Abstract
Background:
Since lobaplatin (LBP) has been approved to treat metastatic breast cancer in China, this study aimed to evaluate the safety and efficacy of LBP-based chemotherapy in clinical practice.
Methods:
This trial was a prospective, open-label, multicenter phase IV clinical trial that enrolled patients with unresectable locally advanced or recurrent/metastatic breast cancer from 34 sites between July 2013 and March 2017. Patients were treated with LBP monotherapy or in combination for four to six cycles. The primary endpoint was safety. Secondary endpoints included progression-free survival (PFS), objective response rate (ORR), and disease control rate (DCR).
Results:
A total of 1179 patients were analyzed; 59 (5.0%) were treated with LBP alone, 134 (11.4%) with LBP plus paclitaxel, 263 (22.3%) with LBP plus docetaxel, 237 (20.1%) with LBP plus gemcitabine, 403 (34.2%) with LBP plus vinorelbine, and 83 (7.0%) with other LBP-based regimens. The overall incidence of adverse events (AEs) was 95.2%, and 57.9% of patients had grade >3 AEs. The most common grade >3 AEs were neutropenia (43.9%), leukopenia (39.4%), anemia (17.8%), and thrombopenia (17.7%). LBP monotherapy showed the lowest incidence of grade >3 AEs (39.0%), followed by LBP plus docetaxel (52.9%), LBP plus paclitaxel (59.0%), LBP plus vinorelbine (62.5%), and LBP plus gemcitabine (62.9%). The ORR and DCR were 36.8 and 77.0%, respectively. The median PFS was 5.5 months (95% confidence interval: 5.2–5.9).
Conclusion:
LBP-based chemotherapy shows favorable efficacy in patients with advanced breast cancer, with manageable safety profile.
Trial registration:
This trial was registered with ChiCTR.org.cn, ChiCTR-ONC-13003471.
Introduction
Breast cancer is the most common cancer in women worldwide and one of the leading causes of cancer-related deaths among women. 1 Despite the substantial progress in patient management over the past few decades, metastatic breast cancer is still considered incurable. In the treatment of metastatic breast cancer, monotherapy is usually preferred due to a lower impact on the quality of life of patients while the combination therapy do not demonstrate an increase in overall survival (OS).2–4 Anthracyclines and taxanes are the mainstay chemotherapy agents for breast cancer, but many patients with metastatic breast cancer have already received these agents in the early course of disease. They cannot use them further due to drug resistance or cumulative toxicities. 5 For these patients, no standard regimen exists and platinum-based chemotherapy is one of the several options available.6–8
The first-generation platinum drug cisplatin (DDP) exhibits severe renal, auricular, neurological, and gastrointestinal toxicities. The second-generation platinum drug carboplatin (CBP) exerts bone marrow suppression and displays high cross-resistance with DDP. These characteristics restrict their long-term clinical application and treatment effect. 9
Lobaplatin (LBP) is a third-generation platinum drug discovered during the screening of platinum compounds for DDP-resistant tumors. LBP in combination with another chemotherapy agent (paclitaxel, pemetrexed, gemcitabine, vinorelbine, or docetaxel) was effective and well tolerated for patients with metastatic breast cancer.10,11
This study aimed to explore the characteristics of LBP treatment in patients with advanced breast cancer and investigate tailored regimens for specific populations. The results reflected the safety and efficacy of LBP-based chemotherapy in patients with advanced breast cancer, which might guide the rational use of LBP in clinical practice.
Methods
Trial design
This trial was a prospective, open-label, multicenter phase IV clinical trial. It was conducted in accordance with the Declarations of Helsinki and Good Clinical Practice, and was approved by the institutional review boards of the participating centers. All patients provided written informed consent. This study was registered with ChiCTR.org.cn, ChiCTR-ONC-13003470 (date of registration: August 11, 2013).
Participants
From July 30, 2013 to March 15, 2017, patients were enrolled from 34 sites in China. The eligible patients were females aged ⩾18 years; had histologically confirmed unresectable locally advanced or recurrent/metastatic breast cancer; had at least one measurable lesion according to the Response Evaluation Criteria In Solid Tumors (RECIST) version 1.1; had an Eastern Cooperative Oncology Group (ECOG) performance status of 0–2; had a life expectancy ⩾3 months; had adequate hematological (absolute neutrophil count ⩾1.5 × 109/L, platelet count ⩾100 × 109/L, and hemoglobin ⩾90 g/L), hepatic [serum total bilirubin and conjugated bilirubin lower than or equal to the upper limit of normal (ULN) × 1.5, and alanine aminotransferase (ALT) and aspartate aminotransferase (AST) ⩽ULN × 2.5 (⩽ULN × 5 for patients with liver metastasis)], and renal [creatinine ⩽ULN and creatinine clearance rate ⩾60 mL/min (Cockcroft–Gault formula)] function; had no plans to prepare for pregnancy during the study period; and were not breastfeeding. Either patients with or without prior treatment with anthracyclines and/or taxanes could be enrolled. Patients with active central nervous system disease or previous resistance to platinum were allowed to be enrolled. Patients with previous allergies to platinum compounds, coagulation dysfunction, or those who were considered unsuitable by investigators were excluded. The molecular subtype of breast cancer was extracted from previous pathological records, or determined using biopsy tissues by immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH). Human epidermal growth factor receptor 2 (HER2)-positive disease was defined as IHC score 3+, or 2+ with gene amplification by FISH.
Procedures
Patients were treated with LBP-based chemotherapy, including but not limited to LBP alone, LBP plus vinorelbine, LBP plus paclitaxel, LBP plus docetaxel, and LBP plus gemcitabine. 12 Investigators could choose one of the LBP-based regimens at their discretion. The dose of LBP was 50 mg/m2 when used alone and 30 mg/m2 when used in combination. Treatment was given for 4–6 cycles, and every 21 days were deemed as a cycle. If disease progression did not occur after 6 cycles, patients would receive tailored maintenance treatment regimens or discontinue treatment at investigators’ discretion. When grade ⩾3 adverse events (AEs) occurred, the necessity of dose reduction or discontinuation was judged by the investigators. Routine supportive care and other concomitant anticancer therapies (such as anti-HER2 therapy, endocrine therapy, radiotherapy, and surgery) were allowed during the treatment period.
Physical examination, electrocardiogram, tumor marker tests, and urinalysis were performed within 1 week before each cycle. Blood routine test and blood chemistry were performed at least once per week. Spiral computed tomography scan or magnetic resonance imaging was used for tumor assessment by investigators according to RECIST 1.1 at baseline (within 2 weeks before treatment), every two cycles during the treatment period, and every 3 months after treatment completion or discontinuation until disease progression or patient withdrawal from study. AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0. Antiemetic drugs were allowed for the prophylaxis and treatment of gastrointestinal toxicity, while granulocyte colony-stimulating factor, thrombopoietin, and interleukin-11 were only allowed for the treatment of hematological toxicity.
Endpoints
The primary endpoint was safety (incidence and severity of AEs). Secondary endpoints were progression-free survival (PFS), objective response rate (ORR), and disease control rate (DCR). PFS was defined as the time from the initiation of treatment to disease progression or death from any cause, whichever occurred first. ORR was calculated as the proportion of patients with complete response (CR) or partial response (PR). DCR was calculated as the proportion of patients with CR, PR, or stable disease.
Statistical analysis
According to the requirements of Chinese registration regulations, the number of valid patients in phase IV clinical trials should be more than 2000. Hence, two phase IV studies of LBP in small-cell lung cancer and metastatic breast cancer were conducted simultaneously, and the number of valid patients in each study was planned to be at least 1000. Considering the 20% dropout rate, the total number of patients in this study was approximately 1200.
Safety analysis was performed in the safety set, defined as all patients who received at least one dose of the study drug and had safety assessment. Baseline characteristics were summarized in the full analysis set, defined as all patients who received at least one dose of study drug. Efficacy was analyzed in the efficacy-evaluable set, defined as all patients who received at least one dose of study drug and had available efficacy evaluation data.
Categorical variables were presented as frequency (percentage) and analyzed using the chi-square test. Continuous variables were tested for normal distribution using the Kolmogorov–Smirnov test. They were presented as mean ± standard deviation (SD) if normally distributed or median (range) if not normally distributed. PFS was estimated using the Kaplan–Meier method, and the 95% confidence interval (CI) of PFS was estimated using the Brookmeyer–Crowley method. Statistical analyses were done with SAS 9.4 (SAS Institute, Cary, NY, USA).
Results
Characteristics of the participants
From July 30, 2013 to March 15, 2017, a total of 1181 patients were enrolled; 1179 patients received LBP-based chemotherapy and were included in the safety and full analysis sets (Figure 1). The mean age was 50.2 ± 9.4 years, and 449 (38.1%) patients were premenopausal women. The majority (94.9%) of patients had an ECOG performance status of 0 or 1. There were 235 (19.9%) patients with unresectable locally advanced breast cancer and 944 (80.1%) with recurrent/metastatic breast cancer; 341 (28.9%) patients had hormone receptor (HR)-positive, HER2-negative breast cancer, 436 (37.0%) had HER2-positive breast cancer, 252 (21.4%) had triple-negative breast cancer (TNBC), and 150 (12.7%) had unknown molecular subtype. Regarding treatment, 59 (5.0%) patients were treated with LBP alone, 134 (11.4%) with LBP plus paclitaxel, 263 (22.3%) with LBP plus docetaxel, 237 (20.1%) with LBP plus gemcitabine, 403 (34.2%) with LBP plus vinorelbine, and 83 (7.0%) with other LBP-based regimens (Table 1). Of 341 patients with HR-positive, HER2-negative breast cancer, 5 (1.5%) received concomitant endocrine therapy. Of 436 patients with HER2-positive breast cancer, 17 (3.9%) received concomitant anti-HER2 therapy (trastuzumab for all these 17 patients). Notably, 242 (20.5%) patients received <4 cycles of LBP-based chemotherapy because of patient refusal [98 (8.3%)], loss to follow-up [54 (4.6%)], switching to radiotherapy, surgery, or systemic therapy without LBP [36 (3.1%)], no clinical benefit [17 (1.4%)], intolerable toxicity [15 (1.3%)], death [7 (0.6%)], allergy to study drug [2 (0.2%)], or unknown reasons [13 (1.1%)]. Before enrollment, 141 (12.0%) patients were pretreated with taxanes, 136 (11.5%) with anthracyclines, 753 (63.9%) with both anthracyclines and taxanes, and 149 (12.6%) had no previous use of anthracyclines or taxanes. Of 436 patients with HER2-positive breast cancer, 25 (5.7%) had previous anti-HER2 therapy.

Patient flowchart.
Baseline characteristics.

Data were expressed as mean ± SD or n (%).
Other LBP-based regimens included LBP plus etoposide, LBP plus S-1, LBP plus irinotecan, LBP plus epirubicin, LBP plus pirarubicin, LBP plus doxorubicin, LBP plus cyclophosphamide, and LBP plus ifosfamide.
ECOG, Eastern Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; LBP, lobaplatin.
Safety
The incidence of any grade AEs and grade ⩾3 AEs was 95.2% and 57.9% in the safety set, respectively. Table 2 summarizes the incidence and severity of common AEs (incidence > 3%).Hematological toxicities showed the highest incidence of any grade AEs (leukopenia: 80.1%; neutropenia: 75.7%; anemia: 67.6%; and thrombopenia: 51.2%), followed by gastrointestinal toxicities (nausea: 30.6%; vomiting: 15.8%) and liver toxicities (elevated AST: 21.9%; elevated ALT: 20.4%). Regarding grade ⩾3 AEs, neutropenia (43.9%) and leukopenia (39.4%) were the most common events. Prophylactic antiemetic drugs were used in 947 (80.3%) patients. No treatment-related deaths occurred.
Incidence and severity of AEs (n = 1179).

Data were expressed as n (%).
AEs, adverse events; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
The subgroup analyses showed that the incidence of any grade AEs and grade ⩾3 AEs was similar among different molecular subtypes of breast cancer (HR-positive, HER2-negative breast cancer: 94.7% and 57.2%; HER2-positive breast cancer: 95.6% and 56.2%; TNBC: 94.4% and 62.3%). LBP monotherapy (83.1% and 39.0%) showed the lowest incidence of any grade AEs and grade ⩾3 AEs, followed by LBP plus docetaxel (93.5% and 52.9%), LBP plus paclitaxel (96.3% and 59.0%), LBP plus vinorelbine (96.0% and 62.5%), and LBP plus gemcitabine (98.3% and 62.9%) (Supplemental Table S1).
In the SS, dose reductions of LBP and its chemotherapy partner due to AEs occurred in 6.2% and 5.9% of patients, respectively. Discontinuation of the whole LBP-based regimen due to AEs was observed in 5.1% of patients, while 0.9% of patients only discontinued the chemotherapy partner of LBP due to AEs. Specifically, patients treated with LBP plus taxanes (docetaxel or paclitaxel) had the lowest dose reduction and discontinuation rates of LBP (3.8% and 2.0%) (Supplemental Table S2).
Efficacy
There were 1023 patients with available tumor evaluation data. In all, 26 patients achieved CR, and 350 achieved PR; the ORR was 36.8%, and DCR was 77.0%. ORR and DCR in different subgroups are shown in Table 3. A low ORR was observed in patients with brain metastasis (17.0%) or ECOG performance status 2 (24.1%). Patients with unresectable locally advanced breast cancer had an ORR of 48.3% and DCR of 82.9%, while those with recurrent/metastatic breast cancer had an ORR of 33.9% and DCR of 75.6%. Different molecular subtypes achieved similar ORR and DCR (HR-positive, HER2-negative breast cancer: 36.4% and 79.8%; HER2-positive breast cancer: 37.0% and 74.2%; TNBC: 36.5% and 74.9%). Among the different chemotherapy regimens, LBP plus taxanes showed the highest ORR and DCR (LBP plus docetaxel: 51.7% and 84.6%; LBP plus paclitaxel: 49.6% and 85.6%), followed by LBP plus gemcitabine (29.4% and 72.1%), LBP plus vinorelbine (28.9% and 76.3%), and LBP alone (26.9% and 63.5%).
ORR and DCR in different subgroups.
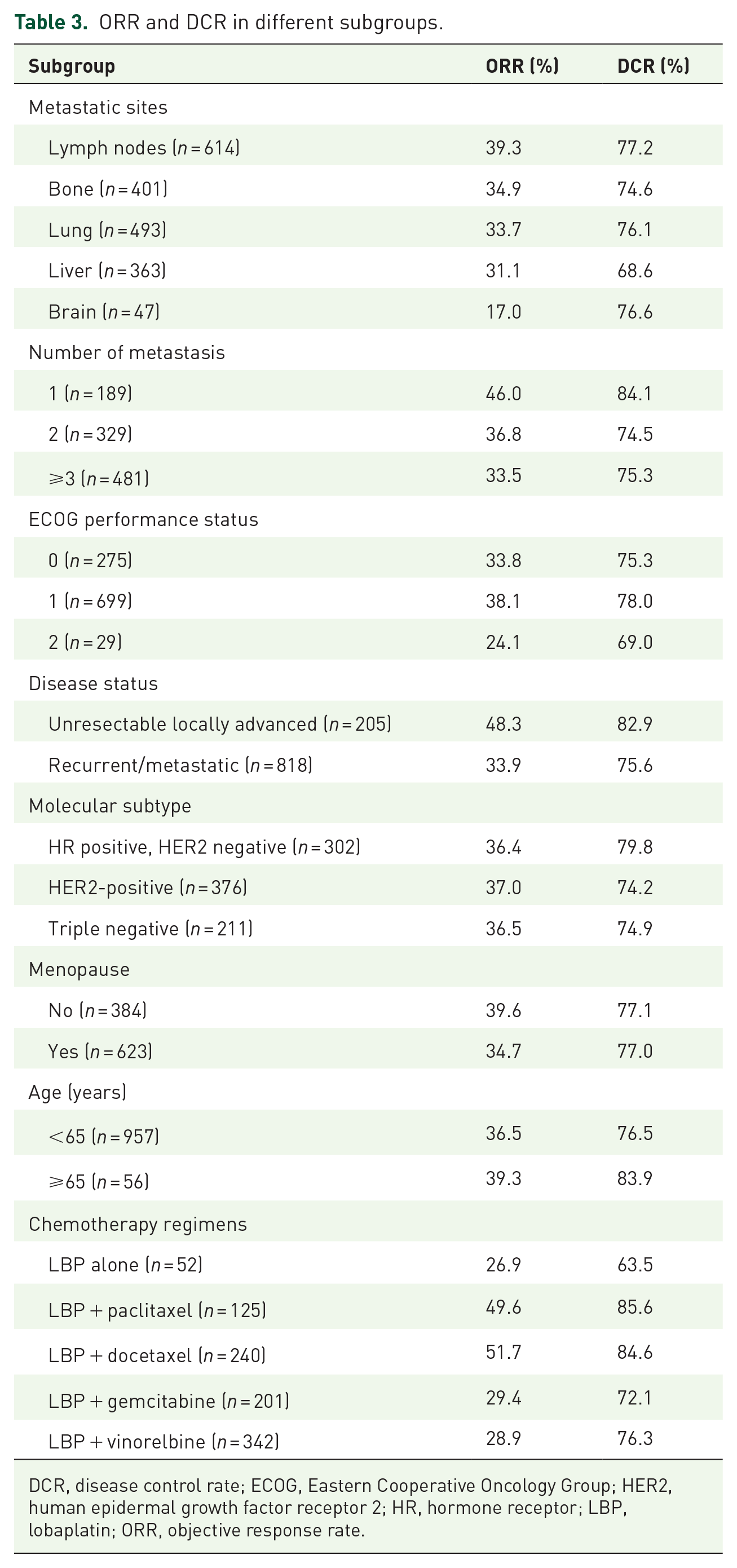
DCR, disease control rate; ECOG, Eastern Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; LBP, lobaplatin; ORR, objective response rate.
By the data cutoff date on May 15, 2019, the median PFS was 5.5 months (95% CI: 5.2–5.9) in the overall population (Figure 2). PFS in different subgroups is shown in Table 4. The median PFS for patients with metastases at lymph nodes, bone, lung, liver, and brain, was 5.7, 5.6, 5.5, 4.2, and 4.2 months, respectively. Patients with only one metastasis showed the highest median PFS (6.6 months), followed by those with two metastases (5.6 months), and those with at least three metastases (5.4 months). The median PFS was 6.4 months in patients with unresectable locally advanced breast cancer and 5.4 months in those with recurrent/metastatic breast cancer. The median PFS was the highest in patients with HR-positive, HER2-negative breast cancer (5.7 months), followed by those with HER2-positive breast cancer (5.4 months), and those with TNBC (4.6 months). Among the different chemotherapy regimens, the median PFS was 8.0 months with LBP plus paclitaxel, 7.0 months with LBP plus docetaxel, 5.1 months with LBP plus gemcitabine, 4.6 months with LBP plus vinorelbine, and 4.5 months with LBP alone.

Kaplan–Meier estimates of PFS.
PFS in different subgroups.

ECOG, Eastern Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; LBP, lobaplatin; PFS, progression-free survival.
Discussion
This phase IV trial investigated the safety and efficacy of LBP-based chemotherapy in a large population with advanced breast cancer. Most of patients had recurrent/metastatic breast cancer (80.1%) and had previous use of anthracyclines and/or taxanes (87.4%). The results confirmed the manageable safety profile of LBP. The efficacy data supported the use of LBP-based chemotherapy in clinical practice.
Patients with recurrent/metastatic breast cancer have poor tolerance and bone marrow function after multiple courses of treatment. Platinum-based chemotherapy should be selected carefully considering its severe side effects. 13 In our study, the incidence of liver toxicities was approximately 20% (grade 3–4: approximately 1%), and renal toxicities and peripheral neurotoxicities were infrequent. The incidence of nausea (30.6%; grade 3–4: 0.5%) and vomiting (15.8%; grade 3–4: 2.0%) was also low, which might be attributed to the high use rate of prophylactic antiemetic drugs (80.3%). Hematological toxicities were the main AEs of LBP-based chemotherapy, which were manageable by symptomatic treatments. No new safety signals were identified. Only a small proportion of patients reduced the dose (6.2%) or discontinued LBP (5.1%) due to AEs. A phase III trial in patients with locoregionally advanced nasopharyngeal carcinoma showed that LBP-based induction chemotherapy plus concurrent chemoradiotherapy resulted in significantly fewer grade 3–4 hematological (neutropenia, leukopenia, anemia) and gastrointestinal (nausea and vomiting) toxicity events than DDP-based therapy. 14 Some other randomized controlled trials in small-cell lung cancer and ovarian cancer demonstrated better tolerance and quality of life with LBP-based chemotherapy than with DDP- or CBP-based chemotherapy.15,16 LBP could be a better option as platinum-based chemotherapy, and our results demonstrated the manageable safety profile of LBP-based chemotherapy in patients with advanced breast cancer.
No more than 10% of breast cancer are metastatic at diagnosis, 17 and the development of anti-HER2 and endocrine therapies has greatly improved the survival of patients with early breast cancer. However, disease recurrence/metastasis and drug resistance still remain challenges in clinical practice, and chemotherapy was still the mainstay treatment for TNBC. The prognosis of metastatic breast cancer was poor, with a median OS of 4–5 years for HR-positive, HER2-negative subtype, 5 years for HER2-positive subtype, and only 10–13 months for TNBC. 17 When most patients with advanced breast cancer had failed with anthracyclines and/or taxanes, DDP- or CBP-based chemotherapy would result in an ORR of 15.7–44.7% and median PFS of 4.3–8.6 months.18–20 LBP-based chemotherapy in our study showed an ORR of 36.8% and median PFS of 5.5 months, within the range of these previous results of platinum-based chemotherapy.18–20 Specifically, a 10-year retrospective study showed an ORR of 38.5% and median PFS of 8.6 months with CBP-based chemotherapy in 148 patients with HR-positive, HER2-negative metastatic breast cancer, and 49.2% and 9.65 months in 65 patients with HER2-positive metastatic breast cancer. 19 However, another retrospective study showed a median PFS of 3.7 months with CBP-based chemotherapy in 48 patients with HR-positive, HER2-negative advanced breast cancer. 20 In our study, LBP-based chemotherapy resulted in an ORR of 36.4% and median PFS of 5.7 months in patients with HR-positive, HER2-negative advanced breast cancer, and 37.0% and 5.4 months in patients with HER2-positive advanced breast cancer. Different patient characteristics and sample size should be considered when indirect comparisons were performed across studies. In addition, it should be noted that a huge proportion of patients in our study did not receive concomitant anti-HER2 therapy, possibly due to financial burden. This indicated that HER2-positive advanced breast cancer would achieve more clinical benefits if anti-HER2 therapy was used in combination with LBP-based chemotherapy. For TNBC, the most aggressive subtype, previous studies showed an ORR of 26.7–56.6% and median PFS of 4.3–8.4 months with platinum-based chemotherapy.19,21–24 The ORR (36.5%) and median PFS (4.6 months) with LBP-based chemotherapy in our study were within the range of these previous results of platinum-based chemotherapy in patients with TNBC.19,21–24 Taken the safety and efficacy results together, LBP could be a good option when platinum-based chemotherapy was selected for the treatment of advanced breast cancer.
Interestingly, our study found that LBP plus taxanes might be the optimal combination, with the best clinical benefits and lowest dose reduction or discontinuation rate, which might deserve further investigations.
There are some limitations in this study. Long-term OS was not analyzed. In addition, quality of life questionnaires were not collected in our study. The impact of toxicities with LBP-based chemotherapy on quality of life in a large population could not be analyzed. However, previous randomized controlled trials have demonstrated better quality of life with LBP-based chemotherapy than with other platinum-based chemotherapies.15,16
In conclusion, this is the largest study of LBP-based chemotherapy in patients with advanced breast cancer, which confirms its manageable safety profile. LBP-based chemotherapy shows favorable efficacy, similar to other platinum-based chemotherapy in patients with advanced breast cancer. LBP could be a good option as platinum-based chemotherapy in clinical practice.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221122715 – Supplemental material for A prospective, open-label, multicenter phase IV clinical trial on the safety and efficacy of lobaplatin-based chemotherapy in advanced breast cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359221122715 for A prospective, open-label, multicenter phase IV clinical trial on the safety and efficacy of lobaplatin-based chemotherapy in advanced breast cancer by Min Yan, Peng Yuan, Quchang Ouyang, Ying Cheng, Guohui Han, Dewei Wang, Li Ran, Tao Sun, Da Zhao, Yuju Bai, Shun’e Yang, Xiaojia Wang, Rong Wu, Xiaohua Zeng, Herui Yao, Xuening Ji, Jun Jiang, Xiaohua Hu, Haifeng Lin, Liping Zheng, Zhitu Zhu, Wei Ge, Junlan Yang, Tongjian Cui, Xiaozhi Zhang, Fangyang Lu, Wenhui Li, Hongyan Xu, Mafei Kang, Ping Gong, Liqun Zou, Jiang Liu, Hongliang Zhang, Hao Yu and Binghe Xu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
