Abstract
Background:
Patients with advanced epidermal growth factor receptor (EGFR)-mutant lung adenocarcinoma (LAD) inevitably experience drug resistance following treatment with EGFR-tyrosine kinase inhibitors (TKIs).
Objectives:
We aimed to analyze the effect of primary tumor consolidative therapy (PTCT) on patients treated with first-line osimertinib.
Design and methods:
This retrospective cohort study was conducted in patients with advanced stage III or stage IV LAD with EGFR-sensitizing mutations (exon 19 deletion or L858R mutation) with disease control after first-line osimertinib. A curative dose of primary tumor radiotherapy or primary tumor resection was classified as PTCT. We compared the progression-free survival (PFS) and overall survival (OS) of patients with and without PTCT.
Results:
This study included 106 patients with a median age of 61.0 years, and of those, 42% were male and 73.6% were never-smokers. Exon 19 deletion was observed in 67.9%, 30.2% had a programmed cell death ligand 1 (PD-L1) tumor proportion score <1%, 33.0% had brain metastasis, and 40.6% had oligometastasis. In all, 53 (50%) patients underwent PTCT. Patients who underwent PTCT demonstrated significantly better PFS [30.3 (95% confidence interval (CI), 24.1–36.4) versus 18.2 (95% CI, 16.1–20.2) months; p = 0.005] and OS [not reached versus 36.7 (95% CI, 32.5–40.9) months; p = 0.005] than patients who did not. A multivariate analysis showed that PTCT was an independent factor associated with better PFS [hazard ratio (HR), 0.22; 95% CI, 0.10–0.49; p < 0.001] and OS [HR, 0.10; 95% CI, 0.01–0.82; p = 0.032]. The PFS benefits of PTCT were consistent across subgroups, and the HR tended to be lower in patients aged <65 years, males, smokers, stage IVB disease, L858R, PD-L1 expression ⩾1%, non-oligometastasis, and brain metastasis.
Conclusion:
Of the patients with advanced EGFR-mutant LAD, those who underwent PTCT had a significantly better survival outcome than those who did not. The survival benefits were consistent across different subgroups.
Introduction
Lung cancer is the leading cause of cancer-related mortality worldwide. 1 For patients with non-small-cell lung cancer (NSCLC), particularly adenocarcinoma, epidermal growth factor receptor (EGFR) mutations are one of the most common driver gene alterations. 2 With superior progression-free survival (PFS) and fewer adverse events than chemotherapy, EGFR-tyrosine kinase inhibitors (TKIs) have been approved as first-line treatment in patients with metastatic EGFR-mutant NSCLC.3–5 Recent studies have demonstrated even better treatment outcomes with third-generation EGFR-TKIs (median PFS, 18.9–19.3 months) than with first-generation TKIs.6,7
Drug resistance to EGFR-TKIs seems to be an inevitable issue. Researchers have promoted a combination with other treatments to delay EGFR-TKI resistance. Combining therapy of a first-generation EGFR-TKI, with either antiangiogenic agents or chemotherapy, has improved PFS in patients with the L858R mutation or exon 19 deletion8–11; however, the combination of osimertinib and bevacizumab failed to demonstrate these survival benefits. 12 Recently, the addition of thoracic surgery or radiation therapy to systemic therapy has been shown to result in better disease control than systemic therapy alone, especially for oligometastatic NSCLC.13–15 For patients with EGFR-mutant NSCLC, the addition of local consolidative therapy, including surgery or radiotherapy, to first-generation EGFR-TKIs has also demonstrated survival benefits.16–19 However, the effects of adding local treatment to third-generation TKIs are unclear.
In this retrospective cohort study, our aim was to analyze the effects of primary tumor consolidative therapy (PTCT), including a curative dose of primary tumor radiotherapy or primary tumor resection, in patients with lung adenocarcinoma (LAD) treated with first-line osimertinib and under disease control.
Patients and methods
Patients
We enrolled lung cancer patients treated at Chung Shan Medical University Hospital, Taichung Veterans General Hospital, National Taiwan University Hospital, and National Taiwan University Cancer Center from November 2017 to May 2021. Patients who were diagnosed with advanced stage III or stage IV LAD with an EGFR-sensitizing mutation (exon 19 deletion or the L858R mutation), treated with osimertinib as first-line systemic therapy, and those with disease control were eligible. Patients who underwent surgical resection for early-stage LAD and who experienced disease recurrence were also included in the analysis. Patients were excluded if they had a comutation with EGFR exon 20 insertion, intolerable side effects of osimertinib, poor performance status [Eastern Cooperative Oncology Group performance status (ECOG PS) ⩾3], or another active cancer. Patients who did not achieve disease control after osimertinib treatment were also excluded. Moreover, patients treated with combinations of chemotherapy, antiangiogenetic agents, or any anticancer drugs with osimertinib were excluded from the analysis. Patients with a history of previous adjuvant or neoadjuvant chemotherapy as a treatment course of curative surgical resection were eligible.
The definition of PTCT includes surgical resection or radiotherapy of the primary tumor. Resection of the primary tumor using any method was regarded as surgical resection, as long as the resection margins were recognized as R0 by pathologists. Radiotherapy methods included stereotactic ablative radiotherapy (SABR) or conventional radiotherapy. SABR was performed for 4–10 consecutive days, with each fractionated dose ranging from 6 to 12 Gy. For conventional radiotherapy, the treatment dose should be 50–70 Gy given in 25–35 fractions. Palliative radiotherapy or surgical resection of metastatic sites was allowed in both groups of patients.
Data records and survival endpoints
Clinical data for analysis included patients’ age, sex, smoking status, ECOG PS, tumor stage, EGFR mutation subtypes, number of metastases, pattern of disease progression, and survival status. Patients with less than or equal to five metastatic lesions were classified as oligometastatic, while patients with more than five metastatic lesions were classified as non-oligometastatic. Lung cancer staging was determined according to the 8th edition of the American Joint Committee on Cancer (AJCC) staging system. 20 Unidimensional measurements as defined by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 were used in this study. 21 Regarding adverse events, we focused on radiation pneumonitis and surgical complications in the PTCT group. This study was approved by the Institutional Review Board of Chung Shan Medical University Hospital, Taichung Veterans General Hospital, National Taiwan University Hospital, and National Taiwan University Cancer Center (IRB No. CS1-20105 and CF20175). Written informed consent to access clinical data records was obtained from all patients.
Statistical methods
In this cohort, patients were classified into PTCT group (treatment group) and non-PTCT group (control group). Propensity score matching was used to generate a matched treatment-control comparison. Data are presented as frequencies (percentages) for categorical variables and medians [interquartile range (IQR)] for continuous variables. To determine the differences between groups, the chi-square test or Fisher’s exact test was applied for categorical variables, and the Mann–Whitney U test was applied for continuous variables. PFS was measured as the time from osimertinib treatment to disease progression or death. Overall survival (OS) was measured as the time from osimertinib treatment to death from any cause. Patients were censored if they were alive at the time of analysis during the last follow-up. The Kaplan–Meier method was used to estimate the survival time. Differences in survival time were analyzed using the log-rank test. Univariate and multivariate analyses of survival outcomes and subgroup analysis of survival were performed with a Cox proportional hazards model. Two-tailed tests with p values <0.05 were considered statistically significant. All analyses were performed using the IBM SPSS Statistics package, version 23 (IBM Corporation, Armonk, NY, USA).
Results
Patients and their demographic data
In all, 131 patients with advanced stage III or stage IV LAD were enrolled at the initial screening. After removing patients who did not meet the inclusion and exclusion criteria, 107 patients were included for propensity-score matching. Finally, 106 patients treated with first-line osimertinib were included. Figure 1 shows the patient enrollment flow chart. The demographics of the patients are summarized in Table 1. The median age of the patients was 61 years. Of them, 44 (41.5%) were male, and 78 (73.6%) were never-smokers. In all, 72 patients (67.9%) had EGFR exon 19 deletion (19del), and 34 (32.1%) had the L858R mutation. Among the patients, 543 (50.0%) underwent PTCT, 27 patients underwent primary tumor resection, and 26 received primary tumor radiotherapy. Patients with PTCT were more likely to have stage IVA disease and stable disease than patients without PTCT.

Flow chart of patient enrollment.
Demographic data.

Categorical data are presented as numbers (%).
p Value: Mann–Whitney U test or chi-square test.
ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, Epidermal growth factor receptor; PD-L1, programmed death-ligand 1; PTCT, primary tumor consolidative therapy.
The survival outcomes and disease progression pattern between patients with and without PTCT
The median PFS of the 106 patients was 26.2 [95% confidence interval (CI), 19.7–32.6] months, and the median OS was 44.0 (95% CI, 36.7–51.4) months (Table 2). The median duration of follow-up was 25.1 (IQR, 20.1–33.5) months. Patients who underwent PTCT demonstrated significantly better PFS [30.3 (95% CI, 24.1–36.4) versus 18.2 (95% CI, 16.1–20.2) months; p = 0.005; hazard ratio (HR), 0.48; 95% CI, 0.29–0.81] and OS [not reached versus 36.7 (95% CI, 32.5–40.9) months; p = 0.005; HR, 0.28; 95% CI, 0.11–0.69] than patients who did not undergo PTCT (Figure 2). Among patients who underwent PTCT, those who received primary tumor radiotherapy and those who underwent primary tumor resection had better PFS and OS than patients treated with osimertinib alone.
Patient outcomes.
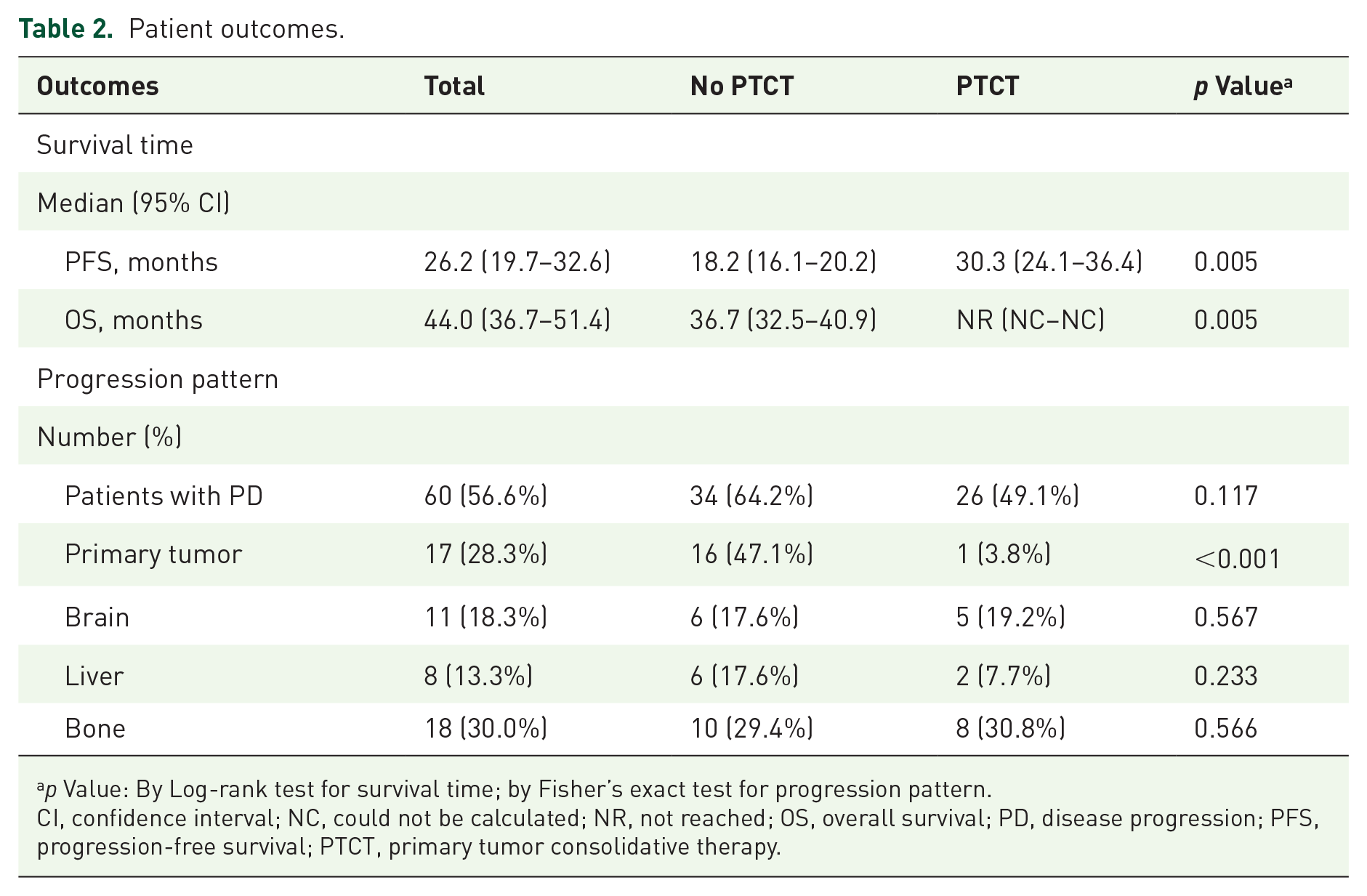
p Value: By Log-rank test for survival time; by Fisher’s exact test for progression pattern.
CI, confidence interval; NC, could not be calculated; NR, not reached; OS, overall survival; PD, disease progression; PFS, progression-free survival; PTCT, primary tumor consolidative therapy.

Progression-free survival (a and c) and overall survival (b and d) were compared by the Kaplan–Meier methods between patients who underwent PTCT or not. Patients who underwent PTCT (either primary tumor radiotherapy or resection) demonstrated significantly better PFS and OS than patients who did not.
A total of 60 patients (56.6%) experienced disease progression (PD); among them, 26 patients were in the PTCT group and 34 patients were in the non-PTCT group (Table 2). Although the rate of PD was similar between patients with and without PTCT (49.1% versus 64.2%; p = 0.117), the rate of primary tumor progression was lower in patients who underwent PTCT than in those who did not (3.8% versus 47.1%; p < 0.001).
Influences of clinical factors on the outcomes and subgroup analysis
The results of the univariate and multivariate survival analyses are presented in Table 3. For PFS, the univariate analysis revealed that male sex, stage IVB, L858R mutation, and no PTCT were associated with a worse prognosis. The multivariate analysis revealed that male sex, stage IVB, L858R mutation, and no PTCT were independently associated with worse PFS. For OS, the univariate analysis demonstrated that those without brain metastasis and PTCT were associated with a better outcome. The multivariate analysis showed that age <65 years, exon 19 deletion, and PTCT were independently associated with better OS.
Univariate and multivariate analyses of the influence of clinical factors on survival.
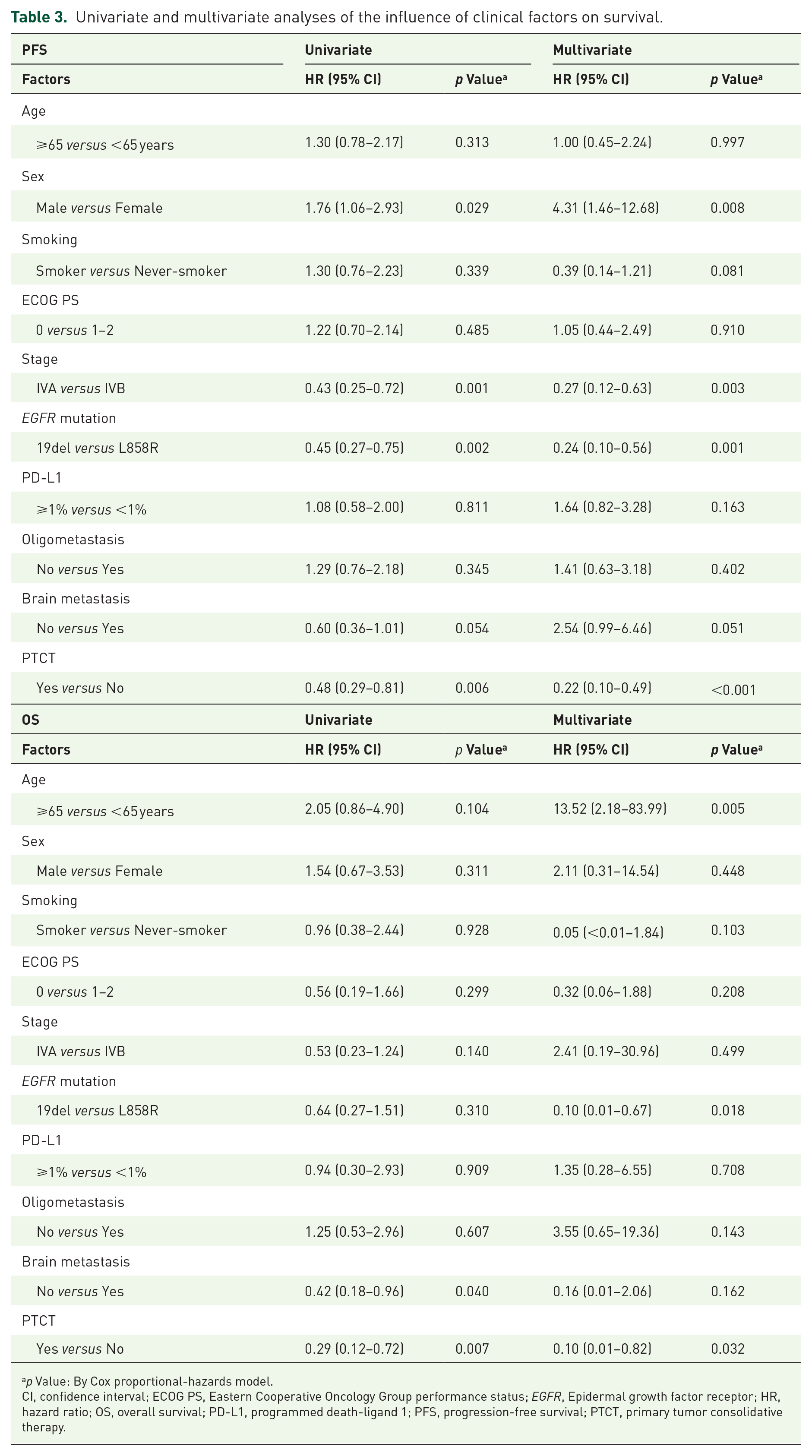
p Value: By Cox proportional-hazards model.
CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; EGFR, Epidermal growth factor receptor; HR, hazard ratio; OS, overall survival; PD-L1, programmed death-ligand 1; PFS, progression-free survival; PTCT, primary tumor consolidative therapy.
The PFS benefits of PTCT were consistent across the subgroups (Figure 3). Although the HR appeared to be lower in certain subgroups, the confidence intervals overlapped. Subgroup analysis was performed for age (⩾65 versus <65 years; HR, 0.81 versus 0.28), sex (male versus female; HR, 0.23 versus 0.71), smoking behavior (smoker versus never-smoker; HR, 0.16 versus 0.64), stage (IVA versus IVB; HR, 0.88 versus 0.38), EGFR mutation subtypes (19del versus L858R; HR, 0.41 versus 0.38), PD-L1 expression (⩾1 versus <1%; HR, 0.30 versus 0.52), oligometastasis (no versus yes; HR, 0.38 versus 0.64), and brain metastasis (no versus yes; HR, 0.64 versus 0.27). Among patients with stage IVB disease, the median PFS was 30.3 (95% CI, 20.4–40.2) months for patients who underwent PTCT and 15.6 (95% CI, 11.4–19.9) months for those who did not undergo PTCT (p = 0.009) (Supplemental Figure A.1). Among patients with the L858R mutation, the median PFS was 23.0 (95% CI, 14.7–31.3) months for those who underwent PTCT and 10.5 (95% CI, 8.5–12.6) months for those did not (p = 0.009). Among patients with PD-L1 expression ⩾1%, the median PFS was 30.3 (95% CI, 21.8–38.7) months for those who underwent PTCT and 13.7 (95% CI, 8.3–19.2) months for those who did not (p = 0.003). Among patients with non-oligometastatic, the median PFS was 31.2 (95% CI, 21.9–40.4) months for those who underwent PTCT and 17.3 (95% CI, 14.7–19.9) months for those who did not (p = 0.006). Among patients with brain metastasis, the median PFS was 31.2 (95% CI, 22.6–39.7) months for those who underwent PTCT and 11.0 (95% CI, 9.9–12.0) months for those who did not (p = 0.003).
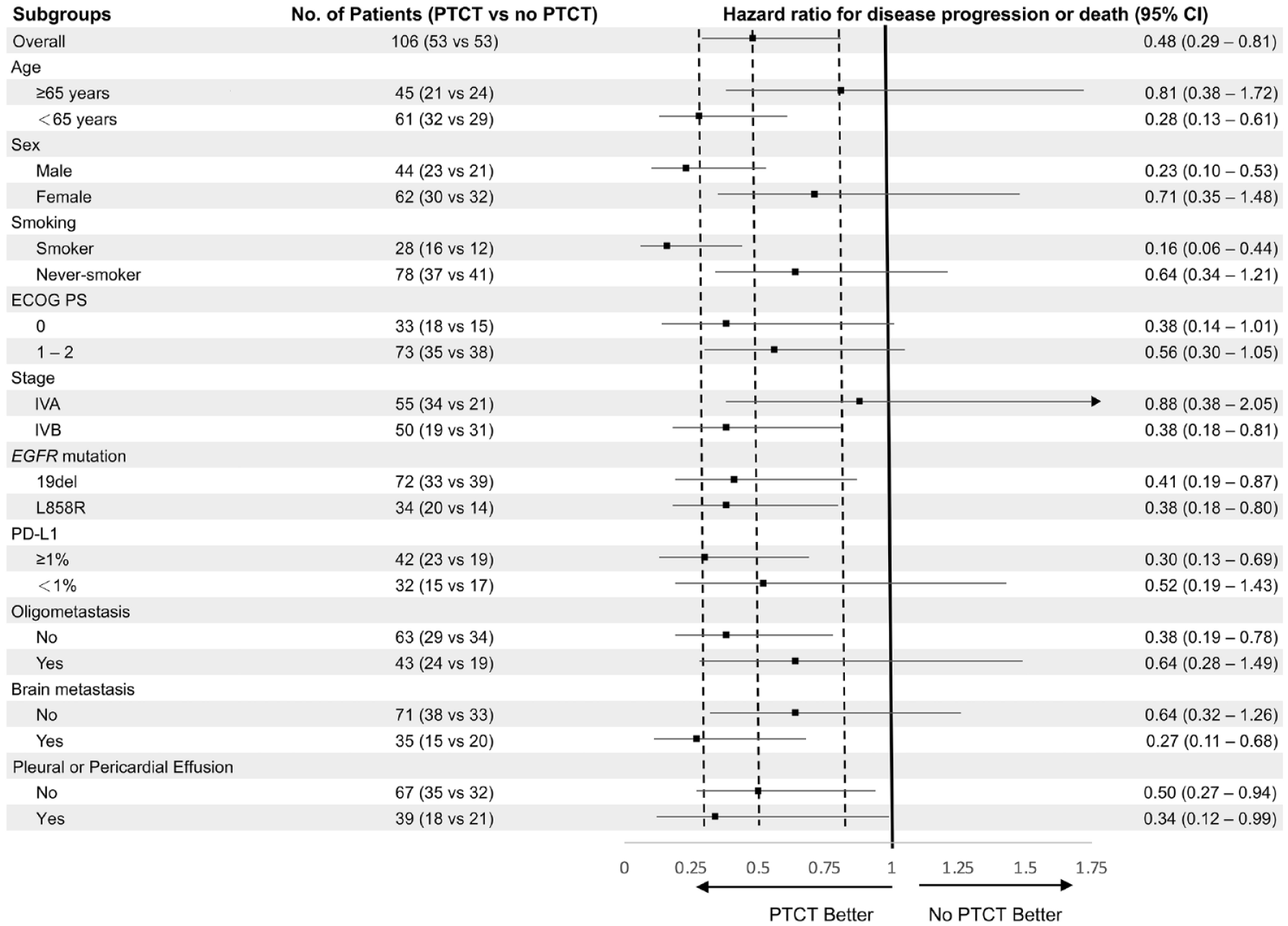
Forest plot of subgroup analysis of progression-free survival. A hazard ratio of less than 1.00 indicates a lower risk of disease progression or death for patients with PTCT than without PTCT.
Adverse events related to PTCT
During the follow-up period, 11 patients in the radiotherapy group experienced clinically significant pneumonitis (Table 4). Among these patients, grade 1 and 2 pneumonitis occurred in 8 (30.8%) and 3 (11.5%) patients, respectively. No significant difference was observed in the rate of pneumonitis between those who received conventional radiotherapy and those who received SABR. Among patients who underwent surgery, four surgical complications occurred, including 2 (7.4%) subcutaneous emphysema, 1 (3.7%) pulmonary artery injury, and 1 (3.7%) chylothorax. No fatal events related to PTCT occurred.
Modalities and adverse events of primary tumor consolidative therapy, number (%).

Radiotherapy methods included stereotactic ablative radiotherapy (SABR) or conventional radiotherapy.
p Value: By Fisher’s exact test.
Discussion
In this study, we demonstrated that PTCT prolonged both PFS and OS in advanced-stage EGFR-mutant LAD patients who achieved disease control with first-line osimertinib. The survival benefits appear to be more significant in certain subgroups, including patients aged <65 years, males, smokers, and those with the L858R mutation, PD-L1 expression ⩾1%, non-oligometastasis, and brain metastasis.
The addition of local consolidative local therapy (LCT) to systemic therapy in patients with oligometastatic NSCLC has been demonstrated to improve survival outcomes.14,22 For patients receiving EGFR-TKI or chemotherapy, patients treated with LCT had better PFS than those not treated with LCT.14–16,23 These studies shared some characteristics. First, patients with more than five metastatic lesions, multiple brain metastases, or pleural/pericardial effusion were ineligible. Second, radiation therapy or surgical resection was performed on all metastatic sites. Although LCT was feasible for those patients, this therapy carried risks for multiple-site radiotherapy. Grade 3–4 radiation pneumonitis and grade 5 treatment-related events have also been reported in previous studies.16,24
Recently, radiotherapy or resection of the primary lung tumor in combination with systemic treatment has been demonstrated to confer survival benefits in patients with metastatic NSCLC.17,19,25,26 Ohtaki et al. demonstrated that salvage surgery was safe and may prolong the OS of patients receiving first-line EGFR-TKI treatments (33 patients) or ALK-TKI (3 patients). 25 In their retrospective study, the 3-year OS and recurrence-free survival rates were 75.1% (95% CI, 55.9–86.9%) and 22.2% (95% CI, 8.6–39.7%), respectively. In the analysis by Tseng et al., 76 of 466 patients with metastatic NSCLC treated with first-line EGFR-TKIs underwent primary tumor resection (PTR). 26 Patients who underwent PTR were shown to have significantly longer PFS (25.1 versus 9.4 months; p < 0.001) and OS (56.8 versus 31.8 months; p = 0.004) than patients who did not. Another study by Kuo et al. also revealed significant survival benefits of adding PTR to first-line EGFR-TKI treatments in patients with stage IV EGFR-mutant NSCLC (PFS, 26.9 versus 13.0 months; p < 0.001). 17 In the study by Hsu et al., primary lung tumor radiotherapy, rather than PTR, was performed following first-line EGFR-TKI treatment. 19 Patients who received radiotherapy had significantly longer PFS (27.5 versus 10.9 months; p < 0.001) and OS (NR versus 38.0 months; p < 0.001) than patients who did not receive radiotherapy. None of the above studies used osimertinib as a first-line treatment.
Primary tumor treatment may improve the effect of systemic therapy by reducing tumor burden and the potential of tumor spreading.18,27 The resistant subclones have been demonstrated to exist in patients treated with EGFR-TKI before disease progression. 28 According to the analysis by Al-Halabi et al., most EGFR-TKI failures were initially from the primary tumor. 29 In the study by Hsu et al., patients who did not receive primary tumor radiation showed a significantly higher rate of primary tumor progression (55.4% versus 25.0%; p = 0.011) than patients who received radiation therapy. In the present study, the rates of primary tumor progression were significantly higher in patients who did not undergo PTCT than in patients who underwent PTCT (47.1 versus 3.8%; p < 0.001). Thus, the addition of PTCT to systemic therapy, either surgery or radiotherapy, is a reasonable approach to achieve better primary tumor control and improve the efficacy of systemic therapy.
Among patients with EGFR-mutant metastatic NSCLC, clinical characteristics, including male sex, smoking status, stage IVB disease, non-oligometastasis, and brain metastasis, have been associated with poor prognosis.30–32 Regarding the molecular aspects, patients with the L858R mutation or high PD-L1 expression have been shown to have a worse response to treatment and worse survival.33–37 In the studies of first- and second-generation EGFR-TKI monotherapy, patients with the L858R mutation had inferior PFS than those with 19del. 33 Although osimertinib resulted in a better PFS and OS than first-generation EGFR-TKIs, a trend of lower efficacy was observed for patients with L858R than for those with 19del (HR, 1.00 versus 0.68). 6 On the contrary, the combination of chemotherapy or antiangiogenetic agents with first-generation EGFR-TKIs has been shown to result in superior PFS benefits in patients with L858R compared with those with 19del. 33 However, in the WJOG9717L study, the combination of osimertinib and bevacizumab failed to demonstrate survival benefits; moreover, for patients with L858R, those treated with combination therapy tended to have inferior PFS than those treated with osimertinib monotherapy. 12 The variety in treatment response may be explained by the distinct cellular behaviors and tumor microenvironments. NSCLC cell lines carrying L858R and 19del have been reported to have different EGFR phosphorylation and downstream signaling pathways.38,39 By contrast, high expression of PD-L1 in patients with EGFR-mutant NSCLC has been associated with more unstable genomic alterations and higher potential for TKI resistance.40,41 In addition, lung tumors with EGFR mutations have been reported to have weak immunogenicity. 42 Therefore, PTCT may help restore immunity, decrease tumor stem cell subclones, and reduce tumor heterogeneity, subsequently improving systemic control by EGFR-TKI.19,26,43–45 In the current study, the addition of PTCT to osimertinib was more beneficial in certain subgroups, including patients aged <65 years, males, smokers, stage IVB disease, and patients with L858R, PD-L1 expression ⩾1%, non-oligometastasis, and brain metastasis. On the contrary, the survival benefits were less significant in patients aged ⩾65 years, which may be due to the higher risks associated with surgery or radiotherapy in elderly individuals. Our results suggest that PTCT improves the efficacy of osimertinib treatment and that the benefits were more significant in patients with a higher risk of drug resistance.
Recently, a retrospective study by Zeng et al. compared the efficacy of adding LCT after treatment of first- or second-line osimertinib in patients with oligo-residual NSCLC. 46 In their study, 14 of 108 patients were classified as the LCT group and received surgery, radiotherapy, or radiosurgery to the primary tumor or metastatic sites. Their study demonstrated a superior PFS (NR versus 12.8 months, p = 0.001) and less primary tumor (12.5% versus 33.0%) and CNS (21.6% versus 50.0%) failure after the addition of LCT to osimertinib. Our study was different in that only patients treated with first-line osimertinib were analyzed. In addition, patients with oligometastasis or non-oligometastasis were included. We focused on the effect of consolidative therapy on primary tumors rather than on metastatic sites.
This study has several limitations. First, since this is a retrospective study, selection bias was inevitable. More patients in the PTCT group were diagnosed with stage IVA disease and had stable disease after osimertinib treatment. These findings may be because some of the patients in the PTCT group had pleural metastases when they underwent PTRs. Although selection bias cannot be excluded, PTCT demonstrated significant survival benefits in the analysis of patients with stage IVB disease. Therefore, the results suggest that PTR or radiotherapy may contribute to the superior outcomes of patients who undergo PTCT. Second, the sample size was not large enough, and some of the comparisons between the subgroups were not statistically significant. Third, the follow-up period may not be long enough, and OS outcomes were not mature when analyzing subgroups (Supplemental Figure A.2). The ongoing phase II trial, NORTHSTAR (NCT03410043) is comparing the efficacy of the addition of radiotherapy or surgery to osimertinib in the treatment of patients with stage IIIB or IV EGFR-mutant NSCLC. 47 We anticipate the study results, which may help to clarify the benefits of combination therapy with PTCT and osimertinib.
Conclusion
Of the patients with advanced EGFR-mutant LAD, those who underwent PTCT had significantly longer PFS and OS than those who did not. The PFS benefits were more significant in patients aged <65 years, males, smokers, patients with stage IVB disease, and in those with the L858R mutation, PD-L1 expression ⩾1%, non-oligometastatic disease, or brain metastasis. However, more prospective studies are needed to validate these findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231220606 – Supplemental material for Primary tumor consolidative therapy improves the outcomes of patients with advanced EGFR-mutant lung adenocarcinoma treated with first-line osimertinib
Supplemental material, sj-docx-1-tam-10.1177_17588359231220606 for Primary tumor consolidative therapy improves the outcomes of patients with advanced EGFR-mutant lung adenocarcinoma treated with first-line osimertinib by Jia-Jun Wu, Jeng-Sen Tseng, Zhe-Rong Zheng, Cheng-Hsiang Chu, Kun-Chieh Chen, Mong-Wei Lin, Yen-Hsiang Huang, Kuo-Hsuan Hsu, Tsung-Ying Yang, Sung-Liang Yu, Jin-Shing Chen, Chao-Chi Ho and Gee-Chen Chang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank NCFB Pharmacogenomics Lab TR6 for technical support, and the National Science and Technology Council for funding support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
