Abstract
Tissue-agnostic indications for targeted therapies have expanded options for patients with advanced solid tumors. The Food and Drug Administration approvals of the programmed death-ligand 1 inhibitor pembrolizumab and the TRK inhibitors larotrectinib and entrectinib provide rationale for next-generation sequencing (NGS) in effectively all advanced solid tumor patients given potential for clinical responses even in otherwise refractory disease. As proof of concept, this case report describes a 64-year-old woman with triple-negative breast cancer refractory to multiple lines of therapy, found to have a rare mutation on NGS which led to targeted therapy with meaningful response. She initially presented with metastatic recurrence 5 years after treatment for a localized breast cancer, with rapid progression through four lines of therapy in the metastatic setting, including immunotherapy, antibody–drug conjugate-based therapy, and chemotherapy. Germline genetic testing was normal. Ultimately, NGS evaluation of cell-free DNA via an 83-gene assay (Guardant Health, Inc.) identified two NTRK3 fusions: an ETV6-NTRK3 fusion associated with the rare secretory breast carcinoma, and CRTC3-NTRK3, a novel fusion partner not previously described in breast cancer. Liver biopsy was sent for whole exome sequencing and RNA-seq analysis of tissue (BostonGene, Inc., Boston, MA, USA), which provided orthogonal confirmation of both the ETV6-NTRK3 and CRTC3-NTRK3 fusions. She was started on the TRK inhibitor larotrectinib with a marked clinical and radiographic response after only 2 months of therapy. The patient granted verbal consent to share her clinical story, images, and data in this case report. This case demonstrates the significant potential benefits of NGS testing in advanced cancer and the lessons we may learn from individual patient experiences.
Introduction
The targeting of gene fusions involving neurotrophic tyrosine kinase (NTRK) 1/2/3 has represented a major milestone in cancer care. In 2018, the TRK inhibitor larotrectinib became only the second targeted cancer therapy approved by FDA for all solid tumors regardless of tissue of origin, based on a genomic alteration in NTRK 1/2/3. The following year, entrectinib was approved for the same tissue-agnostic indication: NTRK 1/2/3 fusions. The approvals were all based on basket trials where enrollment was determined by the existence of NTRK fusions regardless of tissue of origin.1,2 There were few grade ⩾3 adverse events: anemia (11%), neutropenia (7%), AST/ALT elevation (7%), and weight gain (7%) were the most common in larotrectinib. Weight gain (10%) and anemia (12%) were the most common with entrectinib, and nervous system disorders (4%) were the most common of the serious treatment-related adverse events.
NTRK fusions are rare genomic events in breast cancer as a whole, predominantly seen in the very rare secretory breast carcinoma subtype, where the ETV6-NTRK3 fusion is pathognomonic. 3 Secretory breast carcinoma tends to be indolent and it rarely metastasizes. 4 Here, we present the case of a postmenopausal woman with metastatic triple-negative breast cancer (TNBC) with secretory features who had disease progression on multiple lines of therapy. Plasma-based genotyping identified two NTRK fusions via cell-free DNA (cfDNA) analysis: an ETV6-NTRK3 fusion associated with secretory breast carcinoma and an intra-chromosomal CRTC3-NTRK3 fusion not previously reported in the literature, to our knowledge. She was treated with larotrectinib per FDA indications and had a marked radiographic response after 2 months of treatment despite having progressive disease through multiple prior lines of therapy.
Case report
In 2014, a 64-year-old woman palpated a left breast mass, the workup of which led to diagnosis of a grade II, faintly estrogen receptor positive (ER+) at 10%, progesterone receptor negative (PR−), and human epidermal growth factor receptor 2 negative (HER2−) breast cancer. The tumor showed predominantly microcystic architecture with focal solid and papillary areas, polygonal cells with eosinophilic vacuolated cytoplasm, and cystic spaces remarkable for dense eosinophilic secretions. With these findings, the case was signed out as invasive ductal carcinoma (IDC) with secretory features (Figure 1). The mass broadly abutted the pectoralis muscle; thus, she received neoadjuvant dose dense doxorubicin and cyclophosphamide, followed by weekly paclitaxel ×12 (ddACT), and she subsequently underwent left breast surgery with three negative sentinel lymph nodes removed. Final pathology revealed a 2.6 cm mass with no definitive response in the invasive component. The final stage was ypT2ypN0M0. She received adjuvant radiation and completed 5 years of anastrozole.

(a) A diagram of the ETV6-NTRK3 fusion. (b) A diagram of the CRTC3-NTRK3 fusion. (c) The tumor showed a predominantly microcystic architecture. (d) The specimen was composed of polygonal tumor cells with enlarged, round to oval nuclei and vacuolated, eosinophilic cytoplasm, and cystic spaces were remarkable for dense eosinophilic secretions.
In 2020, 5 years after her surgery, she developed painful cervical lymphadenopathy. Positron emission tomography/computed tomography identified recurrent erosive chest wall disease surrounding the manubrium, bilateral pulmonary metastases, and supraclavicular lymph node metastases. Biopsy of the latter confirmed metastatic breast carcinoma, newly ER− and thus TNBC, with 1% programmed death-ligand 1 expression (tumor cells), and Ki-67 20%. Germline genetic testing was negative for inherited mutations in ATM, BRCA1, BRCA2, CDH1, CHEK2, NBN, NF1, PALB2, PTEN, STK11, and TP53.
She subsequently received multiple lines of therapy, after each of which she had disease progression at early interval restaging. Treatments included atezolizumab/nab-paclitaxel (progression after 5 months), gemcitabine/carboplatin (progression after 3 months), sacituzumab govitecan-based therapy (progression after 2 months), and eribulin (progression after 2 months). She underwent two biopsies of her chest wall mass, which showed IDC along with intracytoplasmic mucin.
Before starting sacituzumab govitecan-based therapy, peripheral blood targeted next-generation sequencing (NGS) of cfDNA was performed using the Guardant360 assay, which evaluates clinically significant alterations in 83 genesin cancer. This assay identified an ETV6-NTRK3 fusion at a mutant allele fraction (MAF) of 3.8%. Three months later, after progression on sacituzumab govitecan-based therapy, repeat Guardant testing of a new blood sample showed the MAF of the ETV6-NTRK3 fusion had increased from 3.8% to 10.9%, and a newly detected CRTC3-NTRK3 fusion at a MAF of 3.2% was also identified. She also underwent liver biopsy, which was sent for BostonGene testing, a tissue-based whole exome sequencing and RNA-seq platform. This evaluation confirmed both the ETV6-NTRK3 and CRTC3-NTRK3 fusions on orthogonal tissue-based sequencing (Figure 1).
She was subsequently started on larotrectinib. Repeat staging scans 2 months later showed dramatic improvement in her lung, pleural, lymph node, and chest wall metastases, her first objective response to treatment. CA 15-3, which had been rising, also decreased from a peak of 206 IU/mL to 48 IU/mL. Repeat Guardant testing on a subsequent blood sample showed a reduction in the ETV6-NTRK3 fusion MAF from 10.90% to 0.40%, and the CRTC3-NTRK3 fusion MAF from 3.20% to 0.07% (Figure 2). The patient tolerated the larotrectinib well without any dose reductions or modifications. Anemia and edema were the main side effects noted. The patient continued larotrectinib for 7 months with good disease control. Unfortunately, unrelated to her therapy, she had experienced multiple fractures secondary to her existing osseous metastases, and these led to significant morbidity. Ultimately, she and her family elected to transition to comfort measures, after which she passed away.
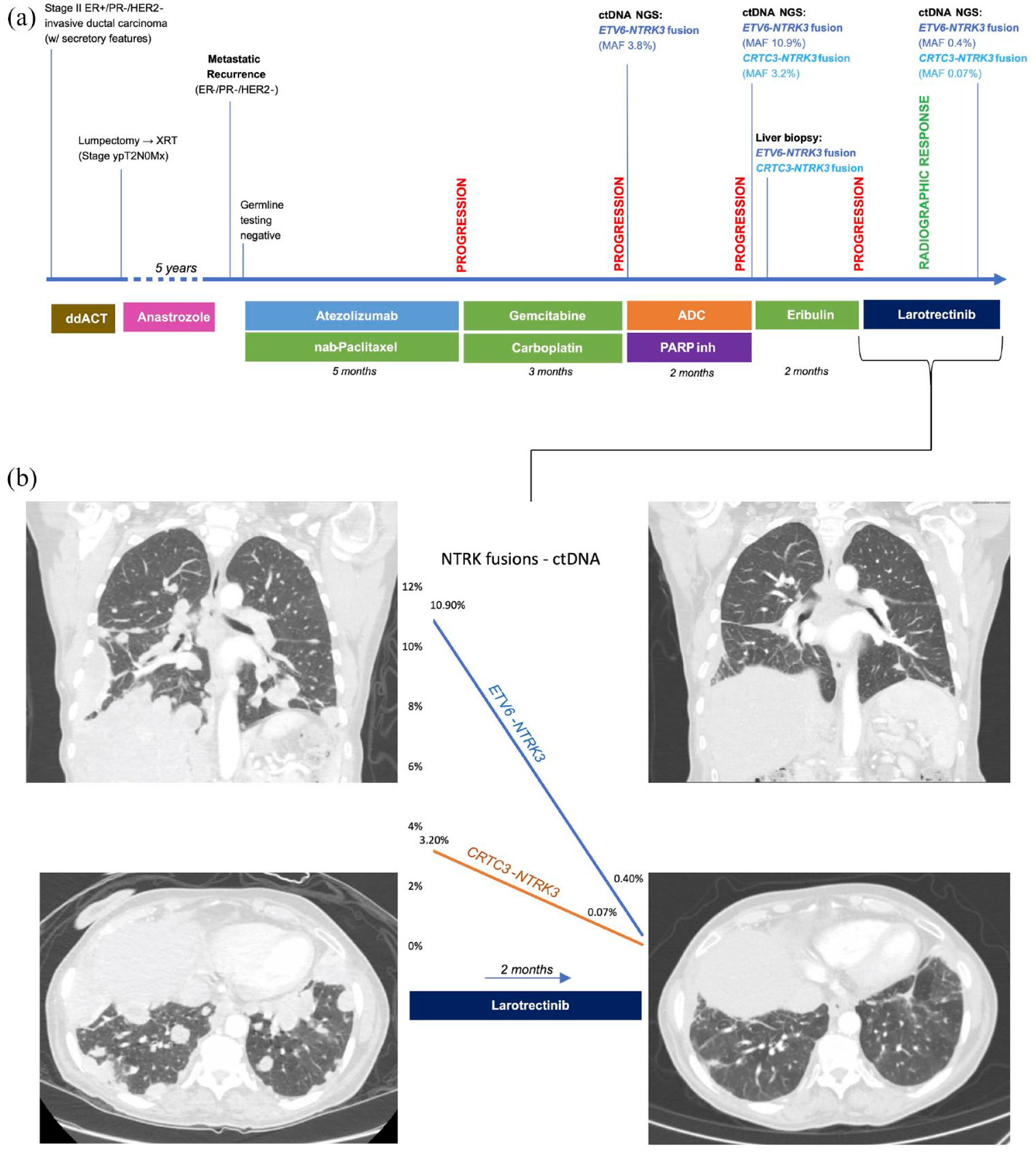
(a) A timeline of the patient’s clinical story, including treatments below the timeline and biopsies and clinical response above the timeline. (b) The left CT chest images demonstrate numerous pulmonary and pleural metastases. The images on the right show marked improvement of these lesions after 2 months of larotrectinib. The decrease in the MAF of the two NTRK3 fusions is demonstrated in between the CT images.
Discussion
This case describes a patient with TNBC who had disease progression on multiple lines of therapy until genomic testing identified two NTRK fusions, for which she was started on a targeted TRK inhibitor with marked clinical response after 2 months of therapy. The identification of NTRK fusions by plasma-based genotyping resulted in matched selection of genotype-directed therapy and should be considered for patients with metastatic breast cancer, particularly the ones with secretory histology.
NTRK fusions are rare genomic occurrences that have now gained significant attention, given they qualify patients for genomically targeted therapy regardless of a cancer’s tissue of origin. However, NTRK fusions are identified in only about 0.3% of adult solid tumors.5–8 NTRK 1/2/3 encode TrkA/B/C, and fusions of these proteins lead to ligand-independent dimerization and subsequent downstream signaling of both the MAPK and PI3K-AKT pathways.3,9–12 A large study of the FoundationCORE® database of >295,000 cancer patients also found NTRK fusions in approximately 0.30% of solid tumors. These were documented in 889 patients in 45 different cancers, and there were 88 unique fusion partners. 5 Out of the 295 cases in adults with known fusion partners, ETV6 was the most common (n = 78, 26.4%). Within breast cancer, ETV6 was the partner in 14 patients (14.7%). Interestingly, the above patient’s CRTC3-NTRK3 fusion involves a fusion partner that does not appear to have been described in these large datasets. ETV6-NTRK3 is an inter-chromosomal fusion t(12;15)(p13;q25), however, CRTC3-NTRK3 is intra-chromosomal. We unfortunately do not have NGS data from our patient’s primary specimen, although given the secretory pathology, it is very likely that the ETV6-NTRK3 fusion was already present. Later, cfDNA analysis detected a low MAF ETV6-NTRK3 fusion at a different fusion point of the ETV6 gene, and a deeper exploration of the cfDNA NGS suggests these genomic alterations may in fact be interleaved, where the exons of one gene may reside in the introns of another, and the multiple fusion reads may reflect a more complex event (Figure 3).

(a) A diagram of the NTRK3, EVT6, and CRTC3 genes. (b) The primary ETV6-NTRK3 fusion identified and reported on cfDNA analysis. (c) A diagram of a second ETV6-NTRK3 fusion identified on cfDNA analysis. (d) A diagram of the CRTC3-NTRK3 fusion identified on cfDNA analysis. (e) An internal NTRK3-NTRK3 fusion identified on cfDNA analysis.
The advent of larotrectinib and entrectinib has generated a new treatment paradigm in oncology. The tissue-agnostic approval of larotrectinib was based on three single-arm basket clinical trials: LOXO-TRK-14001 (NCT02122913), SCOUT (NCT02637687), and NAVIGATE (NCT02576431). In a combined analysis of these three studies, the first 55 patients evaluated with unresectable or metastatic solid tumors had an overall response rate (ORR) of 75% (95% CI: 61–85%), and a subsequent analysis in 153 patients found an ORR of 79% (95% CI: 72–85), where 24 patients (16%) had complete responses.1,2 Approval for entrectinib was similarly based on an integrated study of three single-arm clinical trials: ALKA-372-001 (GO40783), STARTRK1 (NCT02097810), and STARTRK2 (NCT02568267). In this analysis, 54 adult patients had an ORR of 57% (95% CI 43–71%), and 63% of patients had a response duration ⩾6 months.13,14 Interestingly, the approval is not dependent on the identity of the fusion partner. These new tissue-agnostic genomic indications opened a new space in precision medicine and opportunities to learn from our patients as these guidelines are implemented.
There are several important points to learn from this unique case. First, microscopic features observed in this patient’s excision led to a diagnosis of IDC with secretory features, which should be the first clue to pursue further genetic testing. Secretory breast carcinoma histologically and genomically has a greater similarity to salivary cancer than typical IDC; among other features, they both harbor an ETV6-NTRK3 gene fusion.15,16 Furthermore, while NTRK fusions in solid tumors are overall rare, in secretory breast carcinoma, the NTRK-ETV6 fusion is key to diagnosis.3,17 The pathology coupled with a molecularly defining ETV6-NTRK3 fusion helped characterize her tumor as a rare secretory breast carcinoma. To date, there are no published studies that have investigated the use of biomarker results to inform the use of TRK inhibitors in patients with metastatic breast cancer and NTRK fusions.
Secretory breast carcinoma represents fewer than 1% of breast cancers.5–8 This entity was initially described in children and adolescents as ‘juvenile carcinoma’. However, it was later appreciated that similar cancers with the same shared secretory features were reported in older patients from 9 up to 69 years old (median age 25); this prompted its reclassification as ‘secretory breast carcinoma’.18,19 Interestingly, while initial driver mutations in breast cancer have been historically difficult to define, the ETV6-NTRK3 fusion is a known exception and has been invoked as a piece of the genomic puzzle regarding the genomic evolution of breast cancer. 3 Secretory breast cancer is one of the few breast cancers described in children and adolescents.18,20–23
Secretory breast cancers are often triple negative, although they are generally felt to be less aggressive with low metastatic potential compared to their non-secretory counterparts, despite typically being triple negative with a basal-like immunophenotype. 24 This indolent growth trajectory is a feature thought to be shared with salivary cancer. 16 This patient’s tumor showed a more aggressive natural history, with distant metastatic recurrence 5 years after surgery and resistance to multiple lines of systemic therapy. This case highlights an interesting point about rare diseases. It is often difficult to fully characterize the range of natural histories of a rare disease given paucity of data from larger population studies. The question then exists as to whether this patient’s disease was truly atypical, or whether we are seeing a different phenotype less understood in the literature. Furthermore, despite an aggressive course, targeting the NTRK fusion was key to controlling this patient’s otherwise refractory disease.
The above patient’s rapid disease progression on immunotherapy may also be characteristic behavior of NTRK fusion solid tumors. One large-scale, single-institution study involving prospective screening of 26,000 patients identified 76 cases of NTRK fusions, consistent with a 0.28% prevalence in the adult population. This appears to be the largest study of NTRK fusions with concomitant clinical data. Secretory breast cancer comprised a majority of NTRK-positive cases, with non-secretory breast cancers comprising only 0.08% (3/3775) of cases. 8 Across all tumor types, tumor mutation burden tended to be low in patients with NTRK fusions, and overall response to immunotherapy tended to be poor. The one exception was in colorectal cancer, where there appeared to be co-occurrence of spontaneous microsatellite instability with NTRK fusions. 5 Whole transcriptome sequencing via the BostonGene platform also identified a fibrotic tumor microenvironment, which has been associated with poorer immunotherapy response. 25
Multiple studies of NTRK fusions have shown these genomic alterations to be true oncogenic drivers, which tend to be mutually exclusive from other driver alterations. 16 NTRK fusions also frequently appear to be clonal events; the prospective screening cohort above included 17 patients who underwent serial biopsies, and 14 of these patients had newly acquired fusions detected in successive biopies. 8 Subclonal NTRK fusions have also been reported as putative acquired off-target resistance mechanisms, for example in non-small-cell lung cancer with co-occurring EGFR driver mutations emerging after treatment with tyrosine kinase inhibitors. 26
Given the importance of molecularly guided therapy, there has been increasing recognition of the need for highly sensitive methodologies to detect newly acquired molecular events that may inform both biology and response to treatment. There are several approaches for detecting NTRK fusions, including DNA-based NGS, RNA-based NGS, immunohistochemistry (IHC), and fluorescence in situ hybridization (FISH). 27 Among these techniques, RNA sequencing appears to be the most sensitive. In the MSK-IMPACT evaluation described above, DNA testing was unable to identify fusions in 26% (12/46) of cases detectable by RNA testing. All 12 of these cases were due to breakpoints within introns in NTRK2/3 that were too large to be practically included in the MSK-IMPACT DNA-based assay. This discrepancy underlines a limitation of DNA-based NTRK detection. Despite this fact, DNA-based sequencing has a sensitivity of 81.1% and a specificity of 99.9% compared to RNA-based sequencing. 28 IHC has a specificity of 81.1%, but sensitivity for NTRK3 fusions is as low as 79%. Furthermore, specificity of IHC decreases in the setting of high fusion incidence, as in secretory breast and salivary gland carcinomas (82% and 52%). IHC thus is associated with more false negatives and false positives. In a study of 305 TNBC patients, screening with IHC identified 32 (11.1%) positive cases. Confirmatory FISH testing identified 13 total patients with NTRK fusions. However, subsequent NGS and RT-PCR were negative for NTRK fusions in all but one patient. 29 ESMO guidelines call for NGS as the standard diagnostic tool, and if IHC is the initial screening tool, it should be followed by NGS. Similarly, the FDA-approved companion diagnostic for larotrectinib is the FoundationOne test, a tissue-based NGS platform.
The actionability of these rare genomic events makes identifying them critical to individual patients, particularly in heterogeneous diseases such as TNBC. While TNBC historically has been defined as a disease without targetable alterations (e.g. ER, HER2), this case shows that in fact there are patients within the TNBC population who do benefit from targeted therapy, particularly in the case of secretory histology given the potential for excellent clinical response given the inherent NTRK3 fusion. This concept has recently led the American Society of Clinical Oncology to issue updated guidelines on biomarker testing in breast cancer, as well as a provisional recommendation that patients with advanced solid tumors undergo somatic testing if there is potentially more than one biomarker that could lead to targeted therapy.30–32 Given the tissue-agnostic approval of pembrolizumab in patients with microsatellite instability and/or high tumor mutational burden, and a similar approval for larotrectinib and entrectinib in patients with NTRK fusions, this opinion effectively recommends somatic testing in all patients with advanced solid tumors and is reflected in the current NCCN Guidelines Version 3.2022, which includes a recommendation for comprehensive germline and somatic profiling to identify candidates for additional targeted therapies as part of the work up for stage IV (M1) or recurrent breast cancer. As our understanding of the genomic landscape and actionability in breast cancer patients continues to evolve, we look to an individual patient’s genomic makeup as a guide, and its potential for clinical benefit embodied in the above patient’s story.
