Abstract
Background:
Extrapulmonary neuroendocrine carcinoma (EP-NEC) are an aggressive subgroup of neuroendocrine neoplasms (NEN). Advanced EP-NEC is generally treated with platinum-based cytotoxic regimens, but progressive disease occurs rapidly, resulting in a poor prognosis. Genetic alterations in the mammalian target for rapamycin (mTOR) pathway have been identified in NEN, providing a rationale for treatment with the mTOR-inhibitor everolimus.
Methods:
A prospective phase 2 single-arm study included patients with advanced EP-NEC from three Dutch NEN expertise centres between March 2016 and January 2020. Treatment consisted of cisplatin 75 mg/m2 every 3 weeks in combination with daily everolimus 7.5 mg for a maximum of six cycles, followed by maintenance everolimus until disease progression. Primary endpoint was disease control rate (DCR), defined as the sum of overall response rate (ORR) plus the rate of stable disease according to RECIST 1.1, assessed at 9-week intervals. Toxicity was evaluated according to CTCAE version 5.0.
Results:
Thirty-nine patients, with a median age of 64 years (range: 28–74), of whom 20 (51%) were male, were enrolled. DCR was 82.1% (95% confidence interval (CI): 66.4–92.4), with an ORR of 58.9% (CI: 42.1–74.4). Median duration of response was 6.4 (CI: 5.8–7.0) months and median progression-free survival was 6.0 (CI: 4.3–7.8) months. Three patients (8%) had durable responses lasting > 12 months. Median overall survival was 8.7 (CI: 7.8–9.6) months. Most common grade 3/4 toxicities were haematological (36%) and renal (21%).
Conclusion:
Everolimus in combination with cisplatin is an effective first-line treatment option for advanced EP-NEC, especially in highly selected patients.
Trial registration:
Clinicaltrials.gov, NCT02695459, https://clinicaltrials.gov/ct2/show/NCT02695459.
Introduction
Neuroendocrine neoplasms (NEN) are a heterogeneous class of malignancies that may arise in various localizations throughout the body. NEN have been classified according to morphological properties and biological behaviour into well-differentiated neuroendocrine tumours (NET) and poorly differentiated neuroendocrine carcinomas (NEC). 1 This classification and hence the nomenclature of various NEN has been altered several times over the past decades.2–4 The most recent update in 2017 has identified three grades (G1–G3) of NET, and a solitary high-grade (G3) NEC. 1 By definition, NEC show a mitotic count of > 20 per 2 mm 2 and a Ki67-proliferation index of > 20%, although most NEC have a Ki67-index of > 55%.5,6 The majority of NEC are of pulmonary origin in the form of either small-cell lung carcinoma (SCLC) or large-cell neuroendocrine carcinoma (LCNEC).4,7,8 Extrapulmonary NEC (EP-NEC) mostly originate from the gastroenteropancreatic tract (GEP-NEC), accounting for around 35–55% of all NEC. 9 Other primary sites of EP-NEC include the genitourinary tract and the skin (i.e. Merkel cell carcinoma (MCC)).10,11
EP-NEC usually display very aggressive behaviour with up to 85% of patients presenting with locally advanced or metastatic disease at diagnosis.9,12,13 Due to the rarity of the disease, evidence regarding systemic treatment is scarce and is often based on guidelines for treating SCLC.8,9,14 At the time of initiation of this study, a few retrospective studies were available regarding cytotoxic therapies in EP-NEC.13,15 A large retrospective study of 305 patients with EP-NEC indicated that cytotoxic treatment offers survival benefit over a best supportive care (BSC) approach. They observed a median overall survival (OS) of 11 months with palliative chemotherapy, including first to third treatment lines, and a mere 1 month OS in patients who received BSC only. In this series, first-line chemotherapy resulted in an overall response (OR) of 31% and disease stabilization of 33%, summing to a disease control rate (DCR) of 64%. Also, a Ki67-index of > 55% cut-off was found to be significantly associated with worse OS (10 months versus 14 months). 13 More recently, a prospective study of patients with GEP-NEC or unknown primary NEC showed an OR of 50% and stable disease (SD) of 23% for first-line treatment and a progression-free survival (PFS) and OS of 6.2 and 11.6 months, respectively. 16 Most cytotoxic regimens used in the treatment of EP-NEC consist of a platinum backbone, often combined with etoposide or irinotecan.13,16 Currently, the European Society for Medical Oncology (ESMO) guidelines recommend cisplatin/etoposide or carboplatin/etoposide as first-line treatment for advanced EP-NEC. 14 Although highly responsive to these therapies, progression occurs rapidly, often with a strikingly poor prognosis due to the lack of treatment options. To illustrate this, a recent meta-analysis in patients with advanced EP-NEC showed that second-line treatment had very limited efficacy, with a pooled PFS of 2.5 months. 17
Mutations in the mammalian target of rapamycin (mTOR) are present in various cancers, including well-differentiated NET.18,19 mTOR is a serine/threonine kinase that regulates cell growth and proliferation, metabolism and angiogenesis. It has also been implicated in the pathogenesis of NET. Inhibition of the mTOR signalling pathway has shown antiproliferative effects in cell lines and primary cultures of human NET. 18 A therapeutic intervention that specifically targets this mTOR pathway is everolimus. This one-of-a kind anticancer drug has been extensively studied in randomized trials of NET of the pancreas, lung and small intestine.20–22 Based upon superior PFS data, albeit with somewhat disappointing effects on OS, everolimus has become part of standard of care for patients with these tumours.23–25 Preclinical studies have demonstrated synergistic antitumour activity of everolimus in combination with cisplatin, which prompted the necessity to further investigate this combination.26,27 The mechanism underlying this synergistic activity has not been fully elucidated, but it has been suggested that reducing cellular levels of p21, thereby impairing DNA repair, could be an underlying mechanism. 26
The poor efficacy of current treatment options for EP-NEC combined with the abovementioned interactions, as well as the widely accepted anticancer activity of everolimus in patients with NEN, provided the rationale to perform this multicentre phase 2 clinical trial to evaluate the efficacy and safety of everolimus in combination with cisplatin in patients with advanced EP-NEC.
Methods
Study design
A single-arm, open-label, three-centre, national phase 2 clinical trial was designed to assess antitumor activity and safety of cisplatin in combination with everolimus as first-line treatment in patients with advanced EP-NEC. Between March 2016 and January 2020, patients were included from three referral centres in the Netherlands (Erasmus Medical Centre in Rotterdam, Netherlands Cancer Institute (NKI) in Amsterdam and the University Medical Centre Groningen). The study complies with the Declaration of Helsinki rules and the principles of Good Clinical Practice guidelines. This study was approved by the institutional review board (IRB) of the NKI (organizing institute) under IRB-identification number: NL50842.031.15. Written informed consent was obtained from all patients. This trial is registered at clinicaltrials.gov (NCT02695459) on 1 March 2016. The first patient was included in the study on 21 March 2016.
Patients
Eligible patients were adults with histopathologically confirmed locally advanced or metastatic EP-NEC. All pathological samples were classified by a NEN expert pathologist. Inclusion criteria were a World Health Organization (WHO) performance status (PS) of 0–2, adequate bone marrow, liver and renal function (creatinine clearance > 60 ml/min); and an estimated life expectancy of > 3 months.
Patients were excluded if they had received previous chemotherapy for advanced or metastatic EP-NEC or had previously been exposed to everolimus. Neo-adjuvant and peri-operative chemotherapy or chemoradiation with curative intent was allowed if at least 6 months had elapsed between completion of therapy and enrolment in the study.
Study treatment
Study treatment consisted of daily everolimus 7.5 mg/day combined with cisplatin 75 mg/m2 every 21 days, up to a maximum of six cycles, unless withdrawn earlier due to unacceptable toxicity or progressive disease. Everolimus and cisplatin dose was established based on a phase 1 study in patients with advanced head and neck tumours. 28 After six cycles of cisplatin and everolimus were completed, patients continued with single-agent everolimus 7.5 mg/day until disease progression or unacceptable toxicity. On day 1 of every cycle, patients were evaluated for renal function, myelosuppression, ototoxicity and peripheral neuropathy. Cisplatin was switched to carboplatin with an AUC = 4 if creatinine clearance had dropped below 50 ml/min, or when grade 3 or higher ototoxicity or peripheral neuropathy occurred. When creatinine clearance had decreased to 50–60 ml/min, cisplatin was reduced by 20% in the next cycle. Cisplatin was only administered when neutrophils were ⩾ 1.5 × 109/l and thrombocytes ⩾100 × 109/l. When these were below 1.5 and 100, respectively, cisplatin was postponed for 1 week. Similar cut-offs were used if carboplatin was given. After recovery of bone marrow toxicity, cisplatin or carboplatin was given with a 20% dose reduction.
Everolimus doses were adjusted to 5 mg/day (−1 dose level) or 5 mg every other day (−2 dose level) when tolerability issues occurred, including cytopenias, hepatotoxicity, infection, skin toxicity, oral mucositis, pneumonitis, hyperlipidaemia or hyperglycaemia. Dose reductions below −2 dose level were not allowed and patients would go off study.
After disease progression, patients were treated at the discretion of the treating physician and were monitored for survival.
Study endpoints
Primary endpoint of this study was DCR, defined as the sum of overall response rate (ORR) consisting of complete (CR) and partial response rate (PR) plus the rate of SD, according to RECIST 1.1, assessed at 9-week intervals. Patients were evaluable for response if at least one follow-up examination was performed.
Secondary endpoints were PFS, according to RECIST 1.1; duration of response (DOR); OS, defined as death from any cause; and safety of everolimus in combination with cisplatin/carboplatin.
Adverse events (AEs) were defined as any undesirable experience occurring to a subject during the clinical trial, whether or not considered to be related to the investigational drug. AEs were graded according to Common Terminology Criteria for Adverse Events (CTCAE) (version 5.0).
Statistical analysis
A two-stage phase 2 design was set up with response as outcome allowing for early termination should the response rate appear to be (unacceptably) low. Given the evidence in advanced EP-NEC at study initiation, a DCR of 50% or more would warrant further investigation and continuation of the study. Applying the ‘Simon 2-stage minimax’ design, with an α of 0.10 (the probability of rejecting the null hypothesis when it is in fact true) and a power of 90% (the probability of rejecting the alternative hypothesis), 28 patients had to be enrolled in the first stage, with an additional 11 patients to be recruited in the second stage if DCR proved acceptable (to a total of 39 evaluable patients). The combination would be deemed to be effective if the total number of responses exceeded 16.
The primary endpoint DCR was calculated using the Clopper–Pearson method. Patients who experienced early death (within 8 weeks of enrolment in the study) were considered as nonresponders.
Descriptive statistics were used for baseline characteristics: median with ranges and numbers with frequencies for continuous and categorical characteristics, respectively. Kaplan–Meier method was used for time-to-event analysis. DOR was presented for all patients who presented with an objective (complete or partial) response and was measured from the date of treatment initiation until date of documented progression. If a new treatment was started before progression, DOR was censored on the date of new treatment. Analyses were performed using R statistical software version 4.1.1.
Results
Patients
The predefined interim analysis showed a DCR of 78.6% (confidence interval [CI]: 59.0–91.7), so both steps of the ‘Simon 2-stage minimax’ design were completed. A total of 39 patients were included with a median age of 64 (28–74) years. Gender was equally distributed with 20 (51.3%) male patients. Most patients (n = 25, 64.1%) had their primary tumour arising from the gastroenteropancreatic tract; 11 patients (28.2%) had colorectal, 6 patients (15.4%) pancreatic, 4 patients (10.3%) oesophageal, 3 patients (7.7%) gastric and 1 patient (2.6%) had an appendiceal NEC. Five patients had gynaecological primary tumours, of whom four (10.3%) had a NEC of the cervix and one patient (2.6%) had an ovarian NEC. Three patients (7.7%) had a MCC and six patients (15.4%) had a NEC of unknown primary location. Most common metastatic sites were liver in 32 (82.1%) and lymph nodes in 27 (69.2%) patients. Nine (23.1%) patients had undergone previous surgery for their primary tumour, two (5.1%) of which had received postoperative radiotherapy. No prior neo-adjuvant or peri-operative chemotherapy treatment was given. All patients had a poorly differentiated morphology. For one patient, Ki67-index could not be reliably determined. For patients with known Ki67-index (n = 38, 97.4%), median Ki67-index was 80% (40–100%). One (2.6%) patient had a Ki67-index below 55%, 24 (61.5%) patients had a Ki67-index of 55–80% and 13 (33.3%) had a Ki67-index > 80%. All patients had a WHO PS ⩽ 1. Baseline characteristics are summarized in Table 1.
Baseline characteristics of all included patients.

GEP-NEC, gastroenteropancreatic neuroendocrine carcinoma; WHO PS, World Health Organization Performance Score.
Ki67-index for 38 out of 39 patients with known Ki67-index.
Efficacy
Best OR was a CR in 1 (2.6%) patient, PR in 22 (56.4%) patients and SD in 9 (23.1%) patients, with a DCR of 82.1% (CI: 66.4–92.5) and an ORR of 58.9% (CI: 42.1–47.4). For all patients with a response, median DOR was 6.4 (CI: 5.8–7.0) months. Three (7.7%) patients had a DOR of > 12 months. Of these, two patients had an unknown primary NEC, and one had a colorectal NEC. All three patients had liver metastasis at baseline and one patient also had a metastatic lesion in the pancreas; the sum of lesions were 41, 123 and 144 mm. The Ki67-index for these patients was 70–80%. DOR and survival after progression are shown in Figure 1. No differences in responses were found for subgroups, including gender, PS, previous therapies or primary tumour origin. DCR according to subgroups with corresponding confidence intervals can be found in Table 2.

Swimmersplot showing all patients with time to progression and to death or end of follow-up. One patient received immunotherapy after study termination and was alive at end of analysis, 45.9 months after study treatment initiation. Time-axis is interrupted due to the relatively long survival of this patient compared with other study participants. GEP-NEC: gastro-entero-pancreatic neuroendocrine carcinoma.
Disease control rate according to subgroups, with corresponding Clopper–Pearson 95% confidence intervals.
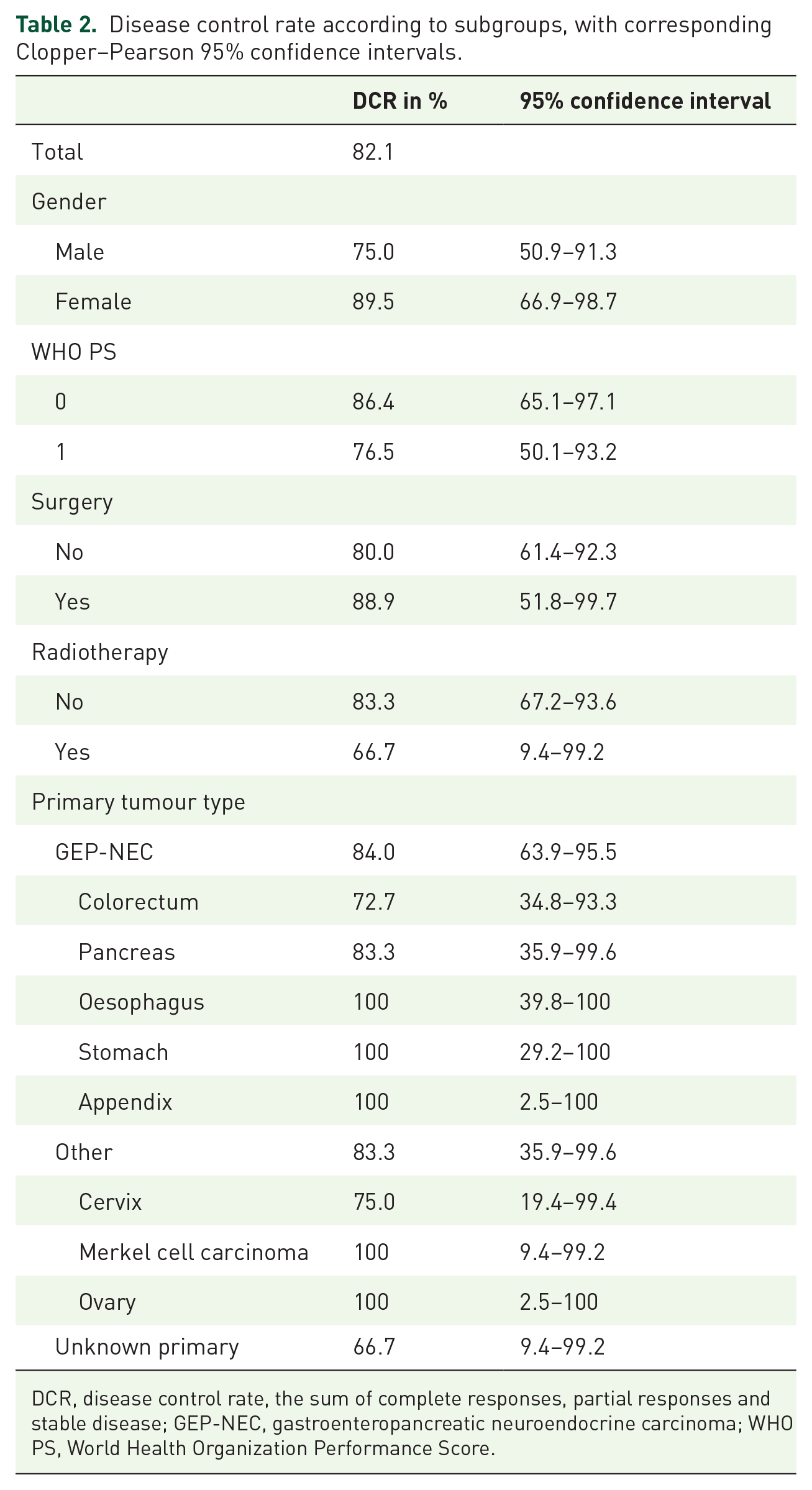
DCR, disease control rate, the sum of complete responses, partial responses and stable disease; GEP-NEC, gastroenteropancreatic neuroendocrine carcinoma; WHO PS, World Health Organization Performance Score.
Regarding the patients with MCC, during the conduct of the study approval for the immune checkpoint inhibitor, avelumab was granted for the treatment of MCC. One patient received avelumab prior to study inclusion, two patients received avelumab following progression on the current study treatment. Of these, one patient was alive with disease at time of study analysis (45.9 months after study initiation).
Median PFS for all patients was 6.0 (4.3–7.8) months and median OS was 8.7 (7.8–9.6) months. PFS and OS are shown in Figure 2.
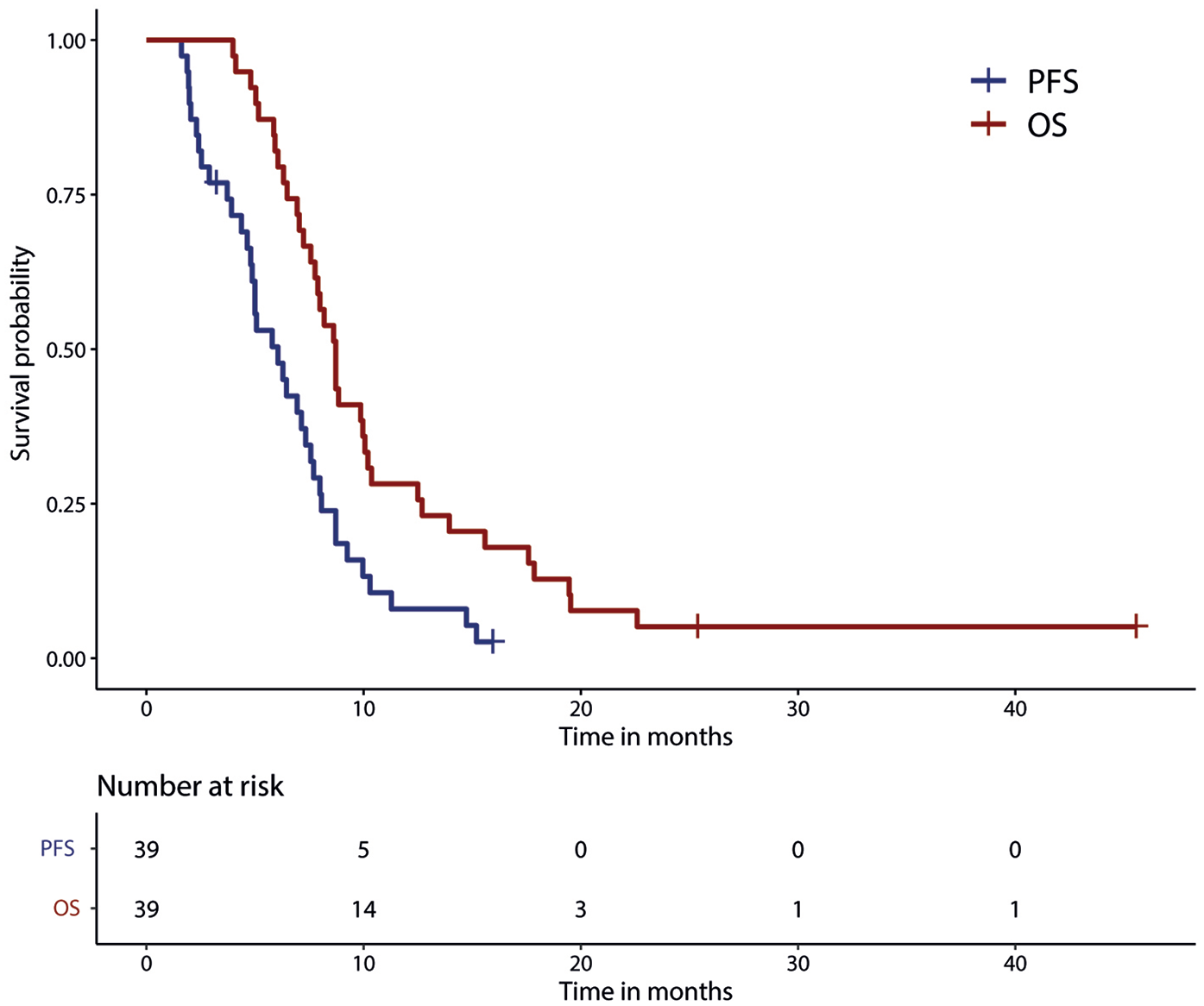
Progression-free survival (PFS) and overall survival (OS) for all patients.
Safety
Patients received a median of 4 (1–6) cycles of cisplatin. Thirteen (33.3%) patients switched to carboplatin, after a median of two cycles (1–5) and received an additional median of three (1–5) cycles (totalling up to a maximum of six platinum-based cycles). Dose reductions of cisplatin occurred in 28 (71.8%) patients. Median everolimus exposure in the entire cohort was 19 (3–57) weeks, and seven (17.9%) patients had dose reductions for everolimus.
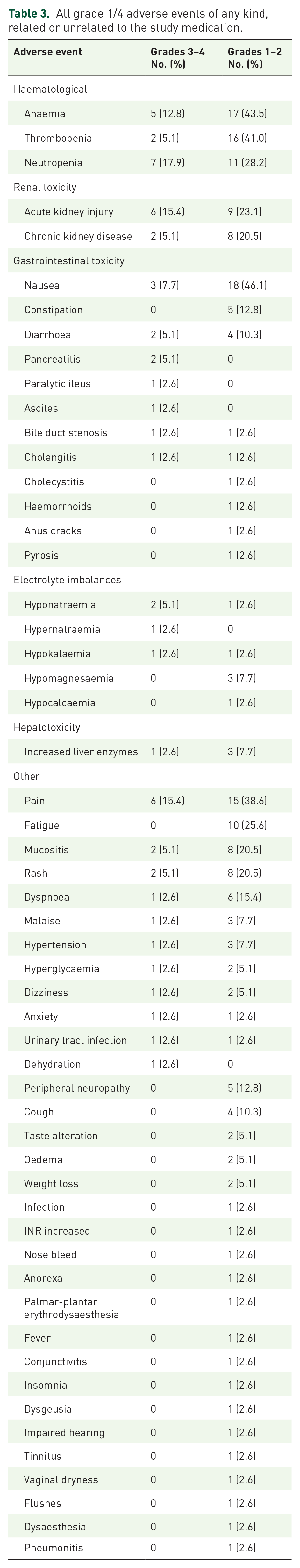
Thirty-five (89.7%) patients experienced grade 1–4 AEs of any kind, related or unrelated to the study medication. Of these, most common grade 1–2 events were nausea in 18 (46.1%) patients, pain in 15 (38.6%) patients and haematological AEs: anaemia in 17 (43.5%); thrombopenia in 16 (41.0%) and neutropenia in 11 (28.2%) patients. Regarding grade 3/4 AEs, 33 (84.6%) patients experienced grade 3/4 AEs of any kind. Most treatment-unrelated AEs consisted of pain (abdominal or other) and were present in six (15.4%) patients. A total of 22 (56.4%) patients experienced treatment-associated grade 3/4 AEs. Haematological toxicity was most common with a total of 14 (35.9%) patients, including 5 (12.9%) patients with anaemia, 7 (17.9%) with neutropenia and 2 (5.1%) with thrombopenia. Renal toxicity occurred in eight (20.5%) patients, treatment-associated gastrointestinal toxicity occurred in five (12.8%) patients, and electrolyte imbalances in four (10.3%) patients. No grade 5 AEs occurred. All grade 1/4 AEs are summarized in Table 3.
All grade 1/4 adverse events of any kind, related or unrelated to the study medication.
Discussion
In this phase 2 clinical trial, everolimus in combination with cisplatin showed to be an effective first-line treatment in patients with advanced EP-NEC.
This study was the first to investigate the combination of targeted therapy with conventional, platinum-based cytotoxic therapy in patients with advanced EP-NEC. Interestingly, the combination of everolimus with cisplatin/carboplatin used in this study showed comparable response rates, DORs, survival and AEs compared with studies investigating platinum-based cytotoxic combination therapies.16,29 For instance, regarding ORR, Walter et al. 16 performed a prospective observational study in patients with advanced EP-NEC and found an ORR of 50% for patients treated with cisplatin and etoposide, comparable with the ORR observed in this study. Other, retrospective studies found ORRs ranging from 28% to 52%.5,30–32
DOR and survival in our study were similar to that observed in the study of Zhang et al. in which patients with advanced GEP-NEC were randomly assigned to receive cisplatin with etoposide (EP) or cisplatin with irinotecan (IP). The study was terminated early due to slow accrual (66 patients of planned 144 included). At premature analysis PFS of 6.4 months and 5.8 months for EP and IP, respectively, was noted. 33 The OS in our cohort was slightly shorter than found in the aforementioned study by Walter et al. (8.7 versus 11.6 months). This might be explained by the fact that a third of the patients included in our study had a Ki67-index > 80%. Previous reports identified a Ki67-index cut-off of 55% to be associated with worse survival. 13 Although Walter et al. 16 found no difference in survival between patients with a Ki67-index < 55% and > 55%, their cohort only included 18% of patients with a Ki67-index > 80. Therefore, the somewhat less favourable outcome in our study might still be due to the higher proportion of more aggressive cancers.
AEs mainly consisted of haematological and renal toxicities, and the percentage of patients that experienced grade 3/4 AEs was comparable with that from studies of cisplatin combined with etoposide. The most striking difference is the absence of alopecia in our study population, as this is a side effect caused by etoposide, and does not occur in cis-/carboplatin or everolimus. Alopecia is known to have a significant impact on the quality of life and wellbeing of cancer patients.34,35 As efficacy of the cisplatin and everolimus combination seems similar to current recommended treatment of cisplatin and etoposide, but an important quality of life related side effect is avoided, the choice of treatment by both clinicians and patients is likely to favour everolimus with cisplatin.
The added survival benefit of everolimus to cisplatin in EP-NEC remains unclear. Although everolimus was able to improve cisplatin-resistance in vitro, 36 our study results show that everolimus was unable to uphold such a resistance for a longer duration than current therapies. Nevertheless, our cohort included three patients that had a DOR of > 12 months, suggesting that highly selected patients might have particular benefit from this combination. Similar results were found a study that investigated everolimus in combination with cisplatin and paclitaxel in LCNEC, with a median PFS of 4.4 months and OS of 9.9 months. There, 34% of 49 patients were alive at 1 year after treatment initiation, again suggesting the possible benefit for highly selected patients with NEC. 37 Unfortunately, as molecular and mutational tumour analyses were not performed in their study as well as in ours, mechanisms underlying these notable effects remain to be elucidated.
Besides acting as a synergistic component to platinum, everolimus has also been investigated as a single-agent after progression on platinum-containing chemotherapy in patients with pancreatic NEC (pNEC) or in SCLC.38,39 This resulted in a disappointing PFS of 1.2 months and an OS of 7.5 months for pNEC, 38 and similarly, a PFS of 1.3 months and OS of 6.9 months for SCLC. 39 Currently, there are a few randomized trials that involve different treatment combinations registered for first-line treatment of patients with EP-NEC (such as NCT04325425 and NCT02595424). These will hopefully further contribute to unveiling the tumour resistance mechanisms and improvement of survival of patients with EP-NEC.
A possible limitation of this study is that patients with MCC were also included in the EP-NEC cohort. This might have increased the heterogeneity of the study cohort, as MCC has a slightly different pathogenesis, including the oncogenic Merkel cell polyomavirus and ultraviolet exposure.40,41 Nonetheless, at the time of study initiation in 2016, no other systemic treatments for advanced MCC were approved, and MCC had a similar treatment regimen and prognosis as all other EP-NEC. And although one patient had received avelumab prior to study initiation, Figure 1 shows that patients with MCC were randomly distributed across the cohort and responses to therapy, hence were very unlikely to influence the results.
A major strength of this study is the prospective, multicentre study design, which is challenging when studying such rare diseases. This study is one of very few that managed to complete predefined accrual and hence is adequately powered. By including patients from three large referral centres in the Netherlands, this study provides insight into the nationwide approach in the treatment of EP-NEC, as well as epidemiological aspects such as the occurrence and survival of patients with EP-NEC.
In conclusion, the combination of everolimus with cisplatin is considered to be an effective treatment option for patients with advanced extrapulmonary NEC. While, in general, treatment-related AEs are in line with those observed in more classic cytotoxic regimens, absence of alopecia could favour this regimen. The observation of some patients obtaining DOR exceeding 12 months urges for additional in-depth analysis of so far unknown predictive biomarkers in this highly aggressive disease. If this research leads to real patient selection, this could be considered to be nothing less than a giant leap forward.
Footnotes
Acknowledgements
The authors thank all the patients and their families, the Dutch patient advocacy group for neuroendocrine cancers ‘Stichting NET groep’ and the investigators of the study and supporting teams at each of the participating centres.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by an unrestricted grant from Novartis [no grant number available].
