Abstract
Positron emission tomography (PET) imaging in prostate cancer has advanced significantly in the past decade with prostate cancer targeted radiopharmaceuticals now playing a growing role in diagnosis, staging, and treatment. This narrative review focuses on the most commonly used PET radiopharmaceuticals in the USA: prostate-specific membrane antigen (PSMA), fluciclovine, and choline. 18F-fluorodeoxyglucose (FDG) is used in many other malignancies, but rarely in prostate cancer. Previous literature is discussed regarding each radiopharmaceutical’s utility in the settings of screening/diagnosis, initial staging, biochemical recurrence, advanced disease, and evaluation prior to targeted radiopharmaceutical therapy and radiation therapy. PET imaging has demonstrated utility over traditional imaging in various scenarios; however, there are few head-to-head studies comparing PET radiopharmaceuticals. PSMA radiopharmaceuticals are the newest tracers developed and have unique properties and uses, especially at low prostate-specific antigen (PSA) levels. However, each PET radiopharmaceutical has different properties which can affect image interpretation. Choline and fluciclovine have minimal urinary activity, whereas PSMA agents can have high urinary activity which may affect locoregional disease evaluation. Of the three radiopharmaceuticals, only PSMA is approved for both diagnostic and therapeutic indications with 177Lu-PSMA. A variety of diagnostic PET radiotracers for prostate cancer allows for increased flexibility, especially in the setting of supply chain and medication shortages. For the time being, keeping a diverse group of PET radiopharmaceuticals for prostate cancer is justifiable.
Keywords
Background
Prostate cancer is the most common cancer in men worldwide, excluding dermatologic malignancies. In the United States, it is the second leading cause of malignancy-related mortality after lung cancer. 1 Diagnosis, prognosis, and treatment are dependent upon the patient’s clinical state along the course of the disease. These disease states range from localized disease, rising PSA without detected sites of metastasis, and clinically evident metastases, and can also be classified based on castrate range testosterone level following treatment. 2 As developments in therapies and changes in practice occur, prostate cancer diagnosis, staging, and treatment have evolved. Prostate cancer imaging has also significantly improved for both local and metastatic diseases. The past decade has been an enlightened period for the development of positron emission tomography (PET) in prostate cancer, from the approval of the first prostate-specific PET agents to the development of specific therapeutic radiopharmaceuticals targeting prostate cancer.
This narrative review will illustrate the clinical utility of PET imaging for prostate cancer and compare the literature surrounding the current most used PET agents, including prostate-specific membrane antigen (PSMA) targeting compounds, choline, and fluciclovine. The utility of these compounds will be compared for visualization in diagnosis, initial staging, localization of metastases in biochemical recurrence and in advanced disease.
PET agents
There are multiple PET agents used for prostate cancer imaging with numerous others in development and clinical trials, including RM2/bombesin receptors (phase IIa) as both diagnostic and therapeutic agents, 3 but for brevity and clinical relevance, we will focus on the most commonly utilized agents in the United States, attempting to highlight the strengths and weaknesses of each agent, and key differentiating features among groups of agents (see also Table 1).
Positron emission tomography.

PSMA, prostate-specific membrane antigen.
Prostate-specific membrane antigen
PSMA is a type II transmembrane glycoprotein found on the surface of the prostate and other cell membranes. 4 PSMA is expressed 100–1000-fold greater on the cell surface of prostate cancer cells than normal prostate cells4,5 making the compound particularly attractive for imaging detection of prostate cancer. Despite the name, PSMA is also found on the surface of other cells, including the gastrointestinal tract, kidneys, and salivary glands and can be overexpressed in malignancies other than prostate cancer. 68Ga-PSMA-11 and 18F-Piflufolast (18F-DCFPyL) are the PSMA-based agents with US FDA regulatory approval, and differences exist in biodistribution, availability, and supply. Both radiotracers have evidence to support comparably high detection of prostate cancer 5 with specific advantages and differences in the diagnostic performance between PSMA-based radiotracers being an active area of research. The United States first approved 68Ga-PSMA in 2020 for the initial staging of high-risk prostate cancer and the detection of metastatic disease in patients with biochemical recurrence. There are several different methods for production, including the commercially available radiolabeling kit LOCAMETZ® (68Ga-PSMA-11, Advanced Accelerator Applications, Millburn, NJ, USA) available to departments across the United States. 6 68Ga is typically produced from a 68Ge generator. It has a half-life of 68 min, which makes transportation over long distances possible but somewhat challenging.18F-Piflufolast PSMA (DCFPyL) PYLARIFY® (Lantheus/Progenics, Billerica, MA, USA) was approved in 2021, also for the initial staging of high-risk prostate cancer and detection of metastatic disease in the setting of biochemical recurrence. This radiotracer is commercially manufactured, has a longer half-life of 110 min, and can be distributed regionally similar to 18F-fluorodeoxyglucose (18F-FDG). Several other PSMA agents are currently in development and investigational use, most with 18F-radionuclides for the distribution and image quality considerations. 5 Normal biodistribution of currently approved PSMA tracers shows the highest uptake in the urinary collecting system as it is renally excreted, which can potentially obscure the vesicoureteral junction (Figures 1 and 2). In addition, there is high physiologic uptake in the salivary and lacrimal glands and lower levels of uptake in the liver, spleen, bowel (predominately jejunum), and autonomic ganglia (Figures 1 and 2). 4
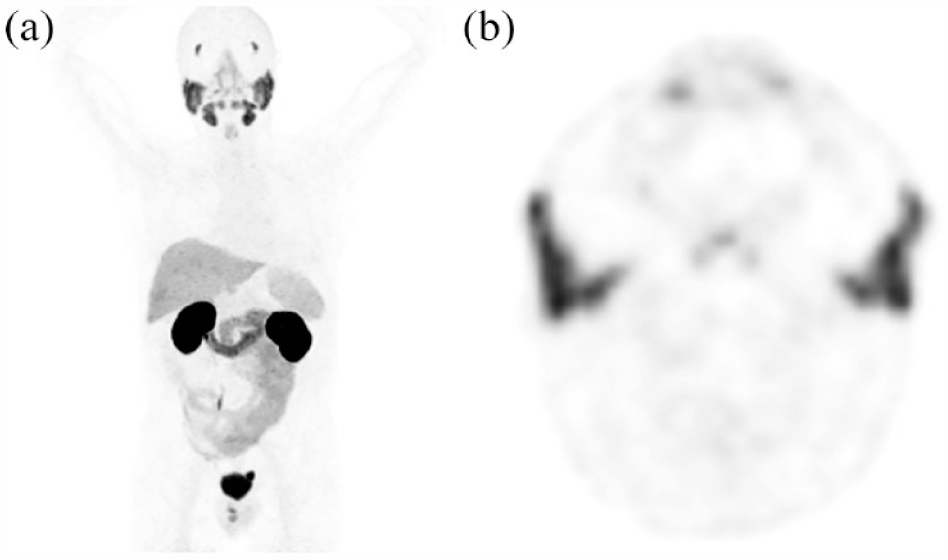
68Ga PSMA-11 PET/CT normal distribution, 76-year-old patient with a history of robotic prostatectomy and adjuvant radiation therapy to the prostate bed and pelvic lymph nodes for Gleason score 4+3 prostate cancer with lymph node metastasis. PSA = 0.6 ng/mL. (a) MIP image demonstrating normal 68Ga PSMA-11 distribution of marked uptake within the lacrimal glands, salivary glands, kidneys ureters and bladder and low to moderate uptake within the liver, spleen, and low uptake in the small bowel. (b) Axial PET image through the level of the parotid gland demonstrating physiologic uptake.

18F-DCFPyL PET/CT normal distribution, 71-year-old patient with a history of definitive prostate radiotherapy initial Gleason 4+4 prostate cancer, local prostate, and seminal vesical recurrence with Gleason 5+5 prostate cancer and subsequent prostate cryoablation, and salvage radiotherapy. At the time of the scan, the patient was on hormonal therapy. PSA: 0.49 ng/mL (a) MIP image demonstrating normal 18F-DCFPyL distribution of marked uptake within the lacrimal glands, salivary glands, kidneys, ureters and bladder and low-to-moderate uptake within the liver, spleen, and low uptake in the small bowel. (b) Coronal PET/CT-fused images demonstrating blooming artifact of the urinary bladder. (c) Sagittal PET/CT-fused image demonstrating physiologic uptake with blooming artifact near the bladder obscuring the vesicoureteral junction.
Choline
Choline is an amino acid analog used as a precursor for the cell membrane phospholipid phosphatidylcholine. As cancer cells divide, choline is incorporated into the cell membrane, increasing radiolabeled choline’s signal in prostate cancer and other malignant and benign processes. 7 11C-choline was approved by the FDA in 2012 as an investigational new drug for the imaging evaluation of suspected metastatic prostate cancer. 18F-Choline, while used in other countries, was never approved in the United States. 7 11C-choline has a short half-life of 20 min, requiring an on-site cyclotron for synthesis, limiting the availability of the radiotracer. 18F-Choline has a longer half-life, allowing for regional distribution and greater use outside large institutions. 7 11C and 18F-choline have similar normal biodistributions with high avidity within the liver, spleen, pancreas, and salivary glands and low levels within the bowel and bone marrow (Figure 3). 7 Owing to uptake time, 18F-Choline has significantly greater urinary bladder activity than 11C-choline, which can obscure disease within the pelvis, bladder, and ureters. 7

11C-Choline PET/CT normal distribution, PSA = 10.5 ng/mL (a) MIP image demonstrating normal 11C-Choline distribution in the same patient as Figures 1 and 3, 1 year after the 68Ga-PSMA PET and 6 months after the Fluciclovine PET. No focus of metastatic disease was identified. Normal distribution of uptake within the salivary glands, kidneys, and moderate uptake within the liver, spleen, and diffusely throughout small bowel. Physiologic uptake is also noted in lymph nodes and bone marrow. (b) Axial-fused PET/CT image through the level of the kidneys demonstrating physiologic uptake within kidneys, bowel, and bone marrow.
Fluciclovine
Fluciclovine is a synthetic amino acid that targets amino acid transporters, specifically alanine-serine-cysteine transporters (ASCT2) and sodium-independent system L (LAT1) which are upregulated in prostate cancer cells. 8 18F-Fluciclovine (Anti-1-amino-3-18F-flurocyclobutane-1-carboxylic acid) Axumin® (Blue Earth Diagnostics, Oxford, UK) was approved by the FDA in 2016 for the identification of metastatic foci in the setting of biochemical recurrence after definitive therapy. 9 The half-life of 110 min allows for regional distribution, as with other 18F-based radiotracers. Normal biodistribution of fluciclovine is most marked in the pancreas, followed by the liver (the critical organ), with lower levels of uptake in the salivary gland, pituitary, and bowel (Figure 4). 8 Fluciclovine kinetics cause it to wash out of organs and marrow and slowly accumulate within skeletal muscle. 10 Fluciclovine is renally excreted, though there is typically a low level of bladder uptake due to early imaging. 9 Fluciclovine does not incorporate into the tumor cells, and the tumor avidity decreases 65% after 90 min, preventing the development of fluciclovine as a theragnostic agent. 5

18F-Fluciclovine PET/CT normal distribution, same patient as Figure 1, 6 months after the 68Ga PSMA scan in Figure 1, PSA 4.8 ng/mL (a) MIP demonstrating normal 18F-Fluciclovine distribution of uptake predominately within the pancreas and liver, and low-level uptake within the bone marrow and skeletal muscle. (b) Axial-fused PET/CT image through the level of the pancreas demonstrating marked physiologic uptake within pancreas and liver with low level in skeletal muscle.
Fluorodeoxyglucose
18F-FDG PET, while useful in many other malignancies, is not routinely used in prostate cancer imaging because well-differentiated prostate cancer does not readily take up 18F-FDG. Significant urinary concentration of 18F-FDG also limits the evaluation of avid locoregional disease. 11 18F-FDG has a potential role in identifying metastatic disease foci in poorly differentiated prostate cancers, such as neuroendocrine (68Ga-Dotatate can also be used) and small cell variants (Figure 5). 5 Additionally, candidates for PSMA-targeted therapies such as 177Lu-PSMA-617 (Pluvicto®, AAA, Novartis, Basel, Switzerland) were evaluated with 18F-FDG PET because FDG avidity would portend a poor response to therapy resulting in exclusion from PSMA radiopharmaceutical therapy in clinical trials.5,12

18F-FDG PET/CT Normal distribution and FDG avid prostate cancer. (a) MIP image demonstrating normal 18F-FDG uptake with marked signal in the brain, collecting system, bladder, and patchy uptake in the myocardium in a 37-year-old male with history of treated rectal cancer. (b) MIP image of an 81-year-old male with incidentally noted sclerotic osseous lesions, which were biopsy-proven metastatic ductal adenocarcinoma of the prostate, Gleason 4+4, PSA: 1.3 ng/mL. Atypical 18F-FDG avid lesions consistent with known high-grade malignancy. (c) Axial-fused PET/CT image demonstrating left femoral metastasis (arrow). (d) Axial-fused PET/CT image demonstrating primary prostatic lesion (arrow).
Protocols
Imaging protocols vary between institutions and radiopharmaceuticals. Previously published protocols with dosages and imaging procedures are described below.
11C-Choline must be synthesized with an on-site cyclotron. Patients receive 370-632 MBq (10–17.1mCi) of 11C-choline. 13 Low-dose CT is performed from the skull base to mid-thighs (120 kvp and 20–160 mA). PET imaging begins approximately 5–10 min after injection of radiotracer. 7
PSMA PET radiotracer imaging protocols are similar. 14 Patients receive 111–259 MBq (3–7 mCi) of 68Ga-PSMA-11. Patients void immediately prior to PET imaging images, which are obtained 50–100 min after injection. Images are acquired from mid-thighs to skull base to mitigate the collecting system and bladder filling. 14 18F-Piflufolast PSMA (DCFPyL) patients receive 296–370 MBq (8–10 mCi), and the uptake time is 60–120 min. 12
18F-Fluciclovine is administered in a dose of 370 MBq (10 mCi). Imaging is performed 3–5 min after administration. Scanning is acquired from the mid-thigh to skull base over 20–30 min. 8 Patients are instructed to avoid significant exercise for 24 h, fast for 4 h, and void 30–60 min prior to imaging. 8
Depending on departmental capabilities, each of the aforementioned radiopharmaceuticals can be performed with PET/CT or PET/MRI.
Initial diagnosis/screening
Initial diagnosis of prostate cancer is made with a combination of a digital rectal exam, PSA level, multiparametric prostate MRI (mpMRI), and biopsy (either systematic saturation biopsy or US-guided fusion biopsy targeted to a specific MRI finding). Although beyond the scope of this review, newer screening and diagnostic laboratory testing, including bioassay risk stratification tests, are being increasingly utilized. 15
In patients without pathologic diagnosis of prostate cancer, there was limited evidence for the use of PET agents until recent studies showed that PSMA PET/MRI could increase the diagnostic accuracy of primary prostatic lesions.16,17 For localization of primary prostate cancer, fluciclovine PET/MRI showed potential for delineation of high-grade
Initial staging in intermediate- and high-risk patients
Traditionally, staging of prostate cancer utilized contrast-enhanced CT imaging, mpMRI, and nuclear medicine bone scintigraphy. The most recent National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology (NCCN Guidelines) recommend bone and soft tissue imaging in patients with intermediate unfavorable, high, and very high-risk prostate cancer based on initial risk stratification guidelines.
24
Data from the landmark ProPSMA trial, a randomized controlled trial comparing 68Ga-PSMA-11 PET
11C-Choline for initial staging has been investigated in several studies with variable results. 27 Combined with other studies, the patient-based sensitivity of 11C-choline ranges from 18.9 to 57%.23,28
18F-Fluciclovine was also examined in preoperative staging, yielding 55.3%
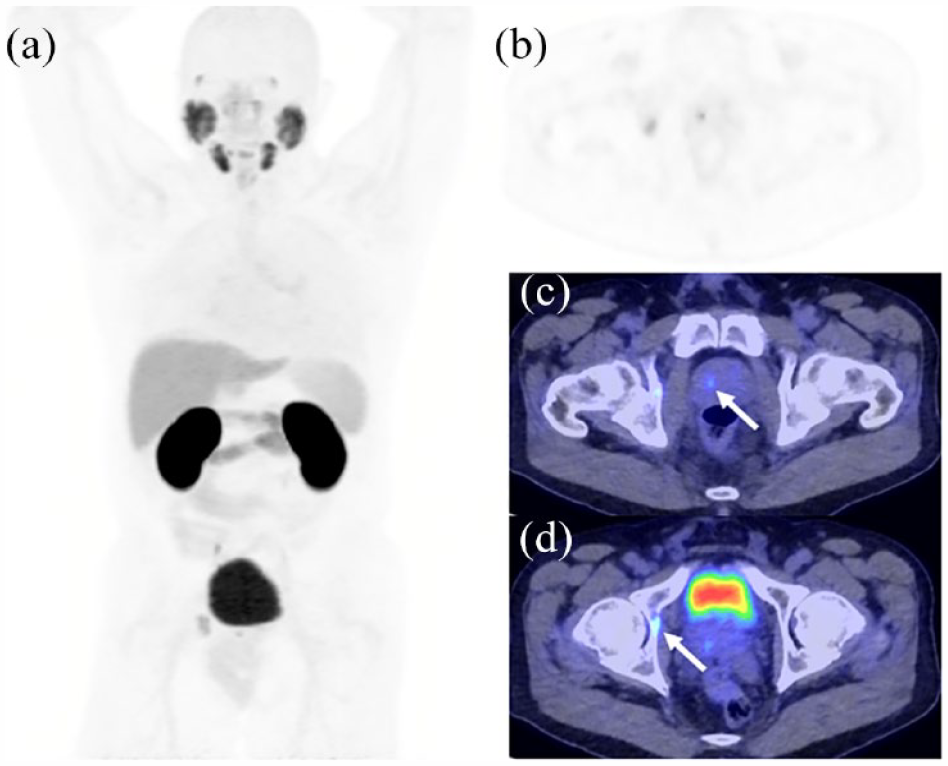
18F-DCFPyL PET/CT performed for initial staging in a 70-year-old patient with Gleason 3+4 prostate acinar adenocarcinoma with metastasis to the right acetabulum. PSA: 17.5 ng/mL (a) MIP image demonstrating an avid lesion in the prostate and right medial acetabulum. (b) Axial PET image centered on the primary prostate lesion with uptake in the acetabulum. (c) Axial PET/CT-fused image at the same location as (b). demonstrating the prostate lesion (arrow). (d) Axial PET/CT-fused image focused on the acetabular lesion (arrow).
Biochemical recurrence
PSMA, fluciclovine, and 11C-choline are all approved in the setting of biochemical recurrence after definitive therapy. As such, these radiopharmaceuticals have been compared in several reviews and meta-analyses in the setting of BCR after prostatectomy or radiotherapy. In the setting of biochemical recurrence, overall pooled sensitivity, and specificity for 11C and 18F-choline have ranged from 85% to 89% and 88% to 97% per patient.22,30–32 Pooled sensitivity and specificity for fluciclovine have been reported at 87% and 66%,
33
and pooled sensitivity and specificity for 68Ga PSMA has been reported at 86–99% and 76–86% with variation in studies making head-to-head comparison difficult.22,34,35 Evans

68Ga PSMA-11 PET/CT performed in the setting of biochemical recurrence. Sixty-eight-year-old patient with history of robotic prostatectomy for Gleason 4+3 prostate cancer. PSA 1.5 ng/mL. (a) MIP image demonstrates PSMA avid osseous lesions involving C7, T11, and L1 vertebrae. There is an additional focus of possible recurrence in the right pelvis. (b) Axial CT image demonstrating the sclerotic metastasis in the L1 vertebral body. (c) Axial PET/CT-fused image showing uptake within the sclerotic L1 lesion.

11C-Choline and 68Ga PSMA PET in the setting of biochemical recurrence. Seventy-year-old patient with history of prostatectomy and bilateral lymph node dissection for Gleason 4+3 prostate adenocarcinoma and salvage pelvic radiation therapy. PSA 0.45 ng/mL. Subsequent biopsy proved metastatic prostate cancer. (a) 68Ga PSMA PET image demonstrating avid left external iliac lymph node (arrow). (b) Axial 68Ga PSMA PET/CT-fused image at the same slice demonstrating PSMA avid left external iliac lymph node (arrow). (c). 11C-Choline PET image (17.8 mCi) demonstrating avid left external iliac lymph node (arrow). (d) 11C-Choline Axial-fused PET/CT image at the same slice demonstrating avid left external iliac lymph node (arrow).
Radiotracer detection in the setting of biochemical recurrence.
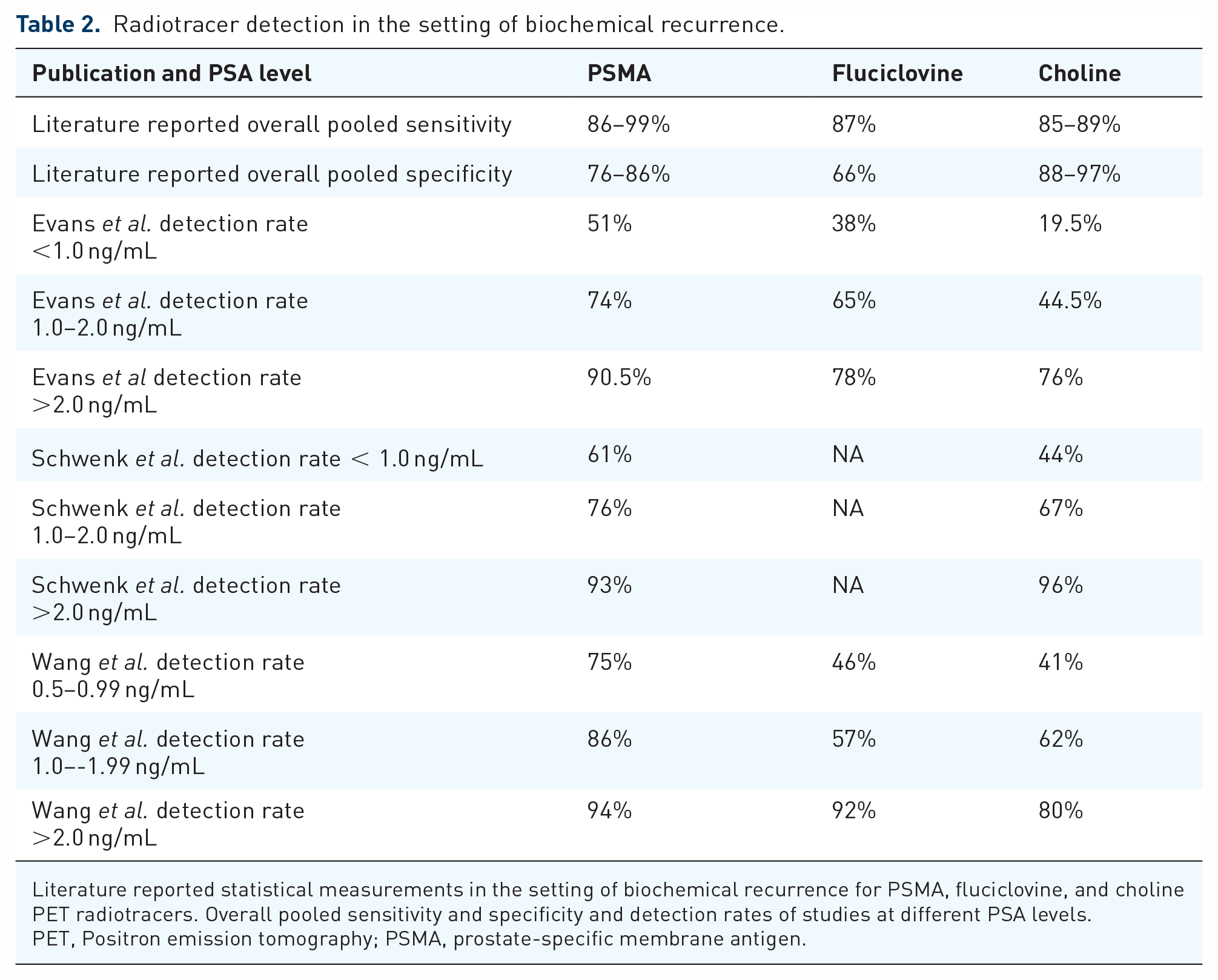
Literature reported statistical measurements in the setting of biochemical recurrence for PSMA, fluciclovine, and choline PET radiotracers. Overall pooled sensitivity and specificity and detection rates of studies at different PSA levels.
PET, Positron emission tomography; PSMA, prostate-specific membrane antigen.
A point of note was made in several of these studies that there are cases of discordant lesions, where lesions are seen only specifically on choline, fluciclovine, or PSMA. In these cases, the tumor’s avidity for specific radiopharmaceuticals may be affected by several factors, including the level of differentiation, hormone-sensitive status, and androgen deprivation therapy (ADT). PSMA has a complex response to ADT with different expressions of the membrane protein depending on the treatment length with current data suggesting PSMA expression is initially increased after beginning ADT (2–6 weeks) and decreases after long-term ADT (3–12 months).40–42
In the post radiotherapy scenario, biochemical recurrence is defined as PSA >2 ng/mL above the nadir. PSMA has been studied prior to this defined cut-off with a pair of studies showing recurrence or metastasis in 84% and 75% of patients with PSA increases below the threshold for biochemical recurrence. Until the definitions for radiotherapy can be updated, PSMA PET may be useful prior to technical BCR in the appropriate clinical setting.43–45
PSMA PET NCCN guidelines are the same in the setting of BCR as they are in initial staging with intermediate or high-risk features.
Advanced prostate cancer
Advanced prostate cancer includes metastatic castration non-resistant, and castration-resistant prostate cancer. The role of prostate PET is well defined in the setting of biochemical recurrence in the search for metastatic lesions. In advanced disease, however, the role is less clear. The use of PSMA PET for response to therapy has been advocated in consensus statements as useful for evaluating response to local and systemic treatments (Figure 9). 46 The Appropriate Use Criteria classifies this indication as ‘may be appropriate’ at this time, citing a lack of clinical trial data to support this use. A key point to note is that PSMA may be less diagnostic in the setting of the recent initiation of systemic therapy for patients with the hormone-sensitive disease. Hence, a consensus statement recommended waiting at least 3 months after initiation of therapy, 14 since ADT can change PSMA expression and alter imaging. 34 FDG/PET may have a role in evaluating treatment response in advanced prostate cancer as these tumors become de-differentiated. Overall, the role of PET agents in advanced prostate cancer is variable and patient dependent. Post-treatment changes and altered tumor metabolism, differentiation, and PSMA expression may lead to discordant PET findings of metastatic disease. In diffusely metastatic castration-resistant disease after systemic therapy, PET can be used if it will somehow change treatment decisions, such as prior to PSMA radiopharmaceutical therapy. The Report of the Advanced Prostate Cancer Consensus Conference 2022 reached consensus recommending PSMA PET in patients with rising PSA after radical radiotherapy of the prostate whose interval to biochemical failure is >18 months and European Association of Urology (EAU) low-risk category. 47 The report also recommended PSMA PET as a first step for imaging in patients with rising PSA after RT therapy of the prostate with interval to biochemical failure <18 months or EAU high-risk category. Further work is needed to determine the best PET imaging agent based on disease characteristics and treatment options. The report made numerous additional recommendations beyond the scope of this review, but based on this report it can be surmised that PSMA is the radiotracer of choice for the European Association of Urology.

Focused prostate MRI and whole body 18F-DCFPyL PET/MR MIP. Seventy-one-year-old patient with metastatic prostate cancer Gleason 5+4 with small cell de-differentiation, prior treatment with Taxotere-based chemotherapy and hormonal therapy. PSA 3.1 ng/mL. (a) MIP PET image which demonstrates only subtle PSMA avid lesions, most noticeable in the liver parenchyma. (b) Axial-fused PET/MR image through the level of the sacroiliac joints demonstrates a soft tissue nodule without PSMA avidity that was biopsy proven metastatic prostate cancer (arrow). A nearby lymph node demonstrates mild PSMA uptake (arrowhead) (c). Coronal MIP PET image demonstrating the PSMA avid left internal iliac lymph node (arrowhead). (d) Coronal PET/MR-fused image demonstrating another view of the soft tissue metastasis (arrow) with nearby internal iliac lymph node metastasis (arrowhead).
PET radiotracers in salvage radiotherapy and oligometastatic disease
Patients with imaging diagnoses of recurrent disease may have different patterns of disease based on location. These patterns have distinct treatment options depending on the site and number of metastases. Salvage radiotherapy is one such therapy for prostate bed recurrence after prostatectomy. The EMPIRE-1 trial was a randomized controlled trial comparing fluciclovine with conventional imaging for planning prostate bed recurrence salvage radiotherapy. This study showed a survival benefit for patients in the fluciclovine arm of the trial. 48 The role of PSMA in salvage radiotherapy (SRT) is currently being investigated with preliminary data from the PSMA-SRT Trial NCT03582774 showing PSMA PET changes SRT planning in over half of patients, and further evaluation of an endpoint of improved outcome will be included after completion. 49 PSMA PET is increasingly being utilized for radiotherapy planning and prognosis. Metastases to lymph nodes detected by PSMA PET are risk factors for poor outcomes in risk prediction nomograms being developed for salvage radiotherapy. 50 A small study of choline PET in the setting of BCR for salvage radiotherapy showed choline PET changed the extent of RT in 13% of patients. However, this was a small (37 patients) retrospective study with no control group. 51 Patients with few metastatic lesions, known as ‘oligo-metastatic disease’, now have an additional treatment option in metastasis-directed radiotherapy (MDT). 52 MDT relies heavily on PET imaging to specifically identify sites of metastasis. A comparison of 18F-choline and 68Ga-PSMA showed patients who underwent PSMA imaging had a longer period free from ADT, suggesting improved visualization and treatment with PSMA. 53 PSMA offers advantages for lower PSA values <1 ng/mL, but in patients with PSA >0.5 ng/mL, no significant difference was found in androgen-free survival. 53 Fluciclovine can also identify oligometastatic disease prior to MDT, though comparative studies with PSMA and choline are needed. 54 High sensitivity and specificity are imperative for patients with oligometastatic disease prior to MDT.
Role of PET agents in radionuclide therapy
For prostate-specific radiotracers, PSMA is the model for theragnostic treatment. The FDA approval and adoption of 177Lu-PSMA (Pluvicto), a PSMA radiopharmaceutical therapy, has solidified PSMA’s role in prostate PET. The VISION trial evaluating 177Lu-PSMA radiopharmaceutical therapy showed a radiographic progression-free survival of 8.7 months
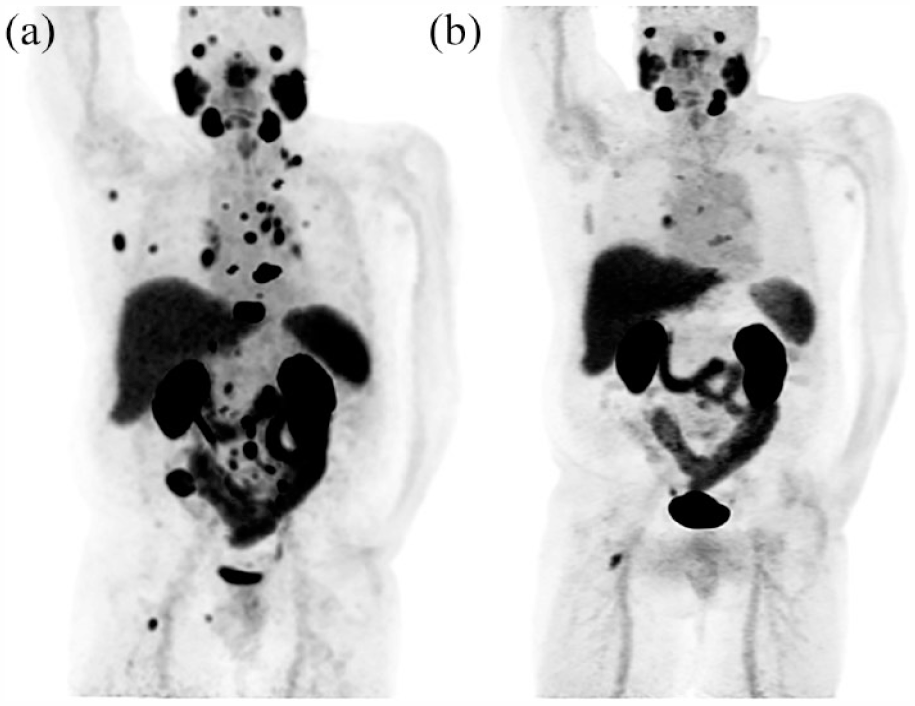
177Lu PSMA radiotherapy pretreatment and mid-treatment imaging. Fifty-six-year-old patient with metastatic prostate cancer Gleason 4+5, prior hormonal therapy, external beam radiation, and Taxotere- and carboplatinum-based chemotherapy. Pretreatment PSA: 13.8 ng/mL. Mid-treatment PSA 0.65 ng/mL. A: 68Ga PSMA-11 MIP image PET/CT prior to 177Lu PSMA radiotherapy demonstrates numerous lymph node and osseous prostate cancer metastases. (b) 18F-DCFPyL PET/CT MIP image PET/CT follow-up scan after two cycles of 177Lu PSMA radiotherapy shows marked improvement in size and number of multiple nodal and osseous metastases.
Choline and fluciclovine have not been developed into theragnostic agents for a variety of reasons, including the low half-life of 11C-choline and the rapid washout of fluciclovine.
PSMA PET has a clear advantage compared with choline and fluciclovine as it is the required radiotracer prior to PSMA radionuclide therapy. Additionally, the development of new PSMA radiopharmaceutical therapy, both with beta and alpha emitters, may change the utilization of PSMA for treatment planning and response.
Conclusion
PET imaging is changing prostate cancer care. There is currently strong evidence for PET imaging in biochemical recurrence, and promising work is being done in locoregional staging, advanced disease, and treatment planning. Furthermore, PSMA radiopharmaceutical therapy is an exciting new treatment that will likely continue to grow.
Comparison of the PET radiopharmaceuticals choline, fluciclovine, and PSMA has been attempted in many prior reviews and meta-analyses, but there is drastic variation in studies and few head-to-head comparisons. Further work is being done to determine the best role for each radiopharmaceutical in different clinical settings. However, PSMA has the significant advantage of being the only FDA-approved agent for diagnostic imaging as well as therapeutic prognostication and evaluation.
Ultimately, a predominant prostate PET radiopharmaceutical will be dictated by multiple factors in addition to improved detection. These include industry pressure, availability, ease of use, and reimbursement. Currently, a variety of PET radiopharmaceuticals allows flexibility and options for patients, which may be lost if only a single agent is available in the future.
