Abstract
Objectives:
A definitive diagnosis of pulmonary sarcomatoid carcinoma cannot be made with small biopsies. In clinical practice, a diagnosis of advanced non-small-cell lung cancer with spindle cell and/or giant cell carcinoma (NSCLCsg), or possible sarcomatoid carcinoma, is acceptable. Therefore, we aimed to investigate the treatment patterns and outcomes of advanced NSCLCsg.
Materials and methods:
Between 01 January 2012 and 01 April 2021, patients with pathologically proven advanced NSCLCsg were enrolled. The choice of treatment was based on clinician discretion.
Results:
In all, 101 patients with advanced NSCLCsg were enrolled. In total, 77 (76.2%) patients received at least one line of systemic therapy; 44 patients (43.1%) had received platinum doublet chemotherapy; 27 (26.7%) patients had been treated with targeted therapies; and 23 patients (22.8%) had been given an immune checkpoint inhibitor (ICI). The median overall survival (OS) was 6.3 months [95% confidence interval (CI): 3.6–9.0 months]. Excluding patients without systemic therapy, patients who had received an ICI had better OS (median: 18.2 months) than those who had not (median 3.8 months, log-rank test p = 0.002). No significant difference in OS was detected between patients who had or had not received platinum doublet chemotherapy (log-rank test p = 0.279), or targeted therapy (log-rank test p = 0.416). Having received any systemic therapy [hazard ratio (HR): 0.33, 95% CI: 0.18–0.61, p < 0.0001) and ICI (HR: 0.38, 95% CI: 0.19–0.78, p = 0.008) were independent factors for better OS. Patients with programmed death ligand-1 (PD-L1) expression ⩾50% had better OS than those with PD-L1 expression <50% (HR: 0.51, 95%: 0.30–0.86, p = 0.012).
Conclusion:
Although advanced NSCLCsg has a poor survival outcome, our results showed that ICI may prolong OS in patients with advanced NSCLCsg. Further prospective studies are warranted to gain more understanding of the role of ICI in this specific patient population.
Keywords
Introduction
Pulmonary sarcomatoid carcinoma (PSC) is a rare type of non-small-cell lung cancer (NSCLC), accounting for only 0.5% of all lung cancers. 1 PSC is defined as a poorly differentiated tumor with non-carcinomatous elements, and can be further divided into five subtypes based on the 2021 World Health Organization (WHO) classification of lung tumors: namely, pleomorphic carcinoma, spindle cell carcinoma, giant cell carcinoma, carcinosarcoma, and pulmonary blastoma. 2 Pleomorphic carcinoma is the most common subtype of PSC. 3 The diagnosis of PSC should be made based on resection specimens, not on small biopsies or cytology specimens. However, most patients with advanced NSCLC were diagnosed based on small biopsy specimens; thus, PSC may be underestimated in advanced NSCLC patients due to the lack of a surgical specimen. Nevertheless, if small biopsy specimens of advanced NSCLC showed sarcomatoid features, such as discohesive giant cells with eosinophilic cytoplasm or spindle cell morphology, the tumor could be described as a non-small-cell carcinoma with spindle cell and/or giant cell carcinoma. 4 For the above reason, we use the term ‘non-small-cell lung cancer with spindle cell and/or giant cell carcinoma (NSCLCsg)’, instead of PSC in our study.
The clinical course of PSC has been reported to be highly aggressive, and the prognosis was poorer than that of other types of NSCLC.3,5,6 Median overall survival (OS) of advanced PSC was only 3–5.4 months.1,3,7 Due to its rarity, randomized controlled trials cannot be conducted to determine appropriate treatment. Therapeutic strategies for advanced PSC follow the general guideline of that for non-PSC NSCLC, but advanced PSC is often chemoresistant.3,6,8,9 Median OS of PSC patients receiving chemotherapy was only 4.3–8.7 months.3,6,8–11 Due to poor response to conventional chemotherapy, optimal advanced PSC-oriented strategies are being investigated. However, there has been no study investigating the prognosis or treatment outcomes of advanced NSCLCsg.
Molecular testing of known genetic abnormalities, including epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK), is recommended in PSC to find appropriate treatments. 4 Nevertheless, targetable mutations are less frequent in advanced PSC than in non-squamous NSCLC. 12 The efficacy of targeted agents in treating driver mutation-positive PSC is controversial due to limited studies. 13 However, immune checkpoint inhibitors (ICIs) have been proven to be an effective treatment for many chemoresistant cancers.14,15 The use of programmed death-1 (PD-1) or programmed death ligand-1 (PD-L1) inhibitors as treatment has had a dramatic response in patients with advanced NSCLC with a high PD-L1 expression.16,17 PSC has been shown to have even higher PD-L1 expression than other types of NSCLC. 18 As of now, the role of ICIs in treating advanced NSCLCsg is unknown.
The purpose of our study was to investigate the treatment patterns and efficacy of chemotherapy, targeted therapy, and ICIs in patients with advanced NSCLCsg in a real-world setting.
Methods
Patients and populations
This retrospective study was conducted at National Taiwan University Hospital, a 2400-bed tertiary-care center in northern Taiwan. Patients with advanced NSCLCsg between 01 January 2012 and 01 April 2021 were identified and reviewed by two thoracic pathologists (M.S.H. and Y.H.L.), based on the 2021 WHO classification of lung tumors (Supplemental Figure 1). 2 We enrolled patients with newly diagnosed stage III/IV or recurrent NSCLCsg who had not undergone curative-intent treatment. Recurrent NSCLCsg is defined as a recurrence of the disease in patients who had complete remission after curative treatment. Patients who were lost to follow-up within 1 month after diagnosis and those who had received ICIs in clinical trials were excluded. Baseline clinical characteristics, including age at diagnosis, sex, initial Eastern Cooperative Oncology Group (ECOG) performance score, smoking and disease status, oncogenic mutations (EGFR mutation by DNA direct sequencing, 19 matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) 20 or cobas EGFR Mutation Test v2 [cobasv2; Roche Molecular Systems, Pleasanton, CA, USA; KRAS and HER2 by MALDI-TOF MS; ALK by VENTANA ALK (D5F3) CDx Assay; 21 ROS1 by next generation sequencing (FoundationOne CDx); BRAF by VENTANA anti-BRAF V600E (VE1) immunohistochemical (IHC) assay; MET exon 14 skipping by reverse transcription-PCR)], tumor PD-L1 expression (Dako PD-L1 22C3 assay or VENTANA PD-L1 SP263 assay), systemic therapy regimen, and mortality, were recorded by chart review. This study was approved by the Institutional Review Board (IRB) of National Taiwan University Hospital (201910045RINC). The need for written informed consent was waived by the IRB because of its nature of retrospective analysis.
Treatment
The choice of treatment strategy was based on clinician discretion. Patients were tested for different kinds of driver mutations, such as EGFR, ALK, ROS1, KRAS, and BRAF, based on the judgment of the primary physicians. Targeted therapy was used for specific mutation-positive advanced NSCLCsg patients. If the treatment duration was less than 2 weeks, it was not counted as a treatment regimen.
Assessment
Tumor response to treatment was evaluated every 3 months based on routine clinical practice at our institute. The primary outcome was OS, which was defined as the duration between the date of diagnosis and the date of all-cause mortality. The survival status of patients who were lost to follow-up during the study period was checked in the Taiwan Death Registry. Progression-free survival (PFS) was defined as the duration between the start of new treatment and the time of disease progression. The best overall response was the best response recorded from the time of treatment initiation to disease progression. Tumor response was assessed using computed tomography every 3 months and was evaluated based on Response Evaluation Criteria In Solid Tumors version 1.1. The data cutoff date was 15 August 2021.
Statistical analysis
Statistical analyses were performed using SPSS 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables were reported as the median (range). Categorical variables were expressed as numbers (percentage), and chi-squared tests were used for comparison between two groups. OS was assessed using Kaplan–Meier analysis, and compared with the log-rank test. Prognostic factors for OS were estimated using Cox proportional hazards analysis. Variables with a p < 0.10 in univariate analysis were introduced into multivariate analysis. Statistical tests were two-sided and significance was set at p < 0.05.
Results
Patient characteristics
A total of 101 patients with primary or recurrent advanced NSCLCsg were enrolled. Of these, 82.2% (83/101) had initial stage IV NSCLCsg, and 17.8% (18/101) had recurrent NSCLCsg. In terms of the histology classifications of the non-sarcomatoid component, 59 patients (58.4%) had adenocarcinoma, 7 patients (6.9%) had squamous cell carcinomas, and 35 patients (34.7%) were classified as not otherwise specified (NOS), according to the 2021 WHO classification of lung tumors. 2 Of these patients, 86.1% (87/101) were tested for EGFR mutations (DNA direct sequencing for 29 patients, MALDI-TOF MS for 25 patients, and the cobas EGFR Mutation Test v2 for 33 patients), 67.3% (68/101) had ALK rearrangement VENTANA ALK (D5F3) CDx Assay testing, 50.5% (51/101) received ROS1 IHC testing, 30.6% (31/101) were tested for KRAS and HER2 mutations, 47.5% (48/101) had BRAF V600E IHC testing, and 82.2% (83/101) had PD-L1 testing (Dako PD-L1 22C3 assay for 46 patients and VENTANA PD-L1 SP263 assay for 37 patients). One patient had pre-existing interstitial pneumonia before any systemic therapy. The baseline characteristics of the 101 patients are shown in Table 1.
Clinical characteristics of 101 patients with advanced NSCLCsg.
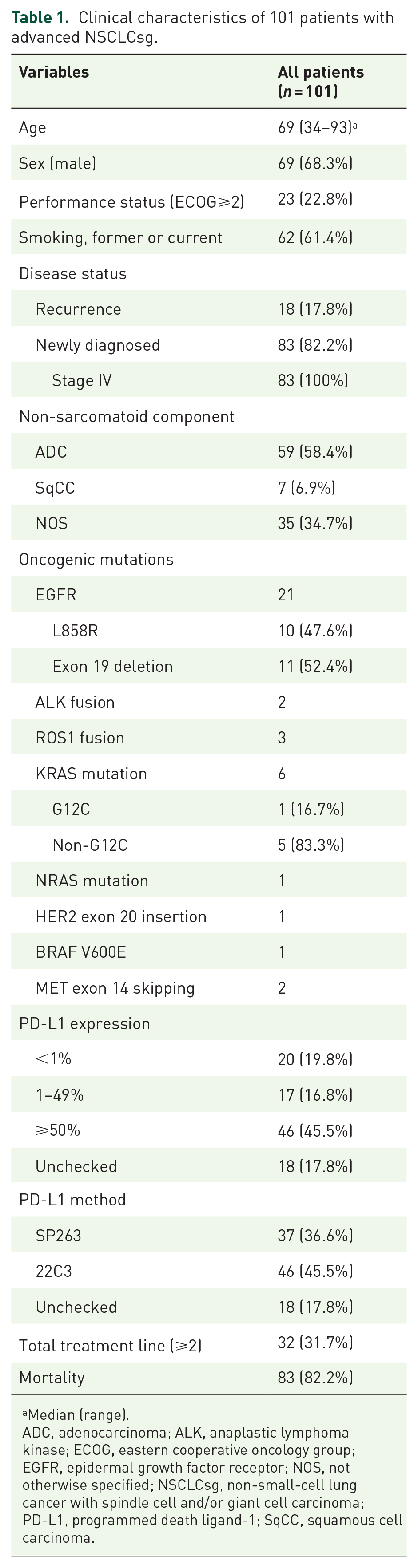
Median (range).
ADC, adenocarcinoma; ALK, anaplastic lymphoma kinase; ECOG, eastern cooperative oncology group; EGFR, epidermal growth factor receptor; NOS, not otherwise specified; NSCLCsg, non-small-cell lung cancer with spindle cell and/or giant cell carcinoma; PD-L1, programmed death ligand-1; SqCC, squamous cell carcinoma.
Systemic therapy
In all, 77 (77/101, 76.2%) advanced NSCLCsg patients received at least one line of systemic anticancer therapy, and the other patients (24/101, 23.8%) were given supportive care only. Of the patients receiving supportive care, 10 had a poor performance status (ECOG ⩾ 2); 7 had rapidly progressive disease; 6 had uncontrolled severe infection; and 1 patient refused systemic therapy. In all, 44 (44/101, 43.1%) advanced NSCLCsg patients received platinum doublet chemotherapy. Among them, 33 patients (33/44, 75%) were treated as first line, 8 (8/44, 18.2%) as second line, and 3 (3/44, 6.8%) as third line (Table 2). Of those patients treated with platinum doublet chemotherapy, 9.5% (4/44 patients) achieved a partial response, 34.1% (15/44 patients) had stable disease, and 56.8% (25/44 patients) had progressive disease. Supplemental Table 1 shows the details of the platinum doublet chemotherapy regimens. Furthermore, 21 patients had EGFR mutations, 6 had KRAS mutations, 2 had ALK rearrangements, 1 had a BRAF mutation, 2 had MET exon 14 skipping mutations, 3 had ROS1 rearrangements, 1 had a NRAS mutation, and 1 patient had a HER2 mutation. In total, 27 (26.7%) patients with driver mutation-positive advanced NSCLCsg were treated with targeted therapies, based on the corresponding genomic alterations. The most commonly used targeted therapy was EGFR tyrosine kinase inhibitors (TKIs) (19 patients, 70.4%); of these, 84.2% (16/19) were treated with EGFR TKIs as first-line treatment. Front-line targeted therapy included EGFR TKI (15.8%, 16/101), ALK (2.0%, 2/101), MET (1.0%, 1/101), HER2 (1.0%, 1/101), and ROS1 (1.0%, 1/101) inhibitors. In all, 13 of 19 (68.4%) patients who received EGFR TKI treatment had a partial response, and the median PFS of EGFR TKI treatment was 4.9 (95% CI: 0–9.9) months (Supplemental Figure 2).
Regimens used to treat patients with advanced NSCLCsg.

ALK, anaplastic lymphoma kinase; EGFR, epidermal growth factor receptor; ICI, immune checkpoint inhibitors, NSCLCsg, non-small-cell lung cancer with spindle cell and/or giant cell carcinoma; TKI, tyrosine kinase inhibitors.
ICI treatment was given to 23 of the 101 (22.8%) patients: 14 (14/23, 60.9%) were given ICI monotherapy and 9 (9/23, 39.1%) had ICI combined with chemotherapy. There was no baseline interstitial pneumonia in any of these 23 patients. The detailed regimens of ICI with/without chemotherapy are shown in Table 3. Most of the ICI monotherapy and ICI in combination with chemotherapy were given as first- or second-line treatments. In all, 32 of the 101 (31.7%) patients received at least two treatment lines. Figure 1 shows the treatment course of the ICI-treated advanced NSCLCsg patients. The median follow-up time after starting ICI was 15.4 months (range: 2.3–36.2). The median PD-L1 expression among patients with ICI treatment was 80% (range: 0 to >95%). Pembrolizumab (17/23, 73.9%) was the most commonly used ICI. Of the advanced NSCLCsg patients treated with ICI, 26.1% (6/23 patients) achieved a partial response, 43.5% (10/23 patients) had stable disease and 30.4% (7/23 patients) had progressive disease. There was no difference in response to ICI among the advanced NSCLCsg patients, with or without (partial response: 11.1% versus 35.7%, p = 0.328) combined chemotherapy. The median PFS for ICI with or without combined chemotherapy was 4.0 months (95% CI: 2.6–5.4) (Table 3). Kaplan–Meier analysis showed no significant difference in PFS between patients treated with ICI with or without chemotherapy [median PFS: 4.0 (95% CI: 1.4–6.6) versus 3.9 (95% CI: 1.2–6.7) months, log-rank test p = 0.155] (Figure 2). Two patients received targeted therapy after ICI [patient 17 (BRAF and MEK inhibitors), and patient 19 (ROS-1 inhibitor)], and one patient received targeted therapy before and after ICI [patient 21 (EGFR TKI)]. No drug-induced interstitial lung disease developed during ICI or targeted therapy in these three patients. Three patients received targeted therapy before ICI [patients 8 (Pan-HER inhibitor), 11 (EGFR TKI), and 15 (EGFR TKI)]. Only one patient (patient 11) developed drug-induced interstitial lung disease during treatment with ICI in combined with chemotherapy.
Characteristics of patients with advanced NSCLCsg who had received an ICI.

ICI with chemotherapy.
ADC, adenocarcinoma; ICI, immune checkpoint inhibitors; NOS, not otherwise specified; NSCLCsg, non-small-cell lung cancer with spindle cell and/or giant cell carcinoma; PD, progressive disease; PR, partial response; SD, stable disease; SqCC, squamous cell carcinoma.

Treatment course of 23 NSCLCsg patients who had received ICIs.

Kaplan–Meier analysis showed no significant difference in PFS between patients treated with an ICI with or without chemotherapy [median PFS: 4.0 (95% CI: 1.4–6.6) versus 3.9 (95% CI: 1.2–6.7) months, log-rank test p = 0.155).
Overall survival
The all-cause mortality rate was 82.2% (83/101). The median OS was 6.3 months (95% CI: 3.6–9.0 months) (Figure 3), with a median follow-up of 9.2 months (range: 0.2–42.1 months). Patients with NSCLCsg and an adenocarcinoma component had better OS than those with NSCLCsg and a NOS component (log-rank test p = 0.001) (Supplemental Figure 3). Advanced NSCLCsg patients that received any systemic therapy (median OS: 10.0 months, 95% CI: 5.7–14.4 months) had better OS than those that received only supportive care (median OS: 1.4 months, 95% CI: 0.8–2.1 months) (log-rank test p < 0.0001) (Supplemental Figure 4). Excluding patients without systemic therapy, Kaplan–Meier analysis showed no significant difference in OS between patients with and without platinum doublet chemotherapy (log-rank test p = 0.279), as well as targeted therapy (log-rank test p = 0.416) (Figure 4(a) and (b); Supplemental Figure 5(a) and (b)). We further divided patients into four groups according to whether they had prior exposure to platinum doublet chemotherapy and/or targeted therapy. Kaplan–Meier analysis also showed no significant difference in OS among the four groups (log-rank test p = 0.813) (Figure 5(a)).
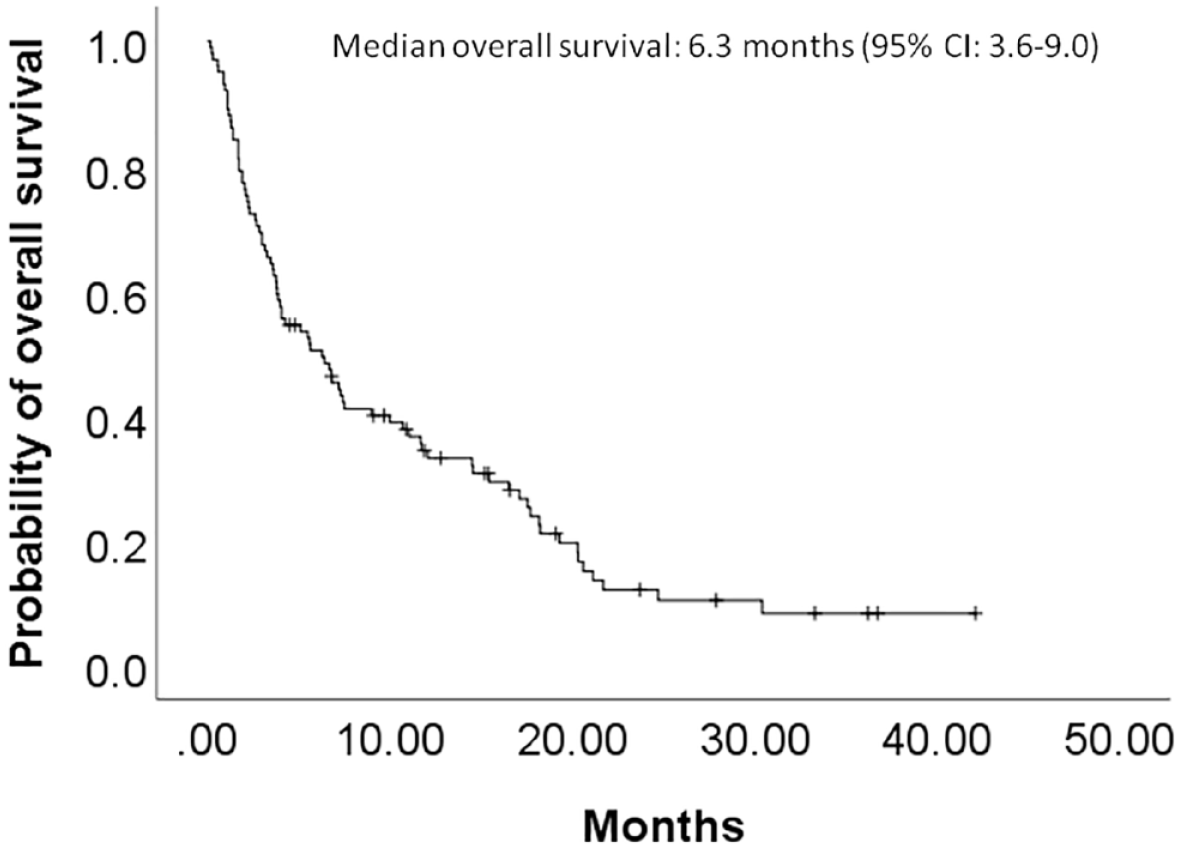
The Kaplan–Meier curve illustrating OS of advanced and recurrent NSCLCsg patients showed a median OS of 6.3 months (95% CI: 3.6–9.0 months).

Kaplan–Meier analysis comparing OS of patients, excluding those without systemic therapy (n = 77), based on different kinds of regimens, including platinum doublet chemotherapy (log-rank test p value: 0.279). (a) and targeted therapy (log-rank test p value: 0.416) (b).

Kaplan–Meier analysis showed no significant difference in OS among patients who had received platinum doublet chemotherapy only, targeted therapy only, platinum doublet chemotherapy, and targeted therapy, or non-platinum doublet chemotherapy only (log-rank test p = 0.813) (a). Kaplan–Meier analysis showed no significant difference in OS among patients with EGFR mutations and those with non-EGFR treatable oncogenic driver-positive NSCLCsg who had received targeted therapy and those who were not treated with targeted therapy (log-rank test p = 0.704) (b).
Since EGFR mutation was the most common driver oncogene in our patients, we categorized the patients with a driver oncogene into EGFR mutation and non-EGFR treatable oncogenic driver groups. Excluding those without systemic therapy, the median OS of the EGFR mutation and non-EGFR treatable oncogenic driver-positive NSCLCsg patients who had received targeted therapy was 17.1 (95% CI: 5.2–29.0) and 11.1 (95% CI: 1.3–20.8) months, respectively, and 7.4 (95% CI: 2.8–12.1) months for those who were not treated with targeted therapy. Kaplan–Meier analysis showed no significant difference in OS among these groups (log-rank test p = 0.704) (Figure 5(b)). However, patients who had received an ICI had better OS than those who had not (log-rank test p = 0.002) (Figure 6(a); Supplemental Figure 4(c)). The median OS of advanced NSCLCsg patients who had received an ICI was 18.2 months (95% CI: 17.0–19.4 months), and for those who had not received an ICI, 3.8 months (95% CI: 3.2–4.5 months, log-rank test p = 0.002). Nevertheless, Kaplan–Meier analysis showed no significant difference in OS between patients treated with ICIs with or without chemotherapy [median OS: 17.7 (95% CI: 11.8–23.5) versus 18.2 (95% CI: 12.4–24.1) months, log-rank test p = 0.911] (Figure 6(b)). Patients with PD-L1 expression ⩾50% had the best OS (log-rank test p = 0.008) (Figure 7). The median OS of patients with PD-L1 expression ⩾50% was 12.1 months (95% CI: 4.5–19.7 months), and that of patients with PD-L1 expression <50% and unchecked was 3.0 (95% CI: 0.9–5.1) and 3.6 months (95% CI: 3.0–4.2 months), respectively.

Kaplan–Meier analysis of OS of patients, excluding those without systemic therapy (n = 77), treated with or without ICIs (log-rank test p value: 0.002) (a). Kaplan–Meier analysis showed no significant difference in OS between patients treated with an ICI with or without chemotherapy (log-rank test p = 0.911) (b).

The Kaplan–Meier curve illustrating OS of NSCLCsg patients with different PD-L1 levels (log-rank test p value: 0.019).
Univariate and multivariate Cox proportional hazard analyses for OS were conducted, and the results are found in Table 4. Having received any systemic therapy (HR: 0.34, 95% CI: 0.18–0.62, p < 0.0001), ICI (HR: 0.45, 95% CI: 0.21–0.94, p = 0.033), and having a PD-L1 expression ⩾50% (HR: 0.45, 95% CI: 0.26–0.78, p = 0.004) were independent factors for better OS.
Univariate and multivariate analyses of covariables associated with OS of patients with advanced and recurrent NSCLCsg.

ADC, adenocarcinoma; CI, confidence interval; ECOG, eastern cooperative oncology group; ICI, immune checkpoint inhibitors; NOS, not otherwise specified; NSCLCsg, non-small-cell lung cancer with spindle cell and/or giant cell carcinoma; OS, overall survival; PD-L1, programmed death ligand-1; SqCC, squamous cell carcinoma.
Discussion
Our study included 101 advanced NSCLCsg patients with a median OS of 6.3 months. Despite the poor prognosis, having received any systemic therapy or ICI were independent factors predicting longer OS (median OS: 10 months and 18.2 months, respectively). In addition, patients with PD-L1 expression ⩾50% (median OS: 12.1 months) had better OS than those with PD-L1 expression <50% (median OS: 3 months).
The incidence of targetable mutations in advanced PSC varies widely in different studies. For example, the incidence of EGFR mutations is 0–28%, depending on the series, but this is surely less frequent than for adenocarcinoma-histologic NSCLC.12,13 The prevalence of MET exon 14 skipping mutations was higher in PSC than in non-PSC NSCLC.22,23 Available studies on the efficacy of targeted therapy for driver mutations in PSC are limited. A few cases of EGFR-mutated PSC treated with TKI have been reported, but the outcomes were controversial.24–26 In our study, targeted therapies did not improve OS in advanced NSCLCsg patients, compared to other systemic therapies. Among 19 EGFR-mutated patients who received EGFR TKI treatment, the median and OS were 4.9 (95% CI: 0–13.6) and 17.1 (95% CI: 5.2–29.0) months, respectively. However, the survival outcome was not in line with that of our previously reported 301 EGFR-mutated advanced NSCLC patients with a predominant adenocarcinoma histology. The median PFS of first-line gefitinib, erlotinib, and afatinib was 12.4 (95% CI: 9.1–15.6), 14.4 (95% CI: 10.8–18.0) and 12.4 (95% CI: 9.8–15.0) months, respectively, with a median OS of 37.0 months in a real-world cohort. 27 Thus, further prospective study is warranted to investigate the efficacy of targeted therapy in advanced NSCLCsg patients with driver mutations.
ICIs that block the PD-1/PD-L1 pathway have been shown to improve the outcomes of PD-L1-positive advanced NSCLC patients.16,17 A few case reports have indicated that advanced PSC was treated effectively by ICIs.28–31 A small retrospective report from New York discussed three advanced PSC patients that received pembrolizumab alone as first-line treatment: two of the three had a partial response and the other had stable disease. Two cases with advanced PSC treated with pembrolizumab alone as a second-line and third-line regimen achieved a complete response and partial response, respectively. All patients had a PD-L1 expression >75%. 32 A retrospective study from French centers reported 37 stage III/IV PSC patients who had progression on first-line platinum-based chemotherapy, and were treated with ICIs alone as second-line or later therapy (nivolumab, 86.5% of cases): 40.5% (15/37 patients) achieved a response, 24.3% (9/37 patients) had stable disease, and 32.4% (12/37 patients) had progressive disease. PFS was 4.89 (range: 0.3–35.7) months. OS from initiation of the ICI was 12.7 (range: 0.3–45.7) months. Of these patients, 2.7% (1/37 patients) had an EGFR mutation and 37.8% (14/37 patients) had a KRAS mutation. In addition, 94.7% (18/19) of the 19 (19/37, 51.4%) patients with a known PD-L1 status had a PD-L1 expression ⩾5%. The median PD-L1 expression was 70% (0–100). A trend toward a higher PD-L1 expression in responsive diseases was found, but without statistical significance. 33
In our study, most of the patients received pembrolizumab alone or in combination with chemotherapy. ICI-treated advanced NSCLCsg patients had a 13.3-month median OS (range: 0.57–33.3) from initiation of ICI; 26.1% (6/23 patients) achieved a partial response, 43.5% (10/23 patients) had stable disease, and 30.4% (7/23 patients) had progressive disease. The median PFS for ICI with or without combined chemotherapy was 4.0 months (95% CI: 2.6–5.4). However, there was no difference in best overall response rate (partial response: 11.1% versus 35.7%, p = 0.328), PFS [median PFS: 4.0 (95% CI: 1.4–6.6) versus 3.9 (95% CI: 1.2–6.7) months, log-rank test p = 0.155], and OS (partial response: 11.1% versus 35.7%, p = 0.328) between patients treated with ICI with or without chemotherapy.
We enrolled all patients with NSCLCsg regardless of prior exposure to ICI or not, and our results demonstrated that patients who had received ICI had better OS than those who had not (log-rank test p = 0.002). Among our patients, 13.0% (3/23 patients) had an EGFR mutation and 13.0% (3/23 patients) had a KRAS mutation, which differed from the French cohort study. The median PD-L1 expression among patients with ICI treatment was 80% (range: 0 to >95%), and 18 out of 23 patients (78.3%) had a PD-L1 expression ⩾5% in tumor specimens. Our study showed that advanced NSCLCsg patients with PD-L1 expression ⩾5% who had been treated with an ICI had better PFS than those with PD-L1 expression <5% (median PFS of PD-L1 ⩾5% versus PD-L1 <5%: 4.0 versus 1.2 months, log-rank test p = 0.014). Furthermore, patients with PD-L1 expression ⩾5% tended to have better OS than those with PD-L1 expression < 5% (median OS of PD-L1 ⩾5% versus PD-L1 <5%: 18.2 versus 14.6 months, log-rank test p = 0.172).
In NSCLC, PD-L1 from different biopsy sites and during different clinical courses had a distinct predictive value for ICI. 34 The cutoff for PD-L1 expression positivity in PSC or NSCLCsg is unknown, making it difficult to compare the results in different clinical data. Further studies designed to investigate the role of PD-L1 expression and ICI in advanced PSC or NSCLCsg are needed.
The rationales for the efficacy of ICIs in treating PSC include the following: First, 24–61% of PSC patients have high PD-L1 expression (PD-L1 ⩾50%).10,35,36 In our study, 45.5% (46/101) of NSCLCsg patients expressed high PD-L1 (PD-L1 ⩾50%) in tumor specimens. PD-L1 expression in tumor cells was related to a response to the anti–PD-1/PD-L1 blockade, due to an immunoactive microenvironment. 37 Second, PSC is related to smoking and has a high mutation burden. 38 PSC had a higher tumor mutation burden than non-PSC NSCLC, 23 and the mutation burdens of the sarcomatous component are even higher than those in the epithelial component. 35 In another study, 40.6% of 32 Chinese PSC patients had a high tumor mutation burden, 39 which was associated with better efficacy for the ICIs. 40
Our study has several limitations. First, the number of advanced NSCLCsg patients enrolled was small because of the rarity of the disease. Second, all data were collected retrospectively, and therefore, any broad generalizability is restricted. Third, previous studies indicated that PD-L1 testing using specimens older than 3 years may lead to underestimation. 41 In our study, the majority of specimens (40.9%, 34/83) used for re-evaluating PD-L1 expression were more than 3 years old.
In conclusion, based on our study results, advanced NSCLCsg patients receiving any treatment had better OS than those with supportive care only. Patients with PD-L1 expression ⩾50% had better OS than those with PD-L1 expression 0–49%. ICI was found to be effective in treating advanced NSCLCsg patients, yielding an 18.2-month median OS. Further prospective studies are warranted to confirm whether the clinical characteristics and outcomes of advanced NSCLCsg are the same as those for PSC, and to investigate the role of ICIs in this specific patient population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221133889 – Supplemental material for Real-world treatment patterns and outcomes among patients with advanced non-small-cell lung cancer with spindle cell and/or giant cell carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359221133889 for Real-world treatment patterns and outcomes among patients with advanced non-small-cell lung cancer with spindle cell and/or giant cell carcinoma by Chia-Ling Chang, Min-Shu Hsieh, Jin-Yuan Shih, Yi-Hsuan Lee, Wei-Yu Liao, Chia-Lin Hsu, Ching-Yao Yang, Kuan-Yu Chen, Jih-Hsiang Lee, Chao-Chi Ho, Tzu-Hsiu Tsai, James Chih-Hsin Yang and Chong-Jen Yu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
