Abstract
Background:
Anlotinib is a novel oral small-molecule receptor tyrosine kinase inhibitor. However, the efficacy and safety of its combined use with chemotherapy remain unclear in patients with advanced ovarian cancer.
Objectives:
To assess the efficacy and safety of the combined use of Anlotinib with chemotherapy in patients with advanced ovarian cancer.
Design:
A multi-center retrospective real-world analysis and a meta-analysis.
Data sources and methods:
We enrolled patients with advanced ovarian cancer who received a combination therapy of Anlotinib and chemotherapy from 15 medical centers. We also searched electronic databases for studies assessing the efficacy and safety of the combined use of Anlotinib with chemotherapy in patients with ovarian cancer. The outcomes of interest included objective response rate (ORR), disease control rate (DCR), and median progression-free survival (mPFS).
Results:
A total of 71 patients, who were predominantly recurrent cases, were included in the real-world study. The ORR and DCR of the included patients were 40.8% and 76.1%, respectively; and their mPFS was 4.6 months. The log-rank test showed that previous antiangiogenic therapy was related to a longer mPFS (p < 0.05). Five studies in total were eligible for meta-analysis. The random-effects meta-analysis model showed that the ORR, DCR, and mPFS were 33.8% [95% confidence interval (CI) 22.7–44.8% from four studies], 90.6% (95% CI 73.6–99.9% from five studies), and 6.6 months (95% CI 4.9–8.4 months from five studies). The most common adverse events were hand-foot syndrome and hypertension.
Conclusion:
The combined use of Anlotinib with chemotherapy showed potential in treating patients with advanced ovarian cancer, with a tolerable safety profile.
Introduction
As one of the most common causes of female cancer death, 1 ovarian cancer affects more than 310,000 women over the world acoording to the World Cancer Report in 2020. 2 Chemotherapy (e.g. platinum-based chemotherapy) remains a standard treatment approach for patients with ovarian cancer. 3 However, approximately 70–80% of patients with ovarian cancer, in particular those with epithelial ovarian cancer, may relapse, requiring further systemic therapy, 4 with the median survival reported to be around 5 years. Recent studies have shown that the use of anti-angiogenic agents in combination with chemotherapy may prolong the progression-free survival (PFS) and improve the response rates to chemotherapy. This is particularly apparent in patients with platinum-resistant or advanced-stage disease, highlighting the therapeutic benefits of anti-angiogenic agents. 5 For instance, the MITO 11 study conducted by Pignata et al. 6 showed that the combined use of pazopanib with paclitaxel achieved a median PFS (mPFS) of 7.5 months, while of only 6.2 months for paclitaxel monotherapy, in patients with ovarian cancer.
Anlotinib is an oral small-molecule receptor tyrosine kinase inhibitor (TKI) that extensively inhibits tumor angiogenesis and growth by targeting vascular endothelial growth factor receptor (VEGFR) 1/2/3, c-Kit, platelet-derived growth factor receptors (PDGFR)-α, and fibroblast growth factor receptor (FGFR) 1/2/3. 7 Anlotinib has now been approved for the treatment of lung cancer, soft tissue sarcoma, and other solid tumors, but not of gynecological malignancies, by the Food and Drug Administration and the China National Medical Products Administration. However, there have already been some attempts to investigate the efficacy of Anlotinib monotherapy and combined therapy in the treatment of advanced ovarian cancer in clinical practice as evidenced by several studies using a real-world study (RWS) design.8 –10 Yet they had very small sample sizes ranging from 4 to 40 and were predominantly conducted at single centers. Moreover, despite promising outcomes in improving the objective response rate (ORR), disease control rate (DCR), and PFS, their results on the benefit of combined use of Anlotinib with chemotherapy in patients with advanced ovarian cancer were inconsistent. For example, the studies by Chen et al. 11 and Ni et al. 10 observed a 100% DCR related to the combination use in patients with advanced ovarian cancer, while in other studies, the magnitudes were around 80–95%.
Given these, we conducted this study with a large sample size from multi-centers to explore the efficacy and safety of Anlotinib combined with chemotherapy in patients with advanced ovarian cancer, based on the RWS design. Moreover, we also conducted a meta-analysis of available evidence to provide a more robust estimate of the effectiveness of Anlotinib in combination with chemotherapy.
Methods
Cohort study
Patients
This was a retrospective multi-center real-world cohort study that enrolled advanced ovarian cancer patients who received Anlotinib treatment in combination with chemotherapy, based on a tumor database constructed by 15 institutions located in East China. This tumor database included patients treated with Anlotinib for gynecologic tumors from August 2018 and thereafter, and collected information on demographic parameters, efficacy evaluations, and treatment outcomes. The database lock-time for this study was on 31 March 2022.
Patients were included if they were: aged ⩾18 years, diagnosed with advanced ovarian cancer (included FIGO stage III–IV, platinum-sensitive, and platinum-resistant recurrent ovarian cancer) based on histopathology and/or cytology, and treated with Anlotinib in combination with chemotherapy because of no response or resistance to standard therapy. Patients were excluded if they: could not be used for efficacy evaluation, or had insufficient follow-up information on the treatment details or a follow-up time-length <1 month prior to the database lock-time (unless disease progression or death occurred).
In this study, chemotherapy regimens included but were not limited to the 21-day cycles of platinum-based chemotherapy, pemetrexed chemotherapy, or any other chemotherapy prescribed by the gynecologist in accordance with the guidelines. Anlotinib was administrated orally at a dose of 12 mg/day for 14 days, 21 days a cycle, until death or progress during the observation period. However, the dosage would be slightly reduced in case of severe adverse reactions.
Outcome definitions
We employed RECIST version 1.1 to evaluate the therapeutic efficacy of the use of Anlotinib combined with chemotherapy in this study.12 The outcomes of interest included ORR, DCR, mPFS, and adverse events (AEs), which were calculated as previously suggested. 13 The timepoint of initializing Anlotinib treatment was designated as the reference timepoint for all assessments of the treatment-related outcomes. AEs are classified into grade 1–2 and grade 3–4 based on the Common Terminology Criteria for Adverse Events version 4.03, including fatigue, pain, hypertension, hand-foot syndrome, rash, diarrhea, anorexia, weight loss, pneumothorax, wound healing problems, oral mucositis, proteinuria, etc.
Statistical analysis
Continuous variables are presented as medians (interquartile ranges), and categorical or binary variables are as numbers (percentages). Clopper–Pearson method was used to estimate the 95% confidence interval (CI) for the binomial proportion. Kaplan–Meier method was used to obtain the survival curve of mPFS with 95% CI. Subgroup analysis for mPFS was conducted based on age (<60 or ⩾60 years), timepoint for initializing Anlotinib treatment (following 1–2 or ⩾3 lines), tumor histology (mucinous carcinoma, serous carcinoma, or clear cell carcinoma), and previous antiangiogenic therapy (yes or no). Statistical analysis was performed using Stata 16 software (Stata Corp., College Station, TX, USA) and SPSS software version 21.0 (SPSS Inc., Chicago, IL, USA), with a two-sided p < 0.05 indicative of significance.
Meta-analysis
Search strategy
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses guideline (Supplemental Table S1). Databases including PubMed, Web of Science, and Cochrane Library were searched from their inception to March 2023, using the words/terms related to ‘ovarian cancer’ and ‘Anlotinib’. The detailed search strategy is shown in Supplemental Appendix S1. We also manually checked the reference lists of relevant articles to identify other potentially eligible studies.
Inclusion and exclusion criteria
We included cohort, case–control, or case series studies that evaluated the efficacy and safety of Anlotinib in combination with chemotherapy for the treatment of advanced ovarian cancer. However, we excluded studies that evaluated the outcomes of Anlotinib as a monotherapy or in combination with anti-programmed cell death-ligand 1 agents. In case of significant overlaps in the enrolled participants from a single research group, we selected the study with the largest sample size for our meta-analysis.
Data extraction
Upon the removal of duplicates, we screened the title/abstracts, followed by the full-text reading, by two independent investigators (XH and SQ), for potentially eligible studies. Disagreements, if occurred, were resolved by discussions with a third investigator (BD).
The following variables were extracted by two independent investigators (XH and SQ): first author, publication year, country of the study, number of cases, study design, study setting, sociodemographic status, tumor type, Eastern Cooperative Oncology Group performance status (ECOG PS), history of chronic disease, history of previous therapy, outcomes including ORR, DCR, and mPFS, and most commonly reported AEs.
Statistical analysis
The random-effects meta-analysis model, which better accounts for the heterogeneity between studies and may provide more conservative results than the fixed-effects meta-analysis model, was used in this study to obtain the summary estimates on the outcomes of interest. Heterogeneity was assessed by I 2 statistic. Sensitivity analysis was performed in this study by removing each study individually. Publication bias was not assessed because of the very small number of included studies. All the analyses were conducted using Stata 16 software (Stata Corp.).
Results
Cohort study
Among the 125 patients with advanced ovarian cancer, we excluded 11 participants due to the missing follow-up evaluation information, and 43 participants due to non-combined use of chemotherapy agents or irregular medication. Finally, a total of 71 patients, who were predominantly recurrent cases, were included in our RWS.
The median age of included patients was 57 years (range: 38–80 years, Table 1). At the initiation of the Anlotinib treatment, most patients had an ECOG PS score of 0–1 and 80.7% (57 cases) were diagnosed with FIGO Stage III–IV disease. Serous carcinoma accounted for the majority of tumor histology (64.8%), followed by mucinous carcinoma (15.5%), and clear cell carcinoma (5.6%). Additionally, platinum-based chemotherapy was administered in 71.8% of the patients while antiangiogenic drugs were used in 45.1%.
Baseline characteristics of enrolled patients in the cohort study.

Patients might be counted more than once because they may have received one of the prior therapies as mentioned.
ECOG PS, Eastern Cooperative Oncology Group performance status; FIGO, The International Federation of Gynecology and Obstetrics.
The ORR and DCR of the included patients were 40.8% (95% CI: 29.1–52.6%) and 76.1% (95% CI: 65.9–86.2%), respectively (Table 2). Complete response was achieved in five cases (7.0%), and the mPFS was 4.6 months (95% CI: 3.2–5.9 months). The log-rank test showed that age (<60 or ⩾60 years), timepoint for initializing Anlotinib treatment, and tumor histology were not related to longer mPFS (all p > 0.05), but previous antiangiogenic therapy was (p < 0.05) (Figure 1).
Analysis of the efficacy of Anlotinib.

CI, confidence interval; CR, complete response; DCR, disease control rate; ORR, objective response rate; PFS, progression-free survival; PR, partial response.
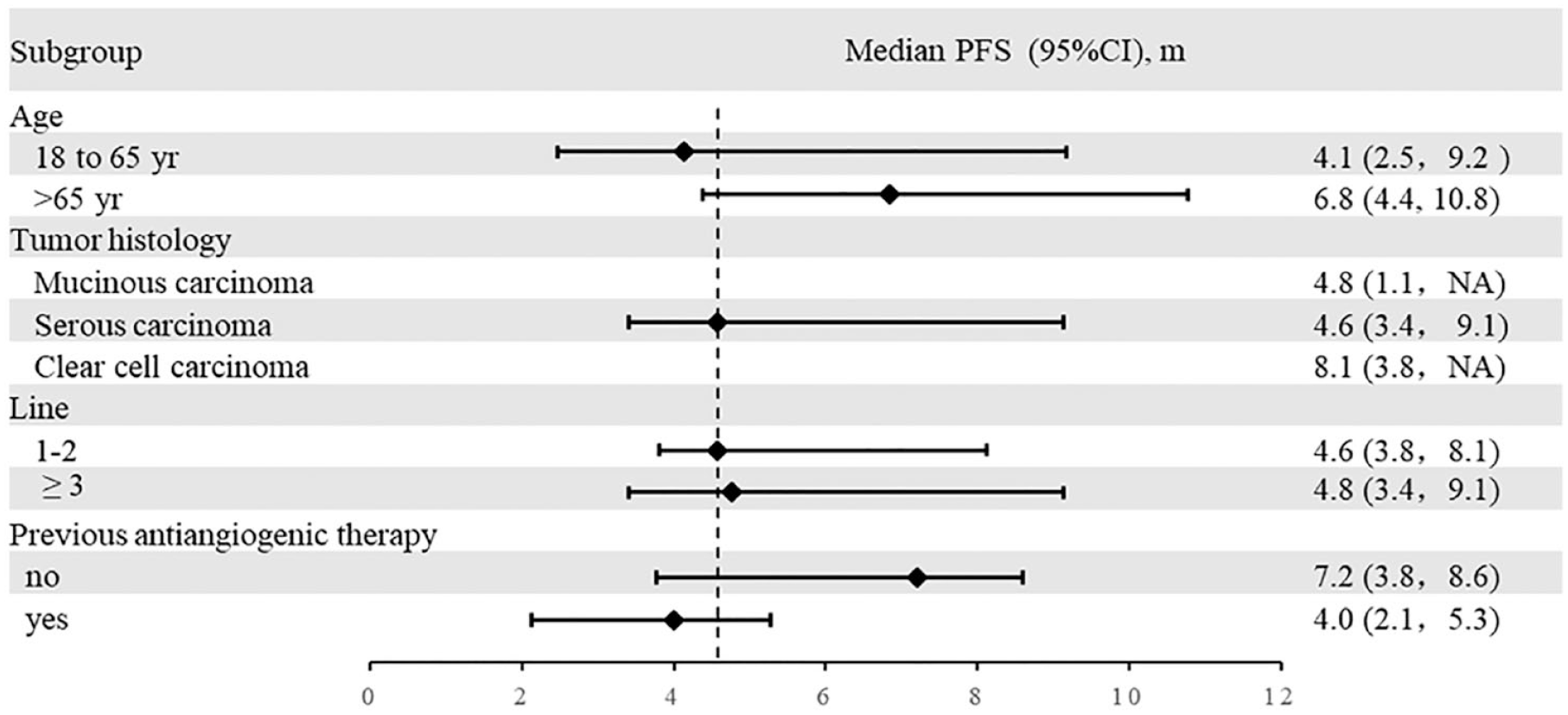
mPFS in different subgroups*.
The most common AEs of any grade included hand-foot syndrome (18 patients, 25.4%), hypertension (7 patients, 9.9%), and nausea, vomiting, or diarrhea (7 patients, 9.9%, Table 3). There were 10 patients reporting grade 3–4 AEs (Supplemental Table S1). Four patients were discontinued due to grade 3–4 AEs, but no treatment-related deaths were recorded.
Treatment-related adverse events for enrolled patients.

Mainly hand and foot pain and arthralgia.
Including oral and vulvar ulcers.
PPE, palmoplantar painful erythema; HFS, hand-foot syndrome.
Meta-analysis
Out of the 34 unique articles retrieved (Figure 2), 4 articles were eligible for inclusion.8,10,11,14 The baseline characteristics of these studies are shown in Supplemental Table S2. Among them, three were retrospectively designed based on a real-world database and the remaining one, a phase II study, was prospectively designed. However, none of the studies had a control group that enrolled patients who only received chemotherapy. The age of the included patients ranged from 22 to 77 years, and their sample sizes varied from 4 to 27. Together with the data from our study, a total of 148 patients (from five studies) with ovarian cancer who were treated with Anlotinib combined with chemotherapy were finally included.
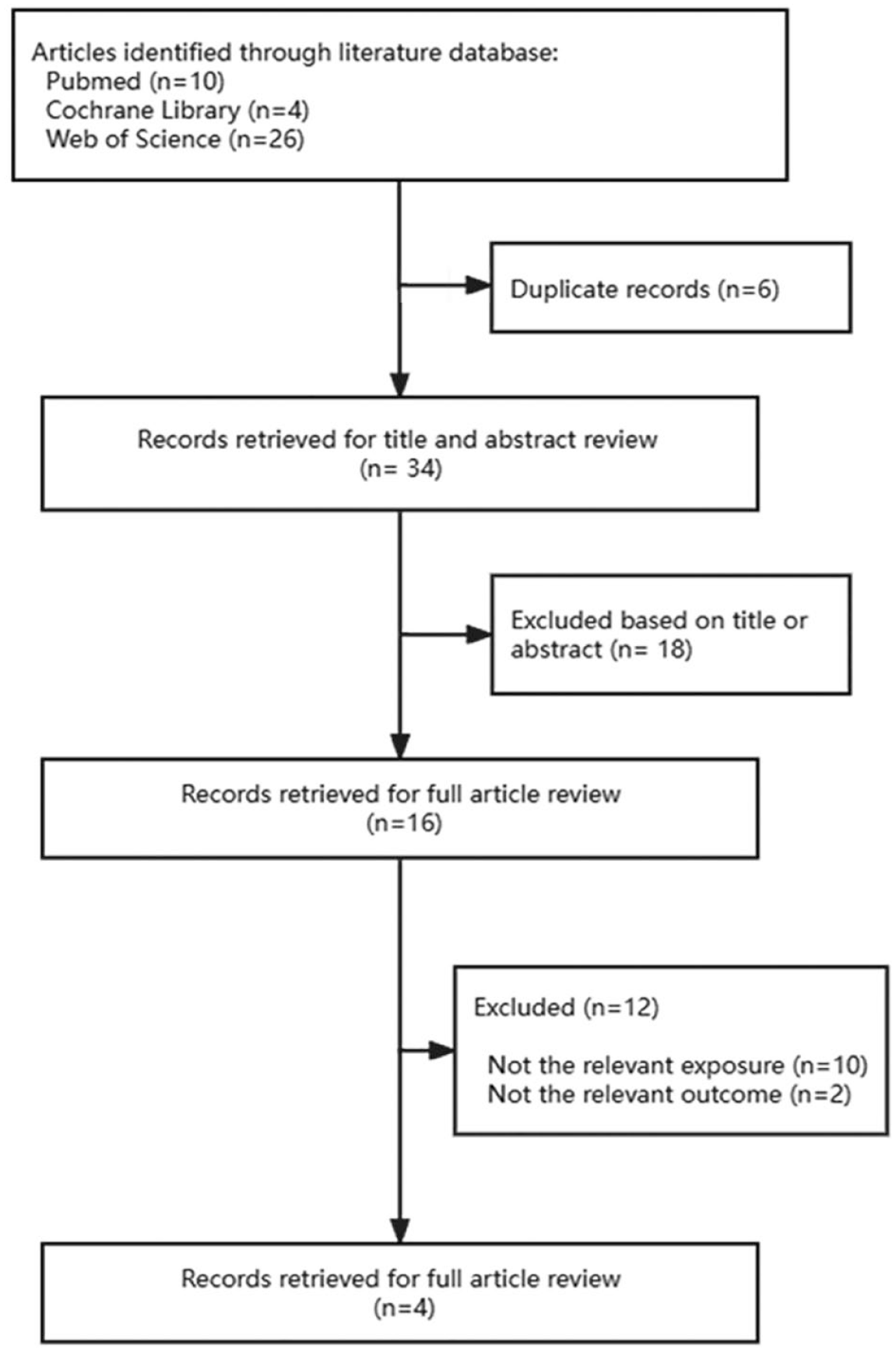
Flowchart of the study selection process.
Meta-analysis showed that the ORR related to the combined use of Anlotinib with chemotherapy in patients with ovarian cancer was 33.8% (95% CI 22.7–44.8%, I 2 = 44.2%, from four studies), the DCR was 90.6% (95% CI 73.6–99.9%, I 2 = 79.1%, from five studies), and the mPFS was 6.6 months (95% CI 4.9–8.4 months, I 2 = 72.0%, from five studies, Figure 3). Sensitivity analysis revealed that the outcomes were minorly changed upon the individual removal of each study. The most common AEs reported in the eligible studies included hand-foot syndrome, hypertension, nausea, fatigue, and rash.

Forest plots of the (a) median progression-free survival, (b) objective response rate, and (c) disease control rate in meta-analysis.
Discussion
Main findings
This study, which comprises an observational retrospective cohort in the real-world setting and a meta-analysis, showed consistently that the combined use of Anlotinib with chemotherapy had a significant anti-tumor activity in patients with ovarian cancer, with an overall ORR of 33.8%, an overall DCR of 90.6%, and an overall mPFS of 6.6 months. This study also showed that the combined use of Anlotinib with chemotherapy exhibited a tolerable safety, with the hand-foot syndrome being the most common AE.
Interpretations and implications
Chemotherapy, in particular platinum-based chemotherapy, is widely recommended for the treatment of patients with advanced ovarian cancer. 15 It is also frequently used in combination with anti-angiogenic agents to maximize its antitumor activity. 16 Anlotinib is a novel oral small-molecule multi-target TKIs that exerts anti-angiogenic benefit by targeting at VEGFR 1/2/3 and FGFR 1/2/3. 17 Recent studies conducted by Wang et al. 18 and Xu et al. 13 reported favorable outcomes of Anlotinib in the treatment of platinum-resistant recurrent ovarian cancer. Moreover, Anlotinib combined with chemotherapy has been also investigated in patients with ovarian cancer as a promising treatment option.19,20 However, there is a lack of evidence with a larger sample size to support the efficacy of their combined use, since the vast majority of the available studies had small sample sizes (ranged from 4 to 40), weakening their robustness potentially. To address this issue, we conducted a multi-center RWS with the largest sample size to date, showing that the combination therapy of Anlotinib and chemotherapy is associated with an ORR of 40.8%, DCR of 76.1%, and mPFS of 4.6 months in patients with advanced ovarian cancer.
Our findings are in general consistent with those from prior studies. For example, the study by Chen et al. 11 showed a comparable outcome in ORR as ours (33.3% versus 40.8%), despite the differences in the baseline characteristics of included patients. However, the mPFS associated with the combined use of Anlotinib with chemotherapy was smaller in our study compared with the one by Chen et al. 11 (4.5 versus 9.3 months) and another one by Cui et al. 15 (4.5 versus 8.0 months). A possible reason for this might be due to the variations in the follow-up periods and sample sizes. To account for these differences, we conducted a meta-analysis by pooling all the available evidence together, showing that the combined use of Anlotinib with chemotherapy led to an overall ORR of 33.8%, an overall DCR of 90.6%, and an overall mPFS of 6.6 months, during the follow-up of 11–34 months in patients with advanced ovarian cancer.
Previous studies suggested that older age and tumor histology were associated with poor outcomes in patients with advanced ovarian cancer. However, in our analysis we found that neither age, nor previous lines of therapy, nor tumor histology were related to a longer mPFS. Notably, the history of the application of anti-angiogenic agents prior to the use of Anlotinib was associated with a longer mPFS. This on the one hand indicates that earlier initialization of the antiangiogenic agents in treating ovarian cancer might bring more benefits. On the other hand, this provides a piece of evidence in support of the effectiveness of shifting from one anti-angiogenic agent to another one, such as Anlotinib, in treating patients with ovarian cancer. In addition to the efficacy, the use of Anlotinib in combination with chemotherapy exhibited a good safety, with the AEs commonly reported to be hand-foot syndrome, hypertension, nausea, fatigue, and rash. Moreover, only a very small proportion of patients may experience AEs that were graded as 3–4 level.
Our study also provided some insights into the current status of Anlotinib use in patients with advanced ovarian cancer in eastern China. Similar to previous studies, the patients in our study were mainly middle-aged and older, 10 with a median age of 57 years. However, it is worth being mentioned that in the real-world clinical settings, patients who were prescribed with Anlotinib could be also at a younger age (e.g. aged at 30 years reported in the study by Cui et al. 8 ). Moreover, we found that the enrolled patients who were given Anlotinib were majorly at a late FIGO stage. It is likely that this might be due to the fact that Anlotinib has not been approved for gynecological cancer by the official authorities. Furthermore, preclinical data suggest the potential synergistic effects when combining antiangiogenic therapies with immunomodulatory agents. The positive findings from the PAOLA-1 study support the combined use of olaparib with bevacizumab in patients with homologous-recombination deficiency, 21 thereby opening up new possibilities for combining poly ADP-ribose polymerase (PARP) inhibitors with antiangiogenic drugs as a therapeutic option. It might be therefore of clinical interest to investigate whether the supplementation of PARP inhibitors to the use of Anlotinib alone or the combined use of Anlotinib with chemotherapy would further improve the health outcomes in patients with advanced ovarian cancer.
Strengths and limitations
The strengths of our study include a multi-center data-analysis, a large sample size, and the use of meta-analysis to obtain more robust outcomes. However, our study has some limitations. Firstly, although the data used in our analysis were retrospectively collected using a pre-designed Excel document, there is the possibility that the follow-up interval and efficacy evaluation criteria may not be consistent from each center due to the non-experimental nature of the RWS. Secondly, our study did not collect adequate information on breast cancer susceptibility gene status and homologous recombination deficiency status. As a result, we could not determine their influences on the outcomes we observed. Thirdly, some heterogeneity was detected for the meta-analyses on DCR and mPFS. Moreover, the lack of sufficient data on chemotherapy regimens or platinum sensitivity/resistance information restricted us to conduct further subgroup analyses to explore their influences on the outcomes of interest. Fourthly, our study and the retrieved four individual studies did not introduce a control group that enrolled patients receiving chemotherapy alone for comparison. This may potentially affect the evidence-level of our study. Finally, most patients in our study (including the four studies in the meta-analysis) were recurrent cases with ovarian cancer. This indicates that the findings might be of more relevance to patients with recurrent ovarian cancer. Moreover, the enrolled patients in this study were mainly Chinese, and the generalization of the present findings to other populations with different ethnics/races remains to be confirmed.
Conclusion
In conclusion, our study showed that the combined use of Anlotinib with chemotherapy had potential in treating patients with advanced ovarian cancer, with a tolerable safety. However, our findings are required to be replicated by future studies using randomized controlled designs or in other populations (e.g. Europeans).
Supplemental Material
sj-docx-1-tam-10.1177_17588359231221336 – Supplemental material for Combined use of Anlotinib with chemotherapy in patients with advanced ovarian cancer: a real-world cohort study and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359231221336 for Combined use of Anlotinib with chemotherapy in patients with advanced ovarian cancer: a real-world cohort study and meta-analysis by XinYi Hong, ShanHu Qiu, Bo Ding, Hao Xu and Yang Shen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
