Abstract
Background:
Primary tumor resection and metastasectomy may be beneficial for many patients with metastatic colorectal cancer (mCRC).
Objective:
To assess the differences in postoperative survival outcomes between adjuvant therapy with chemotherapy alone and chemotherapy plus targeted agents (TAs).
Design:
Retrospective cohort study.
Methods:
Patients with mCRC who underwent surgical resection for primary colorectal tumor and distant metastases and received adjuvant therapy from 1 January 2010 to 31 December 2017 were enrolled in the Taiwan Cancer Registry. We analyzed the overall survival of patients with resectable or initially unresectable mCRC who received adjuvant chemotherapy alone and chemotherapy plus TAs.
Results:
We enrolled 1124 and 542 patients with resectable and initially unresectable mCRC, respectively. Adjuvant chemotherapy plus TAs and chemotherapy alone resulted in similar mortality rates among patients with resectable mCRC [adjusted hazard ratio (aHR) = 1.13; 95% confidence interval (CI), 0.93–1.36]; however, it marginally reduced the mortality rate among patients with initially unresectable mCRC who underwent conversion surgery after neoadjuvant therapy (aHR = 0.81; 95% CI, 0.62–1.06). The subgroup analysis of patients who received more than nine cycles of TAs preoperatively and anti-epidermal growth factor receptor agents revealed aHRs of 0.48 (95% CI, 0.27–0.87) and 0.33 (95% CI, 0.18–0.60), respectively.
Conclusion:
Adjuvant chemotherapy plus TAs may improve survival in patients with initially unresectable tumors who underwent conversion surgery following neoadjuvant therapy with TAs, especially in those who respond well to the targeted therapy. Our study underscores the importance of stratifying patients with mCRC based on tumor resectability when selecting the adjuvant therapy regimen.
Introduction
Colorectal cancer (CRC) is the leading cause of cancer-related mortality worldwide. 1 Approximately 20–25% of patients with CRC develop metastasis during the initial diagnosis, with the liver and lung being the most common sites. 2 In addition, 10–30% of patients with metastatic colorectal cancer (mCRC) have primary colorectal tumors and distant metastases3,4 that can be managed either with staged or simultaneous surgical resection. Conversion surgery can be performed safely in patients with initially unresectable mCRC but who respond to systemic therapy, referred to as neoadjuvant therapy.5,6 The 5-year survival rate of patients who undergo surgery is 35–50%,7,8 but the disease relapse rate is 75%. 9
Patients receiving adjuvant chemotherapy (CT) showed improved survival after complete tumor resection.10,11 The National Comprehensive Cancer Network® (NCCN) guidelines (version 3, 2021) suggest that patients with resectable mCRC should receive neoadjuvant and adjuvant CT, and patients with unresectable mCRC should receive neoadjuvant CT plus a targeted agent (TA). If these patients respond well to TAs, the unresectable tumors at the primary site and the distant metastases can be converted to resectable tumors, and TAs would need to be added to subsequent adjuvant CT after complete surgical resection. However, the guidelines do not indicate the clinical conditions or provide associated evidence to support such recommendations. Conversely, the EPOC trial [the European Organization for Research and Treatment of Cancer (EORTC) 40983] reported that neoadjuvant and adjuvant therapy with cetuximab plus CT can result in a significantly shorter median overall survival (OS) in patients with resectable mCRC than adjuvant CT alone (55.4 versus 81 months). 12 Therefore, the effectiveness of TAs added to CT as adjuvant therapy is yet to be determined.
Although adjuvant CT is frequently indicated in clinical practice for patients who are initially eligible for both primary tumor resection (PTR) and metastasectomy or are undergoing conversion surgery after neoadjuvant therapy, the need and options for TAs in adjuvant therapy should be further explored. In Taiwan’s National Health Insurance (NHI), CT combined with TAs as first-line systemic therapy for patients with mCRC is reimbursed, regardless of whether they have undergone surgical resection. 13 Herein, we aimed to assess the effectiveness of adjuvant therapy with CT alone or CT plus TAs on the survival outcomes of patients with mCRC who underwent PTR and metastasectomy between a resectable group and an initially unresectable group using data from the Taiwan Cancer Registry (TCR) and National Health Insurance Database (NHID).
Materials and methods
Data source
The NHID is a claims database derived from the NHI, which is a single-payer insurance program that covers more than 99.99% of the entire Taiwanese population (Supplemental Data 1).14,15 Complete records of the prescriptions of TAs, including bevacizumab, cetuximab, and panitumumab, and CT, such as fluorouracil, irinotecan, and oxaliplatin, and associated surgical status were extracted from the database.
The TCR database, managed by the Ministry of Health and Welfare, Taiwan, has an excellent coverage rate (97%). The quality of data emanating from the cancer registry is deemed excellent, including a self-check procedure using standardized logic algorithms at the TCR central office to identify and correct potential errors before data submission (Supplemental Data 1). 16 The causes of death were evaluated to obtain mortality data and traced until 31 December 2019. The data were anonymized.
This study adhered to the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of Kaohsiung Veterans General Hospital (KSVGH21-CT2-03) waived the requirement for informed consent because we used a consistent encryption procedure to deidentify the original identification number of each patient in the NHIRD. This study followed the STrengthening the Reporting of OBservational studies in Epidemiology reporting guideline 17 (Supplemental Data 9).
Study population
We conducted a retrospective, cohort study of newly diagnosed patients with mCRC according to the International Classification of Diseases for Oncology, third edition, codes: C180–C189, C199, and C209. Patients who underwent surgical resections of both the primary colorectal tumor and synchronous distant metastases and received adjuvant therapy from 1 January 2010 to 31 December 2017 were identified from the TCR database. Furthermore, we categorized them into groups according to whether their mCRC was resectable or initially unresectable as per the NCCN guidelines. 6 The resectable group was defined as patients who did not receive neoadjuvant therapy or received only CT before surgical resection. The initially unresectable group referred to patients with mCRC who were initially considered unresectable but became eligible for surgical resection after receiving neoadjuvant therapy with TAs. The index date was defined as the date on which the patient received the first cycle of adjuvant therapy after PTR and metastasectomy. We enrolled patients who received at least six cycles of adjuvant therapy with intervals between consecutive cycles shorter than 60 days to compare the effectiveness of adjuvant therapy between CT plus TA and CT alone. The criteria were based on biweekly adjuvant CT for at least six cycles within approximately 3 months. 18 However, in a real-life setting, patients may not be able to regularly receive CT every 2 weeks; therefore, we designated consecutive cycles as being shorter than 60 days. The drugs for treating mCRC were covered by the Taiwan NHI, and the corresponding Anatomical Therapeutic Chemical Classification System (ATC) codes are provided in Supplemental Data 2. Patients were excluded if they had (1) not undergone PTR and metastasectomy, (2) not received systemic therapy after diagnosis, (3) synchronous left- and right-sided CRC, (4) undergone any systemic CT within 1 year before the diagnosis date, and (5) received adjuvant therapy with CT plus TA or CT alone for less than six cycles, with intervals between consecutive treatment cycles longer than 60 days.
Study variables and outcomes
The following demographic variables were examined: year of systemic therapy postoperatively, age, sex, histological grade, primary tumor location, stage (4A and 4B), tumor size, carcinoembryonic antigen (CEA) status, Kirsten rat sarcoma virus (KRAS) status, bowel obstruction, bowel perforation, lymph node status, radiotherapy, the interval between PTR and metastasectomy (the corresponding surgical procedure codes are listed in Supplemental Data 3), metastasectomy type, the interval between resuming adjuvant therapy and surgery, TA type, CT type, Charlson comorbidity index (CCI) score [corresponding International Classification of Diseases, ninth revision (ICD-9) codes],19,20 ICD-10 data (Supplemental Data 4.1), and co-medication 1 year before the operation (the corresponding ATC codes are provided in Supplemental Data 4.2). Regarding the cycles of neoadjuvant therapy, KRAS status, and different TA types, we further analyzed the subgroups as >9 and ⩽9 cycles preoperatively, KRAS wild or mutation type, and TA type with cetuximab/panitumumab and bevacizumab, respectively. The primary outcome was the OS, calculated from the index date to the end of 2019, death, or censorship, of the patients with resectable or unresectable mCRC after PTR and metastasectomy on adjuvant CT alone or CT plus TA.
Statistical analyses
Descriptive statistics were calculated for the demographic and tumor characteristics. A standardized mean difference exceeding 0.2 was used to evaluate differences in baseline covariates between adjuvant CT alone and adjuvant CT plus TA in the resectable and unresectable mCRC groups. In addition, OS was calculated and compared using the Kaplan–Meier method and log-rank test for unadjusted survival differences between adjuvant CT alone and adjuvant CT plus TA in the resectable and unresectable mCRC groups. However, the adjusted survival for comparing adjuvant CT alone and adjuvant CT plus TA was estimated using multivariable analysis by fitting a Cox proportional hazards model. The results were expressed as hazard ratios (HRs) and their corresponding 95% confidence intervals (CIs). All analyses were performed using SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA). For all tested hypotheses, analyzed items with a two-tailed p < 0.05 were considered statistically significant. Missing values were imputed by multivariate imputation using chained equations. The method is based on a fully conditional specification, where each incomplete variable is imputed by a Bayesian model, 21 including histological grade, tumor size, positive lymph node number, CEA status, KRAS status, bowel obstruction, and bowel perforation.
Sensitivity analyses
Two sensitivity analyses were performed to examine the robustness of our findings. In the first sensitivity analysis, we included the covariates in a logistic regression model to generate a propensity score (PS) for the probability of patients receiving treatment. We used a Cox proportional hazards model that was adjusted for the PS and baseline characteristics to compare the survival differences between the two groups. We generated the comparison group for adjuvant CT plus TA using one-to-one PS matching. Furthermore, we estimated the OS after PS matching to control for confounding factors and ensure comparativeness between the adjuvant CT alone and adjuvant CT plus TA groups. Potential confounders and covariates that might be associated with the outcome, including medications, comorbidities, and tumor patterns, were included in the PS model (Supplemental Data 5). In the second analysis, we used the E-value method to assess the unmeasured confounding with KRAS mutational status that was highly associated with different biological agents and prognosis 22 since information on KRAS status was not available in our database.
Results
Cohort characteristics
We identified 24,180 patients with mCRC (Figure 1), among whom 1666 received adjuvant therapy after synchronous or staged PTR and metastasectomy. Among the 1124 patients with resectable mCRC, 326 and 798 received at least six cycles of adjuvant therapy with CT alone or CT plus TA, respectively. Among them, 813 did not receive neoadjuvant therapy, while 311 did. Among the 542 patients with initially unresectable mCRC, 339 and 203 received CT alone or CT plus TA, respectively, as adjuvant therapy. The patient characteristics are presented in Table 1; the CT regimen is provided in Supplemental Data 6 and 7.

Flow chart of cohort selection.
Baseline characteristics of patients with mCRC who received chemotherapy alone or targeted therapy plus chemotherapy after surgery.
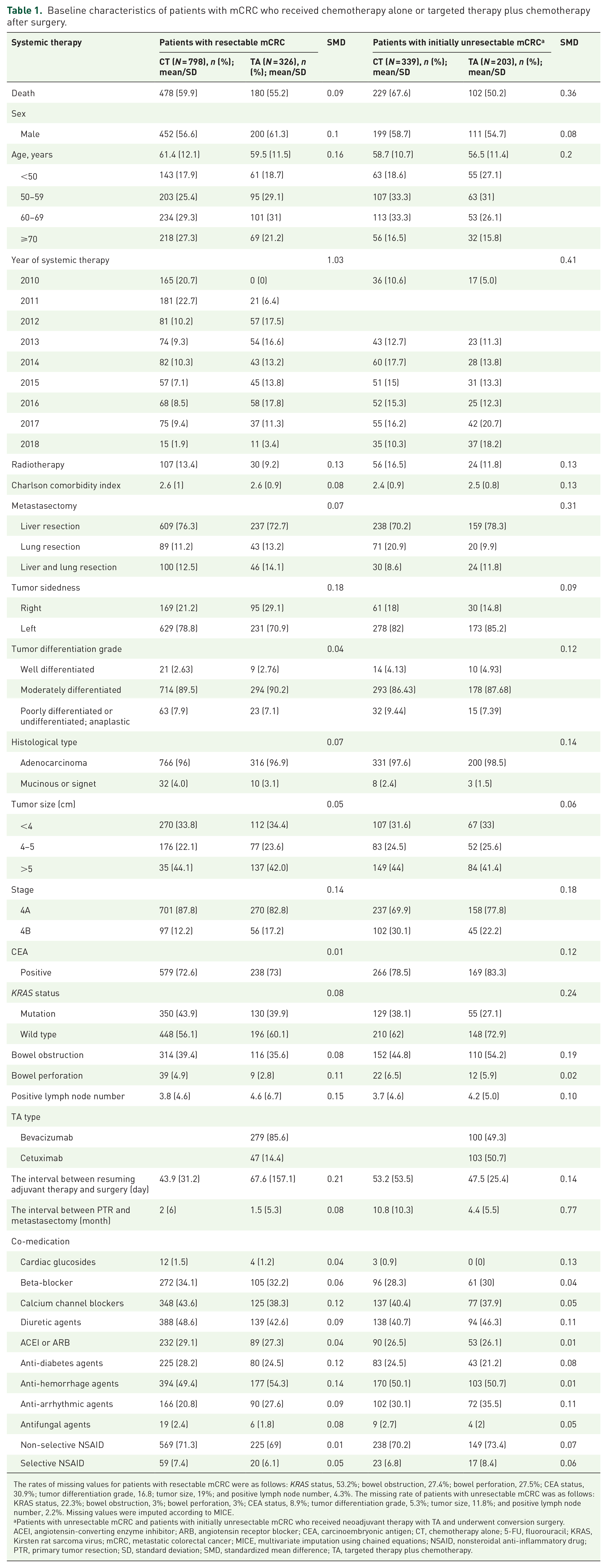
The rates of missing values for patients with resectable mCRC were as follows: KRAS status, 53.2%; bowel obstruction, 27.4%; bowel perforation, 27.5%; CEA status, 30.9%; tumor differentiation grade, 16.8; tumor size, 19%; and positive lymph node number, 4.3%. The missing rate of patients with unresectable mCRC was as follows: KRAS status, 22.3%; bowel obstruction, 3%; bowel perforation, 3%; CEA status, 8.9%; tumor differentiation grade, 5.3%; tumor size, 11.8%; and positive lymph node number, 2.2%. Missing values were imputed according to MICE.
Patients with unresectable mCRC and patients with initially unresectable mCRC who received neoadjuvant therapy with TA and underwent conversion surgery.
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CEA, carcinoembryonic antigen; CT, chemotherapy alone; 5-FU, fluorouracil; KRAS, Kirsten rat sarcoma virus; mCRC, metastatic colorectal cancer; MICE, multivariate imputation using chained equations; NSAID, nonsteroidal anti-inflammatory drug; PTR, primary tumor resection; SD, standard deviation; SMD, standardized mean difference; TA, targeted therapy plus chemotherapy.
Overall survival
A total of 658 (58.5%) patients with resectable mCRC died during follow-up: 478/798 (59.9%) and 180/326 (55.2%) in the CT alone and CT plus TA groups, respectively. The median OS was not significantly different between the CT alone and CT plus TA groups (47.74 versus 44.4 months), with crude and adjusted HRs of 1.03 (95% CI, 0.87–1.22) and 1.13 (95% CI, 0.93–1.36), respectively. Regardless of whether they received neoadjuvant therapy or not, there was no difference in survival between adjuvant CT alone and adjuvant CT plus TA (no neoadjuvant therapy: adjusted HR = 1.2, 95% CI, 0.96–1.49; neoadjuvant CT: adjusted HR = 0.82, 95% CI, 0.54–1.24). Among the patients with initially unresectable mCRC, 331 (61.1%) died during follow-up: 229/339 (67.6%) and 102/203 (50.2%) in the CT alone and CT plus TA groups, respectively. The median OS was marginally different between the CT alone and CT plus TA groups (28.83 versus 35.83 months), with crude and adjusted HRs of 0.67 (95% CI, 0.53–0.85) and 0.81 (95% CI, 0.62–1.06), respectively (Figures 2 and 3).
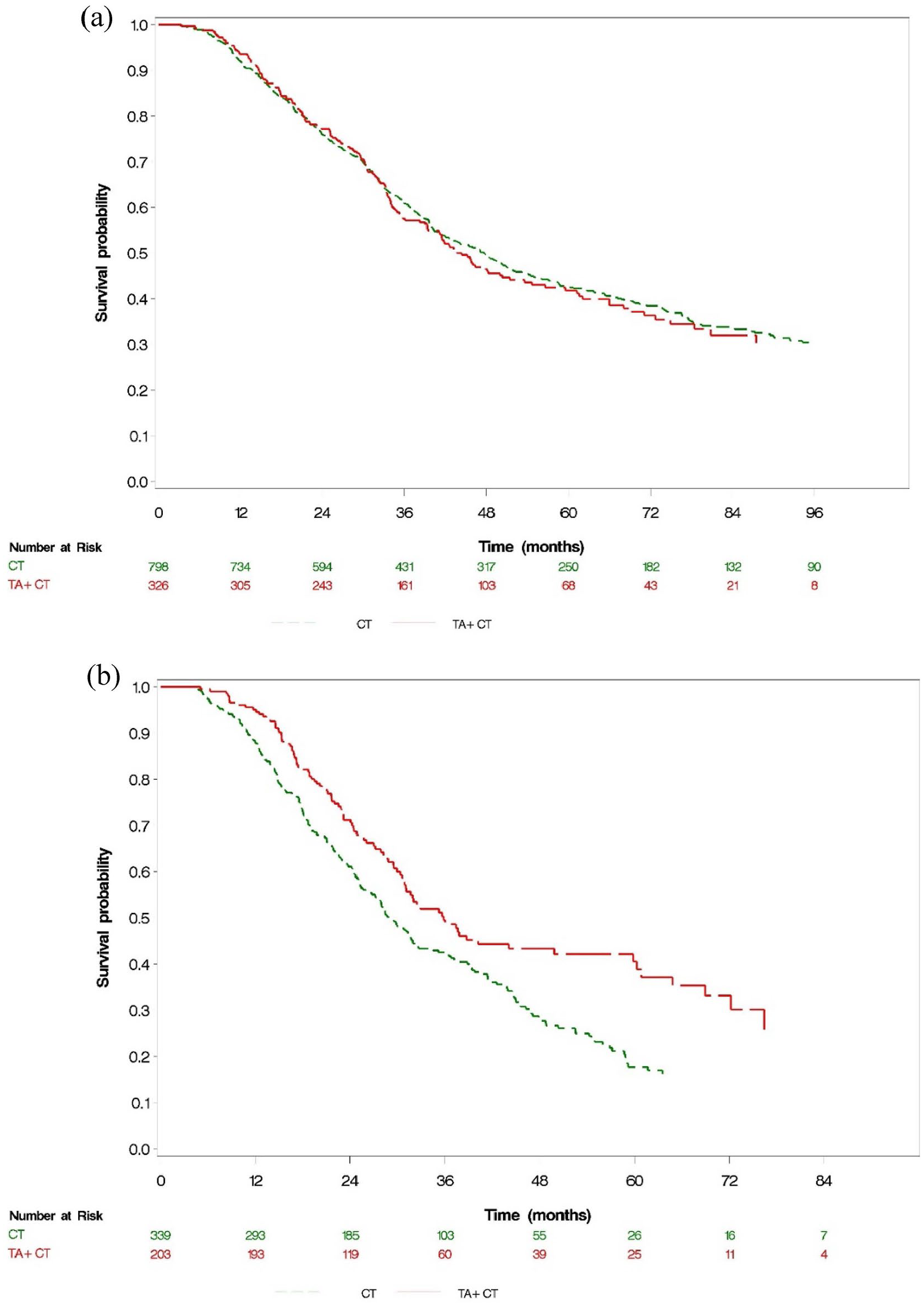
Kaplan–Meier survival curves for OS in (a) patients with resectable mCRC and (b) patients with initially unresectable mCRC who received neoadjuvant therapy with TAs and subsequently underwent conversion surgery.

HRs of overall survival with CT alone versus CT + TA in patients with resectable mCRC and patients with initially unresectable mCRC who received neoadjuvant therapy with TAs and subsequently underwent conversion surgery.
In the initially unresectable mCRC group, for patients who received ⩽9 cycles of TA preoperatively, the crude and adjusted HRs were 0.75 (95% CI, 0.55–1.01) and 0.99 (95% CI, 0.70–1.39), respectively, and for patients who received more than nine cycles of TA preoperatively, the crude and adjusted HRs were 0.58 (95% CI, 0.35–0.98) and 0.48 (95% CI, 0.27–0.87), respectively, in the CT plus TA group. The adjusted HRs were 0.33 (95% CI, 0.18–0.60) and 0.87 (95% CI, 0.62–1.23) for patients who received anti-epidermal growth factor receptor (EGFR) agents and anti-vascular endothelial growth factor (VEGF) agents (KRAS-wild type: 55%), respectively, in the CT plus TA group compared to those in the CT alone group. The adjusted HR was 0.36 (95% CI, 0.19–0.69) for patients who had KRAS wild-type and received anti-EGFR agents.
Sensitivity analyses
Regarding OS, PS adjustment and PS matching between both groups showed HRs of 0.99 (95% CI, 0.82–1.19) and 0.93 (95% CI, 0.75–1.16) in patients with resectable mCRC, respectively (Figure 3). In patients with mCRC who underwent successful conversion surgery, the OS after PS adjustment and PS matching between both groups showed HRs of 0.80 (95% CI, 0.61–1.06) and 0.87 (95% CI, 0.64–1.20) (Figure 3) (Supplemental Data 8). This finding was robust since the results were consistent between the PS-matched and PS-adjusted analyses. The E-values for the estimated HR and upper confidence bound were 3.55 and 1.56, respectively, associated with an unmeasured confounder with the KRAS profile between systemic therapy and OS. Notably, the observed HR of 0.48 could be explained by an unmeasured confounder that was associated with both the treatment and the outcome by a risk ratio of 3.55-fold each beyond the measured confounders; however, it could not be explained by a weaker confounder. The calculation was based on the HR obtained for neoadjuvant therapy with more than nine cycles of TAs in patients who underwent conversion surgery in this study. In a previous study, 23 the OS of the KRAS-wild type compared to that of the KRAS-mutant type was 1.65 (0.96–2.86).
Discussion
Our study is the first to investigate patients with initially unresectable mCRC who, after undergoing neoadjuvant therapy with targeted therapy, transitioned from being initially ineligible for surgery to becoming eligible for resection. Subsequently, after surgery, we compared the efficacy of adjuvant therapy with CT plus TA to that of CT alone. Our study demonstrated that, among patients with initially unresectable mCRC who became eligible for conversion surgery after neoadjuvant therapy, those treated with adjuvant CT plus TA showed a trend toward longer survival than those receiving adjuvant CT alone. This implication remained robust during our sensitivity analyses based on adjusted and matched PS.
The pharmaceutical reimbursement scheme of the NHI covers the 10th–18th cycle of TAs if the patient responded well to the initial nine cycles of systemic therapy, at least according to imaging evidence. Therefore, we selected and analyzed patients who had received CT with TAs for more than nine cycles as the subgroup of good responders to neoadjuvant therapy. In this subgroup, patients who received adjuvant CT plus TAs postoperatively had more favorable outcomes than those who received adjuvant CT only. Our findings support the guidelines suggested by the NCCN that if patients with initially unresectable mCRC respond well to TAs and convert to a resectable status, TAs may be considered for subsequent adjuvant CT after complete PTR and metastasectomy. This result emphasizes the significance of selecting an adjuvant regimen for patients with initially unresectable mCRC who could undergo conversion surgery after neoadjuvant therapy.
In our subgroup analysis of patients who received different types of neoadjuvant therapy with TAs, it was evident that the subgroup that received neoadjuvant therapy with cetuximab after surgery had significantly better survival outcomes than those who received CT alone. Based on the current treatment guidelines and Taiwan’s NHI pharmaceutical reimbursement scheme, cetuximab is only prescribed for patients with RAS-wild-type mCRC. 6 Furthermore, our subgroup analysis specifically of patients with KRAS wild-type tumors who received neoadjuvant therapy with cetuximab after surgery showed that they exhibited significantly better survival than those who received CT alone. However, the subgroups of patients who received neoadjuvant therapy with bevacizumab and those with KRAS wild/mutation type with bevacizumab exhibited no survival benefit. This finding suggests that patients with KRAS-wild-type unresectable mCRC should be treated with CT plus cetuximab as adjuvant therapy after successful conversion to complete surgical resection of the primary tumor and metastases since they respond favorably to the same regimen as that used for the initial neoadjuvant therapy. However, this might be associated with the immune contexture of the tumor microenvironment, which can be altered by cytotoxic and TAs, rendering the cancer cells less sensitive to subsequent therapy.24 –26
In addition, we found that the survival benefits between the adjuvant CT plus TA and CT alone groups were comparable among patients with resectable mCRC who underwent PTR and metastasectomy after adjusting for covariates, including sex, age, date of targeted therapy, metastasectomy before the index date, tumor sidedness, histological type, tumor grade, CEA status, KRAS status, the interval between resuming adjuvant therapy and surgery, the interval between PTR and metastasectomy, bowel perforation and obstruction, CCI, co-medication, and staging. Meanwhile, multivariate Cox regression analyses after PS adjustment and PS matching compared the survival outcomes and yielded HRs of 0.99 and 0.93, respectively, indicating a dominant effect of complete surgical resection for patients with resectable mCRC. Therefore, adding TAs in adjuvant therapy might not be necessary. Compared with the findings of Turan et al.,27,28 we revealed a consistent median survival (44–48 versus 53 months) in patients with mCRC who received adjuvant therapy after metastasectomy in a real-world setting. The similar OS durations supported the reproducibility of adjuvant CT plus TAs after PTR and metastasectomy across different countries and clinical practice settings, regardless of the CT backbone. However, our finding was inconsistent with the results of the new EPOC study, in which patients with resectable mCRC who received CT plus cetuximab or CT alone before surgery showed significantly worse survival than those who received CT alone. 12 Notably, only 10% of patients who received cetuximab as subsequent palliative therapy developed recurrence compared with 30% of patients in the CT-alone group, and this could have led to lower OS because of decreased exposure to cetuximab upon recurrence. 29 By contrast, in our study of patients with resectable mCRC, no neoadjuvant-targeted therapy was administered before surgery. Instead, the efficacy of CT plus cetuximab was compared to that of CT alone in the group of patients with resectable mCRC after undergoing surgery. Meanwhile, the results supported the NCCN guidelines, implying that patients with resectable mCRC may require adjuvant CT alone after complete metastasectomy and PTR. Moreover, our analysis demonstrated that patients who underwent conversion surgery had shorter median survival than those with initially resectable mCRC (median survival: 29–36 versus 44–48 months), consistent with the results of previous studies.30,31 In addition, our results indicated varying proportions of anti-VEGF and anti-EGFR usage in the resectable (86%/14%) and unresectable (49%/51%) cohorts, respectively. The primary rationale suggests that when patients are eligible for immediate surgical resection, representing the resectable group, there is no necessity for early tumor shrinkage. Moreover, the resectable group exhibits a relatively higher proportion of KRAS mutations compared to the initially unresectable group, potentially influencing the choice of bevacizumab as a treatment option for the resectable group. Conversely, patients initially deemed unresectable require aggressive systemic targeted therapy plus CT to facilitate early tumor shrinkage. The FIRE-3 study 32 has also indicated that cetuximab tends to be more effective than bevacizumab in terms of early tumor shrinkage efficacy. In addition, considering the relatively lower proportion of KRAS mutations in the initially unresectable group than in the resectable group, a higher proportion of patients in the initially unresectable group may opt for cetuximab than in the resectable group.
The main strengths of this study are the comprehensive enrolment of patients with mCRC from a nationwide claims database; the availability of complete information on comorbidities, treatments, procedures, and medications; and confirmation of diagnosis via linkage to the TCR. Furthermore, the sample size in our study was larger than that in previous studies.
This study had some limitations. First, the disease severity, number of metastatic organs, and extent of metastatic disease were not available for analysis; we employed the covariate of staging to adjust for the confounding effect. Second, the databases had no information on performance status, nutritional status, surgical quality, or hematologic, hepatic, or renal function. Instead, we balanced the differences between the two groups using the following variables: age, tumor pattern, comorbidities, and co-medication. Third, due to constraints within the available dataset, analyzing progression-free survival or disease-free survival post-curative surgery was unfeasible. Despite this limitation, we successfully performed a rigorous analysis of OS, yielding valuable insights into the comprehensive survival outcomes of patients who underwent surgical resection and received adjuvant therapy.
Conclusion
Our findings suggest that adjuvant CT plus TAs may improve the OS of patients who were initially unresectable but who underwent conversion surgery after neoadjuvant therapy with TA, especially in those who responded well to the targeted therapy. Our results underscore the importance of stratifying patients based on the resectability of their mCRC tumor when selecting the regimen of adjuvant therapy. Further investigations are thus needed.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241246427 – Supplemental material for Assessing the effectiveness of targeted agents in adjuvant therapy for patients with metastatic colorectal cancer undergoing surgical resection: a retrospective cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359241246427 for Assessing the effectiveness of targeted agents in adjuvant therapy for patients with metastatic colorectal cancer undergoing surgical resection: a retrospective cohort study by Yi-Chia Su, Chih-Chien Wu, Yu-Hsun Chen, Chien-Chou Su, Yu-Ching Chang, Meng-Che Hsieh and Yea-Huei Kao Yang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors wish to thank the Health Data Science Center, National Cheng Kung University Hospital, for providing administrative and technical support, and Ms. Hsiao-Ling Chiu of the Cancer Center, Kaohsiung Veterans General Hospital, for her advisory comments on interpreting the results.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
