Abstract
Background:
Involuntary weight loss may occur during systemic anti-cancer therapy (SACT), causing treatment disruption and poorer prognoses. There remain gaps in clinical awareness as to which patients may benefit from nutritional interventions that aim to prevent unintended weight loss during SACT.
We utilised England’s population-level cancer registry data, conducting a pan-cancer assessment of patient weight loss during SACT. We aimed to identify cancers with weight loss-associated treatment modifications, potential beneficiaries of nutritional intervention.
Methods:
This cross-sectional study used England’s Cancer Analysis System database, including SACT-treated adults with one tumour and ⩾2 weight recordings between 2014 and 2018. Binary weight loss (threshold: 2.5%) was derived from patients’ most negative weight change from first SACT weight recording. The Martin et al. body mass index-adjusted weight loss grading system (BMI-WLG) was assigned. We describe binary weight loss, BMI-WLG and treatment modification status by cancer. Multivariate logistic regression models of weight loss (binary and BMI-WLG) and a composite outcome of patient treatment-modification status by cancer were produced.
Results:
Our study population contained 200,536 patients across 18 cancers; 28% experienced binary weight loss during SACT. Weight loss patients were more likely to have multiple types of treatment modifications recorded across all cancers. Regression analyses included 86,991 patients. Binary weight loss was associated (p < 0.05) with higher likelihood of treatment modification in; colon [Odds Ratio (OR) = 1.72, 95% confidence interval (CI): 1.42, 2.07]; gynaecologic (excl. ovarian) (OR = 1.48, 95% CI: 1.08, 2.01); stomach (OR = 1.6, 95% CI: 1.04, 2.06); lung (OR = 1.38, 95% CI: 1.21, 1.58); leukaemia (OR = 1.30, 95% CI: 1.09, 1.55); head and neck (OR = 1.30, 95% CI: 1.02, 1.65) and oesophageal (OR = 1.29, 95% CI: 1.01, 1.64) cancers. In lung, colon, and grouped gastro-intestinal cancers, association between BMI-WLG and treatment modification increased by WLG.
Discussion:
Our study is a wide assessment of weight loss during SACT using England’s cancer registry data. Across different cancers we found patients have weight loss-associated treatment modifications during SACT, a precursor to poorer prognoses. Our findings highlight cancers that may benefit from improved nutritional intervention during SACT.
Keywords
Introduction
Involuntary weight loss is common in patients with cancer and is a reported independent prognostic factor for survival. 1 Weight loss is associated with worsening of patient treatment outcomes and reduced tolerance to systemic anti-cancer therapies (SACTs).1–3
A SACT is defined as chemo-, immuno-, hormonal or targeted biological therapy for treatment of malignancies. SACT treatment can act as an independent driver of patient weight loss, causing toxicities which accelerate a patient’s tendency to lose weight. 4 The compounding effect of weight loss and SACT toxicities can lead to disruptive treatment modifications.
A validated body mass index (BMI)-adjusted weight loss grading system (BMI-WLG) was derived by Martin et al. 1 Through comprehensive assessment over a large multi-country sample population, BMI-WLG was demonstrated to predict patient survival based on weight loss, independent of cancer type, age, sex or performance status. 1 The BMI-WLG system is a recognised tool to assess clinically significant weight loss in cancer patients by current international cancer-nutritional guidelines. 5
Patient weight loss prior to treatment and pre-treatment BMI are standard indicators of patient risk of malnutrition. 6 In the United Kingdom, it is mandatory to screen for the risk of malnutrition in oncology care settings and is advised that high-risk patients receive a supportive nutritional plan early and alongside treatment.6,7 Yet, advice and nutritional support during anti-cancer treatment may vary depending on clinical knowledge of weight loss management and local provider.5,7 Weight loss may be under-treated in cancer patients, in particular smaller, cumulative changes in weight or in obese patients that less obviously appear malnourished. 8
There is need for real-world research into associations between weight loss and SACT modification across cancers to identify gaps in, or priorities for, early nutritional support during treatment. 7
This study utilised England’s large-scale centralised cancer registry data to assess patient weight loss during SACT across 18 different cancer groupings. We aimed to identify cancers susceptible to weight loss-associated treatment modifications, potentially those to prioritise for nutritional intervention. The applicability of the BMI-WLG system was assessed for its ability to identify patient likelihood of experiencing treatment modifications in retrospective real-world data.
Methods
Patient selection
This retrospective cross-sectional study included adult patients with a single primary tumour diagnosis between 1 January 2014 and 31 December 2017 and treated with a SACT between 1 January 2014 and 31 March 2018. Study patients required a minimum follow-up time of ⩾30 days and at least two viable weight recordings.
Patient follow-up time began at date of SACT initiation and finished at date of death, end of study time period or date of inactivity (date when no further patient information was recorded in the dataset for >6 months).
Cancer groupings based on International Statistical Classification of Diseases 10th revision (ICD-10) codes of patients’ primary cancer were; brain/central nervous system (CNS), breast, colon, gynaecologic (excl. ovarian), head and neck, leukaemia, lower gastro-intestinal (GI), lung, lymphoma, myeloma, oesophageal, ovarian, pancreatic, sarcoma, skin (melanoma only), stomach, upper GI (other), and urology.
Data source
Data for this study are based on patient electronic medical records (EMR) collect by the National Health Service as part of the care and support of cancer patients. The data are collated, maintained and quality assured by the National Cancer Registration and Analysis Service (NCRAS), part of Public Health England (PHE). This study worked with EMRs within SACT and Cancer Outcomes and Services Dataset (COSD) datasets from PHE’s Cancer Analysis System (CAS) database.9,10 Access to these data was facilitated by the Simulacrum.
Dataset structure and capture of variables of interest had a decisive effect on study design. Patient weight prior to initiation of SACT treatment is not recorded in CAS and patients’ weight loss status prior to treatment was unknown. Patient weight during treatment can be recorded at start of each SACT regimen or start of each cycle of a SACT regimen in the SACT dataset. Start date of SACT regimen and cycle is available in the SACT dataset.
Treatment modifications are recorded within SACT data as three categorical variables (‘Yes’, ‘No’ or ‘Missing’); dose reduction (DR); time delay (TD) and stopped early (SE). 11 Occurrence of each of these treatment modifications can be recorded only once per SACT regimen. Date of treatment modification is not captured.
Exposures
Per patient, viable weight recordings included those recorded on a date independent of another regimen-level weight recording and within the range of 30–150 kg. If patient weight was recorded at both regimen and cycle-level on the same date, the mean weight was accepted when recordings were within ±10% kg of the lowest value recording.
Exposures: weight loss (unadjusted for BMI)
Patients’ most negative percentage weight change between their first (baseline) and ith SACT weight recording was calculated as below:
(for 2nd ≤ i ≤ nth, where nth is last weight recording)
Patients’ most negative weight change from baseline was made binary at threshold −2.5%. Patients with weight change more negative or equal to −2.5% were identified as weight loss patients.
Further categorisation of weight loss patients was undertaken for descriptive analyses based on percentage thresholds derived by Martin et al.; category 0: non-weight loss (up to 2.5% weight loss); 1: mild weight loss (2.5–5.9%); 2: moderate weight loss (6.0–10.9%); 3: severe weight loss (11.0–14.9%); 4:most severe weight loss (15.0% or greater). 1
Exposures: BMI-adjusted weight loss grade
Patients were assigned BMI-WLGs based on Martin et al. classifications (grades 0–4 of worsening prognosis) (Table 1). 1 Patients’ most negative percentage weight change from baseline was used to define percentage weight loss. BMI was derived from baseline weight recording and any viable measurement of height (range 1.25–2.00 m), recorded during SACT treatment. Of the study population, 10% did not have a viable height recording, so were not assigned BMI-WLGs.
Grid to identify BMI-WLG (0–4) based on most negative percentage weight change from baseline and BMI at start of SACT treatment (Daly, Dolan and Power, 2020) 28
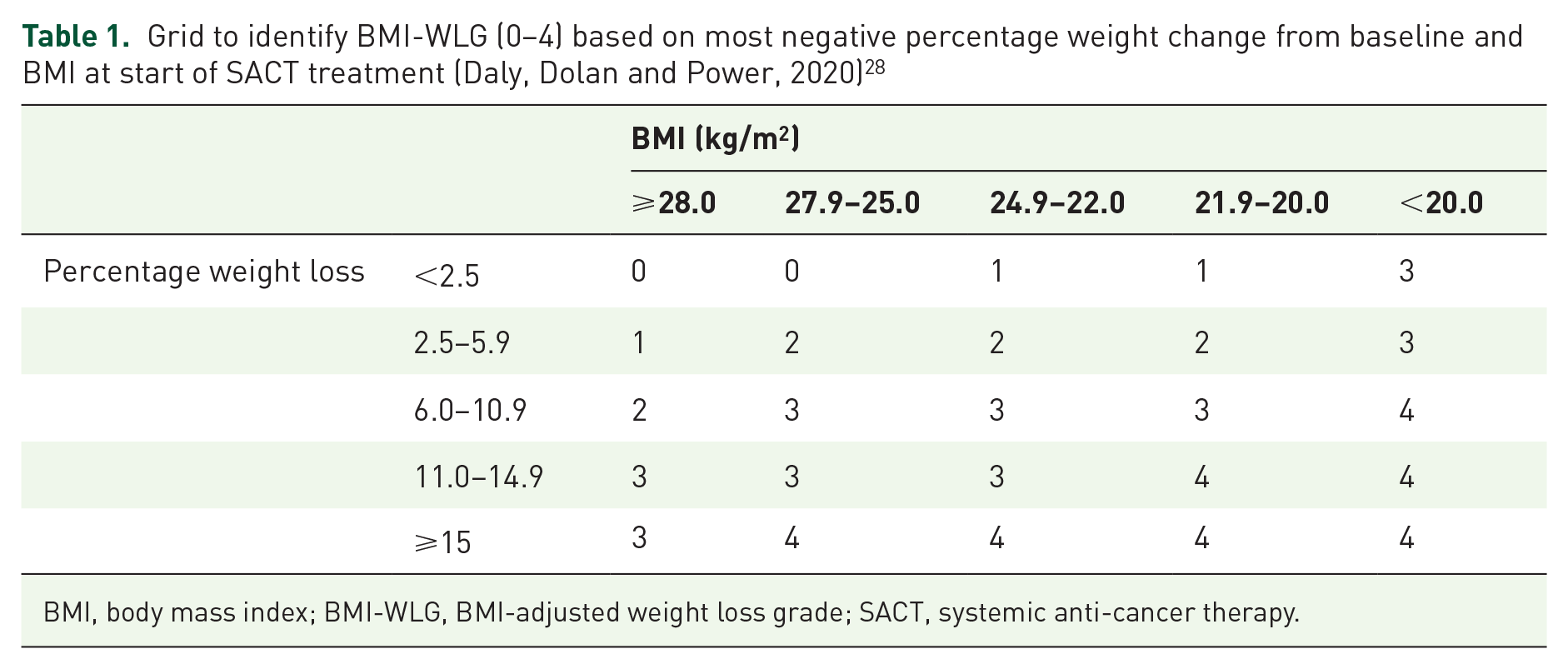
BMI, body mass index; BMI-WLG, BMI-adjusted weight loss grade; SACT, systemic anti-cancer therapy.
Outcomes: categorical measure of treatment modification
Mutually exclusive categories to identify patient experience of treatment modifications were derived:
Patient experienced only one type of treatment modification (DR, TD or SE)
Patient experienced two types of treatment modification (DR, TD or SE)
Patient experienced all three types of treatment modification (DR, TD and SE)
Patient did not experience a known type of treatment modification (‘No’ recorded at least once for all of DR, TD and SE and no ‘Yes’ recordings)
Patient had only ‘Missing’ recordings for all type of treatment modification (DR, TD and SE)
Outcomes: composite outcome of treatment modification
A composite outcome was derived to measure association between weight loss and patient’s first recorded treatment modification. Composite criteria ‘Yes’ patients had at least one ‘Yes’ recorded for DR, TD or SE. Composite criteria ‘No’ patients did not experience a treatment modification (patients had only ‘No’ recorded for all of DR, TD or SE during follow-up time). Patients with only ‘Missing’ treatment modification recordings during follow-up time or with at least one ‘Missing’ recording in a prior SACT regimen to the composite outcome were excluded from association analysis. Figure 1 describes patient eligibility criteria for association analysis.
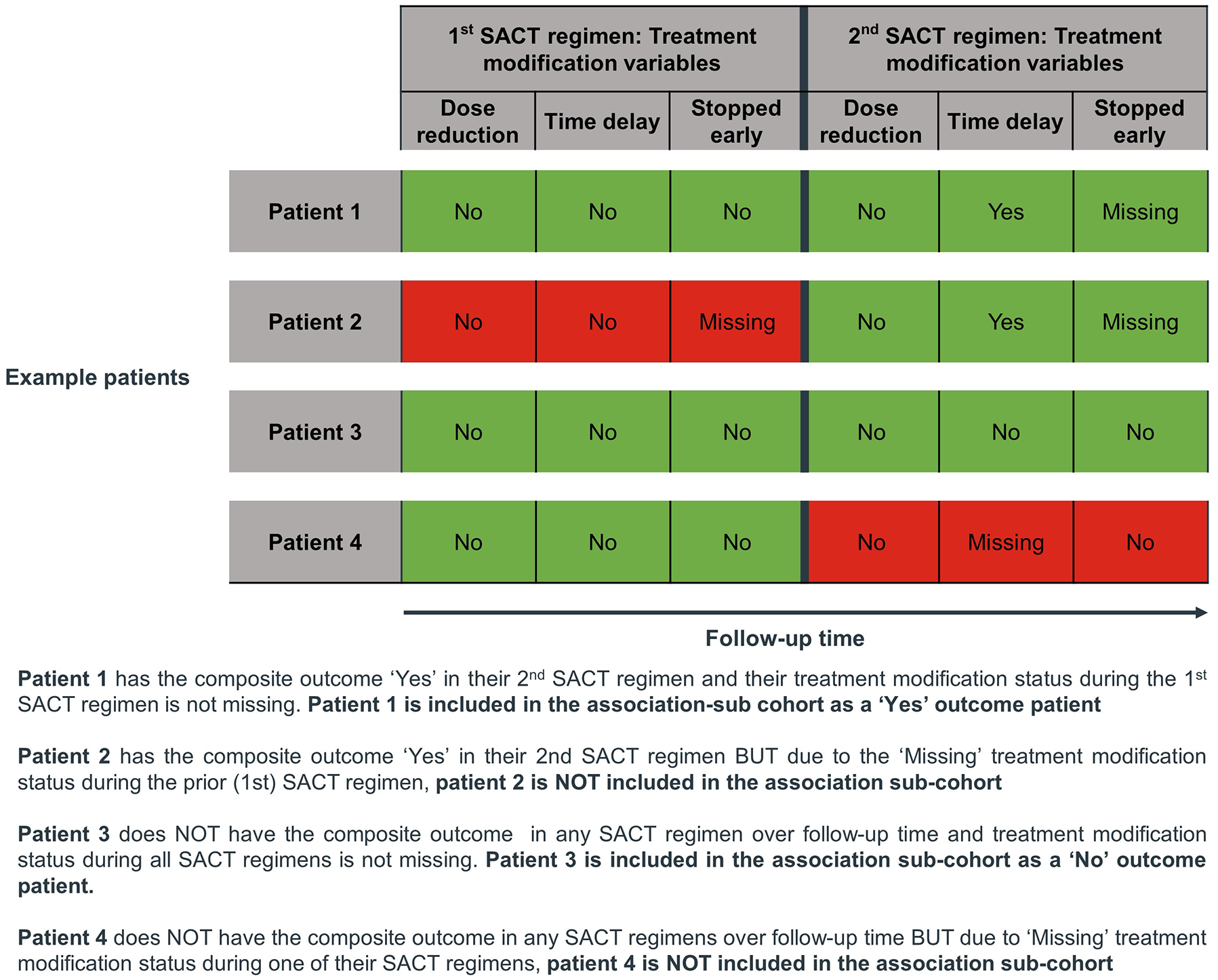
Example sequence of treatment modification data recording in the SACT dataset that was required to identify patients eligible for inclusion in the association sub-cohort.
Date of treatment modification is not captured. To ensure patient weight loss status was proximal to the composite outcome, follow-up time was censored at start date of the SACT regimen immediately after the regimen in which the composite outcome was identified. For association analysis, exposure variables were re-evaluated, inclusive of only weight recordings made prior to composite outcome-censored follow-up time.
Statistical analysis
Statistical analysis was completed using R version 3.6.1 (R Core Team, 2019). Descriptive statistics were used to describe patient demographic and clinical characteristics sub-grouped by binary weight loss status. Exposures were described by cancer and the categorical outcome was described by binary weight loss status and BMI-WLG, sub-grouped by cancer. Mean [with standard deviation (SD) or 95% confidence intervals (CIs) as applicable] were described for continuous variables.
Association analysis was conducted between exposures (binary weight loss status, BMI-WLG) and the composite outcome using crude and adjusted logistic regression modelling. Odds ratios (OR), 95% CIs and p-values were reported. Patients with missing BMI were not included in models where BMI-WLG was the exposure.
A priori confounders, recorded in COSD or SACT data, BMI (at first weight recording), age (at SACT initiation), sex, follow-up time and receipt of concomitant radiotherapy were included in adjusted regression models. BMI was removed as a confounder from the BMI-WLG models. Adjusted models were reported sub-grouped by cancer.
Results
Overall, a total of 200,536 SACT-treated cancer patients were included in our study population. Number of patients in the study population by cancer grouping was as follows; brain/CNS (n = 3723), breast (n = 45,260), colon (n = 18,400), gynaecologic (excl. ovarian) (n = 6505), head and neck (n = 6096), leukaemia (n = 5538), lower GI (n = 10,959), lung (n = 28,469), lymphoma (n = 20,428), myeloma (n = 7521), oesophageal (n = 8095), ovarian (n = 7983), pancreatic (n = 5762), sarcoma (n = 1118), skin (melanoma only) (n = 1502), stomach (n = 4729), upper GI (other) (n = 3548), and urology (n = 14,900).
Most patients (72%) did not experience weight loss during SACT treatment (Table 2). The mean (SD) age of non-weight loss patients (n = 144,271) was 60.7 (13.5) and 43% were male. Weight loss patients’ (n = 56,265) mean age was 62.4 (12.8) and 48% were male. Weight loss patients had a greater proportion (33%) of stage IV cancer patients (at diagnosis) than non-weight loss patients (25%) and more weight loss patients (41%) died during the study time period than non-weight loss patients (26%).
Demographic and baseline characteristics by patient (binary) weight loss status.

%, percentage; BMI, body mass index; CNS, central nervous system; GI, gastro-intestinal; ICD-10, International Statistical Classification of Diseases 10th revision; n, count; SACT, systemic anti-cancer therapy; SD, standard deviation.
Across cancer groupings most patients did not experience binary weight loss during SACT treatment (Table 3). Cancers with the highest percentage of weight loss patients were leukaemia (39%), oesophageal (43%), pancreatic (42%), sarcoma (44%), and stomach (44%) cancers (Table 3). The same cancers (except sarcoma) also contained greatest proportions (~8%) of ‘severe weight loss’ patients.
Description of BMI and non-BMI-adjusted measurements of patient weight loss in the study population by cancer grouping.

%, percentage; BMI, body mass index; BMI-WLG, BMI-adjusted weight loss grade; CI, confidence intervals; CNS, central nervous system; GI, gastro-intestinal; ICD-10, International Statistical Classification of Diseases 10th revision; IQR (interquartile range).
Breast and urology cancer patients saw minimal weight loss during SACT treatment; their mean (95% CI) most negative percentage weight change from baseline was −1.46 (–1.51, –1.41) and −1.77 (–1.88, –1.66), respectively. For stomach and oesophageal cancer patients, cancers with greater proportions of ‘severe weight loss’ patients, the mean (95% CI) most negative percentage weight change from baseline was −4.66 (–4.87, –4.44) and −4.39 (–4.56, –4.23), respectively.
Cancers with a high proportion of ‘severe weight loss’ patients (oesophageal, pancreatic and stomach) also had greatest proportion of patients with BMI-WLG ⩾3; oesophageal (30%), pancreatic (33%) and stomach (32%).
Table 4 summarises categorical assessment of patient experience of treatment modifications by binary weight loss status and BMI-WLG. Across all cancers, (binary) weight loss patients were proportionately more likely to experience two or more types of SACT treatment modification over follow-up time compared with non-weight loss patients (weight loss versus non-weight loss); breast cancer (17% versus 10%), colon cancer (28% versus 21%), pancreatic cancer (27% versus 18%), lower GI cancers (26% versus 18%), myeloma (30% versus 20%), and upper GI other cancers (22% versus 16%).
How patients with SACT treatment modifications are distributed between weight loss and non-weight loss patients by cancer grouping.

‘No’ recorded at least once for all of DR, TD and SE and no ‘Yes’ recordings.
1 In line with small number suppression guidelines as outlined by our Public Health England affiliated data partner, counts <10 and their accompanying percentages have been suppressed and replaced with ‘*’. Complimentary data suppression has also been conducted to remove possibility of patient reidentification.
BMI-WLG, body mass index-adjusted weight loss grade; CNS, central nervous system; DR, dose reduction; GI, gastro-intestinal; ICD-10, International Statistical Classification of Diseases 10th revision; SACT, systemic anti-cancer therapy; SE, stopped early; TD, time delay.
Additionally, across all cancers, a greater proportion of non-weight loss patients did not experience a known treatment modification of any type during SACT treatment compared with weight loss patients. Similar trends were found when comparing patients with high BMI-WLG (⩾2) against patients with low BMI-WLG (0 or 1).
Of the study population, 43% (n = 86,991) were eligible for association analysis. Of these patients, 5% (n = 4706) had missing BMI and were excluded from BMI-WLG association analyses.
Most patients (81%) in the association sub-cohort experienced the composite outcome, 29% (n = 25,371) of patients experienced weight loss (binary) and 66% of patients had a BMI-WLG of 0 or 1, whilst 9%, 16%, and 3% had BMI-WLGs of 2, 3, and 4, respectively Table 5 reports these data by cancer grouping.
Weight loss characteristics and composite outcome of patients in the association sub-cohort by cancer grouping.
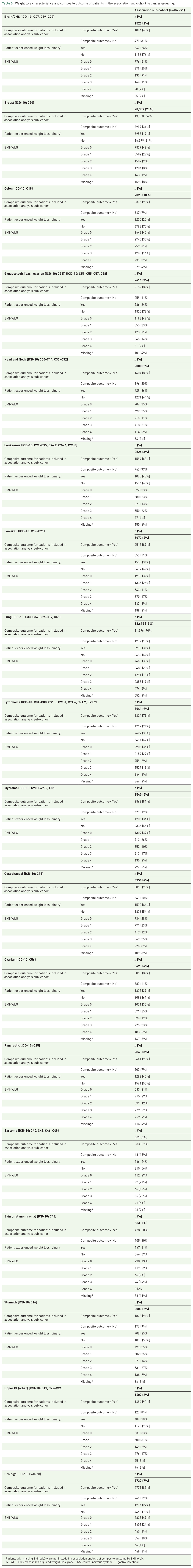
Patients with missing BMI-WLG were not included in association analysis of composite outcome by BMI-WLG.
BMI-WLG, body mass index-adjusted weight loss grade; CNS, central nervous system; GI, gastro-intestinal.
When adjusting for a priori confounders, binary weight loss during SACT treatment was associated with an increased likelihood of patients having a treatment modification in the following cancers: colon (OR: 1.72, 95% CI: 1.42, 2.07); gynaecologic (excl. Ovarian) (OR: 1.48, 95% CI: 1.08, 2.01); stomach cancer (OR: 1.46, 95% CI: 1.04, 2.06); lung (OR: 1.38, 95% CI: 1.211.58); leukaemia (OR: 1.30, 95% CI: 1.09, 1.55); head and neck (OR: 1.30, 95% CI: 1.02, 1.65), and oesophageal (OR: 1.29, 95% CI: 1.01, 1.64) (Figure 2 and Table 6). Weight loss during SACT treatment in breast cancer patients was associated with reduced likelihood of experiencing a treatment modification compared with non-weight loss patients (OR: 0.86, 95% CI: 0.79, 0.93) (Figure 2 and Table 6).

Forest plot of adjusted odds ratios for the association between binary weight loss and the composite outcome (likelihood of experiencing a treatment modification) by cancer grouping in the association sub-cohort. Plot includes number of patients within each cancer grouping of the association sub-cohort and accompanying 95% confidence intervals and p-value of the adjusted odds ratio.
Outputs of crude and adjusted model of association between binary weight loss (yes/no) and composite outcome by cancer grouping.

group includes: Upper GI (other) (ICD-10: C17, C22–C24), Stomach (ICD-10: C16), Pancreatic (ICD-10: C25), Oesophageal (ICD-10: C15), Colon (ICD-10: C18).
95% CIs, confidence intervals; BMI, body mass index; log, logarithmic; CNS, central nervous system; GI, gastro-intestinal; SACT, systemic anti-cancer therapy.
A combined ‘grouped GI’ [grouping inclusive of; upper GI (other), stomach, pancreatic, oesophageal, and colon cancers] was assessed during association analysis. For patients with lung, colon and grouped GI (including colon) cancers, association between BMI-WLGs and experiencing a SACT treatment modification increased with increasing BMI-WLG for both adjusted and crude models (Table 7). In these cancers, patients with BMI-WLG ⩾ 2 had at least 35% greater odds of having a treatment modification over follow-up time compared with patients with a BMI-WLG of 0 (reference; grade of best predicted patient prognosis). 1
Crude and adjusted model of association between BMI-WLG and composite outcome by cancer grouping.

group includes: Upper GI (other) (ICD-10: C17, C22–C24), Stomach (ICD-10: C16), Pancreatic (ICD-10: C25), Oesophageal (ICD-10: C15), Colon (ICD-10: C18).
95% CIs, confidence intervals; BMI-WLG, body mass index-adjusted weight loss grade; CNS, central nervous system; GI, gastro-intestinal; log, logarithmic; SACT, systemic anti-cancer therapy.
Discussion
To our knowledge, this is the largest (n = 200,536) and most comprehensive pan-cancer study of SACT-treated patients in England, achieved using the real-world CAS database. Our findings highlight the common occurrence of weight loss-associated SACT treatment modifications across 18 different cancer groupings and demonstrate how previously validated thresholds of weight loss could be used as early identifiers of patients vulnerable to cancer treatment disruptions.
It is known that treatment toxicity is exacerbated by patient weight loss and such toxicities can lead to chemotherapy treatment disruptions.4,12 Treatment disruption is associated with poorer response to treatment, an increased chance of disease progression, and a decreased survival rate. 12 Our findings show, across studied cancers, weight loss patients were more likely to experience multiple types of treatment modification over follow-up time than non-weight loss patients. Patients with higher BMI-WLGs were also more likely to experience multiple treatment modifications over follow-up time compared with patients with lower grades across the majority of cancers studied.
Cancers in this study with greatest percentages of weight loss patients (oesophageal, pancreatic, and stomach) are consistently noted in the literature as prone to involuntary weight loss during SACT treatment.4,12,13
The strong association we found between weight loss during SACT and experience of treatment modification in GI-related, head and neck, lung, and leukaemia cancer groupings are also consistent with previous literature that identifies these cancers as prone to weight loss-exacerbated treatment toxicities.3,12,14
In lung, colon, and the grouped GI (including colon) cancers, patients with the highest BMI-WLG were those with greatest odds of having a treatment modification recorded during SACT treatment.
Cancers with negligible point estimates of weight loss from baseline, such as urology, saw no association between weight loss and treatment modification during follow-up time in our study. This would suggest avoidance of extreme acute weight changes during SACT mitigated modifications during treatment. Weight stability has been previously found to improve patient survival outcomes and decrease chances of disease progression by maintaining continuity of the patient’s SACT treatment.12,15,16
Nutritional interventions, including dietary counselling and oral nutritional supplements (ONS) have been demonstrated to prevent weight loss during SACT. 17 There is increasing evidence that clinical nutrition (CN) interventions (including ONS, enteral tube feeding and parenteral nutrition) are associated with decreased anti-cancer therapy toxicity, improved relative dose intensity with fewer treatment modifications, and improved treatment continuity.18,19
Patient weight was recorded relatively frequently throughout follow-up time; however, for the majority of weight loss patients their largest individual weight loss from baseline was only 2.5–5.9%.
European guidelines provide clear guidance on CN management of patients with extensive weight loss and malnutrition following diagnosis, but are less clear on management of patients with lesser amounts of weight loss, who may benefit from nutritional support to maintain weight stability during treatment.5–7 Mild weight loss is often overlooked. Referrals to dietitians tend to occur when weight loss is established (5% or greater) and patients have accumulated multiple nutritional barriers to maintaining adequate dietary intake. 7
Our weight loss patients were heavier and proportionally more likely to be obese at start of SACT treatment than our non-weight loss patients.
Healthcare professionals may lack the knowledge to identify weight loss-induced malnutrition or be aware of the impact that weight loss may have on tolerance to SACT in obese or overweight cancer patients, where clinical guidance lacks clarity.8,20 Patients with cancers prone to obesity and high BMI, for example those with colon cancer, often have their weight loss neglected and do not receive nutritional intervention. 8 Patients likewise may not recognise losing weight as having a negative impact on their treatment outcomes. 21
Our results identify cancers with greatest weight loss-associated treatment modification and suggest existing gaps in weight loss management. CN support during SACT treatment could help improve patient treatment outcomes in cancers with traditionally ‘under-recognised’ and ‘under-intervened’ weight loss-related treatment disruptions. Currently, most nutritional support planning focuses on patient weight loss status prior to SACT treatment.
In highlighting colon cancers as susceptible to weight loss-induced SACT treatment modification, our study shows a potential gap in current weight loss management of cancers common in overweight patients. Until 2016, there was no dedicated dietetic service for colorectal cancer patients in the UK. 22 Across cancers, limited CN resources are generally reserved for patients who present with malnutrition at diagnosis, or are being used in patients with advanced disease only or not used at all.8,23,24 Expansion of naïve dietetic services in under-intervened cancers and improving patient accessibility to nutritional support are potential steps to improve patients’ weight loss-associated treatment outcomes, and is recommended in recent UK guidelines for GI cancer treatment.25,26
Strengths and limitations
A key strength of this retrospective study is the large sample size and centralised, routine collection of cancer registry data via the CAS database. Approximately 95% of the population of England with systemic cancer treatments are covered by CAS.9,10 To our knowledge this is the largest (n = 200,536) and most comprehensive population-level study of SACT-treated patients in England, given our analysis of 18 unique cancer groupings.
It is a strength of our study that we were able to analyse patient experience of treatment modifications via the CAS database, as such treatment outcomes usually go under-recorded in EMR data and can remain confined to clinician notes. However, necessary assumptions and restrictions had to be made to our study design to accommodate for limitations in data capture in our real-world datasets. Since date of treatment modification is not recorded within the SACT dataset, we assumed patient weight loss status at time of treatment modification was represented by the patient’s most negative weight change from baseline. A cross-sectional study design was chosen as we cannot confirm weight loss occurred prior to treatment modification. Our results do not imply causality.
We restricted measures of association to between weight loss and our composite outcome as measuring repeated outcome events required temporal treatment modification data, unavailable in the SACT dataset. Furthermore, it is likely repeated treatment modification decisions made by clinicians are not independent and highly correlated. Analysis of total number of occurrences of a single type of treatment modification per SACT regimen could not be measured within this study.
Inability to measure patient weight loss status prior to SACT initiation is noted as a limitation of this study as patient weight loss prior to treatment often determines eligibility for nutrition support. We assume weight loss identified during treatment is predominantly involuntary in our cancer sub-populations given we found it was associated with increased likelihood of treatment modifications. Patient malnutrition or receipt of nutritional intervention is not recorded in CAS. We acknowledge our data cannot directly identify patients with malnutrition, but are able to identify patients with sufficient weight loss that should be an indicator of requirement of nutritional intervention.
Standard real-world evidence limitations apply to this study. The number of weight recordings within the SACT data may not reflect the total number of weight recordings made by the treating clinician. Under-reporting of weight data may lead to misclassification of exposures. Patient weight and height were restricted to a ‘viable’ range to remove infeasible or incorrectly inputted recordings from study.
Multivariate modelling was used to determine association between weight loss and treatment modification. Whether patients received additional treatment, such as radiotherapy or surgery administered independently of SACT, is not captured within the SACT or COSD datasets and is acknowledged as an unmeasured confounder of this study.
Over-stratification within our multivariate models was a problem for cancers with smaller sample sizes post composite outcome-censoring of follow-up time. We identify this as an issue for the interpretability of association within sarcoma and skin (melanoma only) cancers only.
Application of BMI-WLG to predict non-mortality outcomes such as quality of life has been demonstrated in prospective observational studies.27,28 Our descriptive analysis showed a trend of increasing proportions of patients experiencing multiple treatment modifications with increasing BMI-WLGs. However, association between increasing WLG and likelihood of treatment modification in our association sub-cohort appeared only detectable in cancers with substantial sample size (n > 8600).
Conclusion
Our results provide comprehensive, population-level insights into the prevalence of weight loss in SACT-treated cancer patients in England and identify cancers that are prone to weight loss-associated treatment modifications. We highlight potential gaps in awareness and management of patient weight loss during treatment which could be addressed with clearer guidelines of when nutritional interventions may benefit patient treatment outcomes.
Our report begins to demonstrate how clinically relevant weight loss thresholds could be applied to routinely collected patient EMR and could aid clinicians in tracking and treating early presentations of involuntary weight loss in SACT-treated cancer patients. However, the applicability of the BMI-WLG to predict patient likelihood of treatment modification from EMR data requires further exploration given the mixed results of this study. Our study highlights that a wider than expected population of cancer patients are vulnerable to weight loss-associated treatment modifications. Still, future evaluation of the beneficial role of weight stability on patient-reported outcomes and the role of nutritional interventions to maintain weight stability is recommended.
Footnotes
Acknowledgements
Data access and running of the analysis would have not been made possible without facilitation by Health Data Insight (HDI). HDI are a social enterprise affiliated with Public Health England that maintains the CAS database and provided access to the data so that the study could be conducted.
Authors’ note
This study represents independent research supported by the National Institute for Health Research (NIHR) Biomedical Research Centre at The Royal Marsden NHS Foundation Trust and the Institute of Cancer Research, London. The views expressed are those of the authors CS and NS and not necessarily those of the NIHR or the Department of Health and Social Care.
Contributors
AR, EW, and SN led on concept and design of the study. EW analysed and interpreted the data. AR, EW, SN as well as CS and NS actively and substantially contributed to the writing of this manuscript and performed literature searches. All authors read and approved the manuscript.
Conflict of interest statement
AR, EW and SN are employees of IQVIA who performed the study and received sponsorship from Baxter Healthcare Ltd. RW and JS are employees of Baxter Healthcare Ltd. These authors report no other conflict of interest.
NS and CS received no sponsorship for their participation in this study and report no conflict of interest in relation to this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors, AR, EW and SN disclose sponsorship from Baxter Healthcare Ltd for the research and publication of this article. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, and writing and publishing the results.
Statement of ethics and consent
Ethics approval was not sought for the present study. At no point did the authors have access to patient-level data. Authors were provided access to Simulacrum. The Simulacrum is a publicly available dataset that contains artificial patient-like cancer data, imitating the data held securely by the Public Health England’s NCRAS. The data is synthetic and does not contain any identifiable information about real patients, so there is no risk of breaching patient confidentiality.
Code performing the study analysis, adapted in the Simulacrum environment, was provided to Health Data Insight (HDI). HDI are a social enterprise affiliated with Public Health England. Access is highly controlled to ensure that patient confidentiality is always protected and no data is released from PHE without permission of the PHE Office for Data Release and the PHE Caldicott Guardian.
Author access to aggregate results, post running of the analysis by HDI, is classed as secondary users use and access to de-identified data compliant with the Information Commissioner’s Code of Anonymisation.
The National Cancer Registration and Analysis Service (NCRAS) in Public Health England collects data on all patients diagnosed with cancer from across the population of England and has permission to do so without specific patient consent under Section 251 of the NHS Act 2006. The data collected by PHE is then linked with other data to create the Cancer Analysis Service (the CAS).

