Abstract
Molecular imaging, such as positron emission tomography (PET), is increasingly used as biomarker to predict and assess treatment response in breast cancer. The number of biomarkers is expanding with specific tracers for tumour characteristics throughout the body and this information can be used to aid the decision-making process. These measurements include metabolic activity using [18F]fluorodeoxyglucose PET ([18F]FDG-PET), oestrogen receptor (ER) expression using 16α-[18F]Fluoro-17β-oestradiol ([18F]FES)-PET and human epidermal growth factor receptor 2 (HER2) expression using PET with radiolabelled trastuzumab (HER2-PET). In early breast cancer, baseline [18F]FDG-PET is frequently used for staging, but limited subtype-specific data reduce its usefulness as biomarker for treatment response or outcome. Early metabolic change on serial [18F]FDG-PET is increasingly used in the neo-adjuvant setting as dynamic biomarker to predict pathological complete response to systemic therapy, potentially allowing de-intensification or step-up intensification of treatment. In the metastatic setting, baseline [18F]FDG-PET and [18F]FES-PET can be used as biomarker to predict treatment response, in triple-negative and ER-positive breast cancer, respectively. Metabolic progression on repeated [18F]FDG-PET appears to precede progressive disease on standard evaluation imaging; however, subtype-specific studies are limited and more prospective data are needed before implementation in clinical practice. Even though (repeated) [18F]FDG-PET, [18F]FES-PET and HER2-PEt all show promising results as biomarkers to predict therapy response and outcome, for eventual integration into clinical practice, future studies will have to clarify at what timepoint this integration has to optimally take place.
Keywords
Introduction
The prediction and assessment of treatment response is an essential part of the clinical care of patients with breast cancer. Expression of the oestrogen receptor (ER) and human epidermal growth factor receptor 2 (HER2), often measured by immunohistochemistry (IHC), is used as a reference standard to predict response to endocrine- and HER2-targeted treatment, facilitating the selection of patients for these treatments. However, predicting response to systemic therapy based on IHC results is not ideal, because lack of endocrine-responsiveness can occur despite ER positivity, 1 whereas responsiveness to HER2-targeting antibody–drug conjugates (ADCs) has been observed in patients considered to have a HER2-negative tumour. 2 To select patients for and predict response to local therapy, it is essential to know the characteristics and the extent of the disease. The latter is normally done using standard imaging techniques, including ultrasound, computed tomography (CT), magnetic resonance imaging (MRI) and/or [18F]fluorodeoxyglucose positron emission tomography ([18F]FDG-PET). These standard imaging techniques, with the exception of ultrasound, are also used for response assessment.3–5
Usually, response is assessed using the Response Evaluation Criteria for Solid Tumours (RECIST), but they are not universally applicable. 6 In fact, RECIST response cannot be assessed in bone metastases, which is the most prevalent site of metastasis, especially in ER-positive breast cancer. 7 The use of other response criteria (e.g. iRECIST, PERCIST and RANO) is mainly limited to clinical trials.8–10
It would therefore be valuable to have additional biomarkers that can improve treatment response prediction and assessment and provide a rationale for informed de-intensification of surgical and systemic treatment in early- and metastatic breast cancer. This would be especially valuable for new treatment options, such as ADCs, cyclin-dependent kinase 4/6 inhibitors or immunotherapy, that are often accompanied by higher costs and toxicity.
PET imaging is increasingly used to non-invasively measure tumour characteristics throughout the body with an expanding array of tracers. These measurements include tumour metabolism using [18F]FDG-PET, ER expression using 16α-[18F]Fluoro-17β-oestradiol ([18F]FES)-PET and HER2 expression using PET with radiolabelled trastuzumab (HER2-PET). 11 In addition to the standardized uptake value (SUV) of these tracers, which is used as a proxy for the measured tumour characteristic, for [18F]FDG-PET volumetric metrics are often reported either alone as metabolic tumour volume or combined with metabolic activity as total lesion glycolysis (TLG). Baseline scans can provide information on disease extent and presence of tumour characteristics. Repeat imaging can monitor changes in these characteristics induced by treatment.12–17 In this way, molecular imaging can potentially serve as biomarker to predict treatment response and long-term outcome, in addition to response assessment (Table 1a–c). In this review, we provide an overview of the evidence for using [18F]FDG-, [18F]FES- and radiolabelled trastuzumab, as biomarker in early- and metastatic breast cancer.
Overview of PET imaging as biomarker for response prediction and assessment of molecular imaging in breast cancer.

PET imaging with radiolabelled trastuzumab.
FDG, fluorodeoxyglucose; FES, fluoroestradiol; HER2, human epidermal growth factor receptor 2; pCR, pathological complete response; PET, positron emission tomography.
Overview of level of evidence application of molecular imaging as biomarker in early breast cancer.
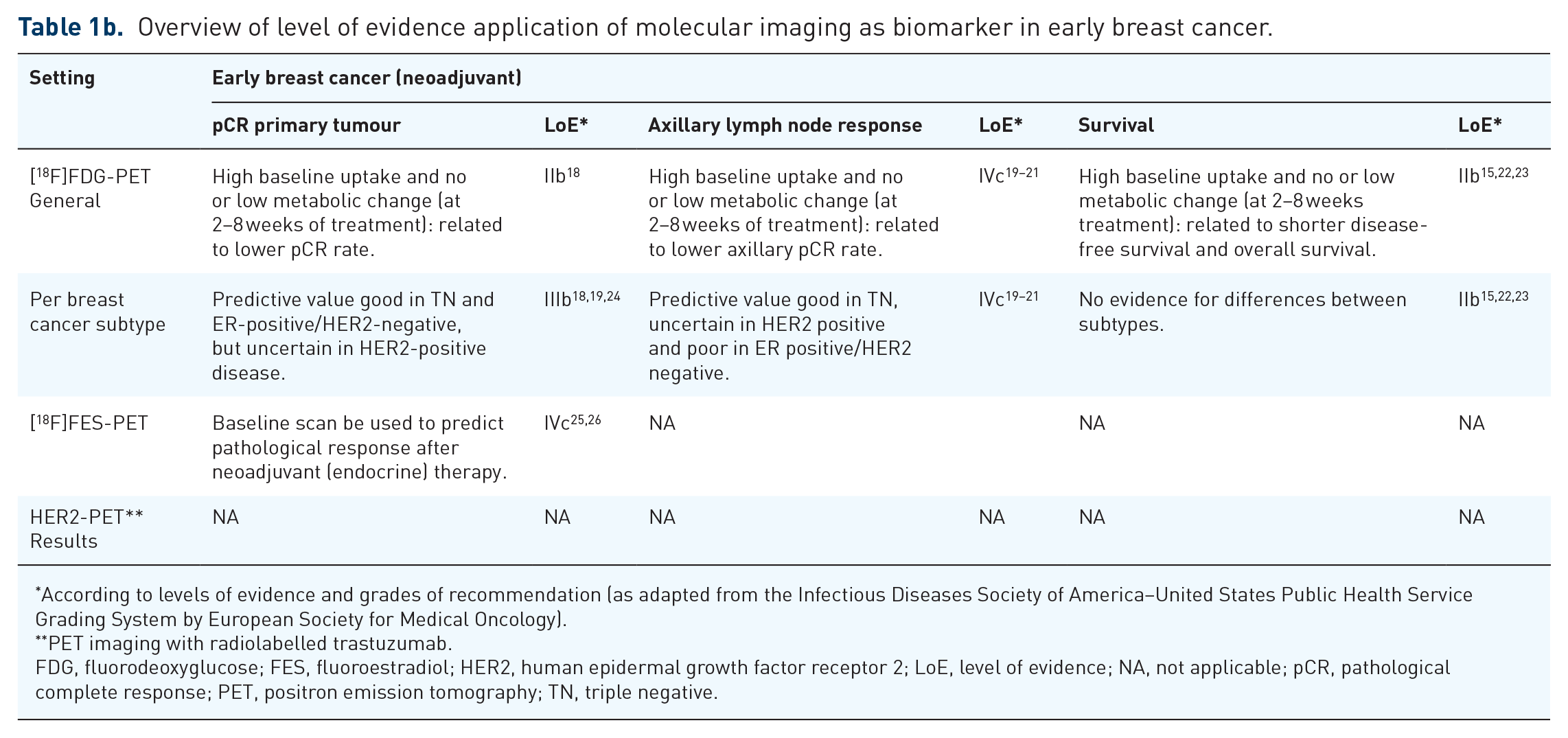
According to levels of evidence and grades of recommendation (as adapted from the Infectious Diseases Society of America–United States Public Health Service Grading System by European Society for Medical Oncology).
PET imaging with radiolabelled trastuzumab.
FDG, fluorodeoxyglucose; FES, fluoroestradiol; HER2, human epidermal growth factor receptor 2; LoE, level of evidence; NA, not applicable; pCR, pathological complete response; PET, positron emission tomography; TN, triple negative.
Overview of level of evidence application of molecular imaging as biomarker in metastatic breast cancer.

According to levels of evidence and grades of recommendation (as adapted from the Infectious Diseases Society of America–United States Public Health Service Grading System by European Society for Medical Oncology).
PET imaging with radiolabelled trastuzumab.
FDG, fluorodeoxyglucose; FES, fluoroestradiol; HER2, human epidermal growth factor receptor 2; LoE, level of evidence; NA, not applicable; PET, positron emission tomography; RECIST, Response Evaluation Criteria for Solid Tumours; TN, triple negative.
Search strategy and selection criteria
A search was performed on PubMed up and until August 2022, using the following search terms or combination(s) of these search terms ‘breast cancer’, ‘breast tumo(ur)’, ‘breast neoplasm(s)’, ‘biomarker’, ‘molecular imaging’, ‘positron emission tomography’, ‘PET’, ‘prediction’, ‘prognosis’, ‘response’, ‘progression(-)free survival’ and ‘(disease) progression’. Studies that were not available in English were excluded. The resulting publications were screened by title and abstract for relevance to this study’s research question. The selected articles were checked for relevant references that had not been included yet. Clinicaltrials.gov and abstracts from annual oncology meetings were also checked for ongoing studies relevant to this review.
[18F]FDG-PET in early breast cancer
Biomarker for pathological complete response in the neoadjuvant setting
The last decade there has been an increasing shift from adjuvant therapy towards neoadjuvant strategies. This offers several advantages such as higher chance on breast conserving surgery, de-intensification of axillary lymph node dissection using marking of axillary nodes by sentinel node procedures and evaluation of treatment response to adjust adjuvant strategies. These strategy adjustments can be based on the presence of a pathological complete response (pCR), which acts as a surrogate for long-term outcome, particularly in triple-negative (TN) and HER2-positive breast cancer. 50 Therefore, reaching a pCR has become an important aim in the neoadjuvant setting. Various studies have evaluated baseline [18F]FDG-PET as biomarker to predict pCR on neoadjuvant chemotherapy (NAC). Increasingly, early metabolic change on repeated [18F]FDG-PET is investigated as a dynamic biomarker for pCR to allow (de-)intensification during NAC (Table 2a). The diagnostic performance of metabolic change measured by repeated [18F]FDG-PET to predict pCR was evaluated in a meta-analysis of 22 studies including 1119 patients. A moderate accuracy for metabolic change during NAC to predict pCR was found with a sensitivity of 82% (76–87%) and specificity of 79% (72–85%). 18 Due to the unequal distribution of subtypes among the included studies in the aforementioned review, differences between subtypes could not be analysed. This might limit the value of these findings in clinical practice since the diagnostic performance is likely to be different per subtype.
Clinical trials evaluating [18F]FDG-PET for pCR prediction in early breast cancer.
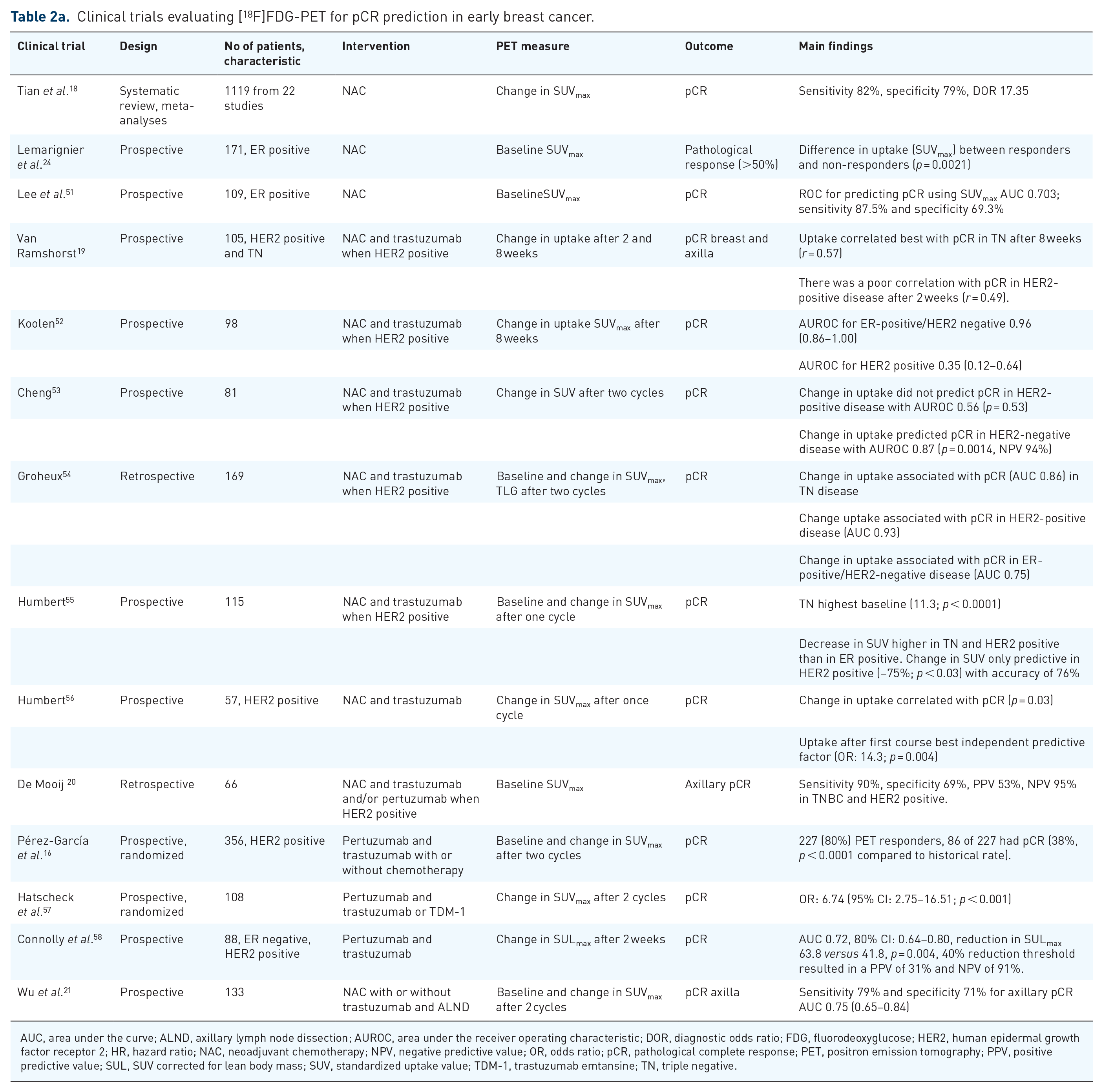
AUC, area under the curve; ALND, axillary lymph node dissection; AUROC, area under the receiver operating characteristic; DOR, diagnostic odds ratio; FDG, fluorodeoxyglucose; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; NAC, neoadjuvant chemotherapy; NPV, negative predictive value; OR, odds ratio; pCR, pathological complete response; PET, positron emission tomography; PPV, positive predictive value; SUL, SUV corrected for lean body mass; SUV, standardized uptake value; TDM-1, trastuzumab emtansine; TN, triple negative.
Biomarker for pCR per breast cancer subtype
Breast cancer subtype is expected to play a role in response prediction with [18F]FDG-PET, because of differences between subtypes in baseline [18F]FDG uptake and different treatment strategies which may induce varying changes in [18F]FDG uptake. The exact role of breast cancer subtypes in this setting is complicated and conflicting reports are available.
Several studies evaluated whether baseline [18F]FDG-PET or early metabolic change during NAC in the primary tumour on repeated [18F]FDG-PET correlated with the final pathology report, that is, pCR (Table 2a). The most robust evidence is available for TN breast cancer, with the reported area under the curve (AUC) for metabolic change to predict pCR ranging between 0.82 and 0.90.19,52,54,60 For ER-positive/HER2-negative breast cancer, multiple studies have also observed a correlation between [18F]FDG-PET and pathological response.24,51,52,59 Although the value of pCR in this subtype is itself low due to its limited relation to survival, 50 a prospective study in 109 patients with ER-positive breast cancer found an adequate AUROC curve of 0.703 for baseline [18F]FDG uptake as defined as maximal SUV (SUVmax) to predict pCR. 51 A smaller study including 46 patients with ER-positive/HER2-negative breast cancer found that metabolic change could predict pCR with an AUROC of 0.90 as well. 52 Ultimately, we can conclude that early metabolic change on repeated [18F]FDG-PET is correlated with pathological response in HER2-negative breast cancer. However, in HER2-positive breast cancer, the role of [18F]FDG-PET is uncertain, since contradictory reports are available. The largest prospective study in 115 patients found that metabolic change (defined as 75% decrease in SUVmax during NAC after the first course) predicted pCR in HER2-positive tumours with an accuracy of 76% (p < 0.03). 55 These findings were confirmed by other studies.41,56–58,61 However, in three large prospective trials, the metabolic change during NAC did not correlate with pCR in HER2-positive tumours.19,52,53 In the largest of these prospective studies including 105 patients with either TN or HER2-positive breast cancer, the best correlation with pCR was found with the absolute [18F]FDG uptake at 2–3 weeks after treatment initiation for patients with HER2-positive tumours with a c-index of 0.62 (p = 0.11). 19 Despite an adequate correlation between [18F]FDG-PET results and response, especially in HER2-negative breast cancer, heterogeneity among study protocols (different thresholds used for metabolic change or different methods of scan acquisition and quantification) complicates establishing optimal timing and interpretation of baseline [18F]FDG-PET and early metabolic change on repeated [18F]FDG-PET for response prediction. Therefore, at this moment, there is not enough evidence to support routine use of (repeated) [18F]FDG-PET to predict treatment response for the different breast cancer subtypes. A large prospective multicentre study with fully standardized protocols would have to be performed to determine the exact accuracy of early assessment of response to neoadjuvant therapy per breast cancer subtype using [18F]FDG-PET. One of these trials is the ongoing DIRECT trial (NCT05710328) which will prospectively evaluate the negative predictive value (NPV) of early metabolic change after 15 days to predict pCR in 235 patients with HER2-positive breast cancer treated with neoadjuvant HER2-targeted therapy. The results of this study will further clarify the role of early metabolic change for pCR prediction in HER2-positive breast cancer.
Biomarker for axillary lymph node response in the neoadjuvant setting
Whether (metabolic change on) [18F]FDG-PET can also predict axillary lymph node response has been evaluated in various trials. In a prospective trial in 105 patients with TN- or HER2-positive breast cancer, the early metabolic change on repeated [18F]FDG-PET after a single course of NAC in axillary lymph nodes and primary HER2-positive tumours combined was more strongly correlated to pCR than metabolic change in the primary tumour alone. Yet, this correlation was still weaker in HER2-positive than in TN breast cancer. 19 A retrospective study in 66 patients evaluated whether axillary uptake at baseline would predict response to neoadjuvant therapy. This study found no significant differences in baseline [18F]FDG uptake at the axillary nodes between responders and non-responders in the ER-positive/HER2-negative group. However, in patients with ER-negative disease (irrespective of HER2 status), axillary residual disease based on the final pathology report could be predicted with a sensitivity, specificity, positive predictive value (PPV), NPV of 90%, 69%, 53% and 95%. 20 Recently, metabolic change after one cycle of NAC to predict axillary pCR was evaluated in 188 patients and resulted in an AUC of 0.73, a sensitivity of 51% and a specificity of 83% among all subtypes. However, only in the HER2 positive and TN subtypes, the axillary response could be predicted with sufficient accuracy. 62 These results show that subtype is relevant in axillary response prediction using baseline or repeated [18F]FDG-PET. Furthermore, it shows an adequate predictive value in the TN and HER-positive subgroup but limited predictive value in the ER-positive/HER2-negative group.
Biomarker for survival in the neoadjuvant setting
While pCR can be used as a surrogate marker for long-term outcome and is approved as such by the Food and Drug Administration (FDA) and European Medicines Agency (EMA)63,64, there are some limitations. 65 The relationship between pCR and long-term outcome is strong in TN and HER2-positive breast cancer, but weaker in ER-positive breast cancer. 19 For this reason, it is of interest to directly evaluate the relationship between [18F]FDG-PET and long-term outcome (Table 2b). Baseline [18F]FDG-PET before NAC and adjuvant endocrine therapy has been found to have prognostic value for long-term outcome in a study including 466 patients with ER-positive/HER2-negative breast cancer. A multivariable analysis showed that high baseline [18F]FDG uptake was correlated with distant relapse-free survival [2.93, 95% CI: 1.62–5.30; p < 0.001] and OS (4.87, 1.94–12.26; p < 0.001). 22 This association has also been prospectively observed in 129 patients with ER-positive/HER2-negative breast cancer that underwent adjuvant chemotherapy with an independent association between baseline [18F]FDG-PET and disease-free survival (DFS) [hazard ratio (HR) = 2.49, 1.06–5.84]. In addition to [18F]FDG-PET at baseline, early metabolic change on repeated [18F]FDG-PET during NAC might also correlate with long-term outcome measures. A meta-analysis of 21 studies in 1630 patients with all breast cancer subtypes evaluated the value of baseline [18F]FDG-PET and early metabolic change on repeated [18F]FDG-PET during NAC, to predict survival. A pooled HR of metabolic change for DFS from 17 studies was 0.21 (95% CI: 0.14–0.32) for interim imaging and 0.31 (0.21–0.46) for post-treatment imaging. For OS, the HRs were 0.20 (0.09–0.44) and 0.26 (0.14–0.51) for interim and post-treatment imaging, respectively. A meta-regression analysis did not find significant influence of pCR or subtype on the HRs, despite overrepresentation of TN and HER2-positive subtypes among the studies. 23 In the TBCRC026 trial, decreased metabolic activity after 2 weeks of treatment was associated with DFS (HR = 0.36; 95% CI: 0.11–1.05; p = 0.06) and OS (HR = 0.14; 0.01–1.24; p = 0.01), although this did not reach statistical significance. 66 In conclusion, these results show that both baseline [18F]FDG uptake as well as metabolic change on repeated [18F]FDG during neoadjuvant therapy are correlated with long-term outcome measures after neoadjuvant therapy and may be of additional prognostic value to pCR. As such, they can potentially serve as biomarker to predict long-term outcome. However, the added value of (metabolic change on) [18F]FDG-PET to pCR to predict long-term outcome, and how this could be used to guide treatment decision-making, should be further elucidated in prospective comparative trials, in all subtypes.
Clinical trials evaluating prognostic value of [18F]FDG-PET for long-term outcome in early breast cancer.

CI, confidence interval; CT, chemotherapy; DFS, disease-free survival; ET, endocrine therapy; FDG, fluorodeoxyglucose; HR, hazard ratio; NAC, neoadjuvant chemotherapy; OS, overall survival; ratio PET, positron emission tomography; SUV, standardized uptake value.
Guiding systemic and local therapy in the neoadjuvant setting
[18F]FDG-PET is increasingly incorporated in studies to support de-intensification strategies in the neoadjuvant setting. These de-intensification strategies are of clear interest when new, expensive, invasive and toxic therapies are considered. In a multicentre trial, patients were randomized to receive trastuzumab and pertuzumab with or without chemotherapy. Patients not receiving chemotherapy were stratified based on metabolic change after two treatment cycles. Patients with 40% or more reduction in SUVmax, continued without chemotherapy, while patients with less metabolic change switched to chemotherapy. Out of 285 patients that received trastuzumab and pertuzumab alone, 227 (80%) showed a metabolic change, and 86 patients obtained a pCR (37.9%, 95% CI: 31.6–44.6; p < 0.0001 compared with the historical rate). In the group that received chemotherapy based on lack of metabolic change pCR was observed in 15 out of 58 patients (25.9%, 15.3–39.0). Ultimately, in the arm receiving chemotherapy from the start 41 out of 71 patients obtained pCR (57.7%; 47.4–69.4) versus 101 out of 287 patients in the experimental arm (35.4%; 29.9–41.3). Patients that did not receive NAC and did not obtain pCR received adjuvant chemotherapy. The usefulness of this interesting de-intensification strategy will have to be become clear after the 3-year DFS becomes available. 16 The value of [18F]FDG-PET to alter surgical management has been explored in one study using baseline [18F]FDG-PET and metabolic change after two cycles of NAC to select patients eligible for targeted axillary dissection. In this study of 133 patients, the axillary pCR was evaluated per subtype. The overall AUC for decreased metabolic activity to predict pCR was 0.75 (95% CI: 0.65–0.84), while the AUC per subtype decreased from 0.85 in ER positive/HER2 positive, to 0.77, 0.66 and 0.55 in TN, ER-positive/HER2-negative and ER-negative/HER2-positive breast cancer, respectively. Identifying non-responders with low [18F]FDG uptake at baseline or limited metabolic changer after NAC could have spared 19 patients an axillary lymph node dissection. 21 Other applications, such as selecting patients for breast conserving surgery, might also be possible. In 77 patients with luminal/HER2-negative breast cancer that underwent [18F]FDG-PET at baseline and after one cycle of NAC, the probability of breast conserving surgery could be predicted using three subgroups with different odds for breast conserving surgery based on volumetric measures (p = 0.001). 72 Even though currently these findings are insufficient to recommend routine implementation in clinical practice, future studies could evaluate selection of patients for surgical intervention with (metabolic change on) [18F]FDG-PET as biomarker to predict local response, based on these results. Another cornerstone of early breast cancer treatment is radiation therapy, which can be given post-operatively after breast-conserving surgery to the breast and lymph nodes to reduce risk of in-breast or in lymph node relapse, respectively. 4 [18F]FDG-PET is currently increasingly integrated in standard of care for planning in radiation therapy, since it improves delineation of target volumes and reduces uncertainty around delineation of tumour sites. 73 This improvement is also illustrated by a retrospective analysis in 31 patients that underwent [18F]FDG-PET. In this study, 32 out of 142 lymph nodes (23%) were only partially within the radiation field and 9 (6%) were totally uncovered with standard contouring according to the radiation therapy oncology group atlas with CT compared to [18F]FDG-PET. 74 One study evaluating the clinical impact of [18F]FDG-PET and multiparametric MRI in 18 centres in the Netherlands found that the highest impact and observer agreement for disease management was found using these imaging techniques for breast cancer compared to other cancer types, influencing disease management in 96% of all cases. 75 However, it is uncertain how (long-term) outcome such as recurrence- and complication rate is exactly affected by the addition of [18F]FDG-PET, as no prospective comparative studies are available.
Conclusions: [18F]FDG-PET as biomarker for early breast cancer in the clinic
There is insufficient evidence to support the use of baseline [18F]FDG-PET as biomarker for pCR to guide neoadjuvant treatment strategy (Table 1b). However, early metabolic change on [18F]FDG-PET is promising as it correlates well with pCR in early breast cancer, in particular in TN breast cancer. Furthermore, for all subtypes, high baseline [18F]FDG uptake plus low or no metabolic change after 2–8 weeks could potentially be used as a biomarker for a low pCR rate. As such, future de-intensification studies should incorporate early metabolic change on [18F]FDG-PET using an adaptive design. Furthermore, baseline [18F]FDG-PET and early metabolic change have prognostic value in predicting long-term outcome measures such as survival, in all breast cancer subtypes.
[18F]FDG-PET in metastatic breast cancer
Biomarker for treatment response
Baseline and early metabolic change on repeated [18F]FDG-PET up to 8 weeks after treatment initiation has been evaluated in several studies as biomarker for treatment response (on reference standard CT measured according to RECIST) (Figure 1; Table 1c). In the metastatic setting, breast cancer subtype plays an essential role due to differences in prognosis and treatment strategies between subtypes. 7 The ability of [18F]FDG-PET to predict RECIST response on first-line subtype-based standard treatment was evaluated in a retrospective analysis in 177 patients. In this study, a higher baseline [18F]FDG uptake was correlated with lower progression-free survival (PFS) after first-line chemotherapy for patients with TN breast cancer (HR = 1.862, p = 0.030). The standard PET parameters at baseline were not predictive for response, neither in patients with ER-positive/HER2-negative breast cancer receiving endocrine therapy or chemotherapy nor in patients with HER2-positive breast cancer receiving HER2-targeted treatment. 27 In 56 patients with advanced HER2-positive disease, early metabolic change after one cycle of trastuzumab emtansine (T-DM1) could predict RECIST response after three cycles, with a NPV/PPV of 83%/96. 28 In exceptional cases, also a metabolic increase on repeated [18F]FDG-PET could be of benefit. In 51 patients with ER-positive metastatic breast cancer, who underwent [18F]FDG-PET before and after 30 mg oestradiol exposure, increase in [18F]FDG uptake was shown in 20.9% of patients with an ultimate RECIST response on endocrine treatment, compared to −4.3% in non-responders. A significantly longer OS was observed in the group with at least a 12% increase in uptake (p = 0.0062). 29 This method was also found to be useful to predict response to oestradiol. 30 Regardless of breast cancer subtypes, early metabolic change after one course of chemotherapy was related to RECIST assessment in a prospective study with 24 patients with metastatic breast cancer. Responders had a larger decrease in [18F]FDG uptake than non-responders (p < 0.001). The metabolic change was also associated with OS in this study (r2 = 0.27, p < 0.01). 33

Early metabolic change on repeated [18F]FDG-PET as biomarker for response on CT according to RECIST.
Concluding, these studies show that high baseline [18F]FDG uptake and low or no metabolic change after 2–8 weeks of treatment could potentially be used as a biomarker to predict response according to RECIST.
In addition to predicting RECIST response, [18F]FDG-PET may itself also plays a role in treatment response assessment.17,34 While RECIST does incorporate [18F]FDG-PET, its role is supportive to CE-CT. 6 Several retrospective studies have found [18F]FDG-PET to be of additional value in response assessment.34–36 The largest of these trials was conducted in 300 patients with metastatic breast cancer and found significantly higher 5-year survival rates in patients who received standard response evaluation after 9–12 weeks based on [18F]FDG-PET (42%) or combined imaging (43%), compared to CE-CT (16%). Detection of first progression was detected 4.7 months earlier when [18F]FDG-PET was used. 36 The increase in survival can probably be explained by the fact that progressive disease is detected earlier, which could have a beneficial effect on the efficacy and tolerability of subsequent treatment lines. Recently, the earlier detection of progression with [18F]FDG-PET has been confirmed in a prospective study in 87 patients. In this study, progression was first observed with [18F]FDG-PET in 43 out of 87 patients (49.4%) compared to 1 out of (1.15%) CE-CT (p < 0.0001). Furthermore, there was a median delay of 6 months for detection of progression with CE-CT compared to [18F]FDG-PET. 37 For novel treatment strategies, [18F]FDG-PET may also be of value for response assessment as a preliminary analysis of 28 patients that were treated with CDK 4/6 inhibitors in the retrospective PUCCINI study, found that the first response assessment using PERCIST after 90 days was associated with longer PFS (HR = 0.16; 95% CI: 0.05–0.55). 76
The performance of [18F]FDG-PET to assess bone metastases might be superior to that of conventional imaging, using CT and bone scan. 77 This is of relevance since bone is the most common site for breast cancer metastases. 7 Furthermore, this facilitates response prediction and assessment in patients with bone-only or bone-dominant disease, who are often excluded from clinical trials when response is assessed according to RECIST, since metastases confined to bone cannot be considered as a target lesion according to RECIST 1.1. 6 Several studies have evaluated the value of [18F]FDG-PET for response assessment in this setting. A prospective study in 24 patients with bone-dominant metastatic breast cancer found that metabolic change measured with repeated [18F]FDG-PET after 4 months was correlated with skeletal events (p < 0.001) and treatment response (p = 0.044), but not with OS. 38 However, a retrospective study in 32 patients did find that baseline [18F]FDG-PET results were associated with PFS and OS in patients with bone metastases. 39 Another prospective study in 23 patients with ER-positive bone-only or bone-dominant metastatic breast cancer found a strong association between [18F]FDG-PET results at 4 and 12 weeks (r = 0.81). Patients in which metabolic change was observed after 4 weeks had a longer PFS (14.2 months versus 6.3; p = 0.53) and OS (44.0 months versus 29.7 months; p = 0.47) in this study. 40 There seems to be a role for [18F]FDG-PET in response prediction and assessment in bone-only and bone-dominant disease; however, the currently available evidence is limited. Prospective studies evaluating the role of [18F]FDG-PET in this specific setting will have to be conducted to clarify its exact value. One such study is the ongoing single arm FEATURE study (NCT04316117) which will prospectively evaluate if [18F]FDG-PET can be used to assess response and predict PFS in 134 patients with bone-dominant disease.
Concluding, the addition of [18F]FDG-PET to CE-CT for response assessment might improve the quality of current treatment assessment because of earlier detection of progressive metastatic breast cancer, especially in patients with bone-only or bone-dominant disease. However, this needs to be further evaluated in a prospective setting.
Biomarker for survival
In the metastatic setting, response outcome measures, such as RECIST, are approved by the FDA and EMA as endpoints for clinical trials. 78 However, surrogate measures such as response, disease control, PFS and time to progression are poorly correlated with survival. 79 This is further complicated by breast cancer subtypes: for instance, PFS is strongly correlated with OS in TN, but not in ER-positive/HER2-negative breast cancer.80,81 For long-term outcome prediction, direct correlation of [18F]FDG-PET findings with survival in the context of breast cancer subtypes is preferable (Table 3b). Also it should be considered that previous (neo)adjuvant treatment, location of metastases and tumour subtype can affect the prognostic value of [18F]FDG-PET in the metastatic setting. The relationship between subtype, timing of first diagnosis, survival and baseline [18F]FDG uptake was evaluated in a large prospective study in 244 patients. This study found that breast cancer subtype independently influences baseline [18F]FDG uptake in previously untreated disease (β = 0.290, p = 0.016). Furthermore, only in patients that did not receive (neo)adjuvant treatment [18F]FDG uptake at baseline was prognostic for PFS (HR = 1.049) and OS (HR = 1.124). 82 Similar findings were reported by a retrospective study. 70 It is conceivable that tumour characteristics change due to treatment, resulting in other [18F]FDG-PET results. Furthermore, the anatomical location of where the uptake is measured could also be of influence, as illustrated by one study in 253 patients that found worse OS with increased uptake (SUVmax) in bone metastases (HR = 3.1, p < 0.01; HR = 2.2, p = 0.02), but not in lymph node, liver or lung. On the other hand, TLG was only associated with worse survival in lymph node and liver metastases. 31 All these studies show that the value of baseline [18F]FDG-PET to predict long-term outcome, dependents on previously received therapy and the method and location of uptake measurement (Table 3b).
Clinical trials evaluating [18F]FDG-PET for response prediction and assessment in metastatic breast cancer.

AUC, area under the curve; CE-CT, contrast-enhanced CT; CMR, complete metabolic response; CT, computed tomography; FDG, fluorodeoxyglucose; HR, hazard ratio; MTV, metabolic tumour volume; NPV, negative predictive value; OS, overall survival; PET, positron emission tomography; PFS, progression-free survival; PPV, positive predictive value; SUL, SUV corrected for lean body mass; SUV, standardized uptake value; TLG, total lesions glycolysis; TTF, time-to-treatment failure.
Clinical trials evaluating prognostic value of [18F]FDG-PET for long-term outcome in metastatic breast cancer.

AUC, area under the curve; CMR, complete metabolic response; FDG, fluorodeoxyglucose; HR, hazard ratio; LN, lymph node; MTV, metabolic tumour volume; NPV, negative predictive value; OS, overall survival; PET, positron emission tomography; PFS, progression-free survival; PPV, positive predictive value; SUL, SUV corrected for lean body mass; SUV, standardized uptake value; TLG, total lesions glycolysis.
Concluding, baseline [18F]FDG-PET and early metabolic change are promising biomarkers to predict treatment response and survival in both early and metastatic breast cancer (Table 1). In early- and metastatic breast cancer, high baseline uptake and low or no metabolic change is related with adverse clinical outcome. Studies taking subtype into account are limited and more prospective data on the additional value of [18F]FDG-PET in this setting is needed before implementation in clinical practice. Also, trials incorporating [18F]FDG-PET as biomarker should be harmonized regarding timing and measurements. To show clinical utility, future studies would randomize patients to undergo [18F]FDG-PET after which stratification takes place based on the result of this scan. The long-term results of such a study will have to clarify how to optimally implement [18F]FDG-PET as biomarker in breast cancer management.
[18F]FES-PET in breast cancer
The ER status of tumours throughout the body can be non-invasively assessed with [18F]FES-PET.83,84 Several studies have evaluated the role of [18F]FES-PET as biomarker (Supplemental Table 1). [18F]FES-PET recently became the first FDA-approved PET imaging agent specifically indicated for breast cancer. 85
Biomarker for therapy response in early breast cancer
The diagnostic performance of [18F]FES-PET in breast lesions is good with a sensitivity and specificity of 86%/76%, respectively. 84 In a pilot study including 18 patients undergoing NAC, the [18F]FES uptake and the [18F]FES/[18F]FDG ratio were lower in responders (1.75 versus 4.42, p = 0.002 and 0.16 versus 0.54, p = 0.002, respectively). 86 However, in early breast cancer predicting response to NAC using [18F]FES-PET is likely to be of limited value since the pCR rate in ER-positive breast cancer is relatively low 50 and long-term response to NAC does not correlate with ER status. 87 Nevertheless, [18F]FES-PET could be of added value in early breast cancer when neoadjuvant endocrine therapy (NET) is considered. In the NEOCENT trial, a prospective feasibility study in ER-positive early breast cancer, the value of baseline [18F]FES-PET to predict response after NAC or NET was evaluated. In this study, 12 patients had low [18F]FES uptake (below SUVmax 7.3) in the primary tumour at baseline. Of these patients, none of five patients that received NET responded, whereas five out of seven patients that received NAC did. 26 In the Neo-ALL-IN study, low [18F]FES uptake (below SUVmax 5.5) was associated with low RECIST response in 24 patients with ER-positive/HER2-positive breast cancer, treated for 18–12 weeks with neoadjuvant letrozole and lapatinib (p = 0.007). 41 These findings suggest that patients with an ER-positive, but [18F]FES-negative tumour might benefit from NAC instead of NET, but the data should be regarded as exploratory due to low patient numbers and unconventionally high SUV thresholds used.
Biomarker for therapy response in metastatic breast cancer
[18F]FES uptake at baseline is related to endocrine therapy response in metastatic breast cancer. Using a threshold of SUV ⩾ 1.5 to define [18F]FES-positive disease on [18F]FES-PET, in a heavily pre-treated group of ER-positive metastatic breast cancer patients, none of 15 patients with [18F]FES-negative disease responded to endocrine therapy, while 11 of 32 (34%) with [18F]FES-positive disease did respond (p < 0.01). When only patients without HER2-positive disease were considered 11 out of 24 patients (46%) with [18F]FES-positive disease responded to endocrine therapy. 12 Endocrine sensitivity based on [18F]FES-PET has also recently been investigated in the ET-FES study. In this international multicentre study, 146 patients with ER-positive metastatic breast cancer were randomized to receive either endocrine therapy or chemotherapy based on [18F]FES-PET results (mean SUV threshold of 2.0). In patients receiving endocrine therapy, median PFS was 7.3 months in patients with a high [18F]FES uptake, versus, 5.2 months in patients with a low [18F]FES uptake. Patients with low uptake who received chemotherapy had a PFS of 7.7 months. 88 Unfortunately, this study was prematurely halted due to the COVID-19 pandemic. However, these results show great promise to identify patients using [18F]FES-PET that benefit more from chemotherapy instead of endocrine therapy, irrespective of a positive ER assay.
[18F]FES-PET combinations and repeated scans as biomarker for therapy response
Combining [18F]FES-PET, with [18F]FDG-PET or other imaging modalities might improve the ability of [18F]FES-PET to predict response. Response is dependent on several tumour characteristics, which can be measured throughout the whole body in the metastatic setting. Several studies have combined [18F]FES-PET and [18F]FDG-PET to predict and assess treatment response. In 90 patients with metastatic breast cancer planned to receive endocrine therapy, the combined [18F]FES and [18F]FDG uptake was evaluated. 42 Patients with high [18F]FDG uptake and high [18F]FES tumour uptake had a better response to endocrine therapy compared to those with high [18F]FDG uptake and low [18F]FES tumour uptake. 42 In a pilot study in 30 patients, the value of the concordance rate between [18F]FES positivity and the entire tumour load, as determined on [18F]FDG-PET and CT, was evaluated in patients that received letrozole and palbociclib. 14 Two retrospective studies have also evaluated the percentage of [18F]FES-positive lesions, rather than the absolute [18F]FES uptake in patients receiving endocrine therapy with or without CDK 4/6 inhibition.89,43 A consistent finding between these studies is that patients with 100% [18F]FES-positive disease have a longer PFS than patients with lower percentages.14,89,43 However, the optimal method to determine the [18F]FES/[18F]FDG ratio and how this relates to clinical outcome remains unclear.42–44 Recently, one study performed in 54 patients with [18F]FES-positive lesions below 100% found that these patients might benefit chemotherapy instead of endocrine therapy. 90 These studies show that the percentage of [18F]FES-positive lesions might ultimately be used to optimize treatment selection the metastatic setting. Combining [18F]FES-PET and [18F]FDG-PET could also be beneficial in specific instances where the performance of [18F]FDG-PET is suboptimal. In invasive lobular breast cancer, the detection rate of [18F]FDG-PET appears to be lower than that in invasive breast cancer of no special type.91,92 In comparative studies, [18F]FES-PET detected locoregional and distant metastases of lobular breast cancer that were not detected with [18F]FDG-PET.93–95 At present, no data are available on how this affects the prediction and assessment of outcome. The IMPACT-MBC trial (NCT01957332) and future lobular cancer-specific initiatives are expected to clarify whether [18F]FES-PET has additional value in this setting. Using repeated [18F]FES-PET changes in ER expression can be assessed over time, which can be used to assess ER occupancy and potentially predict long-term outcome. In light of this, pharmacodynamics of the different types of endocrine therapy have been evaluated in 30 metastatic breast cancer using repeated [18F]FES-PET. 96 These data show that [18F]FES-PET can visualize changes in ER occupancy throughout the body after endocrine therapy. Multiple studies have evaluated this principle in ER-targeting strategies (Supplemental Table 1). However, it is still unclear how the presence of ER occupancy as measured by [18F]FES-PET exactly correlates with clinical outcome and therefore how it can be used to assess treatment response.
In conclusion, [18F]FES-PET provides information that might be used as biomarker for endocrine response. A high [18F]FES uptake at baseline, a decrease in [18F]FES uptake after treatment initiation and a high percentage of [18F]FES-positive lesions could act as biomarker for a higher endocrine response rate. Further studies will have to clarify if an how this information either alone or combined with [18F]FDG-PET can be used to predict treatment outcome and select patients accordingly.
HER2-PET in breast cancer
HER2 expression can be non-invasively measured using PET imaging. Different tracers are available to image HER2 internalization. The most commonly used tracers to visualize whole-body HER2 turnover in breast cancer patients are trastuzumab labelled with 89Zr and 64Cu.97–99 Compared to [18F]FDG-PET and [18F]FES-PET, a limited numbers of studies are available that evaluate HER2-PET results as biomarker for treatment response and outcome. HER2-PET can be used to support treatment decision-making in metastatic breast cancer, in clinical practice.100,101 Some studies have evaluated the ability of HER2-PET to predict and assess response to HER2-targeted treatment. In a study, 10 patients with HER2-positive metastatic breast cancer underwent follow-up [ 89 Zr]Zr-DFO-trastuzumab-PET and received an inhibitor of HSP90, which plays a critical role in protein folding. Change in size on CT after 8 weeks was correlated with local [ 89 Zr]Zr-DFO-trastuzumab uptake after 3 weeks in this study (r2 = 0.69; P = 0.006). 102 Furthermore, two studies have evaluated the influence of TDM-1 on tracer uptake and how this correlates with response. One of these studies was conducted in 56 patients with HER2-positive metastatic breast cancer that underwent a baseline [18F]FDG-PET and [ 89 Zr]Zr-DFO-trastuzumab-PET prior to receiving T-DM1. Dichotomization of groups into either a positive or negative result based on 50% or more [ 89 Zr]ZR-DFO-trastuzumab-positive lesions compared to the entire tumour load as visualized by [18F]FDG-PET was performed prior to the analyses. This resulted in two groups of 16 (29%) patients with a negative and 40 (71%) with a positive [ 89 Zr]Zr-DFO-trastuzumab-PET. The time-to-treatment failure (TTF) was 11.2 months (95% CI: 8–15 months) in patients with a positive [ 89 Zr]Zr-DFO-trastuzumab-PET scan and 3.5 months (95% CI: 1.4–7.6) when the scan was considered negative. 28 The other study was performed in 10 patients with HER2-positive metastatic breast cancer that underwent a [ 64 Cu]Cu-DOTA-trastuzumab-PET prior to treatment initiation with TDM-1. A higher minimum [ 64 Cu]Cu-DOTA-trastuzumab uptake (SUVmax) on day 1 (5.6 versus 2.8, p < 0.02) and day 2 (8.1 versus 3.2, p < 0.01) was observed in responders according to PERCIST than in non-responders. Using an optimal threshold of SUVmax 5.5 for [ 64 Cu]Cu-DOTA-trastuzumab uptake on day 2, responders could be distinguished relatively well from non-responders with a TTF of 2 versus 28 months (HR = 0.1, p = 0.02). 49 These studies show that there is potential for HER2-PET to predict and assess response to TDM-1 in patients with HER2-positive disease. In light of response to next-generation anti-HER2 targeting agents such as trastuzumab deruxtecan seen in patients with previously considered HER2-negative disease it will be of great interest to see if HER2-PET is able to predict and assess response in patients with HER2-negative diseased based on pathology results. However, all studies that evaluate treatment response so far have been conducted in HER2-positive breast cancer and no data addressing this is available so far.
Concluding, HER2-PET may potentially be used for a biomarker to predict and assess response to HER2-targeted treatment in patients with HER2-positive disease. Studies like the IMPACT-MBC trial (NCT01957332), will have to provide additional insight into the clinical utility of HER2-PET, also in HER2-negative disease.
Future perspectives
Even though [18F]FDG-PET, [18F]FES-PET and HER2-PEt all show promising results for eventual integration into clinical practice, still important issues need to be addressed. To optimize [18F]FDG-PET as biomarker for prediction of therapy response and survival, studies should be subtype specific. Also optimal timing for early metabolic change measurement is likely subtype dependent. In the metastatic setting, the superiority of [18F]FDG-PET to CT to predict survival will have to be confirmed in a prospective setting. In ER-positive metastatic breast cancer, heterogeneity on [18F]FES-PET (combinations) is emerging as an important biomarker for endocrine sensitivity. Whether this can lead to improved patient outcome needs to be further explored.
Until now, HER2-PET has mostly been investigated in the metastatic breast cancer setting and almost exclusively in HER2-positive disease. It will be of interest to investigate whether HER2-PET can help to clarify why response to HER2-targeting therapies is observed in patients HER2-negative breast cancer. Expansion of the evaluation of HER2-PET into the early breast cancer setting could also be of interest in light of increasing HER2-targeted neoadjuvant therapy possibilities and the disappointing [18F]FDG-PET results in this setting.
While HER2 and ER remain indispensable for treatment decision-making, other therapeutic targets have become increasingly important in breast cancer care. In addition to [18F]FDG, [18F]FES and HER2 visualizing tracers, other tracers visualizing other characteristics of breast cancer have been investigated to some extent in clinical trials.11,103
Proper incorporation of PET imaging in clinical trials and practice is dependent on advancements in validation and qualification of these biomarkers. Recommendations regarding these advancements have recently been detailed in a imaging biomarker roadmap for cancer studies. 104 Currently, the technical validity of [18F]FDG-PET and [18F]FES-PET is established, with sufficient repeatability and reproducibility. 11 To advance clinical implementation of imaging biomarkers, guidance documents are needed to support technical and clinical validation processes, such as auditing and quality control. Essential guidance documents that are suitable for this are the currently available appropriate use criteria105,106 and European Association of Nuclear Medicine (EANM) procedure guideline for [18F]FDG-PET 107 as well as the upcoming Society of Nuclear Medicine and Molecular Imaging and EANM [18F]FES-PET guideline. 106 At present, the clinical utility of PET imaging still remains to be established further, since most studies so far have focused on technical and clinical validity. Future studies will have to clarify which tracers have sufficient clinical utility to be considered for implementation into routine care and how this needs to be done. 11 Ideally, implementation of PET imaging into clinical practice will be studied further by stratifying patients based on PET results and assign treatment accordingly as has been done with follow-up [18F]FDG-PET in early breast cancer. 16 In addition, future studies will have to focus on cost-effectiveness of these biomarkers.
Conclusion
Evidence supporting the additional value of PET imaging as a biomarker to predict treatment response in patients with early or metastatic breast cancer is increasing. While [18F]FDG-PET is already widely incorporated in current clinical practice, PET imaging with other tracers outside of clinical trials is still uncommon. In this review, we have identified the evidence for [18F]FDG, [18F]FES and HER2 PET results as biomarkers for treatment response and survival, and described for which issues additional data are needed. Even though (repeated) [18F]FDG-PET, [18F]FES-PET and HER2-PEt all show promising results as biomarkers to predict therapy response and outcome, still generally low-level evidence is available. For eventual integration into clinical practice, optimal scan timing and thresholds for differences need to be established. Subtype-specific trial designs may support PET-based de-intensification to prevent over- and undertreatment. This can ultimately contribute to improved outcome in breast cancer patients.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231170738 – Supplemental material for Molecular imaging as biomarker for treatment response and outcome in breast cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359231170738 for Molecular imaging as biomarker for treatment response and outcome in breast cancer by Jasper J. L. van Geel, Erik F. J. de Vries, Michel van Kruchten, Geke A. P. Hospers, Andor W. J. M. Glaudemans and Carolina P. Schröder in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
