Abstract
Background:
This prospective, multicentre, observational INVIDIa-2 study is investigating the clinical efficacy of influenza vaccination in advanced-cancer patients receiving immune-checkpoint inhibitors (ICIs), enrolled in 82 Italian centres, from October 2019 to January 2020. The primary endpoint was the incidence of influenza-like illness (ILI) until 30 April 2020. All the ILI episodes, laboratory tests, complications, hospitalizations and pneumonitis were recorded. Therefore, the study prospectively recorded all the COVID-19 ILI events.
Patients and methods:
Patients were included in this non-prespecified COVID-19 analysis, if alive on 31 January 2020, when the Italian government declared the national emergency. The prevalence of confirmed COVID-19 cases was detected as ILI episode with laboratory confirmation of SARS-CoV-2. Cases with clinical-radiological diagnosis of COVID-19 (COVID-like ILIs), were also reported.
Results:
Out of 1257 enrolled patients, 955 matched the inclusion criteria for this unplanned analysis. From 31 January to 30 April 2020, 66 patients had ILI: 9 of 955 cases were confirmed COVID-19 ILIs, with prevalence of 0.9% [95% confidence interval (CI): 0.3–2.4], a hospitalization rate of 100% and a mortality rate of 77.8%. Including 5 COVID-like ILIs, the overall COVID-19 prevalence was 1.5% (95% CI: 0.5–3.1), with 100% hospitalization and 64% mortality. The presence of elderly, males and comorbidities was significantly higher among patients vaccinated against influenza versus unvaccinated (p = 0.009, p < 0.0001, p < 0.0001). Overall COVID-19 prevalence was 1.2% for vaccinated (six of 482 cases, all confirmed) and 1.7% for unvaccinated (8 of 473, 3 confirmed COVID-19 and 5 COVID-like), p = 0.52. The difference remained non-significant, considering confirmed COVID-19 only (p = 0.33).
Conclusion:
COVID-19 has a meaningful clinical impact on the cancer-patient population receiving ICIs, with high prevalence, hospitalization and an alarming mortality rate among symptomatic cases. Influenza vaccination does not protect from SARS-CoV-2 infection.
Introduction
Since the first outbreak of the COVID-19 pandemic in China at the end of 2019, up to mid-July 2020, more than 14,000,000 cases of infections from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have been diagnosed worldwide. 1 Clinical factors associated with progression to a more severe disease stage were elderly age, male sex, low lymphocyte count, and presence of comorbidities such as hypertension, diabetes and chronic obstructive pulmonary disease.2,3 Patients with cancer have been deemed as having high risk of severe events from COVID-19, and their high mortality rates have been reported in recent wide retrospective analyses, showing that 30-day all-cause mortality was independently associated with both general risk factors (older age, smoking status, comorbidities) and risk factors unique to oncological patients, like an Eastern Cooperative Oncology Group performance status (ECOG PS) of 2 or higher, and active cancer. 4 Although an exact estimation of lethality is difficult due to the high risk of under-diagnosis of asymptomatic and mild cases, the mortality rate reported up to today for cancer-patient populations developing COVID-19 was between 11% and 39% in wide case series from the United States, Italy and China.4–6
No data are available on the prevalence of COVID-19 in this patient population, given the lack of a denominator in the cited retrospective studies, only reporting the cancer-patient population known to be affected by SARS-CoV-2 and their outcome. Statistical estimates suggested that, at least in male population, patients with cancer have a higher risk of SARS-CoV-2 infection [odds ratio (OR) 1.79; confidence interval (CI) 1.62–1.98, p < 0.0001]. 7
On the other hand, case series from different countries heavily affected by the pandemic showed a high prevalence of cancer patients in the population with COVID-19.3,8 The Italian Medical Council described the characteristics of COVID-19 patients dying in Italy, based on data available on 30 March 2020, showing that 16.5% were patients being treated for a malignancy in the past 5 years. 9 These data indirectly suggest that, beyond their likely worst prognosis when developing COVID-19, the risk of being infected with SARS-CoV-2 could be higher for cancer patients. Of course, during the peak of the pandemic, the need for hospital visits and treatment administrations can be considered a risk factor in itself.
Beyond the higher risk of infections for patients receiving cytotoxic chemotherapy, 10 the possible correlation of SARS-CoV-2 infection with the type of systemic cancer therapy has not been clarified. Patients with cancer undergoing treatment with anti-programmed cell-death protein 1/anti-programmed cell-death ligand 1 (anti-PD-1/anti-PD-L1) or anti-cytotoxic T-lymphocyte-associated protein 4 (anti-CTLA-4) immune-checkpoint inhibitors (ICIs), currently used in the routine clinical practice to treat many solid tumours, constitute a growing oncological population, and their specific susceptibility to bacterial or viral infections has not been investigated. Some concerns have been recently expressed about the possible risk of administering ICIs in the middle of the COVID-19 outbreak, essentially due to two major issues: the potential overlap between the COVID-related interstitial pneumonia and the uncommon immune-related lung toxicity from ICIs, and the hypothesis of a synergy between ICI mechanism and COVID-19 pathogenesis in terms of cytokine-release syndrome. 11 On the other hand, considering that immunotherapy is able to restore the cellular immunocompetence, the patient undergoing ICI could be more immunocompetent than cancer patients undergoing chemotherapy, as our data previously suggested in the case of influenza infection. 12
With the aim of exploring these hypotheses and investigating the prevalence and the true clinical impact of COVID-19 in patients with advanced cancer treated with ICIs, we exploited the prospective data from the multicentre, observational INVIDIa-2 study, which is currently ongoing, performing a non-prespecified preliminary COVID-19 analysis.
The study is investigating the clinical efficacy of influenza vaccination in advanced-cancer patients receiving ICIs enrolled in 82 Italian centres from October 2019 to January 2020 and prospectively observed until April 2020. All the influenza-like illness (ILI) episodes, laboratory tests, complications, hospitalizations, and pneumonitis were prospectively recorded according to the study protocol. Therefore, the INVIDIa-2 study prospectively detected all the symptomatic COVID-19 events in the study population during the peak of the SARS-CoV-2 spread in our country.
Methods
INVIDIa-2 study design and participants
The INVIDIa-2 study is a multicentre, prospective, observational study, still ongoing in the follow-up phase. The primary objective is the assessment of the utility of vaccine administration in patients with advanced cancer undergoing systemic treatment with ICIs, by evaluating the impact of the influenza vaccination on the incidence of ILI. The primary endpoint is the incidence of ILI from 1 October 2019 until 30 April 2020.
The secondary objectives include the description of the vaccine coverage and its impact in terms of reduction of severity and fatality of ILI, data about pharyngeal and/or nasal swabs used in clinical practice for the differential diagnosis of ILI, and serological laboratory test confirmations. In order to measure the entity of the indication bias deriving from the observational nature of the study, the characteristics of the population in the two groups of vaccinated and non-vaccinated patients, have been collected and compared.
All patients with advanced solid tumours receiving therapy with ICI (alone or in combinations) from 1 October 2019 to 31 January 2020 (corresponding to the duration of the influenza vaccine season) were eligible. The influenza season, from October 2019 to April 2020, constituted the ILI observation period, with a cut-off date on 30 April 2020 for both the primary endpoint and the secondary endpoints regarding the severity and fatality of ILI. The timing of the original study is shown in Supplemental Figure S1.
The study involves 82 Italian centres; the enrolment target was of 974 patients according to the sample size calculation (details not shown, original protocol available in the supplemental materials); an over-accrual of 30% was provided, based on the expected rate of screening failures.
ILI, according to the European Centre for Disease Prevention and Control (ECDC), has been defined as a sudden and rapid onset of one or more of the following general symptoms: fever or low-grade pyrexia, malaise and/or fatigue, headache, myalgia; and at least one of the following respiratory symptoms: cough, sore throat, dyspnoea. 13 ‘Sudden and rapid’ is taken to mean to the extent that the date of symptom onset can be clearly identified. Confirmed influenza or other confirmed infection was referred to cases confirmed by pharyngeal/nasal swab and/or serology tests.
The trial is registered in the European electronic database of clinical trials [EudraCT no.: 2020-002603-18]. Local institutional review board approval was required for each centre for inclusion in the study. Written informed consent was obtained for all enrolled patients. All study procedures were in accordance with the precepts of good clinical practice and the Declaration of Helsinki.
COVID-19 analysis methods
Despite the COVID-19 analysis obviously not being prespecified, due to the unawareness of the SARS-CoV-2 outbreaks at the time of the protocol designing, the protocol provided the differential diagnosis of ILI. All the ILI episodes, laboratory tests, complications, hospitalizations, and pneumonitis were collected in a validated electronic case report form (eCRF) platform (OpenClinica). Therefore, the study prospectively recorded all the COVID-19 ILI events within the enrolled patient population.
The primary endpoint of this non-prespecified COVID-19 analysis was the prevalence of confirmed COVID-19 cases in the exposed population, detected as ILI episodes with laboratory confirmation of SARS-CoV-2 infection, occurring between 31 January 2020 and 30 April 2020. This time frame was selected as corresponding to the COVID-19 outbreak in Italy, and includes the peak of incidence, reached in March 2020.
As co-primary endpoint, we assessed the overall COVID-19 prevalence, including cases with clinical-radiological diagnosis of COVID-19 without laboratory confirmation (not performed, or performed and resulting in negative for SARS-CoV-2), defined as COVID-like ILIs.
The secondary endpoints included the hospitalization rate and the mortality rate of COVID-19 cases; their clinical characteristics compared with those of patients without COVID-19 occurrence; their overall survival (OS) compared with that of patients developing non-COVID ILI. Each secondary endpoint was evaluated both for the confirmed COVID-19 population and in the overall COVID-19 group.
The inclusion criterion for this non-prespecified COVID-19 analysis was the potential exposure to SARS-Cov-2 infection, namely, the patients must be alive on 31 January 2020 (this date has been chosen conventionally, corresponding to the declaration of national emergency by the Italian government, before the first local case was officially diagnosed). The prevalence of COVID-19 was assessed among patients with ILI symptoms. Confirmed COVID-19 cases were defined with SARS-CoV-2 nasal and/or pharyngeal swab positivity according to the local clinical practice. COVID-like cases were defined as epidemiological, clinical and radiological characteristics with high suspicion for COVID-19, irrespective of the laboratory confirmation, considering on one hand the high pre-test probability of SARS-CoV-2 infection, and on the other hand, the suboptimal sensitivity of real-time polymerase chain reaction (RT-PCR) tests, with a high percentage of false-negative findings. 14
Statistical analysis
Data related to quantitative variables were reported using the median and interquartile range (IQR), and absolute count and percentages when referring to categorical items. Association between categorical variables was evaluated with the χ 2 test. Differences in lymphocyte distribution were tested with the Friedman one-way repeated measures analysis of variance by ranks. OS was calculated from the onset of ILI symptoms to the date of death or censored at the last available follow-up date. Survival curves were estimated by the Kaplan–Meier method and compared with the log-rank test.
Role of the funding source
The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the publication.
Results
The INVIDIa-2 study prospectively enrolled 1257 advanced-cancer patients receiving ICIs. According to the inclusion criteria, 955 patients were eligible for the present COVID-19 analysis. Their characteristics are summarized in Table 1.
Characteristics of patients included in the COVID-19 analysis of the INVIDIa-2 study.
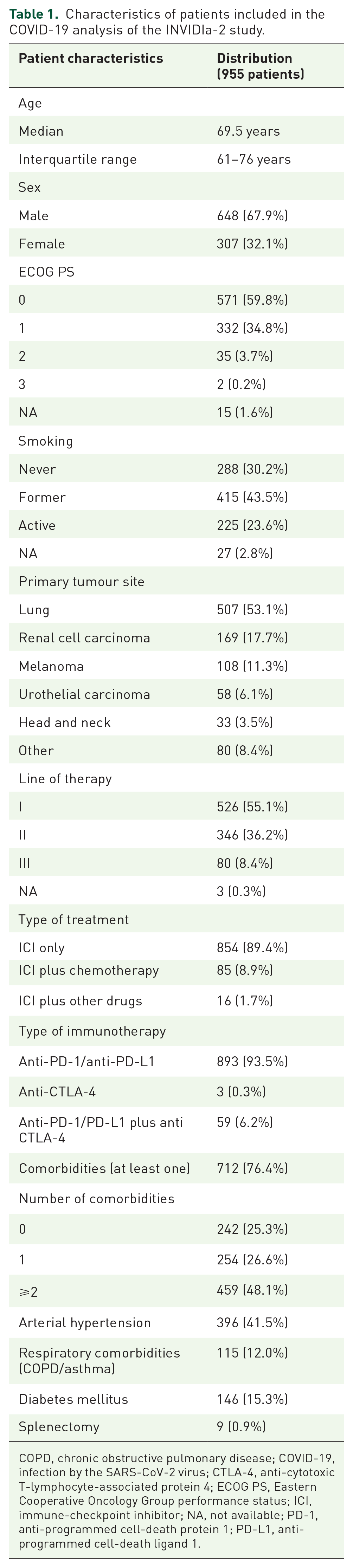
COPD, chronic obstructive pulmonary disease; COVID-19, infection by the SARS-CoV-2 virus; CTLA-4, anti-cytotoxic T-lymphocyte-associated protein 4; ECOG PS, Eastern Cooperative Oncology Group performance status; ICI, immune-checkpoint inhibitor; NA, not available; PD-1, anti-programmed cell-death protein 1; PD-L1, anti-programmed cell-death ligand 1.
Patients who received influenza vaccination were 482, versus 473 who were not vaccinated: the characteristics were unbalanced between the two groups, since vaccination was more frequently administered in males (p = 0.009), smokers (p < 0.0001), lung cancer patients (p = 0.003), patients with comorbidities (p < 0.0001) and elderly (p < 0.0001).
There were 66 patients with ILI episodes occurring after 31 January 2020. The confirmed COVID-19 ILIs were 9, with prevalence of 0.9% (95% CI: 0.3–2.4; 9/955 cases) of the study population. Including COVID-like ILIs, diagnosed in five patients, the overall COVID-19 prevalence in advanced-cancer patients treated with ICIs was 1.5% (95% CI: 0.5–3.1; 14/955 cases). Figure 1 shows the distribution of ILI cases after 31 January 2020. The probability that an ILI episode was COVID related in the considered time frame for this patient population ranged from 13.6% to 21.2%. The positivity rate of the performed swabs in the ILI population was 34.6% (nine positive swabs out of 26 performed in the ILI population).

Pie graph representing the distribution of patients developing influenza-like illness (ILI) after 31 January 2020.
Confirmed COVID-19
For nine patients, the clinical diagnosis of COVID-19 was confirmed by positive RT-PCR on nasal and/or pharyngeal swab. Their characteristics were reported in Table 2. For all patients, the ICI treatment was an anti-PD-1/anti-PD-L1 antibody.
Characteristics of patients developing influenza-like illness with laboratory confirmation of SARS-CoV-2 infection (defined as confirmed COVID-19).

COPD, chronic obstructive pulmonary disease; COVID-19, infection by the SARS-CoV-2 virus; ECOG PS, Eastern Cooperative Oncology Group Performance Status, collected at immunotherapy initiation; F, female; HIV, human immunodeficiency virus; ICI, immune-checkpoint inhibitor; M, male.
None of the characteristics among sex, smoking status, primary tumour, or comorbidities were related to confirmed COVID-19 occurrence, except splenectomy (11.1% in splenectomised patients versus 0.8% in non-splenectomised, p = 0.002; Supplemental Table S1). The influenza vaccination was not related to the confirmed COVID-19 prevalence, which was 1.2% (95% CI: 0.2–3.7) in vaccinated patients versus 0.6% (95% CI: 0.1–1.8) for unvaccinated (p = 0.33).
The clinical features of the confirmed COVID-19 ILIs are described in Table 3: almost all patients presented fever and dyspnoea; all patients developed interstitial pneumonia, diagnosed at the computed tomography (CT) scan or with chest X-ray (RX). The hospitalization rate was 100%; seven of nine patients died due to COVID-19, with mortality rate of 77.8% (95% CI 40.1–96.1 with continuity correction). The median OS (mOS) of patients developing COVID-19-related ILI was 10 days (95% CI: 2–20); it was significantly shorter compared with that of non-COVID ILI, which was not reached [p < 0.0001; Figure 2(a)].
Clinical features of influenza-like illness episodes with confirmed diagnosis of COVID-19.
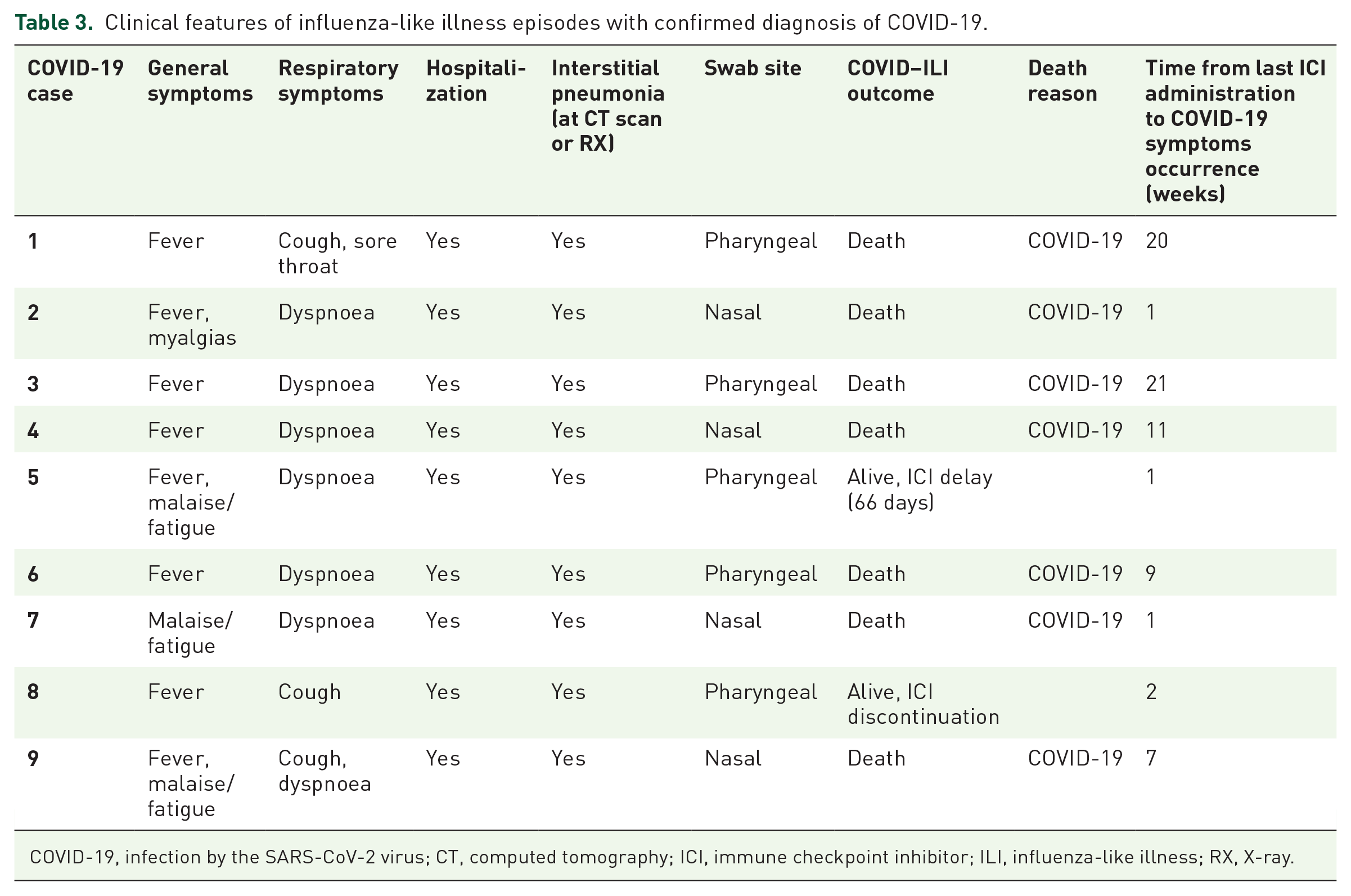
COVID-19, infection by the SARS-CoV-2 virus; CT, computed tomography; ICI, immune checkpoint inhibitor; ILI, influenza-like illness; RX, X-ray.

Kaplan–Meier survival curves for patients with confirmed COVID-19 influenza-like illness (ILI) and patients with non-COVID ILI.
Overall COVID-19
For five further patients, whose characteristics were reported in Supplemental Table S2, the diagnosis of COVID-19 was reached considering the clinical features (summarized in Supplemental Table S3), the radiological finding of interstitial pneumonia in all five cases, and the epidemiological context of the COVID-19 outbreak peak in Italy and in the specific region of the respective participating centre. In all five cases, nasal/pharyngeal swab for SARS-CoV-2 was performed with negative results.
Considering the COVID-like ILIs together with the confirmed COVID-19, the overall COVID-19 ILI group (14 patients) had similar behaviour to that previously reported for the confirmed cases, without any correlation to clinical variables other than splenectomy (p = 0.016; Supplemental Table S1). The influenza vaccination was not related to the overall COVID-19 prevalence, which was 1.2% among vaccinated patients versus 1.7% for unvaccinated (p = 0.56).
The hospitalization rate was 100% also in this subgroup, and two of five patients died due to COVID-like ILI in addition to deaths from confirmed COVID-19, with mortality rate in the overall COVID-19 population of 64.3% (95% CI: 35.6–86.0 with continuity correction).
The mOS of patients with COVID-like ILI was not reached, but life expectancy was shorter compared with that of patients with confirmed COVID-19 ILI and longer than that of non-COVID-ILI patients [p < 0.0001; Figure 2(b)].
The lymphocyte count of the overall COVID-19 cases, assessed at baseline before immunotherapy start, during immunotherapy and finally close to the COVID-19 ILI development, showed a statistically significant downward trend (Figure 3; p = 0.035).

Spider plot representing the trend of the lymphocyte count in patients with confirmed COVID-19 or COVID-like ILI at three timepoints.
Discussion
The prevalence of COVID-19 in a patient subgroup from the overall population is a time- and site- dependent statistic, even in a pandemic scenario, difficult to interpret without its epidemiological contextualization. The COVID-19 outbreak in Italy started at the end of January 2020 with the first two cases imported from China, leading to the subsequent local transmission of the infection, with increasing incidence from February 2020 until the plateau reached in April 2020, after the peak of the Italian outbreak, occurred in March 2020. On 30 April 2020, in Italy, over a population of 60,317,000 inhabitants, the confirmed cases of COVID-19 were 205,463, of whom 13.6% were dead, 0.8% were inpatients in intensive care units, 8.8% were hospitalized due to severe symptoms, 39.8% were outpatients with active illness and 37% were healed. 15 The mortality rate of 13.6% for COVID-19 in our country at the end of April was calculated considering the overall denominator of asymptomatic and symptomatic infections, mainly detected by RT-PCR swab screening, carried out in leopard spots in the national population. On the other hand, if the nationwide incidence of SARS-CoV-2 infection in Italy was about 0.34% according to the previously cited data, when considering symptomatic cases only, it was likely around 0.21%, as also confirmed by regional reports, and the estimated mortality rate of symptomatic COVID-19 was 21.6%.7,15
In this epidemiological context, the prevalence of 0.9% for confirmed COVID-19, prospectively found in our study population, can be interpreted as high. Furthermore, this incidence was even probably underestimated, missing asymptomatic cases.
If, on one hand, the hospitalization rate of 100% could be interpreted as a deliberate choice of the clinicians to keep under close control these frail patients when developing even mild forms of this harmful infection, on the other hand, the upsetting mortality rate, almost reaching 78% of cases with mOS of only 10 days from the onset of symptoms, overcame our expectations. The underestimation of the denominator, missing the share of patients with asymptomatic SARS-Cov-2 infections, can partially justify such a high mortality. Moreover, the mortality estimate could lack precision due to the relatively small number of cases. Beyond these considerations, our findings provide a clear demonstration that COVID-19 has a statistically significant and clinically meaningful worse OS when compared with other influenza-like infections in the advanced-cancer patient population [see Figure 2(a)]. The intermediate OS of patients with COVID-like ILI episodes [shown in Figure 2(b)] could subtend the likely mixed etiopathogenesis of such illnesses, clinically attributed to SARS-CoV-2 due to their presentation and radiological features, but possibly partially due to other viral infections, including the classic influenza virus (of note, none of the COVID-like cases received the flu vaccine, while the majority of confirmed COVID-19 was vaccinated) or even to ICI-related pneumonia. 16
The results of the present analysis should be read in light of our patient characteristics. Before considering their cancer treatment, the patients in this study population are affected by advanced cancer, with advanced median age (69.5 years), male prevalence (2:1), significant comorbidity, and more than 50% of patients having lung cancer. All these variables have been related to high risk of severe events and death from COVID-19.2–4,7,17,18 Furthermore, also within the population of cancer patients, significant difference in the morbidity from SARS-CoV-2 infection has been previously seen in retrospective subgroups, with higher risk of death (OR: 5.58; 95% CI: 1.71–18.23; p = 0.01) and severe events reported for patients with metastatic cancer, but not for patients with non-metastatic tumours, when compared with patients without cancer. 19 Of note, the INVIDIa-2 study included only active advanced cancers, negatively selecting the patient population irrespective of the systemic treatment administered. The treatment with ICI constitutes another concern about the possible risk factors for COVID-19, as recently argued with controversial rationale.11,20,21
On the other hand, the currently available data seem to testify in favour of better outcome of cancer patients with COVID-19 when previously treated with immunotherapy rather than with chemotherapy,19,22,23 and despite the severity of the disease developed by lung cancer patients, PD-1 blockade exposure has not been associated with increased severity of COVID-19. 24 With lack of current prospective evidence about COVID-19 incidence and severity in cancer patients treated with chemotherapy or any other targeted cancer drugs, and without a control arm in our study, definitive conclusions about the possible contribution of the systemic treatment with ICI to COVID-19 morbidity cannot be drawn.
The study findings about the influenza vaccine do not show association between vaccination and COVID-19 morbidity. On the other hand, the characteristics of vaccinated patients were strongly in favour of a high risk of severe COVID-19. Even in the lack of direct advantage from flu vaccine administration with respect to SARS-CoV-2 infection, while a COVID-19 vaccine is still missing, safe and quite effective influenza vaccines are available. Considering the possible convergence of a new outbreak of COVID-19 simultaneously with the epidemic of seasonal influenza, the widespread use at least of the flu vaccine will be even more crucial, both reducing the probability of viral pneumonia and facilitating the differential diagnosis of ILI.
Mention is deserving of the significant decrease of lymphocyte count found in our COVID-19 patients from the beginning of immunotherapy to the development of infection. It has been reported that CD4+ and CD8+ lymphocytes are significantly low in severe/critical COVID-19 patients. 17 The phenomenon of lymphocyte depletion has demonstrated unfavourable prognostic implications both in cancer and COVID-19.25,26 Furthermore, recent data demonstrated that surviving T cells during COVID-19 are functionally exhausted. 25 The lymphocyte decrease shown in our patients developing COVID-19 was consistent with the poor prognosis of the disease in our population.
The present study has some limitations: the COVID-19 analysis was not prespecified; as a consequence, the prospectively recorded data lacked useful COVID-specific details, for example, extra-ILI symptoms (such as anosmia and dysgeusia) and the specific therapeutic approach to COVID-19 used in our patients. Another relevant limitation is the lack of information about asymptomatic COVID-19: the study was not designed to detect subclinical infections and did not include any collection data from laboratory tests performed as screening in the absence of symptoms. In addition, in Italy, during the study period, the laboratory tests were performed only in the case of severe symptoms according to the World Health Organization definition. 27 This approach could have favoured late diagnosis, increasing the need for hospitalization and leading to the high mortality rate in this frail patient population. Even more, the healthcare system in Italy was in an heavy emergency state during the conduct of the study, likely negatively impacting on the outcome of these patients.
The strength of the present study is the prospective nature, providing reliable data about the impact of COVID-19 in advanced-cancer patients treated with immunotherapy, and also constituting the only prospective report on the occurrence of symptomatic SARS-CoV-2 infection in a wide cancer-patient population during the peak of the COVID-19 outbreak. Prospective data about patients receiving other cancer therapies would be warranted in the upcoming further outbreak, given both the high risk of bias from retrospective data collection and the difficulty of the post hoc identification of an unbiased denominator.
In conclusion, according to the risk factors previously demonstrated in the literature, the prevalence, hospitalization and mortality from symptomatic COVID-19 in our study population of advanced-cancer patients treated with ICI was meaningful, likely mainly due to metastatic disease and to general risk factors peculiar to cancer patients. In the current lack of curative treatment for COVID-19, a wider use of laboratory diagnostic tests in advanced-cancer patients treated with immunotherapy should be considered, aimed at the early detection of infection and offering the chance for preventing its progression to severe forms.
Supplemental Material
Supplementary_Material_-_online_only – Supplemental material for Symptomatic COVID-19 in advanced-cancer patients treated with immune-checkpoint inhibitors: prospective analysis from a multicentre observational trial by FICOG
Supplemental material, Supplementary_Material_-_online_only for Symptomatic COVID-19 in advanced-cancer patients treated with immune-checkpoint inhibitors: prospective analysis from a multicentre observational trial by FICOG by Melissa Bersanelli, Diana Giannarelli, Ugo De Giorgi, Sandro Pignata, Massimo Di Maio, Elena Verzoni, Alberto Clemente, Valentina Guadalupi, Diego Signorelli, Marcello Tiseo, Raffaele Giusti, Marco Filetti, Marilena Di Napoli, Lorenzo Calvetti, Alessandro Cappetta, Paola Ermacora, Diego Zara, Fausto Barbieri, Cinzia Baldessari, Vieri Scotti, Francesca Mazzoni, Antonello Veccia, Pamela Francesca Guglielmini, Marco Maruzzo, Ernesto Rossi, Francesco Grossi, Chiara Casadei, Alessio Cortellini, Francesco Verderame, Vincenzo Montesarchio, Mimma Rizzo, Manlio Mencoboni, Fable Zustovich, Lucia Fratino, Saverio Cinieri, Giorgia Negrini, Maria Banzi, Mariella Sorarù, Paolo Andrea Zucali, Gaetano Lacidogna, Antonio Russo, Nicola Battelli, Giuseppe Fornarini, Claudia Mucciarini, Sergio Bracarda, Andrea Bonetti, Debora Pezzuolo, Lucia Longo, Donata Sartori, Mauro Iannopollo, Luigi Cavanna, Fausto Meriggi, Davide Tassinari, Claudia Corbo, Angela Gernone, Veronica Prati, Simona Carnio, Pasqualina Giordano, Angela Maria Dicorato, Claudio Verusio, Francesco Atzori, Francesco Carrozza, Stefania Gori, Antonino Castro, Sara Pilotto, Vanja Vaccaro, Elisabetta Garzoli, Francesco Di Costanzo, Evaristo Maiello, Roberto Labianca, Carmine Pinto, Michele Tognetto and Sebastiano Buti in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We gratefully acknowledge the patients, their caregivers and their general practitioners, contributing to the data collection during the time frame of the Italian lockdown. Moreover, we acknowledge the professionalism of the staff of FULLCRO for the outstanding management of the trial, and of DataRiver for the management of the eCRF validated platform.
Author contributions
Melissa Bersanelli had the original idea for the study and provided the study conception and design. She also coordinated and supervised the study conduct. Sebastiano Buti, Diana Giannarelli, Ugo De Giorgi, Sandro Pignata and Massimo Di Maio, contributed to the study design. Francesco Di Costanzo, Evaristo Maiello, Roberto Labianca, Carmine Pinto, and Michele Tognetto contributed to the study design and coordination and provided the administrative support. Alberto Clemente, Diana Giannarelli, Sebastiano Buti and Melissa Bersanelli contributed to the acquisition and analysis of data, and together with Ugo De Giorgi, Sandro Pignata and Massimo Di Maio, contributed to their interpretation. Melissa Bersanelli drafted the first version of the manuscript; Sebastiano Buti, Diana Giannarelli, Ugo De Giorgi, Sandro Pignata and Massimo Di Maio, contributed to the final version and revised it critically for important intellectual content. Melissa Bersanelli, Ugo De Giorgi, Sandro Pignata, Massimo Di Maio, Elena Verzoni, Valentina Guadalupi, Diego Signorelli, Marcello Tiseo, Raffaele Giusti, Marco Filetti, Marilena Di Napoli, Lorenzo Calvetti, Alessandro Cappetta, Paola Ermacora, Diego Zara, Fausto Barbieri, Cinzia Baldessari, Vieri Scotti, Francesca Mazzoni, Antonello Veccia, Pamela Francesca Guglielmini, Marco Maruzzo, Ernesto Rossi, Francesco Grossi, Chiara Casadei, Alessio Cortellini, Francesco Verderame, Vincenzo Montesarchio, Mimma Rizzo, Manlio Mencoboni, Fable Zustovich, Lucia Fratino, Saverio Cinieri, Giorgia Negrini, Maria Banzi, Mariella Sorarù, Paolo Andrea Zucali, Gaetano Lacidogna, Antonio Russo, Nicola Battelli, Giuseppe Fornarini, Claudia Mucciarini, Sergio Bracarda, Andrea Bonetti, Debora Pezzuolo, Lucia Longo, Donata Sartori, Mauro Iannopollo, Luigi Cavanna, Fausto Meriggi, Davide Tassinari, Claudia Corbo, Angela Gernone, Veronica Prati, Simona Carnio, Pasqualina Giordano, Angela Maria Dicorato, Claudio Verusio, Francesco Atzori, Francesco Carrozza, Stefania Gori, Antonino Castro, Sara Pilotto, Vanja Vaccaro, Elisabetta Garzoli, Carmine Pinto, and Sebastiano Buti enrolled patients and provided substantial contributions to the acquisition of data for the work. All authors revised the work critically for important intellectual content, approved the version to be published and agreed to be accountable for all aspects of the work.
Availability of data and material
The study protocol and the validation certificate for the eCRF platform will be made immediately available by the corresponding author upon request (
Conflict of interest statement
The Federation of Italian Cooperative Oncology Groups (FICOG) received funding for the present study by Seqirus and Roche S.p.A.; the Federation also received funding during the conduct of the present study by Astra Zeneca, Bristol Myers Squibb (BMS), Sanofi. Melissa Bersanelli received funding for the present study by Seqirus and Roche S.p.A. (FICOG as Institution, no personal fees). She also received, outside the present work, research funding from Pfizer and Novartis (Institution), honoraria as speaker at scientific events (personal fees) by Astra Zeneca, Bristol Myers Squibb (BMS), Novartis and Pfizer; as consultant for advisory role (personal fees) by Novartis, BMS and Pfizer. Ugo De Giorgi received honoraria from AstraZeneca, Roche, MSD, Pfizer, GSK, Clovis, Incyte and research funding from Roche, AstraZeneca, MSD, Pfizer. Dr Di Maio reports personal fees from Bristol Myers Squibb, personal fees from Merck Sharp & Dohme, personal fees from AstraZeneca, personal fees from Janssen, personal fees from Astellas, personal fees from Pfizer, personal fees from Eisai, personal fees from Takeda, grants from Tesaro GSK, outside the submitted work. Sebastiano Buti received honoraria as speaker at scientific events and advisory role by BMS, Pfizer; MSD, Ipsen, Roche S.p.A., Eli Lilly, AstraZeneca and Novartis; he also received research funding from Novartis. Marcello Tiseo received honoraria (personal fees) by MSD, BMS, Boehringer (BI), Takeda, AstraZeneca, and research funding by AstraZeneca (Institution). Vieri Scotti participated, with personal fees, to advisory boards and speaker’s bureaus for Roche S.p.A. Dr Cortellini reports grants from AstraZeneca, grants from MSD, grants from BMS, grants from Roche, during the conduct of the study; grants from AstraZeneca, grants from MSD, grants from BMS, grants from Roche, grants from Novartis, outside the submitted work. Saverio Cinieri declared international board for Eli Lilly international. Paolo Andrea Zucali acts in a consultant or advisory role for Sanofi, BMS, Pfizer, MSD, Astellas, Janssen, Ipsen, Novartis, all outside the scope of work. Sergio Bracarda declares to have acted as advisory board member (uncompensated) for: Janssen, Astellas, Pfizer, MSD, BMS, Merck, AstraZeneca, AAA, Ipsen, Bayer, Roche/Genentech. Francesco Carozza declared personal fees from Janssen. Sara Pilotto reports personal fees from AstraZeneca, Eli Lilly, Boehringer Ingelheim, Merk & Co, Roche, outside the submitted work. All remaining authors have declared no conflicts of interest.
Consent for publication
All authors approved the manuscript and gave their consent for publication.
Ethics approval and consent to participate
The trial is registered in the European electronic database of clinical trials [EudraCT no.: 2020-002603-18]. Local institutional review board approval was required for each centre for the inclusion in the study. Written informed consent was obtained for all enrolled patients. All study procedures were in accordance with the precepts of good clinical practice and the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Federation of Italian Cooperative Oncology Groups (FICOG, which is also the promoter of the study), Roche S.p.A., Seqirus.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
