Abstract
Prognosis of metastatic triple negative breast cancer (mTNBC) remains poor despite recent advances in therapeutic options. Trastuzumab deruxtecan (T-DXd) has shown promising efficacy in patients with human epidermal growth factor receptor 2 (HER2)-low breast cancer, which is defined by immunohistochemistry (IHC) 1+ or 2+ and lack of HER2 amplification by fluorescence in situ hybridization (FISH) testing. The purpose of the study is to evaluate the safety and initial evidence of efficacy of intratumoral administration of CF33-hNIS-anti-PD-L1 (CHECKvacc) against mTNBC. Oncolytic virus CHECKvacc intratumoral injection is currently undergoing investigation in patients with mTNBC as a single agent (NCT05081492). The patient was enrolled on the clinical trial CHECKvacc for the Treatment of Metastatic Triple Negative Breast Cancer, received a single dose of CHECKvacc, and discontinued the study due to lack of immediate response. We report a case of a patient with mTNBC who was heavily pretreated and presented with extensive dermal metastasis. Two dermal metastasis biopsies in 2021 showed HER2 0 by IHC. The patient received a single dose of CHECKvacc and discontinued the study due to lack of immediate response. Twenty-five days later, the patient received treatment with T-DXd, and her tumor regressed significantly. The patient’s disease-free survival was 10 months (December 2021–October 2022). The sequential treatment with intratumoral injection of CHECKvacc followed by T-DXd may have significant clinical activity in select patients with heavily pretreated mTNBC. ClinicalTrials.gov NCT05081492.
Background
Triple negative breast cancer (TNBC), defined by lack of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2), accounts for approximately 15–20% of all breast cancers and is associated with poor prognosis once the disease becomes metastatic or inoperable. 1 Cytotoxic chemotherapy remains the mainstay of standard-of-care therapy, however, with limited efficacy. Recently, there have been advances in therapeutic choices in selected subpopulations of TNBC, including poly-ADP ribose polymerase (PARP) inhibitors olaparib and talazoparib approved for the treatment of metastatic TNBC (mTNBC) with germline BRCA1 or BRCA2 mutation 2 based on progression-free survival (PFS) benefit only.1,2 Other newer therapies for mTNBC patients include the immune checkpoint inhibitor (ICI) pembrolizumab 3 as first-line therapy in PD-L1-positive mTNBC, and the antibody–drug conjugate (ADC) sacituzumab govitecan (SG) 4 as second-line therapy in mTNBC, which has shown modest overall survival (OS) benefit. Despite recent approvals and availability of new therapeutic options, the median OS of mTNBC remains limited at 18–24 months leaving opportunity for more effective therapies.
Oncolytic viruses (OVs) are natural or genetically modified viruses that can infect and selectively kill cancer cells while leaving normal cells unharmed. OV therapy has been accepted as a standard injectable immunotherapy since talimogene laherparepvec (T-VEC, Imlygic®) approval by the Food and Drug Administration (FDA) and European Medicines Agency for melanoma therapy in 2015. With the maturation of recombinant DNA technology, recent advances in viral cellular and genomic structure, as well as a better understanding of the interplay of tumor initiation, evolution, and host immunity have provided the foundation for using OVs as cancer therapy. 5 At least nine different families of viruses, including both DNA and RNA viruses, have been successfully transitioned from preclinical studies into early phase clinical trials, 5 and several of those are focused on breast cancer (Table 1). CF33-hNIS-anti-PD-L1 (CHECKvacc) is a novel chimeric orthopoxvirus encoding two transgenes: human sodium-iodide symporter (hNIS) and anti-PD-L1 with robust anti-cancer activity in TNBC xenografts. TNBC cells infected with this virus express functional hNIS and anti-PD-L1 proteins. 6 Virus-encoded hNIS allows tracking of virus by 99mTc single-photon emission computed tomography. Studies in mouse models have shown that CHECKvacc is safe and is effective against TNBC xenografts at doses as low as 1 × 103 plaque forming unit (PFU). 6 A first-in-human phase I, single-center, single-arm clinical trial evaluating the safety and tolerability of CHECKvacc intratumoral injection in patients with mTNBC is currently ongoing (NCT05081492).
Clinical trials evaluating OV therapy in breast cancer.

CR, complete response; mo, month; mPF, median progression-free survival; ORR, objective response rate; OV, oncolytic virus; PR, partial response; RCB, residual cancer burden; SD, stable disease; SBRT, stereotactic body radiotherapy.
Trastuzumab deruxtecan (T-DXd) is a novel HER2-targeted ADC with a topoisomerase I inhibitor payload, which has shown promising clinical activity and has recently been granted FDA approval for treatment of HER-2 amplified metastatic breast cancer (BC) 10 and HER2-low BC, which is defined by HER2 score of 1+ or 1+ on immunohistochemistry (IHC) and negative by fluorescence in situ hybridization (FISH). In the phase III DESTINY-Breast04 trial targeting HER-2 low BC, median PFS was 9.9 months in the T-DXd group and 5.1 months in the physicians’ choice group (HR, 0.50; p < 0.001), and OS was 23.4 months and 16.8 months, respectively (HR, 0.64; p = 0.001). 11 T-DXd resulted in an objective response rate (ORR) of 53.3% in contrast with 16.3% in the physician’s choice chemotherapy arm, 11 respectively. This trial showed that T-DXd provided an efficacious, novel, targeted treatment for advanced HER2-low BC patients who have progressed on prior lines of therapy.
Herein, we report a case of a patient with mTNBC who was heavily pretreated and received a single dose of CHECKvacc intratumoral injection, followed by subsequent treatment with T-DXd, and achieved a remarkable response.
Case presentation
Exceptional responder following treatment on the clinical trial, CHECKvacc for the Treatment of Metastatic Triple Negative Breast Cancer (NCT05081492): Patient is a 74-year-old woman with a history of mTNBC with extensive dermal metastasis who progressed after multiple lines of therapy including neoadjuvant dose dense adriamycin, cyclophosphamide, and paclitaxel (ddAC-T); adjuvant capecitabine; radiation therapy; pembrolizumab (pembro); pembro + sacituzumab govitecan (SG); pembro + p53MVA vaccine; and eribulin. Her skin biopsy on 15 September 2020 showed TNBC with HER2 1+ by IHC. A later skin biopsy on 9 September 2021 confirmed TNBC with HER2 0 by IHC. The patient enrolled in the clinical trial NCT05081492. Again, baseline biopsy re-confirmed TNBC with HER2 0 by IHC and negative for FISH. The patient received one dose of CHECKvacc at 1 × 105 PFU on 12/2/21 (cycle 1 day 1, C1D1). Tumor biopsy was taken 24-h posttreatment (C1D2). On 16 December 2021 (C1D15), it was noted that the injected area appeared to be more erythematous, but no tumor regression was observed. The patient elected to end treatment on the study, and subsequently received T-DXd on 27 December 2021. The patient experienced a significant reduction in dermal metastasis and ultimately achieved complete regression of dermal metastasis as documented with photography in March 2022 (Figure 1). A significant reduction in cancer antigen 15-3 (CA 15-3) was documented over the course of treatment (Figure 2). Patient remained disease-free for 10 months, but recurrence of skin metastasis with increased tumor marker CA 15-3 was observed in November 2022 and patient went on to receive subsequent therapies.

Baseline and posttreatment dermal metastases. Complete resolution of dermal metastasis after 1 dose of CHECKvacc on December 2 2021 and T-DXd from 27 December 2021 to 25 March 2022 was documented with photography.
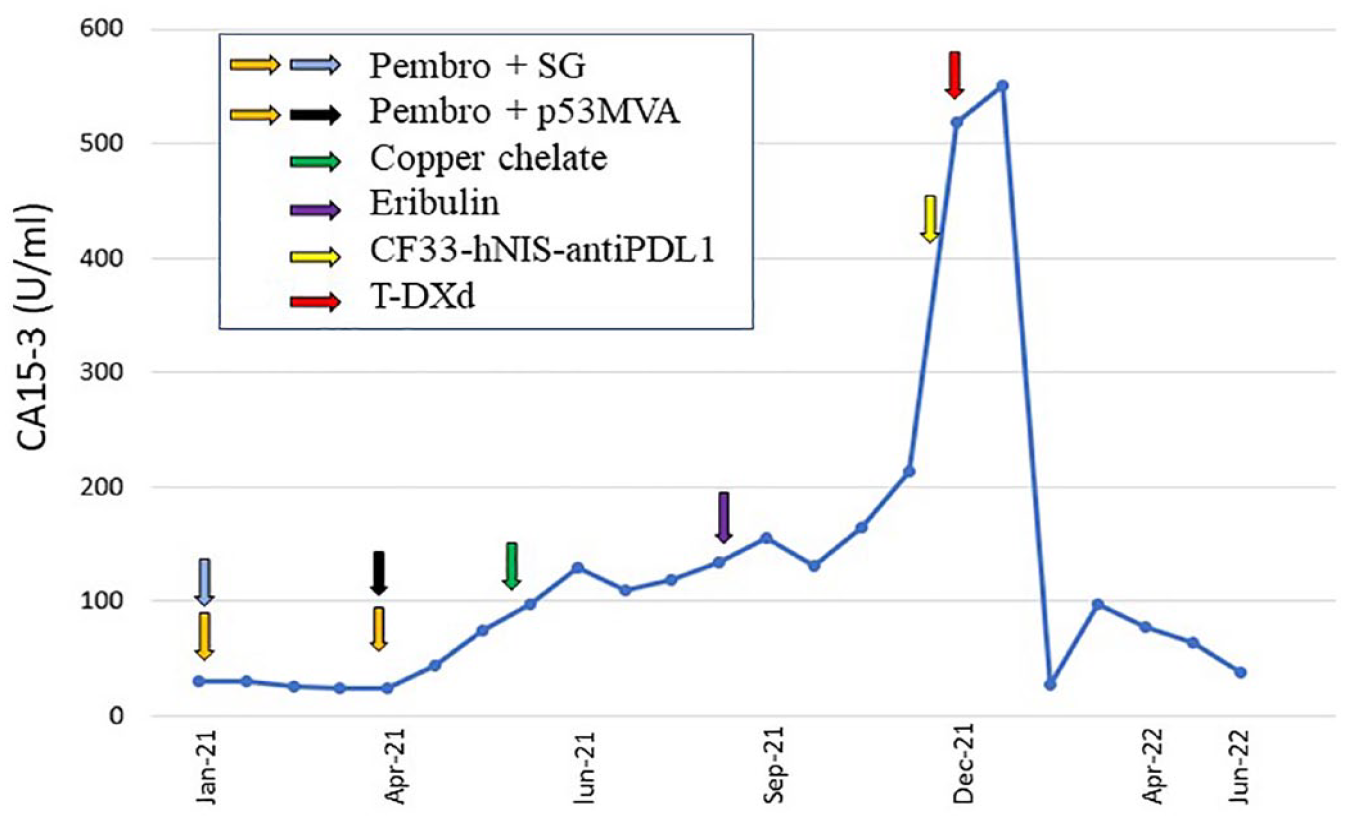
CA 15-3 tumor antigen levels (U/mL) and treatment regimen.
Detailed treatment history
This is a 74-year-old female patient who initially presented with a palpable left breast mass in February 2019. Biopsy of the mass on 2/19/2019 showed early-stage disease, clinical staging cT2N0M0, infiltrative ductal carcinoma, grade 3, ER 4%, PR negative, HER2 negative. She received neoadjuvant dose dense Adriamycin and cyclophosphamide from 20 March 2019 to 1 May 2019, with a minimal response. She then proceeded to neoadjuvant weekly paclitaxel from 14 May 2019 to 6 August 2019. Carboplatin was added on week 3 (5 June 2019) due to continued lack of response to paclitaxel alone. During this time, the breast mass had increased to 4.9 cm × 4.7 cm × 2.7 cm. She then underwent bilateral mastectomies with sentinel lymph node biopsy on 27 September 2019. Pathology showed residual invasive ductal carcinoma, grade 3, ypT2N0, 4.5 cm, extensive lymphovascular invasion, 40% overall cancer cellularity, 10% of cancer consistent with in situ disease, 0/3 nodes involved, ER negative, PR negative, and HER2 IHC 0. Genetic testing (Invitae) on 8 March 2019 showed variants of uncertain significance (VUS) (BRCA2, c.7759C > T; SMARCE1, c.1115A > C). In October 2019, the patient declined adjuvant radiation. She received four cycles of adjuvant capecitabine from October 2019 to February 2020.
Patient was found to have a left chest wall recurrence in August 2020, with biopsy-proven TNBC. Staging scans in September 2020 were negative for distant metastasis. The patient underwent left chest wall resection and left axillary lymph node dissection on 15 September 2020. Pathology showed metastatic carcinoma in the skin, dermal lymphatics, and skeletal muscle lymphatics. Lymphovascular invasion was present, and 1/10 lymph nodes were positive. Biomarkers were ER negative, PR negative, HER2 1+ by IHC, and negative by FISH. Surgical margins were clear. From 26 October 2020 to 1 December 2020, the patient received radiation to bilateral chest wall, followed by treatment with pembrolizumab from 16 November 2020 to 21 January 2021 with progression of dermal involvement at the right chest wall. SG was added on 25 January 2021, with continued progression of dermal metastasis. Patient then received three cycles of pembrolizumab plus p53MVA vaccine as compassionate use based on NCT02432963, 12 given every 3 weeks from 29 April 2021 to 6 June 9 2021 with progression of disease. Patient resumed SG on 23 June 2021. Due to further disease progression with dermal metastasis involving bilateral chest, upper abdomen, and back, the patient started alternative therapy with oral tetrathiomolybdate oil for copper chelation in June 2021. On 9 September 2021, chest wall skin biopsy confirmed progression of disease, still with TNBC features and HER2 0 by IHC. Next-generation sequencing showed TP53 p.R248Q (32% VAF) and tumor mutation burden (TMB) 6.3 mut/Mb. Patient received eribulin on 16 September 2021 due to progression of disease. On 2 December 2021, the patient enrolled to NCT05081492 at City of Hope. Baseline biopsy reconfirmed TNBC, HER2 0 by IHC. She received cycle 1 day 1 (C1D1) intratumoral injection of CHECKvacc at 1 × 105 PFU on 2 December 2021 (cycle 1 day 1) on 2 December 2021. On 16 December 2021, she was noted to have increased erythema of her dermal lesions. Consequently, the patient elected to end treatment on study. She received the next line of therapy T-DXd on 27 December 2021 and was noted to have an excellent clinical response after initial dose with complete resolution of dermal metastasis documented in March 2022 after four doses of T-DXd (Figure 1). The complete regression was maintained until October 2022 with a total of 13 doses of T-DXd given. A significant reduction in CA 15-3 from peak 550–64.4 U/mL was noted (Figure 2). Patient’s disease-free survival (DFS) was 10 months (December 2021 to October 2022). Patient was noted to have recurrence of skin metastasis with increased tumor marker CA 15-3 in November 2022 and went on to receive subsequent therapies. Additional genomic testing showed a trend of increased TMB and more somatic alterations. Guardant360 ctDNA (November 2022) revealed CCNE1 amplification and TP53 R248Q mutation; TMB 11.48 muts/Mb; and microsatellite instability (MSI)-high not detected. Guardant360 ctDNA (March 2023) showed TP53 R248Q mutation, ATM loss, NOTCH1 E1949fs, MYC amplification, AR amplification, CCNE1 amplification, EGFR amplification, ESR1 amplification, and FGFR1 amplification; TMB 22.97 muts/Mb, and MSI-High not detected. Tempus XT (03/2023) showed TP53 R248Q missense mutation; TMB 5.8 muts/Mb; homologous recombinatoin defect (HRD) positive; PD-L1 <1% by 22C3; with multiple VUS detected: SMC3, BCORL1, WT1, SOX9, HNF1B, BCOR, STAG2, FANCA, and PRKDC.
Increase in tumor immune cell infiltration was noted within 24 h of CHECKvacc injection
Pretreatment and C1D2 tumor biopsies were available for multiplex immunofluorescence staining. There was noticeable increase in T-cell population (CD4+ T cells and CD8+ T cells) cells as well as PD-L1+ cells (Figures 3 and 4) on C1D2 posttreatment biopsy. Whole slide images of segmented tissue and cell phenotypes are shown in Figure 3. The pretreatment tumor microenvironment (TME) displayed a modest density of CD4+ and CD8+ T cells, with few PD-L1+ cells identified in the tumor stroma [Figure 4(a)–(e)], whereas C1D2 TME showed an increase in both CD4+ and CD8+ T cells, as well as an increase of PD-L1+ cells [Figure 4(f)–(j)].
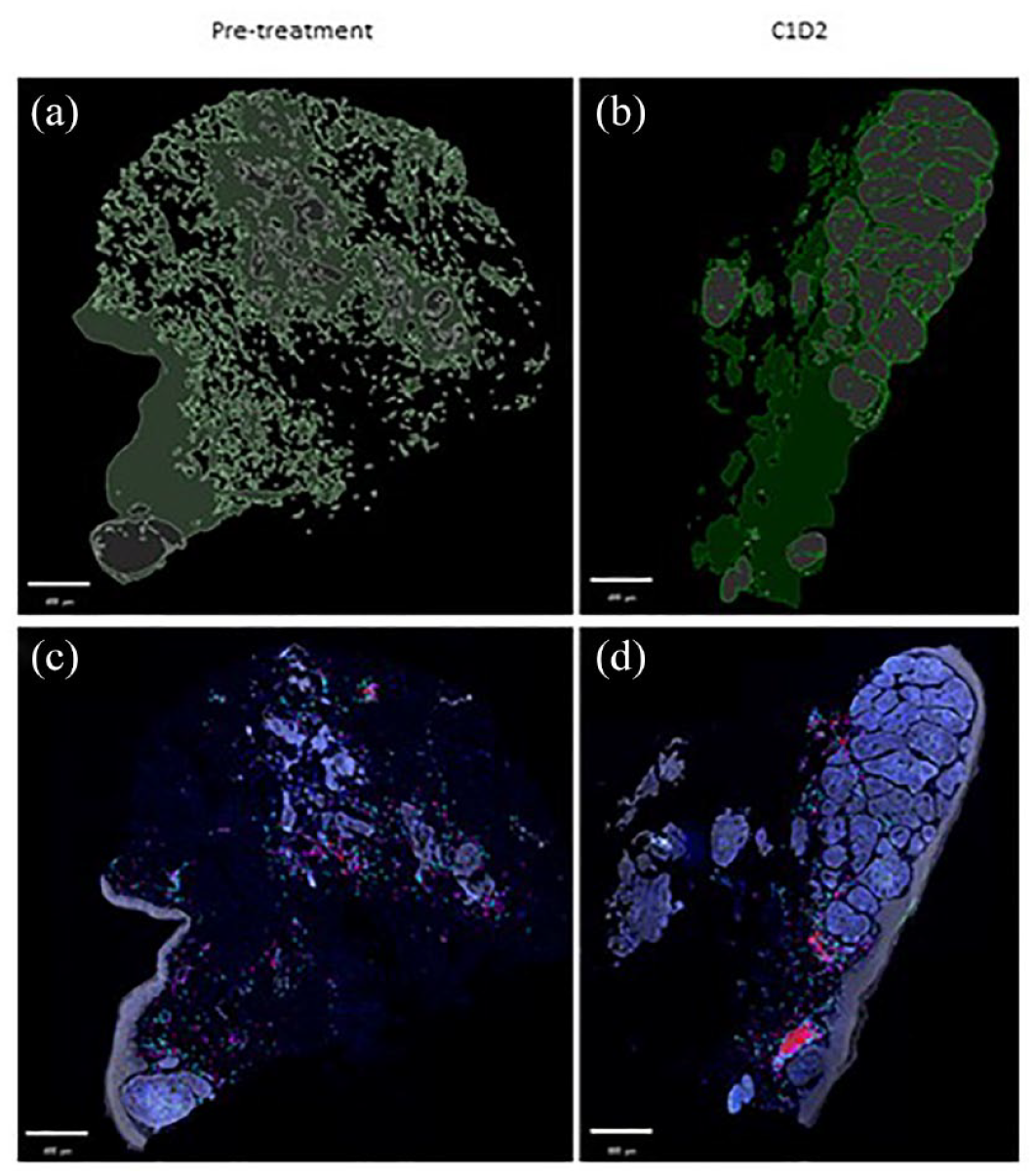
Visualization of immune infiltrates in the tumor microenvironment (pretreatment and 1-day posttreatment). Tumor biopsies from pretreatment and 1-day posttreatment (C1D2) were stained for immune cells (CD3+, CD8+, T cells) and PD-L1+ cells using multiplex immunofluorescence. Tumor stroma (green) and cancer nests (gray) were classified using supervised machine learning (a and b). Cell phenotypes of CD4+ T cells (cyan; CD3+ CD8−), CD8+ T cells (magenta; CD3+ CD8+), and PD-L1+ cells (red; any PD-L1+ cell) were quantified using supervised machine learning (c and d). Whole slide composite images with identified cell phenotypes, pan-cytokeratin staining (gray), and DAPI staining (blue) are depicted. Scale bars are shown.

Increased immune infiltration in CF33-hNIS-anti-PD-L1-treated tumor microenvironment. Patient biopsies were assessed by multiplex IF for changes in T-cell and PD-L1+ cell densities between pretreatment (a–e) and 1-day posttreatment (C1D2; f–j). Composite images of whole tissues were stained for markers CD3 (cyan; b, g), CD8 (magenta; c, h), PD-L1 (red; d, i), pan-cytokeratin (gray), and DAPI (blue). Cell phenotypes of CD4+ T cells (CD3+ CD8−), CD8+ T cells (CD3+ CD8+), and PD-L1+ cells (any PD-L1+ cell) were quantified within the tumor stroma of tissues. The location of higher magnification images is marked by the white box. Densities of identified immune phenotypes were calculated and compared between pretreatment and C1D2 (k–m). Scale bars are shown.
Discussion and conclusion
The patient presented in this case experienced a complete response (CR) to sequential treatment of CHECKvacc and T-DXd after failing multiple lines of prior systemic therapy. While HER2-low expression level was detected in the year 2020, both tumor biopsies in 2021 (first in September and second in December at time of CHECKvacc administration) showed HER2 0 (HER2-zero). After exhausting all the available therapeutic options, the patient was enrolled in our study; however, she self-discontinued therapy after the first dose of virus due to lack of response and lesions appearing more erythematous. Twenty-five days after receiving the OV, the patient went on to receive T-DXd treatment due to historical HER2 1+ in 2020 despite HER2-zero at time of CHECKvacc administration. The decision to receive T-DXd therapy was based on an earlier phase IB trial that showed an initial activity of T-DXd in HER2-low BC reported by Modi et al. 13 In Cohort 3 of the DAISY trial, T-DXd showed an ORR of 29.7%, median PFS of 4.2 month in patients with HER2-zero expression (N = 37). 14 Our patient achieved complete tumor regression which was confirmed by CT imaging. The clinical CR lasted for 7 months (March 2022–October 2022) with a DFS of 10 months. To date, no CR in a patient with HER2-zero metastatic BC receiving T-DXd has been reported. In the DESTINY-Breast04 trial targeting HER-2 low BC, only 3.6% of patients in the T-DXd group achieved CR. 15 No patients achieved CR in the HER2-zero cohort of the DAISY trial, although this could be due to limited sample size (N = 37). Ongoing T-DXd trials in HER2-zero or HER2-ultra low populations may provide more insight (NCT05950945). The activity of single-agent T-DXd in this exceptional responder cannot be ruled out. Precaution needs to be taken when the case report data is interpreted. The current case report is hypothesis-generating and the true impact of CHECKvacc in combination with T-DXd can only be adequately addressed through a well-designed clinical trial. The tumor regression observed in this patient was unlikely to be derived from T-DXd alone based on the available data. It is possible that the OV that was given 25 days before the T-DXd treatment contributed to the overall therapeutic benefit. Because the patient self-discontinued after C1D15, no further longitudinal tumor biopsies were performed, which limited our ability to provide direct evidence. Nevertheless, even with the very limited tumor biopsies (pretreatment and 1-day posttreatment), our data showed positive immune modulation in the TME by the virus treatment. Multiplex staining of the biopsies showed a 2–3-fold increase in the numbers of CD4+ T cells and CD8+ T cells (Figure 4). Likewise, there was an increase in the number of PD-L1+ cells. It is important to note that we observed these immune changes within 24 h of treatment with a very low dose (1 × 105 PFU) of virus. From clinical studies with other poxvirus 16 or T-Vec, 17 we know that it takes several weeks to achieve optimal immune modulation in the TME (CD4+ and CD8+ T-cell population in tumors). Therefore, the fact that we saw a considerable increase in the number of T cells in the tumors within 24 h of virus injection suggests virus-mediated increase in T-cell population at the time of T-DXd treatment (25 days after virus injection) was likely at its optimal timepoint. The observed increase in PD-L1+ cells in the TME after OV treatment agrees with our data in an animal model of TNBC. 18 Also, studies with other OVs have shown similar increases in PD-L1+ cells, however at later time points.17,19 Given the fact that CHECKvacc encodes a functional anti-PD-L1 antibody, it is likely that the virus-encoded anti-PD-L1 may have acted against the elevated PD-L1 and contributed to the anti-tumor immune activation. 6 Furthermore, T-DXd has been found to be synergistic when combined with a checkpoint inhibitor by increasing T-cell activity and upregulating PD-L1 in preclinical murine models 20 and the combination of nivolumab and T-DXd is currently being evaluated in the phase Ib DS8201-A-U105 trial (NCT03523572). A preliminary report showed that the addition of nivolumab did not improve outcomes over historical controls treated with T-DXd monotherapy in patients with metastatic HER2-positive or HER2-low BC. 21 It remains possible that the virus, along with the virus-encoded checkpoint inhibitor (anti-PD-L1), may have contributed to T-DXd exerting its full potential in this patient.
In view of the patient’s complex treatment history and prior exposure to immunotherapy including pembrolizumab and p53MVA, one may speculate that these agents may have impacted the host’s immune composition to subsequent response to CHECKvacc and T-DXd. Due to the limited nature of this case report and lack of peripheral blood mononuclear cells flow cytometry or T-cell receptor analysis, these intriguing temporal immunotherapy impact questions cannot be adequately addressed here. A well-designed, biomarker-enriched prospective trial is required to fully answer these questions. OVs are considered a new class of immunotherapy. Similar to other immunotherapeutics, OVs could also cause ‘pseudo-progression’, especially after local delivery and before measurable clinical benefits are observed.22–24 In oncolytic virotherapy, pseudo-progression is thought to arise from the immune changes within the TME, including infiltration of immune cells and inflammation.25,26 The pseudo-progression may last several weeks before actual regression in tumor is observed. 27 Through their ability to induce immunogenic cell death, OVs are thought to break immune tolerance to tumor cells and induce long-term memory, ultimately reducing the risk of disease recurrence. 28 However, despite anti-tumor response after sequential treatment with CHECKvacc and T-DXd, the subject experienced relapse after 10 months of PFS. The return of disease within a year suggests that any tumor-specific memory induced by the treatments was not potent enough to prevent residual cancer cells from forming tumors. Future studies are necessary to better understand the mechanism of combination therapy with OV and ADCs.
In our patient with HER2-zero expression, the remarkable CR in this heavily pretreated patient with extensive dermal metastasis warrants further investigation. The increase in PD-L1+ cells, CD4+ T cells, and CD8+ T cells in the TME after a single dose of CHECKvacc demonstrated that CHECKvacc may increase immune infiltration and immune activation in the originally immune-cold TME. Pretreatment with CHECKvacc may have contributed to the exceptional response to T-DXd in this patient. This observation warrants further studies to determine if CHECKvacc could be used sequentially or in combination with T-DXd in treatment-refractory HER2-zero TNBC patients.
Materials and methods
Patient was treated through a phase I study of intratumoral administration of CHECKvacc in patients with advanced or metastatic TNBC. This study began in October 2021 and is currently undergoing investigation in patients with mTNBC as a single agent with City of Hope Institutional Review Board (IRB) approval in accordance with the World Medical Association Declaration of Helsinki, International Conference on Harmonization Good Clinical Practice guidelines, and the U.S. code of federal regulations. Informed consent was signed by patient prior to study entry. This study is registered at the ClinicalTrials.gov under number NCT05081492.
Eligibility criteria
For the phase I clinical trial, main eligibility criteria were: TNBC defined as ER and PR ⩽10% by IHC and HER2 negative, per ASCO/CAP guidelines; ECOG ⩽2; measurable disease by RECIST 1.1; patients must have progressed or been intolerant of at least two prior lines of systemic chemotherapy for advanced/metastatic disease; and patient must have superficial tumor (cutaneous, subcutaneous), breast lesion or nodal metastases amenable to safe repeated intratumoral injections per treating physician and interventional radiologist review. Main exclusion criteria included chemotherapy, biological therapy, immunotherapy, or investigational therapy within 14 days prior to Day 1 of protocol therapy; major surgery or radiation therapy within 28 days of study therapy; vaccination within 30 days of first study injection; another malignancy within 3 years, except non-melanomatous skin cancer; and clinically unstable brain metastases. Patients may be enrolled with a history of treated brain metastases that are clinically stable for ⩾4 weeks prior to the start of study treatment.
Multiplex immunofluorescence tumor analysis
Formalin-fixed paraffin-embedded (FFPE) tissue specimens were cut at 4 µm sections and baked onto glass slides. FFPE slides were then deparaffinized in 10-min xylene washes and rehydrated in washes with decreasing ethanol concentration. Heat-induced antigen retrieval was performed using AR9 buffer, 10× (pH 9) (AR9001KT, Akoya Biosciences, Marlborough, MA, USA) in a microwave oven for 20 min. Blocking was performed for 10 min using Antibody Diluent with Background-Reducing Components (S302283-2, Agilent, Santa Clara, CA, USA) to minimize non-specific background staining. Primary antibodies were incubated for 1 h on a shaker at room temperature followed by a 10-min incubation of horseradish peroxidase (HRP)-conjugated secondary antibody (Mach 2 Rabbit or Mouse HRP-Polymer) (RHRP520 L or MHRP520 L, Biocare Medical). Immunofluorescent labeling of antibodies was achieved using the OpalTM 7-color fluorescence IHC Kit (Akoya Biosciences, Marlborough, MA) at a 1:100 dilution for 10 min. To perform multicolor immunofluorescent staining, the slides were serially stained with the microwave incubation acting to remove previous antibodies while simultaneously exposing the next epitope of interest. After staining the final marker, cell nuclei were stained with DAPI (FP1490, Akoya Biosciences) and the slides were mounted with ProLong Gold Antifade Reagent (P36930, ThermoFisher Scientific, Waltham, MA, USA). Primary antibody and opal combinations used were used in the following staining order: CD8 (Leica, Clone 4B11) Opal 520, PD-L1 (Abcam, Clone SP142) Opal 620, CD3 (Dako, polyclonal) Opal 540, pan-cytokeratin (Dako, Clone AE1/AE3) Opal 540, Akoya Biosciences, Marlborough, MA, USA.
Tissue slides were scanned using the Vectra 3.0 automated quantitative pathology imaging system (Akoya Biosciences). After image capture, the MSIs were spectrally unmixed with inForm® tissue analysis software (Akoya Biosciences) and component TIFFs were exported. Using quantitative pathology and bioimage analysis (QuPath) software, the component TIFFs were stitched for tissue segmentation, cell segmentation, and cell phenotyping using supervised machine learning. 29 Identified cell phenotypes were consolidated as follows: CD4+ T-cell (any CD3+ CD8-T-cell), CD8+ T-cell (any CD8+ T-cell), PD-L1+ -T-cell (any PD-L1+ T-cell). Counts of PD-L1+ cells and T-cell phenotypes were not mutually exclusive. Cell densities were calculated by dividing cell phenotype counts by the total area of segmented tissue and plotted using GraphPad Prism 9.4, GraphPad Software, Boston, MA.
