Abstract
Introduction:
Selected patients with locally advanced or metastatic soft tissue and bone sarcomas (STBS) may benefit from intensive local treatment, such as stereotactic radiotherapy (SRT). This study aimed to summarize the utilization and outcomes of SRT in STBS and to identify predictive factors for progression and survival.
Materials and methods:
Consecutive patients with advanced STBS who underwent STBS in a sarcoma tertiary center were identified. We collected tumor- and treatment-related factors. Endpoints comprised time to local progression (TTLP), local progression-free survival (LPFS), time to progression, progression-free survival, and overall survival (OS). The Cox proportional-hazards model was used to identify prognostic factors.
Results:
We identified 141 patients who underwent 233 SRTs. Median follow-up was 21 months. Local and distant progression occurred after 19 and 163 SRTs, respectively. SRT for lung metastases was predictive for better TTLP and LPFS (hazard ratio, HR = 0.12, p = 0.007 and HR = 0.42, p = 0.002, respectively). Bone sarcoma (HR for TTLP = 3.18, p = 0.043; HR for LPFS = 1.99, p = 0.028) and lower administered dose (HR for TTLP = 0.98, p = 0.007; HR for LPFS = 0.99, p = 0.012) were predictive for worse TTLP and LPFS. SRT for oligometastases (HR = 0.46, p = 0.021) and lung metastases (HR = 0.55, p = 0.046) was predictive for better OS, whereas diagnosis of bone sarcoma (HR = 2.05, p = 0.029) was predictive for worse OS.
Conclusion:
SRT provides excellent local control in STBS patients without significant toxicity. Patients with oligometastatic disease, lung metastases, and soft tissue sarcomas benefit the most from SRT. The dose escalation moderately enhances local control; however, it does not translate into better survival.
Keywords
Introduction
Soft tissue and bone sarcomas (STBS) are a heterogeneous group of rare cancers that require multidisciplinary treatment in high-volume tertiary centers. Due to the hematogenous pattern of spread, the most frequent site of distant metastases is lungs; in practice, however, STBS may metastasize into any part of the body. 1 In the past, the only treatment for unresectable or metastatic disease was systemic therapy supplemented with palliative radiotherapy. The wide introduction of new paradigms in oncology – namely, oligometastatic and oligoprogressive disease – and the introduction of modern systemic therapies opened new possibilities for patients with unresectable or metastatic STBS. 2 Intensive local treatment may be effectively combined with systemic therapy to provide prolonged control of the disease.3,4 Stereotactic radiotherapy (SRT) may be a local treatment option for patients with advanced STBS. In addition, SRT may delay starting or changing the systemic treatment. 5
Randomized clinical trials should be considered as the golden standard to validate new treatment proposals, but in rare cancers like sarcomas, this may be unfeasible. Due to the rarity of STBS and internal heterogeneity of this group, no randomized study nor large retrospective studies on this topic exist. The choice of candidates for SRT and the selection of proper fractionation regimens remain challenges. On one hand, STBS are considered radioresistant. Thus, the higher total dose will be delivered, the better local control should be. On the other hand, the higher dose could increase the risk of toxicity and limit further use of SRT in the case of recurrence near the irradiated volume. Interestingly, new studies on their radiosensitivity gave surprising results.6,7 For example, myxoid liposarcomas present excellent pathological response to low-dose preoperative radiotherapy with 100% of 2-year local control rate in two phase II clinical trials.8,9 Moreover, even within the same STBS pathological subtype, the radiosensitivity may vary. 7 The analysis of real-world evidence could provide valuable insights into outcomes of SRT in routine practice and help to optimize the treatment. 10
The objectives of this study were to summarize the utilization and outcomes of SRT in the management of advanced STBS, identify the patients who benefit the most, and check whether the given dose matters.
Hence, we report the results of a large retrospective cohort analysis from a high-volume sarcoma tertiary center.
Materials and methods
Endpoints
To investigate our objectives, we planned to provide the descriptive statistics of utilization, safety, and efficacy of SRT for patients with STBS and identify the sarcoma-related and SRT-related predictive factors for progression and survival in the form of time to local progression (TTLP), local progression-free survival (LPFS), time to progression (TTP), progression-free survival (PFS), and overall survival (OS).
Definitions
All patients were discussed at the sarcoma multidisciplinary tumor board meeting.
We defined oligometastatic disease as one to five metastatic lesions with controlled primary tumor that can be effectively and safely managed with local treatment. 11 According to the National Comprehensive Cancer Network Guidelines for Soft Tissue Sarcoma, version 2.2021, SRT is one of the treatment options of oligometastatic sarcomas. 12 In our sarcoma center, SRT for oligometastases was offered to the patients who were candidates for definitive treatment but not amenable to curative surgery or refused it. Metastasectomy may be considered unfeasible in the case of marginal resectability or unresectability, predictable adverse functional outcomes or high risk of postoperative morbidity, for example, associated with comorbidities.
Oligoprogressive disease is a novel concept in oncology, where after an initially effective systemic therapy for polymetastatic disease, a limited volume of metastases present progression whereas other lesions remain stable or respond to systemic therapy. 13 Minimally invasive treatment, such as SRT, may ablate drug-resistant clones, slow further progression, and allow continuation of the current effective line of systemic therapy. Retrospective studies showed a clinical benefit of such an approach, although there is no prospective studies on this topic.5,14–18 In our cohort, oligoprogressive disease was defined as limited progression of the disseminated disease during active systemic therapy that can be effectively and safely managed with local treatment. We included patients who had evidence of response to systemic treatment, namely, at least stable disease in one follow-up imaging after treatment initiation, prior to the diagnosis of oligoprogression.
SRT was not offered to the patients who were candidates for radiotherapy with purely palliative intent, for example, painful multiple bone metastases that exceed the definition of oligometastatic disease or bulky bleeding locally advanced tumor in a patient in poor performance status.
There is no established definition of SRT in the literature. The term ‘stereotactic radiosurgery’ is usually used in the case of brain lesions, whereas ‘stereotactic body radiotherapy’ describes treatment of extracranial volumes. Conservatively, SRT is described as a method of external beam radiotherapy that delivers a high radiation dose to target volume in one or few treatment fractions. 19 However, several studies and reviews defined SRT as a treatment delivered in even more than few fractions but maintaining a high EQD2 (equivalent dose in 2-Gy fractions), precise radiation delivery, and proper quality assurance.20–23 Thus, in our institution, we defined SRT as highly conformal radiotherapy delivered in 10 or fewer fractions using daily image guidance, without extensive elective volume. The borderline biologically effective dose to qualify radiotherapy as SRT was 50 Gy.
A single patient may receive SRT several times in various time intervals. To provide clear and reliable results, we tried to classify all treatment sequences with the manner described below. Multiple SRT procedures could be performed simultaneously or sequentially. All SRT procedures performed within 3 months were classified as a single SRT cycle. A single SRT procedure was defined as a plan with a single isocenter with a separate dose prescription for a single or multiple planned target volumes (PTVs). This classification was visualized in Supplementary Figure S1.
Best local response and the incidence of progression were assessed with Response Evaluation Criteria in Solid Tumors 1.1 (RECIST 1.1). Local progression was a progression of the disease within the PTV. Distant progression was defined as a progression according to RECIST 1.1, which does not fulfill the definition of local progression.
SRT-related toxicity was reassessed using Common Terminology Criteria for Adverse Events 5.0 (CTCAE).
Collection of data
We performed a retrospective analysis of consecutive adult patients with STBS treated between 2010 and 2021 in our sarcoma tertiary center using MedStream Designer software (Transition Technologies, Poland). We included all consecutive patients who underwent linac-based SRT for primary, recurrent, or metastatic STBS. Medical records were screened individually. All extracted data were verified by the two authors and encoded in the OpenClinica internal server (www.openclinica.com, the United States).
Analyzed parameters
The following parameters were extracted from the hospital’s electronic medical records system CliniNET (CompuGroup Medical, Poland): gender, age, date of primary diagnosis, primary tumor characteristics and site, date of distant metastases diagnosis, indication for SRT, SRT site, treatment completion, concomitant systemic therapy, early and late SRT toxicity, local response (best result of SRT and the incidence of local progression), overall response (the incidence of distant progression), and date and reason of death (if applicable). The missing dates of deaths were obtained from the Polish National Cancer Registry.
SRT data were obtained from the Varian Eclipse 15.5 treatment planning software. That included SRT start and stop dates, number of target volumes in a single plan, treated volumes, technique, motion control, supportive imaging for fusion (magnetic resonance and positron emission tomography), total dose, fraction dose, dose prescription method, covering isodose, and percentage of mean dose in PTV or PTV sum.
Dose calculations
We assumed the alpha/beta ratio of STBS as 4 Gy to calculate EQD2, based on literature data suggesting its values for sarcomas between 0.4 and 5 Gy.24–26 To unify different fractionation regimens and dose prescription methods, we calculated mEQD2 (mean equivalent dose in 2-Gy fractions) for each treatment plan by multiplying EQD2 by percentage of mean dose in PTV or PTV sum.
Statistical analysis
Median follow-up was estimated by Kaplan–Meier analysis using OS data with the reversed meaning of the status indicator, that is, we used the time from the date of the first SRT to the last follow-up or death (censored).
TTLP was calculated from the day of the first SRT to the last follow-up (censored), death without local progression of the irradiated tumor (censored), or confirmed local progression. LPFS was calculated from the day of the first SRT to the last follow-up (censored), death without local progression of the irradiated tumor, or confirmed local progression. TTP was calculated from the day of the first SRT to the last follow-up (censored), death without local or distant progression (censored), or confirmed local or distant progression. PFS was calculated from the day of the first SRT to the last follow-up (censored), death without local or distant progression, or confirmed local or distant progression. OS was calculated from the day of the first SRT to the last follow-up (censored) or death.
The Kaplan–Meier method was used to estimate survival. Multivariate and univariate Cox proportional-hazards models were used to estimate hazard ratios (HRs). The Mann–Whitney U test was used to evaluate differences between the two groups. All p values <0.05 were considered significant. Statistical analyses were performed using R, version 4.1.0 software (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patients’ characteristics
A total of 141 patients who underwent SRT for STBS were identified. Among them, 30 received multiple SRT cycles (see an example in Figure 1).

The plans fusion of multiple stereotactic radiotherapy cycles for lung oligometastases in a patient with myxoid liposarcoma.
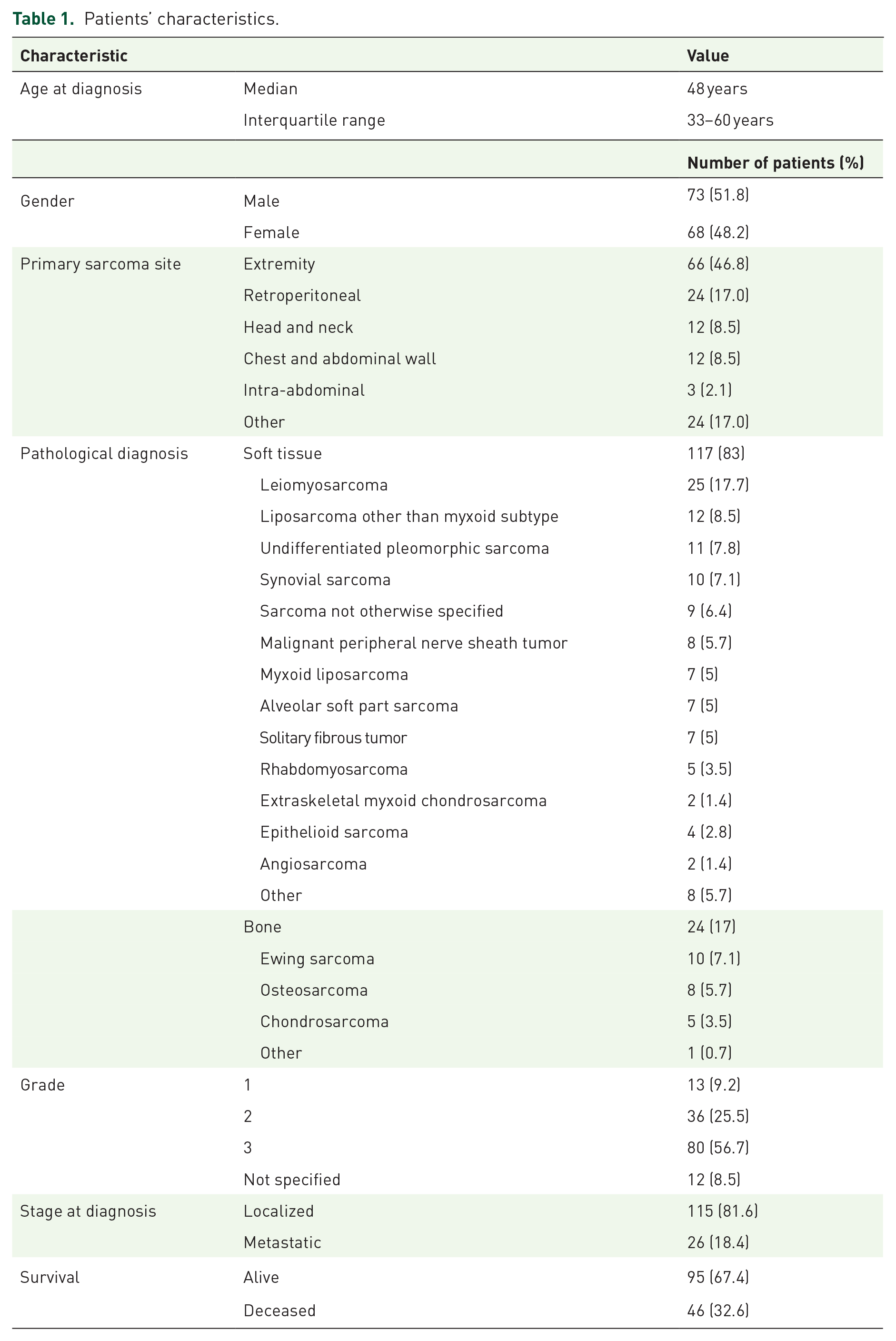
The patients’ characteristics are presented in Table 1. The median follow-up duration was 20.9 months (95% confidence interval, CI = 17.6–24.5 months) with minimum 1 month and maximum 73 months.
Patients’ characteristics.
SRT characteristics
All included patients underwent in total 233 SRT procedures after multidisciplinary tumor board decision. The treatments’ characteristics are presented in Table 2. The most common technique of SRT delivery was volumetric modulated arc therapy (95.3%). In the vast majority of SRTs, the dose was specified to isodose (88%). Median EQD2 was 96 Gy (38–198 Gy). Calculated median mEQD2 in target volumes was 107 Gy (interquartile range, IQR: 79–155 Gy) with minimum 38 Gy and maximum 249.5 Gy. Three patients received SRT to the remaining tumor volume after previous surgery. Systemic treatment was administered concomitantly or within 3 months pre- or post-SRT in 133 SRT procedures (57%). The used regimens are presented in Supplementary Table S1. After 82 SRTs (35.2%), we observed an objective (partial or complete) response.
Treatment characteristics.

4D, four-dimensional; SRT, stereotactic radiotherapy.
Not amenable for metastasectomy due to refusal of surgery (n = 13); marginal resectability, unresectability, or predictable adverse functional outcomes (n = 60); and high risk of postoperative morbidity (n = 11). Twenty patients had solitary oligometastasis (a single lesion without prior local definitive therapy for other distant metastases).
SRT tolerance was very good. We found only one case of significant grade 3 early toxicity and one case of grade 3 late toxicity. Detailed data regarding SRT-related toxicity are presented in Supplementary Table S2.
Predictors of disease progression and survival
Those calculations were performed for all patients (n = 141). At the moment of the analysis, 95 (67.4%) patients were alive. Local progression occurred after 19 SRTs in 15 patients. Most local progression occurred after SRT for radioresistant pathological subtypes (bone sarcomas and malignant peripheral nerve sheath tumors), bone lesions, and locally recurrent head and neck STBS. Exact data of patients who experienced local progression are presented in Supplementary Table S3. Distant progression was diagnosed after 163 SRTs in 91 patients. The proportionality assumption required for Cox analysis was fulfilled for all the studied factors. The results of treatment benefit, survival estimates, and significant factors identified in univariate analyses are presented in Table 3. The relevant figures for TTLP, LPFS, TTP, PFS, and OS, and related univariate Cox analyses are presented in Supplementary Figures S2, S3, S4, S5, and S6, and Table S4.
The summary of treatment efficacy and significant factors in univariate analysis.

CI, confidence interval; GTV, gross target volume; LPFS, local progression-free survival; m, months; mEQD2, mean equivalent dose in 2-Gy fractions; NA, not applicable; NR, not reached; OS, overall survival; PFS, progression-free survival; TTLP, time to local progression; TTP, time to progression.
In an exploratory OS analysis, we performed a multivariate analysis that included all significant and borderline significant factors found in the univariate analysis. In the multivariate survival analysis, SRT for oligometastatic disease was associated with better OS, whereas diagnosis of bone sarcoma increased the risk of death (see Figure 2).

Hazard ratios for death with 95% confidence intervals and p values calculated from a multivariate Cox proportional-hazards model.
We found that the irradiated site strongly correlated with mEQD2; thus, it was not included in the multivariate analysis. Thus, we performed an additional exploratory analysis using Mann–Whitney U test. The median mEQD2 differed between SRTs for lung lesions and SRTs for other lesions (p < 0.001). The results are visualized in violin plot in Supplementary Figure S7. Moreover, to better assess the dose–response relationship in our cohort, we calculated a dose–response curve for local control (see Figure 3). 27

Dose–response curve for 1-year local control after stereotactic radiotherapy.
Due to the focus on sarcoma- and SRT-related factors, calculation of survival times from the day of the first SRT, variety of agents with different mechanisms of action, and various number of previous lines of therapy, systemic treatment was not included as a factor in the univariate and multivariate analyses. However, we performed additional exploratory analysis of oligometastatic patients who may or may not receive systemic therapy based on multidisciplinary tumor board decision. We found a worse OS in a subgroup who received systemic therapy (median = 39.2 months) concomitantly or within 3 months pre- or post-SRT than in those who did not (median OS not reached, p = 0.033). Survival curves are presented in Supplementary Figure S8.
Discussion
We presented the outcomes of the largest STBS cohort treated with SRT. We observed only 19 local progressions after treatment and only two events of significant toxicity, although our group comprised 17 SRT reirradiations. Thus, the long-term efficacy and tolerability of SRT for locally advanced or metastatic STBS in selected patients have been confirmed. In a retrospective study from Karolinska University Hospital, Stragliotto et al. 25 presented the outcomes of SRT for distant metastases in 46 patients with STBS with similar median follow-up time as in our cohort. They used more conservative definition of SRT regarding the number of fractions (five or less) but included regimens with low EQD2, for example, 5× 4 Gy or 1× 10 Gy. Nevertheless, the authors reported overall response rate, defined as all responses except local progression, as high as 88%, like this achieved in our cohort (95%). In the aforementioned study, progressing tumors presented tendency to have larger volumes and lower mean doses. However, no significant correlations were found in the planned Spearman’s correlation analysis. Only after unplanned grouping of tumors with stable disease and partial response, the correlation of best response with administered EQD2 became significant. In our study, higher EQD2 and lower tumor volume increased modestly local control over time (TTLP and LPFS), but this effect did not translate into better OS, probably due to high occurrence of distant progression being the cause of death. Nevertheless, the dose–response curve for local control showed that the most reasonable borderline EQD2 for satisfactory 1-year local control (over 90%) is almost 100 Gy (see Figure 3).
The indication for SRT mattered for local control. Our study showed that SRT for localized disease gave the worse results in TTLP. Our exploratory analysis revealed that those patients had more often bone sarcomas, were previously irradiated, had larger tumors, and received significantly lower EQD2 (see Supplementary Table S5). Thus, the poorer efficacy of SRT for those lesions is explainable. However, the negative predictor for OS was the SRT for oligoprogression. That could be explained with the less advanced disease in the case of oligometastases than in the case of oligoprogression. Similar observation was reported in the study with a cohort of metastatic breast cancer patients treated with SRT. 5 Interestingly, in another exploratory analysis, we found that survival of oligometastatic patients after first SRT was worse when they received systemic treatment. Probably, chemotherapy was administered for patients with more aggressive sarcoma subtypes associated with poorer survival. However, deeper exploration of this topic may provide false results, mostly due to heterogeneity of used systemic treatments in our cohort.
We have shown that SRT for lung lesions was correlated with better prognosis. First, isolated lung metastases are known independent favorable prognostic factor for OS in STBS. 28 Second, the resection of lung metastases significantly improves patients’ outcomes.29,30 Thus, we may assume that SRT for lung metastases has the same influence on survival as metastasectomy due to its high local efficacy.
The strong predictor for worse local control and survival was the diagnosis of bone sarcoma. Advanced bone sarcomas, regardless of the pathological diagnosis, are associated with poor prognosis. Chondrosarcoma and osteosarcoma are considered radioresistant. Results of a small retrospective study on SRT for advanced Ewing sarcoma and osteosarcoma showed a long-term benefit of SRT in this group of patients, namely, estimated 85% of local control at 2 years.31–33Importantly, most of the local progressions in the aforementioned study occurred in patients with osteosarcomas. Thus, higher EQD2 might be considered in patients with potentially radioresistant bone sarcomas.
Importantly, SRT could be safely repeated after development of new STBS metastases or used even in the case of progression after prior radiotherapy. Thirty patients in our cohort underwent multiple SRT cycles, whereas 17 received SRT on previously irradiated volumes. No events of significant toxicity were observed.
Our study has weaknesses. It may contain a selection bias caused by the retrospective nature of the analysis. To minimize the effect of selection, all data were reviewed by two authors (M.J.S and A.B.) and encoded in a dedicated OpenClinica server. Retrospective data retrieval also poses a significant risk of incomplete or misinterpreted data, especially those related to toxicity. However, the significant toxicity, as opposed to mild toxicity, is usually noted in medical records. In our study, we observed only one event of grade 3 early toxicity and grade 3 late toxicitiy. Thus, we may conclude that SRT was a well-tolerated treatment. Moreover, a relatively short follow-up time – namely, 21 months – may be a limitation for evaluating local control. Despite above-mentioned limitations, this study can provide valuable data on this important topic due to the large sample size with this very rare entity.
To sum up, we can say that SRT may be effectively and safely used in patients with STBS, especially those with oligometastatic disease, isolated lung metastases, and soft tissue sarcomas. Our findings suggest that regardless of the used fractionation regimens, the local efficacy of SRT in STBS patients is high. Thus, it is reasonable to use risk-adapted strategies, trying to deliver high EQD2 but also considering a risk of further progression near the irradiated volume and necessity to repeat SRT. The effect of dose escalation to the TTLP was modest (Table 3) and has no influence on OS. Many issues remain unsolved, including the optimal timing of SRT, benefit of concomitant systemic therapy, and the true benefit of SRT on survival. Some of those questions will be answered in the recently opened phase III clinical trial OligoRARE (NCT04498767). 34
Conclusion
SRT provides excellent local control of irradiated lesions in STBS patients without a risk of significant toxicity. It should be offered to selected patients, especially those with oligometastatic disease not amenable for surgery, lung metastases, and soft tissue sarcomas. The dose escalation provides a moderate benefit in local control; however, it does not translate into better survival.
Supplemental Material
sj-docx-10-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-docx-10-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-12-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-docx-12-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-1-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-1-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-2-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-2-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-3-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-3-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-4-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-4-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-5-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-5-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-6-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-6-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-7-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-7-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-8-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-png-8-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xlsx-11-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-xlsx-11-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xlsx-13-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-xlsx-13-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xlsx-9-tam-10.1177_17588359211070646 – Supplemental material for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence
Supplemental material, sj-xlsx-9-tam-10.1177_17588359211070646 for Stereotactic radiotherapy for soft tissue and bone sarcomas: real-world evidence by Mateusz Jacek Spałek, Paweł Teterycz, Aneta Borkowska, Jan Poleszczuk and Piotr Rutkowski in Therapeutic Advances in Medical Oncology
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The processing charge cost was covered by the Maria Sklodowska-Curie National Research Institute of Oncology, Warsaw, Poland.
Ethics statement
Each patient, at the beginning of treatment, provided routine informed consent for the use of their treatment and data processing. The study was conducted according to the guidelines of the Declaration of Helsinki. The paper does not report on the use of experimental or new protocols. This study, as part of a project series, was approved by the Local Ethics Committee at Maria Sklodowska-Curie National Research Institute of Oncology (approval number KB/9/2011) to release these data without additional patient consent as patient consent was deemed unnecessary.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
