Abstract
Background:
In July 2019, bevacizumab-awwb and trastuzumab-anns were marketed in the USA as the first therapeutic oncology biosimilars. We aimed to investigate the initial real-world use of bevacizumab-awwb and trastuzumab-anns for cancer management in US oncology practices.
Methods:
A retrospective, observational analysis of data from US cancer patients (⩾18 years of age) was carried out to describe the use of bevacizumab-awwb and trastuzumab-anns during the first 12 months following their market entry, using structured data from the Flatiron Health electronic health record-derived database.
Results:
A total of 2952 and 2997 patients with recorded use of bevacizumab-awwb and trastuzumab-anns, respectively, were included in the analysis. The first use of bevacizumab-awwb and trastuzumab-anns was in a patient with metastatic colorectal cancer (mCRC) within 10 days of market availability and in a patient with early stage breast cancer (eBC) within 4 days, respectively. The use of these biosimilars was observed across all approved cancer indications; 68% of bevacizumab-awwb users were those diagnosed with mCRC and 72% of trastuzumab-anns users were those diagnosed with eBC. Approximately half the patients were previously exposed to reference product (RP) prior to initiation of bevacizumab-awwb or trastuzumab-anns. Among pre-exposed patients, the majority received the biosimilars [bevacizumab-awwb (63–85%) or trastuzumab-anns (75–81%)] within 28 days of the last infusion of the RP. For both biosimilars, no major differences were observed in patient characteristics between RP-naïve and pre-exposed patients.
Conclusion:
Initial evidence from the first 12 months following market entry suggests rapid clinical adoption of bevacizumab-awwb and trastuzumab-anns across all approved tumor types. Usage of these two biosimilars was observed in both RP-naïve patients and patients who were previously treated with RP, with no distinctive differences in patient characteristics between the two groups.
A video abstract is available for this article as part of the Kanjintionline supplemental material.
Keywords
Introduction
In recent years, the development of biosimilar medicines has provided an opportunity for patient utilization of these biological treatments at a lower cost, particularly for oncology patients for whom biologicals play a dominant role in providing effective therapeutic and supportive care. 1 Since 2015, US Food and Drug Administration (FDA)-approved biosimilars of hematopoietic growth factors (epoetin and filgrastim) have been used in oncology as supportive treatment.2,3 However, therapeutic biosimilars of monoclonal antibodies have only recently started to become available in the US market. In July 2019, bevacizumab-awwb (MVASI, Amgen Inc.) and trastuzumab-anns (KANJINTI, Amgen Inc.) were marketed in the US as the first therapeutic biosimilars in the field of oncology, 4 with several other bevacizumab and trastuzumab biosimilars entering the market shortly after (Table 1). Bevacizumab-awwb, the first FDA-approved biosimilar to Avastin (bevacizumab), is a vascular endothelial growth factor (VEGF) inhibitor indicated for the treatment of metastatic colorectal cancer (mCRC), non-squamous non-small cell lung cancer (NSCLC), recurrent glioblastoma (GBM), metastatic renal cell carcinoma (mRCC), and cervical cancer (CC).5,6 Trastuzumab-anns, a biosimilar to Herceptin (trastuzumab), was approved for the treatment of human epidermal growth factor receptor 2 (HER2) overexpressing breast cancer (BC) and HER2 overexpressing metastatic gastric or gastroesophageal adenocarcinoma (GC). 7
List of FDA-approved bevacizumab and trastuzumab biosimilars currently available in the US market.

FDA, US Food and Drug Administration.
In the US, the approval of biosimilars involves the rigorous assessment of data from extensive analytical and clinical studies designed to show similarity to the reference product (RP) and to demonstrate that there are no clinically meaningful differences in safety, purity, or potency between the biosimilar and the RP.8–10 As many biologicals have multiple indications, the FDA permits extrapolation of indications for a biosimilar if the ‘totality of evidence’ supports biosimilarity with regard to the mechanism of action, target-binding properties, pharmacokinetics, and immunogenicity, and also satisfactorily addresses potential differences in toxicity and efficacy.11–13 This extrapolation allows the approval of a biosimilar for all the eligible indications for which the RP is authorized, without requiring clinical trial data for each approved disease state or tumor type. For example, the initial clinical comparative efficacy and safety studies for bevacizumab-awwb and trastuzumab-anns were carried out in patient populations with NSCLC 14 or histologically confirmed HER2-positive early stage BC, 15 respectively, and demonstrated the clinical bioequivalence of these biosimilars to the respective RPs. Approval was granted for the full range of indications of the originator product via extrapolation of efficacy and safety data from the ‘totality of evidence’.16–20
The market entry of biosimilars provides more affordable treatment options for patients with cancer. 21 Significant cost-savings resulting from biosimilar use has been reported in European countries,22–24 and the projected cost-savings over the next 5 years from biosimilar usage is estimated to be over $100 billion in the US market. 25 However, barriers for biosimilar adoption exist and are predominantly attributed to the gap in awareness among patients and healthcare providers regarding terminologies, evolving regulatory guidance, and prescription patterns in real-world settings. 26 Healthcare providers have voiced concern about the lack of indication-specific clinical data and have been hesitant to accept extrapolation of data for biosimilar use in multiple indications.27,28 In 2018, the American Society of Clinical Oncology released a statement supporting the use of biosimilars in clinical practice by clarifying the difference between biosimilars and generic drugs, explaining the regulatory framework for biosimilar approval, and renewing its commitment to provide educational resources to increase prescriber and payer confidence in biosimilars. 29 The statement also emphasized the importance of post-marketing evidence to enhance patient and provider confidence in biosimilars and for optimizing drug use in a diverse patient population. 29 Real-world evidence demonstrating the extent of biosimilar use in clinically studied and extrapolated indications can offer physicians a better understanding of utilization patterns and patient characteristics and increase their comfort level when adopting biosimilars in their practices. 30 Therefore, in the present study, we aimed to describe the real-world use of bevacizumab-awwb and trastuzumab-anns, the first two therapeutic oncology biosimilars in the US, across all approved cancer indications in the initial 12 months following their market entry.
Methods
Study design and study population
This was a retrospective, cohort study to describe the demographic and clinical characteristics and treatment patterns of US patients who received bevacizumab-awwb or trastuzumab-anns for cancer management during the first 12 months after their market entry as recorded in the Flatiron Health electronic health record (EHR)-derived database. The Flatiron Health EHR is a nationally representative and longitudinal database that includes structured patient-level data (e.g. demographics, laboratory values, prescribed drugs) from over 280 cancer clinics (~800 sites of care), representing more than 2.2 million US cancer patients primarily receiving care in community-based oncology practices. The data provided by Flatiron Health are de-identified and provisions are in place to prevent re-identification in order to protect patient confidentiality. Institutional review board (IRB) approval with waiver of informed consent was obtained from the WCG IRB (registration number: IRB00000533, approval number of the parent protocol: 420180044–https://www.wcgirb.com/) by Flatiron Health prior to the conduct of this data analysis. 31
Inclusion/exclusion criteria
Patients who were new users of bevacizumab-awwb for the management of mCRC, NSCLC, GBM, mRCC, or CC, and patients who were new users of trastuzumab-anns for the treatment of BC (both early stage and metastatic) or metastatic gastric cancer (mGC) between 19 July 2019 and 30 June 2020 and met the eligibility criteria were included in the analysis. Patients who were <18 years of age at the time of initiation of bevacizumab-awwb or trastuzumab-anns and patients who received trastuzumab-anns with negative HER2 status were excluded.
Study measures and variable definitions
The date of initiation of bevacizumab-awwb or trastuzumab-anns was defined as the index date. Patient medical history was assessed based on data in the electronic medical record collected from 1 January 2011. Determination of cancer type was made using the International Classification of Disease (9th and 10th revision) clinical modification (ICD-9/10-CM) diagnostic codes (Supplemental File) recorded most proximal to the index date between 1 January 2011 to 30 days following the index date. Metastatic status required a documented stage IV value or the presence of the ICD-9/10-CM diagnostic code indicating a secondary malignant neoplasm (Supplemental File).
Patient demographic and clinical characteristics evaluated in this study included age, sex, race, practice type, payer category, body weight, body mass index (BMI), tumor stage, Eastern Cooperative Oncology Group (ECOG) performance status, time from cancer diagnosis to initiation of the biosimilars, exposure to bevacizumab or trastuzumab RP prior to initiation of the biosimilars, and time from the last infusion of RPs to initiation of biosimilars among pre-exposed patients. In addition, HER2 status, lymph node status, and hormone receptor status were evaluated for patients who received trastuzumab-anns.
Patients with GBM did not have documented disease staging information as glioblastomas are graded histologically using the World Health Organization (WHO) classification of tumors of the central nervous system (CNS) and are not staged based on the American Joint Committee on Cancer (AJCC)’s tumor–node–metastasis (TNM) staging system, and histology data were not available for analysis for patients with GBM.
Statistical analysis
The study was descriptive in nature and no formal hypotheses were tested. Patient characteristics were assessed using the non-missing value most proximal to the index date. If more than one value was recorded on the day most proximal to the index date, the average of these values for continuous variables (e.g. body weight, BMI) and the highest value for categorical variables (e.g. ECOG performance status, tumor stage) was selected. For patient characteristics presented continuously, summary statistics (e.g. median, minimum, maximum) were calculated after excluding missing values; and for categorical variables with missing data, a category of ‘missing/unknown’ was presented.
Stratified analysis was conducted to describe patient characteristics based on prior exposure to bevacizumab or trastuzumab. For the bevacizumab cohort, ‘pre-exposed’ patients were defined as those with documented prior use of any marketed bevacizumab product, including the bevacizumab RP and the bevacizumab biosimilar Zirabev (bevacizumab-bvzr), at any time prior to the initiation of bevacizumab-awwb. For the trastuzumab cohort, ‘pre-exposed’ patients were defined as those with recorded previous use of any marketed trastuzumab products, including the trastuzumab RP (Herceptin), subcutaneous formulation of trastuzumab (Herceptin HYLECTA),32,33 and other trastuzumab biosimilars Ogivri (trastuzumab-dkst), Trazimera (trastuzumab-qyyp), Herzuma (trastuzumab-pkrb), and Ontuzant (trastuzumab-dttb), at any time prior to the initiation of trastuzumab-anns. Bevacizumab or trastuzumab-naïve patients were defined as those with no recorded previous use of the above-mentioned bevacizumab or trastuzumab products (including RPs and biosimilars) prior to the initiation of bevacizumab-awwb or trastuzumab-anns. ‘Naïve’ and ‘pre-exposed’ patients are mutually exclusive categories.
Results
Bevacizumab-awwb
Overall bevacizumab-awwb use and patient characteristics
The first use of bevacizumab-awwb was observed within 10 days following market availability in a patient with mCRC and first use in other approved indications, including NSCLC, GBM, and CC, was also seen within 2 weeks post-launch. Among a total of 2952 eligible patients who received bevacizumab-awwb treatment, the majority were patients with mCRC (68%), followed by patients with NSCLC (14%), patients with GBM (12%), patients with CC (5%), and patients with mRCC (1%) [Figure 1(a)]. Across indications, most patients were White (61%), ⩾55 years of age (78%), treated at community-based oncology practices (95%), and covered by commercial health insurance plans with or without support from patient assistance programs (PAPs) (43%), although proportions slightly varied across indications (Table 2). Patient clinical characteristics are also presented in Table 2. The majority of patients had stage IV cancer and an ECOG score of 0/1.
Demographic, clinical, and treatment characteristics of patients who received bevacizumab-awwb during the time period between 19 July 2019 and 30 June 2020, stratified by cancer indication.

Other races includes Hispanics/Latino, Black/African American, and Asian.
All patients with previous use of bevacizumab were treated with the reference product prior to initiation of bevacizumab-awwb.
CC, cervical cancer; ECOG, Eastern Cooperative Oncology Group; GBM, glioblastoma; mCRC, metastatic colorectal cancer; M/M/G, Medicare, Medicaid, or other government programs; mRCC, metastatic renal cell carcinoma; NA, not applicable; NSCLC, non-squamous non-small cell lung cancer; PAP, patient assistance program.
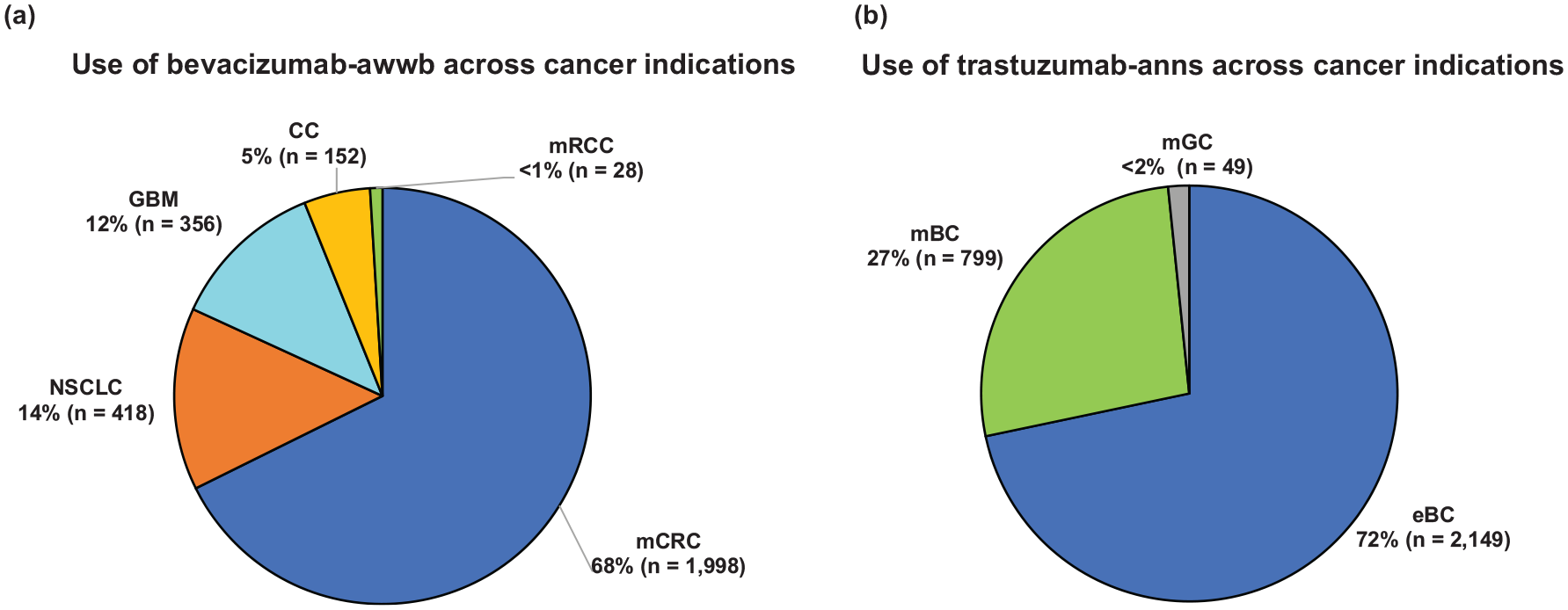
Use of (a) bevacizumab-awwb and (b) trastuzumab-anns across cancer indications between 19 July 2019 and 30 June 2020.
Patient characteristics stratified by bevacizumab treatment history
Overall, approximately half the patient population reported previous use of the bevacizumab RP prior to initiation of bevacizumab-awwb, including 55% of patients with mCRC, 45% of patients with NSCLC, 42% of patients with GBM, 46% of patients with mRCC, and 38% of patients with CC (Table 2). Among these pre-exposed patients, the majority (63% of patients with mCRC, 70% of patients with NSCLC, 83% of patients with GBM, 85% of patients with mRCC, and 67% of patients with CC) were transitioned to bevacizumab-awwb within 28 days of receiving the last infusion of RP [Figure 2(a)].

Time interval in days from end of (a) bevacizumab RP and (b) trastuzumab RP use to initiation of bevacizumab-awwb or trastuzumab-anns, respectively, among pre-exposed patients transitioned to biosimilars.
Across indications, patient demographic and clinical characteristics appeared to be comparable between RP-naïve and pre-exposed patients (Table 3 and Supplemental Table 1). Among patients with mCRC, an increased proportion of pre-exposed patients, particularly those covered by commercial health insurance plans, were covered in combination by PAPs (pre-exposed: 189/464, 40.7% versus RP-naïve: 90/399, 22.6%). A higher proportion of pre-exposed patients with GBM were younger (34% versus 23% being <55 years) and had been treated in oncology practices in an academic setting (32% versus 21%) when compared with RP-naïve patients with GBM.
Demographic and clinical characteristics of patients with mCRC who received bevacizumab-awwb between 19 July 2019 and 30 June 2020, stratified by prior bevacizumab exposure.

Other races includes Hispanics/Latino, Black/African American, and Asian.
BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; mCRC, metastatic colorectal cancer; M/M/G, Medicare, Medicaid, or other government programs; PAP, patient assistance program.
Trastuzumab-anns
Overall trastuzumab-anns use and patient characteristics
The first use of trastuzumab-anns was seen 4 days after its market entry and occurred in a patient with early stage breast cancer (eBC). The first use of trastuzumab-anns in metastatic breast cancer (mBC) and metastatic gastric cancer (mGC) was also observed within 2–3 weeks following market availability. Among the 2997 eligible patients treated with trastuzumab-anns, the majority had a diagnosis of BC (71.7% eBC, 26.7% mBC) and the remaining 1.6% had a diagnosis of mGC [Figure 1(b)]. Most patients with BC were White (57.6%), female (>99%), ⩾55 years of age (69.5%), treated at community-based oncology practices (>99%), and primarily covered by commercial health insurance plans with or without support from PAPs (53% eBC, 46% mBC). Among patients with eBC, the majority had stage I (57%) or stage II (28%) disease at the time of initiation of trastuzumab-anns and were lymph node negative (59%). Over 80% of eBC and mBC patients had an ECOG score of 0/1. Patients with eBC and mBC were mostly hormone receptor positive (68% and 65%, respectively), and 93% of eBC and 86% of mBC patients had confirmed HER2 positive status (Table 4).
Demographic, clinical, and treatment characteristics of patients who received trastuzumab-anns between 19 July 2019 and 30 June 2020, stratified by cancer indication.

Other races includes Hispanics/Latino, Black/African American, and Asian.
Patients with regional lymph nodes status recorded as ‘NX’, which is ‘regional lymph nodes cannot be assessed (for example, previously removed)’.
All patients with previous use of trastuzumab were treated with the reference product prior to initiation of trastuzumab-anns, except that one patient with eBC was previously treated with another trastuzumab biosimilar, Ogivri (trastuzumab-dkst), prior to initiation of trastuzumab-anns.
BMI, body mass index; eBC, early breast cancer, ECOG, Eastern Cooperative Oncology Group; mBC, metastatic breast cancer; mGC, metastatic gastric cancer; M/M/G, Medicare, Medicaid, and other government program; NA, not applicable; PAP, patient assistance program.
Patient characteristics stratified by trastuzumab treatment history
More than half the eligible patients reported prior use of trastuzumab RP prior to initiation of trastuzumab-anns, including 50% of patients with eBC, 76% of patients with mBC, and 59% of patients with mGC (Table 4). The majority of these pre-exposed patients (81% eBC, 75% mBC, 79% mGC) were transitioned to trastuzumab-anns within 28 days of receiving the most recent treatment with trastuzumab RP [Figure 2(b)].
Overall, among patients with eBC and mBC, there appeared to be no major differences in demographic and clinical characteristics between RP-naïve patients and pre-exposed patients (Table 5). However, we observed that an increased proportion of pre-exposed patients, especially those covered by commercial health insurance plans, were supported by PAPs when compared with RP-naïve patients (256/518, 49% versus 167/614, 27% for eBC, 146/274, 53% versus 27/92, 29% for mBC). The characteristics of patients with mGC, stratified by prior trastuzumab treatment history are shown in Supplemental Table 2.
Demographic, clinical, and treatment characteristics of patients with eBC or mBC who received trastuzumab-anns between 19 July 2019 and 30 June 2020, stratified by prior trastuzumab exposure.
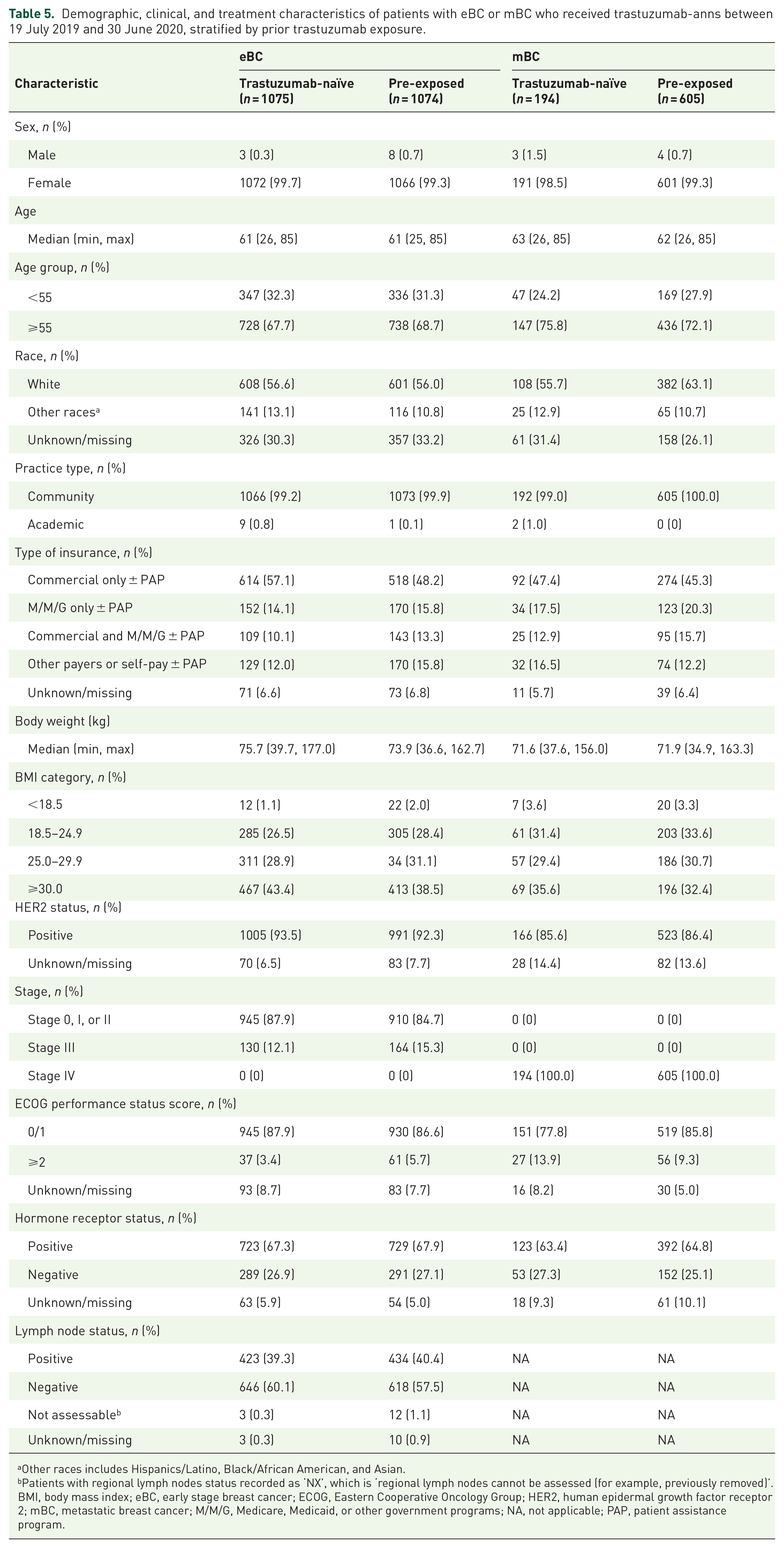
Other races includes Hispanics/Latino, Black/African American, and Asian.
Patients with regional lymph nodes status recorded as ‘NX’, which is ‘regional lymph nodes cannot be assessed (for example, previously removed)’.
BMI, body mass index; eBC, early stage breast cancer; ECOG, Eastern Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; mBC, metastatic breast cancer; M/M/G, Medicare, Medicaid, or other government programs; NA, not applicable; PAP, patient assistance program.
Discussion
To the best of our knowledge, this is the first study to describe the real-world utilization of bevacizumab-awwb and trastuzumab-anns during the first 12 months following their market entry.
We observed that the two biosimilars were quickly integrated into clinical practice, with a recorded use of trastuzumab-anns and bevacizumab-awwb within 4 and 10 days of market availability, respectively. Approximately half the patients in the study were receiving the RP prior to initiating treatment with the biosimilars, and among these pre-exposed patients, the majority were transitioned to the biosimilars within 28 days of receiving the most recent treatment with the RPs. This rapid post-market adoption observed in our study might indicate that physicians were comfortable initiating or transitioning patients to these biosimilars. Support from pharmaceutical manufacturers through PAPs, 34 which can help to reduce out-of-pocket costs for patients, could have also contributed to patient and provider adoption of these medicines. We observed that 15–20% of mCRC and BC patients covered by the commercial health insurance plans received support from PAPs, and the proportion of patients receiving PAPs support was even higher among patients who were previously treated with RPs and subsequently transitioned to these biosimilars. In addition, formulary management by payers to direct stakeholders towards lower priced biosimilar medicines may also have substantially impacted uptake. 35 However, we were unable to elucidate whether this rapid adoption was driven by physicians, patients, or payers owing to the lack of such data for analysis. As the biosimilar market is fast evolving, future studies to understand the key potential drivers of biosimilar adoption will be informative for enhancing patient access to more affordable biological treatment options.
Although biosimilars are lower-cost therapeutic alternatives to branded biological drugs, the willingness of patients and physicians to adopt oncology biosimilars with curative intent for a life-threatening disease might differ from their willingness to adopt biosimilars as supportive care and as treatment for chronic disease. 36 Physicians have voiced their concern about the use of biosimilars in indications approved by data extrapolation without direct clinical comparison. 27 Educational programs for patients and healthcare providers to outline the regulations required for biosimilar approval can, at least in part, address this concern given that extrapolation across indications is based on the totality of the scientific evidence showing that the biosimilar is comparable to the RP, and that decisions regarding extrapolation are made on a case-by-case and indication-by-indication basis rather than by an automatic extension of approval to a biosimilar for all indications of the reference drug.37,38 Provision of evidence on post-marketing utilization patterns can help support the acceptability of biosimilars among patients and providers. Our study provides initial evidence that providers may be comfortable using biosimilars in extrapolated indications, as we observed a non-trivial amount of use of bevacizumab-awwb (~70%) in patients with mCRC (an indication approved based on data extrapolation), which was remarkably higher compared with its use in patients with NSCLC (14%) – a clinically studied indication. However, continuous education of stakeholders, supported by up-to-date real-world data, remains critical for improving the use of these therapeutic biosimilars in a medically appropriate and cost-effective way to treat cancer patients.
Our current findings, descriptive in nature, provide a preliminary view of bevacizumab-awwb and trastuzumab-anns utilization during the first 12 months following their market entry in the US, but are likely to be subject to several limitations. First, although the Flatiron Health EHR-derived database is a nationally representative, geographically, and demographically diverse database, the majority of patients included in the network were treated at community-based clinics. Thus, the patient characteristics and utilization patterns reported in our analysis may not be generalizable to patients who receive treatment outside of the community setting. Second, the classification of cancer types was carried out using the ICD-9/10-CM diagnostic codes available in the Flatiron Health database. The lack of a specific ICD code for differentiating GBM from other types of brain tumors could have led to the inclusion of some patients with non-GBM tumors in our GBM patient group. Finally, information regarding line of therapy was not available for analysis. We were thus unable to evaluate whether physicians were likely to transition a patient to these biosimilars when initiating a new line of therapy or within an ongoing line of therapy.
Despite these limitations, our study provides the first insights into the use of the first bevacizumab and trastuzumab biosimilars in the US. Use of these biosimilars has the potential to deliver considerable cost-savings for patients and payers. Although we observed rapid adoption of the two biosimilars by oncology practices, there is still a compelling need for continuous patient and provider education supported by post-market, real-world evidence to provide valuable information on the utilization patterns, especially in extrapolated indications. Clear evidence from real-world settings on the safety and efficacy of those biosimilars from future long-term follow-up studies will also be warranted when longitudinal data become available.
In conclusion, our findings provide evidence suggesting the early adoption in the US of these two therapeutic oncology biosimilars in both clinically studied and extrapolated cancer indications. Patient characteristics appeared to be comparable between RP-naïve and pre-exposed patients for both biosimilars. Safety and efficacy data from future studies with longitudinal follow-up of these patients will be critical for guiding clinical practice.
Supplemental Material
sj-docx-1-tam-10.1177_17588359211041961 – Supplemental material for Clinical and treatment characteristics of patients treated with the first therapeutic oncology biosimilars bevacizumab-awwb and trastuzumab-anns in the US
Supplemental material, sj-docx-1-tam-10.1177_17588359211041961 for Clinical and treatment characteristics of patients treated with the first therapeutic oncology biosimilars bevacizumab-awwb and trastuzumab-anns in the US by Ran Jin, Reshma L. Mahtani, Neil Accortt, Tatiana Lawrence, Darcie Sandschafer and Arturo Loaiza-Bonilla in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359211041961 – Supplemental material for Clinical and treatment characteristics of patients treated with the first therapeutic oncology biosimilars bevacizumab-awwb and trastuzumab-anns in the US
Supplemental material, sj-docx-2-tam-10.1177_17588359211041961 for Clinical and treatment characteristics of patients treated with the first therapeutic oncology biosimilars bevacizumab-awwb and trastuzumab-anns in the US by Ran Jin, Reshma L. Mahtani, Neil Accortt, Tatiana Lawrence, Darcie Sandschafer and Arturo Loaiza-Bonilla in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xlsx-3-tam-10.1177_17588359211041961 – Supplemental material for Clinical and treatment characteristics of patients treated with the first therapeutic oncology biosimilars bevacizumab-awwb and trastuzumab-anns in the US
Supplemental material, sj-xlsx-3-tam-10.1177_17588359211041961 for Clinical and treatment characteristics of patients treated with the first therapeutic oncology biosimilars bevacizumab-awwb and trastuzumab-anns in the US by Ran Jin, Reshma L. Mahtani, Neil Accortt, Tatiana Lawrence, Darcie Sandschafer and Arturo Loaiza-Bonilla in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors acknowledge Flatiron Health for database consultation support and M. Brad Rechkemmer for providing dedicated data programming support. Medical writing support was provided by Sukanya Raghuraman (Cactus Life Sciences, part of Cactus Communications) and funded by Amgen Inc.
Conflict of interest statement
RJ, NA, TL, and DS are employees of and own stock in Amgen Inc. RLM discloses receipt of consulting fees from Amgen, Agendia, AstraZeneca, BioTheranostics, Daiichi Sankyo¯, Eisai, Genentech, Immunomedics, Eli Lilly, Novartis, Puma Biotechnology, Sanofi, Seattle Genetics, and discloses carrying out contracted research for Genentech. AL-B reports receipt of consulting fees from Amgen, PSI CRO, Bayer, Blueprint, AstraZeneca, Medidata, Taiho, QED, Cardinal Health, BrightInsight, The Lynx Group, Boston Biomedical, Guardant, Natera, Eisai, Ipsen, Merck, Pfizer, BMS, Daiichi Sankyo¯, Genentech, and owns stock in Massive Bio.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study and its analyses were funded by Amgen Inc.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
