malignancies or solid tumors and anemia (n = 367) in two real-world, non-interventional studies conducted in Germany and France.32,33 Additionally, a recent prospective study conducted in South Korea reported a significant increase in Hb levels following treatment with FCM in patients with solid cancers receiving chemotherapy. 19
Recently, there has been active research into predictive biomarkers for CIA treatment with IV iron supplements. The NCCN and ESMO guidelines define iron store status using ferritin and transferrin saturation levels.1,2 However, CIA closely resembles anemia of chronic disease, with patients frequently exhibiting elevated serum ferritin levels due to chronic inflammation, even in iron-deficient status.10,11 Consequently, a new biomarker is needed to identify patients who require iron supplementation. Several studies have suggested that low serum hepcidin levels may be helpful in identifying patients with iron deficiency who are more likely to benefit from IV iron supplementation.19,20,30
This study had some limitations. First, it was a retrospective analysis from a single institution, and the results cannot be generalized. Also, baseline characteristics differed between the FCM and DA groups. These differences in baseline characteristics may have influenced the differences in CIA treatment efficacy between two groups. However, no difference in Hb response was observed according to the primary tumor site, stage, intent of chemotherapy, and chemotherapy regimen of cytotoxic agents over a relatively short period of 3 months. Second, since our study analyzed patients treated in real practice; anemia-related parameters such as iron levels, total iron binding capacity, vitamin B12, folate, and adverse events of FCM or DA, were not systematically measured. Finally, this study did not include patients treated with IV iron supplements in conjunction with ESA therapy. There is insufficient data to routinely recommend IV iron monotherapy without ESA for treating functional IDA. The NCCN and ESMO guidelines suggest that IV iron supplementation combined with ESA therapy should be considered in patients with functional IDA.1,2 Since our study was based on data from real clinical practice, very few patients were treated with both IV iron and ESA, and most were administered sequentially in our institution. However, recent studies have reported that some patients with functional IDA or high ferritin levels (even if ferritin ⩾500 ng/mL) responded to IV iron therapy alone.19,20,30–33 Our study showed that the mean Hb level increased from a baseline of 9.3 g/dL to a maximum of 11.3 g/dL after FCM monotherapy and did not require additional CIA treatment. Additionally, for cancers with a high risk of cancer-associated thromboembolism, using only IV iron without ESA, which poses a thrombotic risk, could be more beneficial. In the future, a prospective comparative study of IV iron alone and IV iron with ESA therapy is needed in cancer patients with functional or non-functional IDA who are receiving palliative chemotherapy.
Conclusion
Our preliminary results suggest that IV iron monotherapy with FCM can improve the Hb response in patients with esophagogastric or pancreaticobiliary cancer undergoing myelosuppressive chemotherapy compared with ESA with DA. Further prospective trials are warranted to demonstrate the efficacy and safety of IV iron supplementation in these patients with well-defined accrual criteria at high risk of CIA.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241265209 – Supplemental material for Efficacy of ferric carboxymaltose or darbepoetin alfa for chemotherapy-induced anemia in patients with esophagogastric or pancreaticobiliary cancer: a retrospective comparative study
Supplemental material, sj-docx-1-tam-10.1177_17588359241265209 for Efficacy of ferric carboxymaltose or darbepoetin alfa for chemotherapy-induced anemia in patients with esophagogastric or pancreaticobiliary cancer: a retrospective comparative study by Minkwan Cho, Eunkyung Park, Yong-Pyo Lee, Hongsik Kim, Hee Sue Park, Hee Kyung Kim, Yaewon Yang, Jihyun Kwon, Ki Hyeong Lee and Hye Sook Han in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
