Abstract
Background:
Invasive pulmonary aspergillosis (IPA) can negatively impact cancer patients’ survival. It remains uncertain whether IPA’s impact on patient outcomes varies by treatment approach in advanced lung cancer.
Objectives:
To explore the association between IPA and outcomes in patients with advanced lung cancer receiving different treatments.
Design:
A retrospective cohort study.
Methods:
We enrolled patients with advanced-stage lung cancer between 2013 and 2021 at a college hospital in Taiwan and used the 2021 European Organization for Research and Treatment of Cancer/Mycoses Study Group Education and Research Consortium consensus for IPA diagnosis. Multivariable logistic regression was used to identify the IPA risk factors. We compared overall survival (OS) and postgalactomannan (GM) test survival between the IPA and control groups using multivariable Cox proportional hazards regression and the Kaplan–Meier method with propensity score matching (PSM).
Results:
Among 2543 patients with advanced-stage lung cancer, 290 underwent a GM test, of which 34 (11.7%) were diagnosed with IPA. Patients undergoing chemotherapy (HR = 4.02, p = 0.027) and immunotherapy [hazard ratio (HR) = 3.41, p = 0.076] tended to have IPA. Compared to the control group, the IPA group had shorter median OS (14.4 versus 9.9 months, p = 0.030) and post-GM test survival (4.5 versus 1.9 months, p = 0.003). IPA was associated with shorter OS (log-rank p = 0.014 and 0.018 before and after PSM, respectively) and shorter 1-year and 2-year survival post-GM test (HR = 1.65 and 1.66, respectively). Patients receiving chemotherapy or immunotherapy had a shorter post-GM test survival if they had IPA.
Conclusions:
IPA tended to be diagnosed more frequently in patients receiving chemotherapy or immune checkpoint inhibitors. Patients diagnosed with IPA are associated with shorter survival. Larger cohort studies are needed to verify the observations.
Introduction
Invasive pulmonary aspergillosis (IPA) is a severe pulmonary Aspergillus infection commonly found in patients with immune deficiency, critically ill status, structural lung disease, or severe viral infections.1,2 Even with adequate antifungal treatment, IPA is associated with poor prognosis in different populations, including critically ill patients with COVID-19 3 or influenza4,5 and HIV patients. 6 Traditionally, the IPA diagnosis was based on the presence of septate, branching fungal hyphae with lung tissue invasion, and positive culture results for Aspergillus. 7 However, lung biopsy is an invasive procedure that increases the risk of complications for patients with structural lung disease, 8 limiting the availability of studies on IPA prevalence in patients with pulmonary disease and its effect on their survival. Currently, nontissue-based methods, such as the galactomannan (GM) test, are widely used clinically to diagnose IPA with different specimen types for various patient populations. However, the sensitivity and specificity of IPA diagnosis vary based on different GM levels. 9 In order to overcome this difficulty, the 2021 European Organization for Research and Treatment of Cancer (EORTC)/Mycoses Study Group Education and Research Consortium (MSGERC) Consensus Definitions of Invasive Fungal Diseases established a diagnostic cutoff value for IPA, 9 enabling investigations into IPA prevalence and survival in patients with pulmonary disease.
Lung cancer is among the most prevalent and leading causes of cancer-related deaths worldwide. 10 While many new tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICIs) have been developed for advanced-stage lung cancer, 11 the overall 5-year survival rate for patients with lung cancer and distant metastasis is only about 7%. 12 Based on the epidemiology study, IPA is most frequently found in those with lung cancer among patients with solid cancer.13,14 From a clinical perspective, patients with lung cancer may have an increased risk of IPA due to factors such as lung tissue destruction and cavitation caused by invading cancer cells, as well as immune deficiency resulting from cancer treatment. A previous epidemiological investigation using the Taiwan Cancer Registry database found a crude IPA incidence of 0.12 per 100 patients, and patients with aspergillosis were also associated with higher mortality. 15 To our knowledge, few biology-based and treatment-stratified studies have explored IPA’s prevalence in patients with advanced-stage lung cancer and their survival or IPA risk factors. While chemotherapy is a well-known risk factor for IPA, 9 there is limited data available on the association between the use of ICIs and the risk of IPA.16,17 No published study has demonstrated an association between TKIs and IPA in patients with lung cancer. The impact of IPA on the survival of lung cancer patients receiving these cancer treatments also remains unclear.
In the present study, we hypothesized that advanced lung cancer with IPA had a shorter survival after IPA diagnosis than those without IPA and that IPA incidence may vary according to treatment strategy. Therefore, we performed a retrospective cohort study using a long-term advanced lung cancer cohort in a college hospital in south Taiwan to investigate the association between IPA and survival and identify risk factors for IPA in patients with advanced lung cancer.
Methods
Patient enrollment and study design
This study was conducted at the National Cheng Kung University Hospital (NCKUH), a college hospital and tertiary referral center for 1.86 million inhabitants in Tainan City, Taiwan, as of 2022. Patients aged ⩾20 years with newly histologically or cytologically diagnosed advanced-stage lung cancer (stage 3B, 3C, and 4) were enrolled between 1 January 2013 and 31 December 2021. Patients were excluded if they: (1) did not undergo the GM test in blood or bronchoalveolar lavage (BAL) after their lung cancer diagnosis; (2) had a previous pulmonary Aspergillus infection; (3) had missing diagnostic or therapeutic information about lung cancer. The remaining patients are referred to as the ‘primary cohort’. Each patient with IPA in the primary cohort was matched 1:2 by age, sex, cancer stage, cancer histology, and treatment lines with patients without IPA. These patients are referred to as the ‘matched cohort’.
IPA diagnosis
The IPA diagnosis was based on the GM test’s optical density index (ODI). GM is a polysaccharide with a mannose backbone and variable galactofuran side chains that is the major component of the cell wall for Aspergillus spp. 18 IPA was diagnosed based on the diagnostic criteria in the 2021 EORTC/MSGERC Consensus Definitions of Invasive Fungal Diseases. 9 Briefly, patients with a serum GM ODI ⩾ 1.0, BAL GM ODI ⩾ 1.0, or a combination of serum GM ODI ⩾ 0.7 and BAL GM ODI ⩾ 0.8 were diagnosed with IPA. The specificity for IPA was 0.90–0.94 and 0.94–0.95 when using a GM ODI > 1.0 in serum and BAL samples, respectively. 9 The GM ODI was determined using the Platelia Aspergillus Test (Bio-Rad Laboratories, Marnes-la-Coquette, France). Medical charts, chest radiologic examinations, and laboratory results were reviewed to diagnose IPA for every patient in the cohort. Patients with airway symptoms and suggestive lung lesions of IPA such as macronodule, cavitary lesion, or consolidation with halo sign 19 are candidates for GM tests in our hospital. We usually performed BAL to diagnose IPA. However, for patients at a high risk of bronchoscopy-related complications, we instead checked the serum GM level. Positive GM test result is not repeatedly tested due to limited laboratory availability in our hospital. Instead, there are restrictive strategies for initiating antifungal treatment in IPA patients, using a high cutoff value for GM, confirming chest imaging, and requiring infectious disease specialist review for every antifungal prescription. Additionally, GM testing is not routinely monitored in patients receiving antifungal treatment for IPA.
Data collection, variables, and outcomes
Patient information, such as age, sex, cancer stage, histology, types and lines of lung cancer treatment, survival status, GM test sample and value, and known IPA risk factors, were collected from electronic medical records in the NCKUH database. The American Joint Committee on Cancer’s stage classification for lung cancer (eighth edition) was used to stage lung cancer. 20 Cancer histology was classified into adenocarcinoma (adeno), squamous cell carcinoma (SqCC), small cell lung cancer (SCLC), and others. Steroid use was defined as a patient receiving systemic steroids within 7 days before their IPA diagnosis. Structure lung disease encompasses conditions such as emphysematous lung, bronchiectasis, cavitary lung lesions, and pulmonary fibrosis. Patient survival status was defined as alive, dead, or lost follow-up. This study’s primary outcome was the survival period post-GM test, and its secondary outcomes included 1-year, 2-year, and overall survival (OS).
Statistical analysis
The collected data were presented as number (percentage), mean (standard deviation), or median [interquartile range (IQR)] according to the feature. Independent t-tests and Fisher’s exact tests were used to analyze the continuous and categorical variables, respectively. Multivariable Cox proportional hazards regression analysis was performed to investigate the 1-year and 2-year survival after the GM test or lung cancer diagnosis. Age (⩾65 versus <65), sex (male versus. female), cancer stage (4 versus 3), cancer type (SqCC, SCLC, and other versus adeno), treatment lines (⩾2 and 1 versus 0), cancer treatment (TKI, chemotherapy, ICI, and chest radiotherapy versus without), and IPA risk factors (structure lung disease and prolonged neutropenia) were controlled. Associations were established using hazard ratios (HRs) and 95% confidence intervals (CIs).
Propensity scores were calculated with a logistic regression model including age, sex, cancer histology, cancer treatment, treatment lines, and the duration from lung cancer diagnosis to GM test and used for 1:2 matching between the IPA and no-IPA groups. Kaplan–Meier estimation was used to compare the survival curves for IPA in the primary and matched cohorts. Analyses of subgroups of patients with different cancer histologies and treatments were conducted. Univariate and multivariate logistic regression was conducted to explore IPA risk factors in patients with advanced-stage lung cancer. Odds ratios (ORs) and 95% CIs were used to establish associations. All p values were calculated two-sided, and a p < 0.05 was considered statistically significant. All statistical analyses were conducted using SAS software (version 9.4; SAS Institute, Cary, NC, USA).
Sensitivity analysis
Several sensitivity analyses were conducted to investigate the robustness of the study results. First, because the sensitivity and specificity varied by different GM diagnostic thresholds, we tested the result by changing the GM value cutoff for IPA diagnosis from 0.5 to 1.5. Second, to avoid confounding by previously undiagnosed IPA, we excluded patients with a GM test within the first 30 days of their lung cancer diagnosis. Third, to avoid confounding by untreated status or hospice care for lung cancer, we excluded patients not receiving any cancer-related treatment at the time of their GM test.
Results
Between January 2013 and December 2021, 2543 patients were newly diagnosed with advanced-stage lung cancer. Of these, 2253 did not undergo a GM test after their lung cancer diagnosis, 26 had missing data, and five had a history of IPA. Their removal left 290 patients in the primary cohort (Figure 1). The characteristics of patients in the primary cohort are presented in Table 1. Of the 290 patients who received a GM test, 159 (54.8%) received a BAL GM test, and 190 (65.5%) received a serum GM test. Among them, 34 (11.7%) were diagnosed with IPA according to the EORTC/MSGERC Consensus Definitions, representing 1.3% of the total population (34/2543). Among the 34 IPA patients, 27 received antifungal treatment, while 7 did not. The reasons for not receiving treatment were as follows: three patients died before test results were obtained, two patients were under hospice care, and two patients were not treated based on the physician’s decision. Out of the treated patients, 26 received voriconazole and 1 received amphotericin B. The median (IQR) day to initiate IPA treatment was 1 (1) day. The median survival post-GM test for treated and untreated IPA patients was 73 and 12 days, respectively (Supplemental Table 1). These 34 patients with IPA were matched 1:2 to patients without IPA (matched cohort).

The algorithm for patient enrollment and propensity score matching.
Patient characteristics of the primary cohort.

Fisher exact test and independent t test were used to calculate continuous variables and category variables, respectively.
Adeno, adenocarcinoma; BAL, bronchoalveolar lavage; COVID-19, corona virus disease 2019; CT, chemotherapy; GM, galactomannan; IPA, pulmonary aspergillosis; ICI, immune check point inhibitor; IQR, interquartile range; RT, radiotherapy; SCLC, small cell lung cancer; SD, standard deviation; SMD, standardized mean difference; SqCC, squamous cell carcinoma; TKI, tyrosine kinase inhibitor.
We compared demographic and clinical characteristics of the patients between the IPA group and non-IPA group in the primary cohort (Table 1). Patients with IPA were more likely to have SCLC, receive an ICI, receive chemotherapy, have structural lung disease, and experience prolonged neutropenia. Only a few patients in this cohort contracted influenza or COVID-19. The median OS was shorter in patients with IPA than without IPA (9.9 months versus 14.4 months; p = 0.030), as was the median time from the GM test to death (1.9 months versus 4.5 months, p = 0.003). In the matched cohort, there were no significant differences in age, sex, cancer stage, cancer histology, lines of treatment, or time from cancer diagnosis to GM test between patients with and without IPA (Supplemental Table 2).
Predictors of IPA in lung cancer patients
We then performed univariable and multivariable logistic regression analyses using the primary cohort to identify risk factors for IPA (Table 2). In the univariable analysis, the crude ORs for chemotherapy and ICI were 2.81 (95% CI: 1.37–5.70) and 3.98 (95% CI: 1.31–12.14), respectively. In the multivariable analysis, the adjusted OR for chemotherapy was 4.02 (95% CI: 1.17–13.83). Structural lung disease and prolonged neutropenia were associated with an increased IPA risk. There was a trend of increased IPA risk for patients receiving ICIs (OR = 3.41, 95% CI: 0.88–13.18, p value = 0.076).
Logistic regression analysis for the risk factors of invasive pulmonary aspergillosis in enrolled patients.

Adeno, adenocarcinoma; CT, chemotherapy; CI, confidence interval; COVID-19, corona virus disease 2019; GM, galactomannan; HR, hazard ratio; ICI, immune check point inhibitor; IPA, invasive pulmonary aspergillosis; Ref., reference; RT, radiotherapy; SCLC, small cell lung carcinoma; SqCC, squamous cell carcinoma; TKI, tyrosine kinase inhibitor.
Survival analysis
By Kaplan–Meier survival analyses, we compared the survival between the IPA group and non-IPA group in the primary and matched cohorts respectively (Figure 2). IPA was associated with shorter survival post-GM test in both the primary (log-rank p = 0.014) and matched (log-rank p = 0.018) cohorts. There was a trend toward shorter OS for patients with IPA in the primary cohort (log-rank p = 0.056). We further performed the subgroup analysis of different cancer treatments (Supplemental Figure 1). Among patients receiving chemotherapy, those with IPA had shorter post-GM test survival (before PS matching log-rank p = 0.027; after PS matching log-rank p = 0.004); there was no significant difference in OS. Among patients receiving ICIs, those with IPA had shorter OS after PS matching (log-rank p = 0.049) and shorter survival post-GM test (before PS matching log-rank p = 0.033; after PS matching log-rank p = 0.014). There were no significant differences in survival among patients receiving TKIs or with different cancer histologies (Supplemental Figures 2–4).
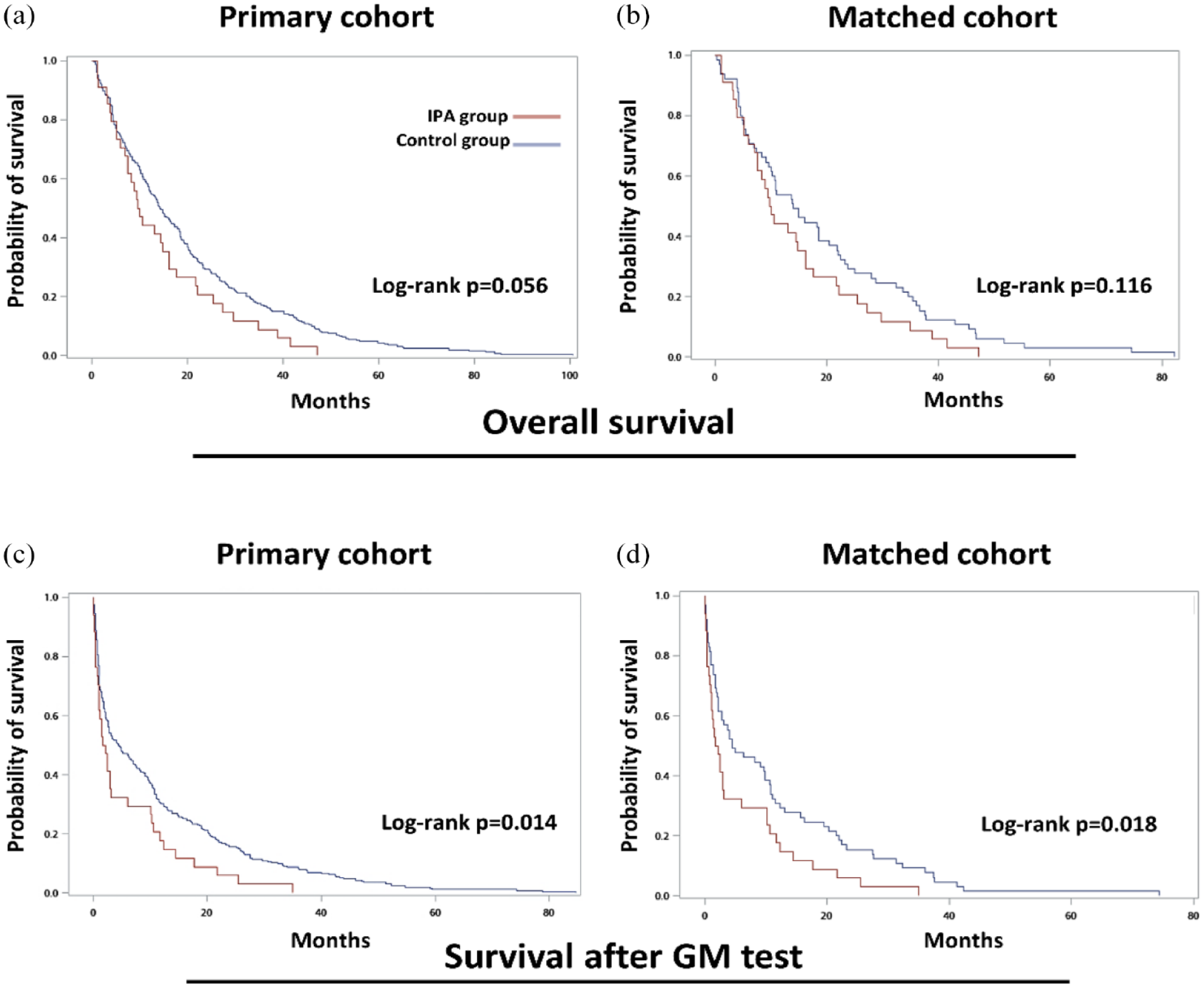
The Kaplan–Meier plot and log-rank test for survival in the IPA and control groups. Overall survival for (a) all patients and (b) PS-matched patients. Survival after GM test for (c) all patients and (d) PS-matched patients.
Using multivariable Cox proportional hazards regression analysis, IPA was an independent poor prognostic factor for 1-year (HR = 1.65, 95% CI: 1.13–2.42) and 2-year (HR = 1.66, 95% CI: 1.19–2.31) survival post-GM test (Table 3). However, IPA was not an independent prognostic factor for shorter 1-year or 2-year OS. In the subgroup analyses of patients with different cancer histologies or who received treatment, patients with IPA receiving chemotherapy or ICIs had shorter survival post-GM test, while patients receiving ICIs also had a shorter 2-year OS (Supplemental Table 3).
Multivariate Cox proportional hazards regression analysis for the survival of enrolled patients.
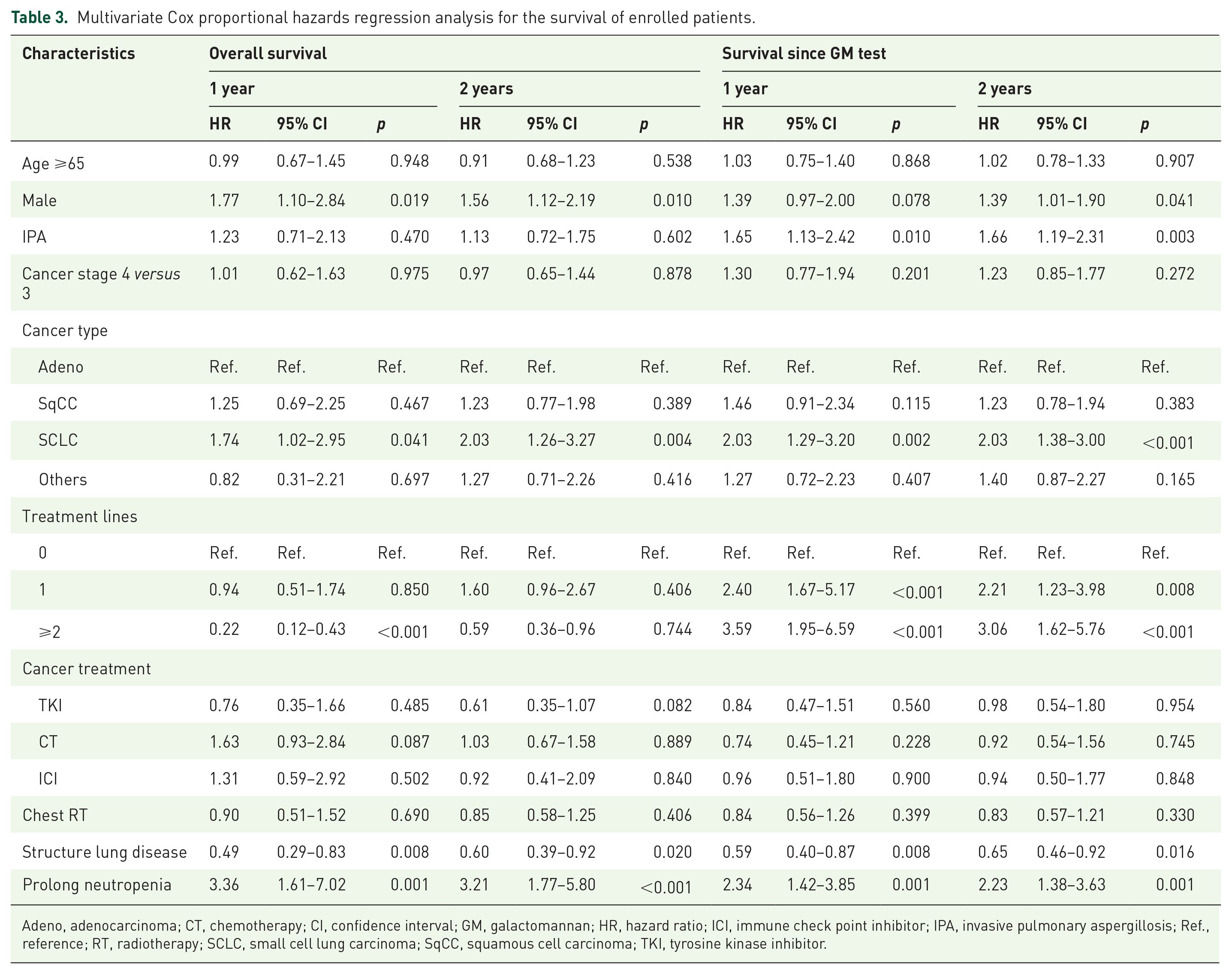
Adeno, adenocarcinoma; CT, chemotherapy; CI, confidence interval; GM, galactomannan; HR, hazard ratio; ICI, immune check point inhibitor; IPA, invasive pulmonary aspergillosis; Ref., reference; RT, radiotherapy; SCLC, small cell lung carcinoma; SqCC, squamous cell carcinoma; TKI, tyrosine kinase inhibitor.
Sensitivity analysis
Firstly, we conducted sensitivity analyses by adjusting the GM diagnostic criteria for IPA (Supplemental Table 4). For 1-year survival post-GM test, IPA was an independent negative prognostic factor before adjusting the BAL or serum GM level to 1.0. IPA remained an independent negative prognostic factor for 2-year survival post-GM test even after adjusting the BAL or serum GM level from ⩾0.5 to ⩾1.5. Secondly, after excluding patients receiving the GM test within 30 days of their lung cancer diagnosis, the HRs for 1-year and 2-year survival post-GM test were 1.71 (95% CI: 1.12–2.62) and 1.75 (95% CI: 1.18–2.58), respectively (Supplemental Table 5). Finally, after excluding patients not receiving cancer treatment at the time of their GM test, the HRs for 1-year and 2-year survival post-GM test were 1.91 (95% CI: 1.12–3.24) and 2.12 (95% CI: 1.29–3.48), respectively (Supplemental Table 6).
Discussion
Our study showed that mean OS and post-GM test survival were shorter in patients with lung cancer and IPA. Multivariable Cox proportional hazard regression analysis was performed to control for age, sex, cancer histology, cancer stage, cancer treatment, structural lung disease, and neutropenia. We found that patients diagnosed with IPA had lower 1-year and 2-year survival after GM testing, confirmed by Kaplan–Meier estimation after propensity score matching. In the subgroup analyses, patients receiving chemotherapy or ICIs had a shorter survival post-GM test if they had IPA, contrasting with patients receiving TKIs. Other associated risk factors included neutropenia and structural lung disease.
The prevalence of IPA in our advanced lung cancer cohort with GM testing was 1.3%. Only a few published studies have examined the epidemiology of pulmonary aspergillosis in patients with lung cancer. Rønberg et al. conducted a retrospective single hospital cohort study of 1200 Danish patients with suspected lung cancer using chest imaging and the pathology, microbiology, and serology criteria of the European Society for Clinical Microbiology and Infectious Diseases and European Respiratory Society to diagnose chronic pulmonary aspergillosis (CPA). They found that 16 of 1200 (1.6%) patients met the CPA diagnosis criteria. 21 Chen et al. 15 showed that the incidence rate of pulmonary aspergillosis among male and female patients with all stages of lung cancer was 12.52 and 7.53 per 10,000 person-years, respectively. In our study, the prevalence may be underestimated since only 290 of 2543 advanced lung cancer patients received GM testing. Further studies are needed to estimate IPA’s prevalence in patients with lung cancer accurately.
We found that patients with advanced lung cancer who underwent chemotherapy, had structural lung disease, or had prolonged neutropenia were at increased IPA risk, consistent with previous studies in other populations. 7 While not statistically significant, there was a trend toward increased IPA risk for patients receiving ICIs. A recent review article and case report also emphasized that immunotherapy may induce infection complications due to dysregulated immunity.15,16 The European Society of Clinical Microbiology and Infectious Diseases proposed a consensus statement that immunosuppressive therapy to treat immune-related adverse events contributes to the risk of opportunistic infections compared to using ICIs alone. 22 In our study, there was only a nonsignificant trend in increasing IPA risk with steroid use. However, there was increasing evidence of a second mechanism contributing to ICI-associated infection independent of immunosuppression. In some cohort studies, immunosuppressive agent uses, or immunosuppressive diseases such as diabetes mellitus were not risk factors for post-ICI infection.23–25 These infections were referred to as immunotherapy infections due to dysregulated immunity, where inhibiting immune checkpoints contributes to an excessive host immune response by counterintuitively favoring the pathogen. To our knowledge, no published studies have shown an association between different treatment strategies and IPA in patients with lung cancer. Further studies with larger cohorts investigating the interaction mechanism between ICI treatment effectiveness and pulmonary aspergillosis are needed.
GM is now widely used to diagnose IPA in clinical patients. Many published studies support using serum or BAL GM tests to diagnose IPA.26,27 However, there are some disadvantages to using GM. First, sensitivity and specificity to IPA vary by GM ODI cutoffs. 9 Nevertheless, our study results remained robust after adjusting the GM ODI cutoff from 0.5 to 1.5. Second, in the past, many medical products, such as gluconate or β-lactam antibiotics, were fermented by Aspergillus spp. 28 A false positive test result may be made after oral or parenteral administration of these products. However, the production of these products has been revised to reduce GM.29,30 The risk of false positive results is minimal because we only enrolled patients treated at our hospital between 2013 and 2021.
We observed a trend of shorter OS and a statistically significant difference in survival post-GM test in patients with advanced-stage lung cancer and IPA. IPA is a poor prognostic factor for populations with different diseases.3,4,6,31 There are several possible reasons for shorter survival in these patients. First, IPA is a sign of compromised patient immunity. The immune cells killing Aspergillus also play a role in host immunity against lung cancer.32,33 Second, metabolites from Aspergillus modulate the host’s immunity. 34 Gliotoxin is one mycotoxin secreted by Aspergillus, and previous studies have shown that it kills dendritic cells to inhibit the proliferation of cluster of differentiation 8 (CD8)+ T cells. 35 Gliotoxin also blocks the activation of nuclear factor kappa B (NFκB) in CD8+ T cells by preventing NFκB inhibitor alpha degradation to inhibit cytotoxic T cell-mediated cytotoxicity. 36 Interestingly, our subgroup analysis showed that patients receiving ICIs had shorter survival after GM testing.
This study had several limitations. First, the patients were retrospectively enrolled at a single center in Southern Taiwan; the sample size was small, and the prevalence of IPA was low in our cohort. Therefore, the results should be generalized with caution. Second, the decision to perform a GM test was based on clinical assessment. Since patients with airway symptoms and causal lung lesions are more likely to undergo GM testing, our study may have overestimated the prevalence of IPA in patients with advanced lung cancer. Third, the IPA diagnosis was based on the GM test rather than microbiologic and pathologic evidence, and some patients did not receive both serum and BAL GM tests. GM’s sensitivity and specificity varied with different ODI cutoffs and sample types. Nevertheless, the results remained robust in sensitivity analyses, changing the serum and BAL GM ODI from 0.5 to 1.5. Fourth, we did not include the treatment effect of IPA in the survival analysis.
Conclusion
In this study, IPA was detected in 1.3% of patients with advanced lung cancer and was associated with shorter postdiagnosis survival, especially in patients receiving chemotherapy or ICIs. Larger cohort studies are needed to identify high-risk patients for IPA and evaluate the impact of IPA on the survival of lung cancer patients receiving different treatments.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231198454 – Supplemental material for Navigating the challenges of invasive pulmonary aspergillosis in lung cancer treatment: a propensity score study
Supplemental material, sj-docx-1-tam-10.1177_17588359231198454 for Navigating the challenges of invasive pulmonary aspergillosis in lung cancer treatment: a propensity score study by Chin-Wei Kuo, Chien-Yu Lin, Sheng-Huan Wei, Yun-Tse Chou, Chian-Wei Chen, Jeng-Shiuan Tsai, Po-Lan Su and Chien-Chung Lin in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
